Abstract
Background:
Anti-CD38 monoclonal antibodies (mAbs) have significantly changed the multiple myeloma treatment landscape. This meta-analysis compared the efficacy and safety of anti-CD38 mAb-based therapy versus standard therapy in newly diagnosed multiple myeloma (NDMM) patients.
Methods:
We performed a comprehensive literature search on PubMed, the Cochrane Database, and ClinicalTrials.gov. The primary outcomes were progression-free survival (PFS) and minimal residual disease (MRD) status. Dichotomous outcomes were pooled using risk ratio (RR) along with the 95% confidence interval (CI) in RevMan 5.4. Subgroup analysis and meta-regression analysis were performed. The RoB 2.0 tool was used to assess the risk of bias.
Results:
Our meta-analysis included 11 randomized controlled trials. There were 5270 patients; 3040 TEs and 2230 TIEs. Anti-CD38 mAbs significantly improved MRD negativity (RR 1.94, 95% CI: 1.59–2.37; p < 0.00001) and PFS (RR 0.51, 95% CI: 0.45–0.58; p < 0.00001). Subgroup analyses revealed better outcomes for both the TE (MRD: RR 1.52, 95% CI: 1.37–1.68; PFS: RR 0.43, 95% CI: 0.34–0.54) and TIE (MRD: RR 3.49, 95% CI: 2.65–4.61; PFS: RR 0.55, 95% CI: 0.47–0.64) populations. Meta-regression revealed that Eastern Cooperative Oncology Group (ECOG) score 0 significantly influenced MRD status (β = −0.015, p < 0.05), whereas ECOG scores 1 and 2 lacked statistical significance. Subgroup analysis revealed that PFS was significantly different between standard (RR 0.47) and high (RR 0.81) cytogenetic risk groups.
Conclusion:
In NDMM patients, anti-CD38 mAb-based therapy significantly improved MRD status, and PFS compared with standard therapy alone, in both TE and TIE patients, suggesting a favorable benefit–risk profile.
Plain language summary
Why was this study conducted? Anti-CD38 monoclonal antibodies (mAbs) have improved the course of treatment for multiple myeloma (MM), a type of blood cancer. These medications may provide better results since they target particular MM cells. In patients recently diagnosed with multiple myeloma (NDMM), this study compared the safety and efficacy of these novel treatments with standard therapy. What did the researchers do? Data from 11 clinical trials with 5,270 NDMM patients were examined by the researchers. They examined two primary outcomes: minimal residual disease (MRD), which looks at the remaining cancer in the body after treatment, and progression-free survival (PFS), which measures how long patients live without the disease getting worse. Patients were separated into two categories: those who qualified for a stem cell transplant (TE) and those who did not (TIE). What did the researchers find? The results showed that anti-CD38 mAbs significantly improved patient outcomes. More patients achieved MRD negativity (lower cancer levels) and had longer PFS compared to those on standard therapy. For TE patients, anti-CD38 mAbs improved MRD by 52% and PFS by 57%. TIE patients saw even greater benefits, with a 249% increase in MRD negativity and a 45% improvement in PFS. What do these results mean? This study demonstrates that, regardless of a patient’s eligibility for a stem cell transplant, anti-CD38 monoclonal antibodies are useful in the treatment of recently diagnosed multiple myeloma. These results imply that this treatment may slow the course of the disease and lower cancer levels in a large number of patients, demonstrating a positive benefit–risk profile for potential future therapeutic strategies.
Keywords
Introduction
Multiple myeloma (MM) is the second most common hematological malignancy and is responsible for 20% of deaths related to blood cancers.1,2 MM is distinguished by the presence of monoclonal immunoglobulins and different clinical features and complications.3–5 Traditionally, treatment for newly diagnosed multiple myeloma (NDMM) involved a combination of chemotherapy, corticosteroids, and newer agents such as proteasome inhibitors (PI) and immunomodulatory drugs (IMIDs). National Comprehensive Cancer Network recommendations for transplant-eligible multiple myeloma (TEMM) patients advocate the use of combinations such as bortezomib, lenalidomide, and dexamethasone, along with alternatives such as carfilzomib or daratumumab-based regimens. For transplant-ineligible multiple myeloma (TIEMM) patients, initial therapies consist of comparable combinations, with supplementary alternatives including melphalan, prednisone, or cyclophosphamide in conjunction with bortezomib or daratumumab.6–10 While these approaches have improved patient outcomes, they also have significant limitations and associated toxicities. 11
Monoclonal antibodies (mAbs) against CD38 have surfaced in recent years as promising therapeutic options for NDMM. These antibodies target the CD38 protein, which is highly expressed in myeloma cells, and enhance the ability of the immune system to combat the disease by promoting cell death and inhibiting growth. 12 Daratumumab and isatuximab are the two FDA-approved anti-CD38 mAbs that, when used alone or in combination with other therapies, have demonstrated effectiveness in clinical trials.13,14
This meta-analysis seeks to answer the following primary question: How do anti-CD38 mAbs plus PI/IMIDs compare with other standard therapies in terms of efficacy, safety, and overall survival rates for patients with NDMM? The specific goals include comparing overall response rates (ORRs) between anti-CD38 mAbs-based therapy and other standard therapies, analyzing the incidence of side effects for every treatment approach, and assessing progression-free survival (PFS) and overall survival. The results of this study could significantly influence treatment guidelines and improve outcomes for NDMM patients, providing new hope for those facing this difficult disease. Moreover, the findings may identify areas that need further research and development.
Methods
The International Prospective Register of Systematic Reviews has registered the study protocol, and this systematic review and meta-analysis were carried out in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, PROSPERO (CRD42024588755). 15
Search strategy
Several databases including PubMed, the Cochrane Database of Systematic Reviews, and ClinicalTrials.gov, which cover publications up to July 18, 2024, were searched electronically. The MeSH terms and keywords that were used were “Multiple Myeloma,” “Daratumumab,” and “Isatuximab.” Relevant articles were also identified through in the bibliographies of all the included papers. The full search strategy is provided in Supplemental Table 1.
Study selection and eligibility criteria
After every reference found during our search was imported into EndNote v20, we eliminated duplicates. Independently, two authors (MHK and SK) reviewed each study’s abstract and title to weed out any studies that did not fit our inclusion requirements. The remaining articles were assessed for eligibility by reviewing their full texts. Discrepancies were discussed and settled with a senior author (MO). The following were eligible for inclusion: abstracts and randomized controlled trials (RCTs) comparing the effects of anti-CD38-based therapy with those of standard therapy in patients with NDMM).
Data extraction and outcomes
Two authors (SK and AT) extracted the data into Microsoft Excel, and in cases of discrepancies, a third author was consulted. The data included the trial name, first author, publication year, study setting, digital object identifier (DOI), number of patients in the study, and each treatment group. Patient characteristics included age and sex, while treatment characteristics included regimen, maintenance therapy, dose of anti-CD38 in cycle 1, and route of administration. The outcomes assessed were minimal residual disease (MRD) status, PFS, stringent complete response (sCR), complete response (CR), overall response, MRD negative status regardless of response, and very good partial response (VGPR), VGPR or better. The hematological adverse events recorded were neutropenia, thrombocytopenia, anemia, and lymphopenia. Nonhematological adverse events included diarrhea, pneumonia, upper respiratory tract infection, constipation, peripheral sensory neuropathy, fatigue, infusion-related reactions, pyrexia, peripheral edema, nausea, cough, asthenia, back pain, and second primary cancer. MRD negative status and PFS were identified as the primary outcomes.
Quality assessment
Using the Cochrane “risk of bias” tool (RoB 2.0) for RCTs, two independent authors (AT and SK) evaluated the risk of bias in the included studies. 16 They evaluated the randomization process for deviations from the intended interventions, outcome measurements, missing outcome data, and reporting bias, and the studies were classified as having low risk, some concerns, or high risk of bias. A third author (MO) helped resolve any disagreements that arose during the discussion of the bias assessment.
Statistical analysis
The number of events and total number of patients were extracted for dichotomous variables, whereas hazard ratios (HRs) were extracted for PFS. For dichotomous outcomes, pooled risk ratios (RRs), whereas for variables involving time such as PFS, pooled HRs were determined. The I2 statistic was utilized to assess heterogeneity among the included studies. 17 Subgroup analyses for primary outcomes were based on Transplant Eligible (TE) and Transplant in Eligible (TIE), and (disease characteristics (for PFS only)). Meta-regression was performed for MRD based on Eastern Cooperative Oncology Group (ECOG) criteria. For adverse events, subgroup analyses were conducted based on any grade and grades 3 or 4. The results are displayed in forest plots. Statistical analyses were carried out with the Review Manager (RevMan, Version 5.4; The Cochrane Collaboration, Copenhagen, Denmark). The Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) assessment was performed for the degree of certainty. The funnel plots made through R software were visualized for publication bias.
Results
Search results
The preliminary search yielded 3043 articles. After filtering out 296 duplicates, we evaluated the remaining 2747 articles to determine their eligibility. A total of 2633 publications were excluded after the titles and abstracts were reviewed. The remaining 114 articles were assessed for eligibility criteria. In the end, we included a total of 11 studies. Figure 1 provides an in-depth summary of the screening process.
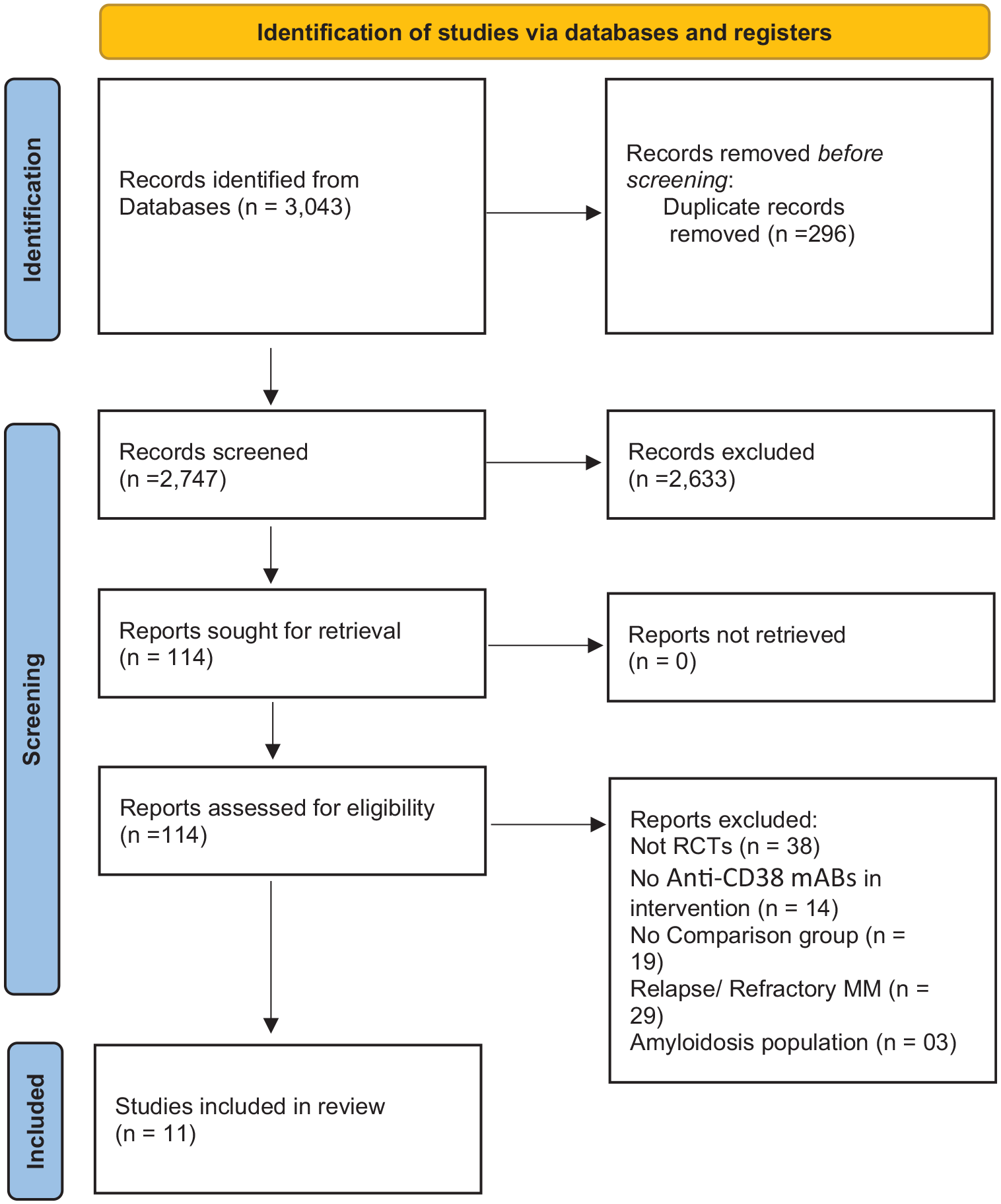
PRISMA flow chart of included studies.
Study characteristics
This meta-analysis included a total of 11 RCTs.18–28 The publication years spanned from 2017 to 2024. These studies included a total of 5272 patients, with 2712 receiving anti-CD38 mAb-based therapy and 2560 receiving standard therapy. Among the patients, 3058 (58%) were male (Tables 1 and 2).
Baseline characteristics of included studies.

ECOG, Eastern Cooperative Oncology Group; ISS, International Staging System; IV, Intravenous; MM, multiple myeloma; NA, Not Applicable or Not Available; NR, Not Reported; SC, Sub cutaneous; TEMM, transplant-eligible multiple myeloma; TIEMM, transplant-ineligible multiple myeloma.
Subgroup analysis of efficacy outcomes based on transplant eligibility and ineligibility.

CR, complete response; MRD, minimal residual disease; OR, odds ratio; ORR, overall response rate; RR, risk ratio; TEMM, transplant-eligible multiple myeloma; TIEMM, transplant-ineligible multiple myeloma; VGPR, very good partial response.
Risk of bias in included studies
The Cochrane Risk of Bias tool was used to evaluate the risk of bias in the included studies. Three of the included studies showed some concern for bias. This bias risk was caused by the domains of deviations from the intended intervention and outcome measurement. The remaining eight studies were assessed to be at a low risk of bias. The summary graph for the quality assessment is displayed in Supplemental Figure 1. The GRADE assessment shows moderate certainty for the majority of the outcomes and high certainty for PFS (Table 3).
GRADE assessment.

The higher i2 (82%) was due to the difference in the transplant eligibility of the included studies, the subgroup analysis based on the transplant eligibility reduces the I2 significantly.
High i2 (85%), subgroup analysis based on transplant eligibility reduces the i2 to 0% in the TIEMM and remains 86% in the TEMM subgroup.
High i2 of 85%, subgroup analysis based on the transplant eligibility reduces the I2 in TEMM to 0%.
High i2 of 58%.
Although the CI crosses 0, but the number of events is above 1000, the observed effect may be due to high heterogeneity.
High I2 of 79%, subgroup analysis reduces the i2 value in the TIEMM to 3%.
The 95% CI crosses the 0 and the number of events is also small.
GRADE PRO software generate the table in these shades.
The risk in the intervention group (and its 95% CI) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
CI, confidence interval; CR, complete response; HR, hazard ratio; mAbs, monoclonal antibodies; MRD, minimal residual disease; NA, not applicable; ORR, overall response rate; PD, Progressive diseases; PFS, progression-free survival; RCT, randomized controlled trial; RR, risk ratio; sCR, stringent complete response; TEMM, transplant-eligible multiple myeloma; TIEMM, transplant-ineligible multiple myeloma; URTI, Upper resiratory tract infection; VGPR, very good partial response.
Meta-analysis of primary efficacy-related outcomes
MRD negative status regardless of response
Nine studies involving 2963 patients (1484 anti-CD38 mAb vs 1479 standard therapy) reported this outcome. The results revealed that the pooled RR for achieving MRD negativity was 1.94 (95% CI: 1.59–2.37; p < 0.00001; I2 = 82%) favoring anti-CD38 mAb-based therapy.
Subgroup analysis
Subgroup analysis based on transplant eligibility revealed a significant difference between TE (RR 1.52, 95% CI: 1.37–1.68; p < 0.00001) and TIE (RR 3.49, 95% CI: 2.65–4.61; p < 0.00001) in terms of MRD negative status (Figure 2; Supplemental Table 2).

Forest plots of the PFS.
Regression analysis based on ECOG
The meta-regression analysis highlighted a significant association between ECOG 0 score and MRD, unlike higher ECOG scores. For ECOG-0, the intercept was 1.7023 (p < 0.0001), with a significant negative regression coefficient of −0.0030 (p = 0.0025), and no residual heterogeneity (I2 = 0%). For the ECOG-1 score, the intercept was 1.6404 (p < 0.0001), with a nonsignificant negative regression coefficient of −0.0029 (p = 0.0735), and mild residual heterogeneity (I2 = 17.34%). In contrast, for the ECOG-2 score, the intercept was 1.2428 (p < 0.0001), with a nonsignificant regression coefficient of 0.0002 (p = 0.9781), and considerable residual heterogeneity (I2 = 57.23%). These results demonstrate a robust association for ECOG-0 score, whereas higher ECOG scores lack significant associations with MRD (Supplemental Figure 2A–C).
Progression-free survival
Eight studies, involving a total of 4231 patients (2195 anti-CD38 mAb vs 2036 standard), showed a significantly high PFS for the anti-CD38 mAbs-based therapy compared with the standard therapy (HR 0.51, 95% CI: 0.45–0.58; p < 0.00001, I2 = 0%).
Subgroup analysis based on TE and TIE
Subgroup analysis based on transplant eligibility revealed no significant difference between the TE (HR 0.43, 95% CI: 0.34–0.54; p < 0.00001) and TIE groups (HR 0.55, 95%CI: 0.47–0.64; p < 0.00001; Figure 3).

Forest plot of the MRD negative status.
Subgroup analysis based on disease characteristics
Subgroup analysis of PFS was performed based on the disease characteristics. There was statistically significant difference between the standard (RR 0.47) and high (0.81) cytogenetic risk groups (test for subgroup difference p = 0.03; Supplemental Figure 3A).
Subgroup analysis of PFS was also performed based on ISS, ECOG criteria, type of Ig, and creatinine clearance, but none of these subgroups were significantly different (Supplemental Figure 3B–E).
Secondary efficacy-related outcomes
In the analysis of secondary efficacy-related outcomes, the ORR favored anti-CD38 mAbs with a RR of 1.09 (Supplemental Figure 4). For the sCR anti-CD38 mAb group was superior, with an RR of 1.69 (Supplemental Figure 5), whereas the two groups’ CRs had a similar RR of 1.14 (Supplemental Figure 6). The rates of VGPR were comparable between the two groups (Supplemental Figure 7). Furthermore, CR or better and VGPR or better were significantly greater for the anti-CD38 mAbs group (Supplemental Figures 8 and 9). The subgroup analysis of these outcomes based on TE and TIE is shown in Supplemental Table 2.
Adverse effects
Hematological
The outcome of neutropenia was reported by 4088 patients (2130 anti-CD38 mAbs vs 1958 standard), and the analysis revealed that the risk of neutropenia was markedly increased with anti-CD38 mAbs-based therapy (RR 1.20). Additionally, the risk of thrombocytopenia was substantially elevated by anti-CD38 mAbs-based therapy (RR 1.17).
Other hematological outcomes, such as lymphopenia and anemia, did not appear to differ significantly between the two groups. Additional details are provided in Supplemental Table 3.
Subgroup analysis of hematological adverse events
Subgroup analysis comparing any grade and grades 3, 4 was conducted, but there was no statistically significant variation in the hematological outcomes. Additional information regarding subgroup analysis can be found in Supplemental Table 4.
Nonhematological adverse events
The analysis revealed significant differences between the anti-CD38 mAbs-based therapy and standard therapy for outcomes such as diarrhea (RR 1.14), pneumonia (RR 1.84), upper respiratory tract infection (RR 1.43), cough (RR 1.75), nausea (RR 1.17), and back pain (RR 1.24).
Subgroup analysis of nonhematological adverse events
Subgroup analysis was also done based on the grade of disease, but there was no statistically significant variation in the hematological outcomes. A detailed analysis is presented in Supplemental Table 4.
Sensitivity analysis
The I2 value of MRD negative status was 82% which was resolved after the subgroup analysis was performed on the TE (45%) and TIE (0%) groups, revealing that the main difference between the studies was transplant eligibility. Similarly, the PFS had an I2 value of 64% which decreased to 0% after performing the subgroup analysis based on transplant eligibility (Figures 2 and 3).
Publication bias
The funnel plot of all the variables shows a symmetrical distribution with no visual asymmetry, indicating that there was no publication bias in any variable. The funnel plots of all the variables are given in the Supplemental File (Supplemental Figures 10–17).
Discussion
In this meta-analysis, 11 RCTs involving 5272 patients were included. Anti-CD38 mAb-based therapy significantly improved MRD (RR 1.94) and PFS (HR 0.51) in TEMM and TIEMM patients. The secondary outcomes included a superior ORR, sCR, and VGPR or better with anti-CD38 mAbs-based therapy, while the CR was comparable between the groups.
Anti-CD38 mAb therapy, using Daratumumab and Isatuximab, has revolutionized NDMM treatment.29,30 These mAbs impact myeloma cells in distinct ways. They can directly cause cell apoptosis, activate the complement system, modify the immune environment by reducing immunosuppressive cells, and inhibit CD38 exoenzymatic activity.30–32 Adding anti-CD38 mAbs to standard regimens on the frontline improves PFS, MRD status, ORR, and sCR without compromising patient safety. They may increase the risk of hematological toxicities, including neutropenia and thrombocytopenia, as well as nonhematological toxicities such as diarrhea, pneumonia, and respiratory infections.
Our meta-analysis focused on reporting MRD status and PFS as the primary outcomes. It was found that anti-CD38 mAb-based therapy significantly improved MRD status in NDMM patients compared to standard therapy alone, with a pooled RR of 1.94. This aligns with findings from Moreau et al. 18 who emphasized the importance of achieving MRD negativity. This is particularly significant, as achieving MRD negativity is a strong predictor of long-term outcomes and survival in MM.
TEMM patients had a pooled RR of 1.52 for MRD negative, favoring anti-CD38 mAb treatment. Even with autologous stem cell transplantation, anti-CD38 mAbs may increase the possibility of a deeper response, improving posttransplant outcomes.
A substantial benefit was found in the TIEMM patient research, with a pooled RR of 3.49. This unexpected finding suggests that anti-CD38 mAb therapy can significantly increase MRD status in a cohort with few therapeutic options and worse prognoses. Low heterogeneity (I2 = 0%) indicates a constant benefit across investigations, indicating reliability. San-Miguel et al. 33 reported that daratumumab increased MRD status for 6–12 months (D-Rd, 14.9% vs Rd, 4.3%; D-VMP, 15.7% vs VMP, 4.5%) and improved PFS from 6 to 12 months compared with normal controls in TIEMM patients.
Patients who were treated with the anti-CD38 mAb experienced a significant improvement in PFS, as shown by an HR of 0.51. Our study found that treatment with anti-CD38 mAbs markedly enhanced PFS in both TEMM patients (HR 0.43) and TIEMM patients (HR 0.55). These findings support Jakubowiak et al. 34 and Mateos et al., 35 who showed that anti-CD38 therapies improved PFR rates. Jakubowiak et al.’s trial reported a 0.59 HR for PFS with a 95% CI of 0.41–0.85. Daratumumab, reduced disease progression and mortality by 41% relative to those in the control group. These strong improvements imply that anti-CD38 antibodies should be the recommended therapy for MM regardless of transplantation eligibility, improving patient outcomes.
The secondary effectiveness-related outcomes of our study revealed that anti-CD38 mAbs-based therapy were better than standard therapy in terms of ORR, with an RR of 1.09. The results revealed that compared to patients treated with standard therapy, those given anti-CD38 mAbs-based therapy had a significantly higher ORR. Similar results were reported by Jakubowiak et al., 34 who reported that anti-CD38 mAb-based therapy improved overall response in TIEMM patients with a high risk (RR 1.24).
In the sCR analysis, the group treated with anti-CD38 mAbs had a higher RR of 1.69. The results showed that anti-CD38 mAbs had a considerably greater sCR rate than standard treatment. These results support the findings of Chari et al., 36 who demonstrated that patients who received anti-CD38 mAbs had a higher rate of sCR than those who received standard therapy. Another study revealed that 29% of patients receiving anti-CD38 mAb-based therapy achieved a sCR, compared to 20% of patients receiving standard therapy (p = 0.0010). 37
The anti-CD38 mAbs group presented significantly higher rates of CR or better (RR 1.41) and VGPR or better (RR 1.23), similar to the findings of the GRIFFIN trial, 19 in which increased VGPR or better increased from 28% in the control group to 52% in the anti-CD38 group. The increase can be linked to the specific ways in which they work. As stated by Bisht et al., 38 these antibodies may attack cells, increase immune responses, and change the tumor environment. The combined effects improve the outcomes.
Both the anti-CD38 mAb-based therapy and standard therapy groups presented identical PD risks. The anti-CD38 mAb group had a 35% lower incidence of PD (RR 0.65). Mateos et al. 21 reported that daratumumab reduced PD, particularly in terms of high-risk cytogenetic characteristics, whereas our study did not reveal any significant difference. Stable disease (SD) risk was considerably reduced in the anti-CD38 mAb group, with an RR of 0.33. The anti-CD38 mAb-based therapy reduced the SD risk by 67%. These findings suggest that NDMM may respond better to anti-CD38 mAbs. Validating these findings and understanding the processes requires further investigation.
Anti-CD38 mAbs such as daratumumab and isatuximab increase the likelihood of adverse hematological effects, including neutropenia, in MM patients.19,39 Our meta-analysis also revealed that anti-CD 38 mAb-based treatment increased neutropenia and thrombocytopenia with (RR 1.20) and (RR 1.17), respectively. In studies of isatuximab, thrombocytopenia was more common.19,40 These findings emphasize the significance of blood count monitoring during therapy. The anti-CD38 group also had an increased incidence of diarrhea (RR 1.14), pneumonia (RR 1.84), and URTIs (RR 1.43). A comparison of our data with those of previous studies revealed that the anti-CD38 group had a greater risk of pneumonia and diarrhea. These patients were also more likely to have URTIs. 41
Limitations
The inclusion of only RCTs and the exclusion of cohort studies, which could provide further insights and real-world data, restrict this meta-analysis. The study did not consider the effects of maintenance therapy with anti-CD38 therapy. By addressing these limitations, a more thorough understanding of the consequences of anti-CD38 mAbs in patients with NDMM can be achieved.
Conclusion
In conclusion, studies have shown that treatment regimens targeting CD38 greatly enhance positive outcomes, such as achieving MRD negativity and improving PFS, while also reducing the occurrence of certain adverse events in patients with NDMM. This is particularly beneficial for people with typical risk profiles and those in the early stages of the illness.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251314289 – Supplemental material for Efficacy and safety of anti-CD38 monoclonal antibodies-based therapy versus standard therapy in newly diagnosed multiple myeloma patients: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251314289 for Efficacy and safety of anti-CD38 monoclonal antibodies-based therapy versus standard therapy in newly diagnosed multiple myeloma patients: a systematic review and meta-analysis by Muhammad Osama, Muhammad Haris Khan, Safeena Khan, Amna Hussain, Ammara Tahir, Mehran Ullah, Abdullah Afridi, Ubaid Ullah and Wajeeh Ur Rehman in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
We acknowledge the contributions of Muhammad Ibrahim, and Wajeeh Ur Rehman regarding continued guidance throughout this meta-analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

