Abstract
Dasatinib is a second-generation tyrosine kinase inhibitor employed for chronic myeloid leukemia (CML) treatment that achieves high rates of prolonged and complete molecular responses (MR). Among the adverse effects reported, it has been associated with hemorrhagic complications, mainly due to its inhibiting effects on platelet functions. In addition, immune alterations induced by dasatinib may elevate the risk of bleeding and cytomegalovirus (CMV) infection, particularly in the gastrointestinal tract, thus contributing to the development of hemorrhagic colitis. In this case report, we highlight three cases of CML receiving treatment with dasatinib where CMV hemorrhagic colitis occurred. All of them exhibited iron deficiency anemia as a premature clinical manifestation in the absence of intestinal symptoms, unlike cases previously reported in the literature. CMV infection was confirmed with stool samples or tissue quantitative polymerase chain reaction and/or immunohistochemistry staining in colon biopsies. All three cases could be managed with valganciclovir and iron supplements in an outpatient setting. Management strategies of dasatinib during and after CMV infection varied, as they are not yet established and need to be individualized based on the gravity of symptoms and disease state. Iron deficiency anemia during dasatinib treatment should raise suspicion for the potential presence of CMV colitis, prompting endoscopic studies to rule out this complication, even if intestinal symptoms are not present.
Introduction
The discovery of tyrosine kinase inhibitor (TKI) has led to deeper molecular responses (MR) and a significant improvement in overall survival for chronic myeloid leukemia (CML) patients. 1 Compared to imatinib, therapy with second-generation TKI induces a faster deep MR. 2 However, these TKIs have been associated with new adverse effects. It is of paramount importance to decide the best choice of treatment depending on patient characteristics, aim of therapy, toxicity, and CML risk profile. 2
Hemorrhagic colitis is described as a potential adverse effect related to the bleeding diathesis induced by dasatinib through different mechanisms of action. It also seems that dasatinib could increase the risk of cytomegalovirus (CMV) infection affecting the colon, which may lead to gastrointestinal bleeding, being bloody diarrhea as the characteristic warning sign.3–5
Here, we describe three cases of chronic phase CML under treatment with dasatinib in which mild-to-moderate iron deficiency anemia was the first clinical sign of CMV-induced hemorrhagic colitis in the absence of intestinal symptoms.
Case reports
Case 1
A 55-year-old female was diagnosed with chronic phase CML in August 2020 with a high-risk Sokal and intermediate risk (European Treatment Outcome Study (EUTOS) score and was started on dasatinib 100 mg once a day achieving major MR after 16 months of treatment. At the 18th month of follow-up, she developed asthenia, and iron deficiency anemia was detected with a hemoglobin level of 8.9 g/dL. The patient was therefore started on intravenous iron supplementation and a colonoscopy was requested. In the following weeks, the patient presented two episodes of watery diarrhea with hematochezia that was self-limiting after some days. The colonoscopy revealed a friable mucosa with spontaneous bleeding and rectal aphthous erosions, all suggestive of pancolitis of inflammatory/infectious origin (Figure 1). Biopsy samples were taken, and histopathological examination revealed positive CMV immunohistochemistry (IHC) staining (Figure 2). Blood CMV quantitative polymerase chain reaction (qPCR) was negative.

Pancolitis of inflammatory aspect. Tarnished, patchy colon mucosa with aphthous erosions, pseudopolyps, and spontaneous bleeding.
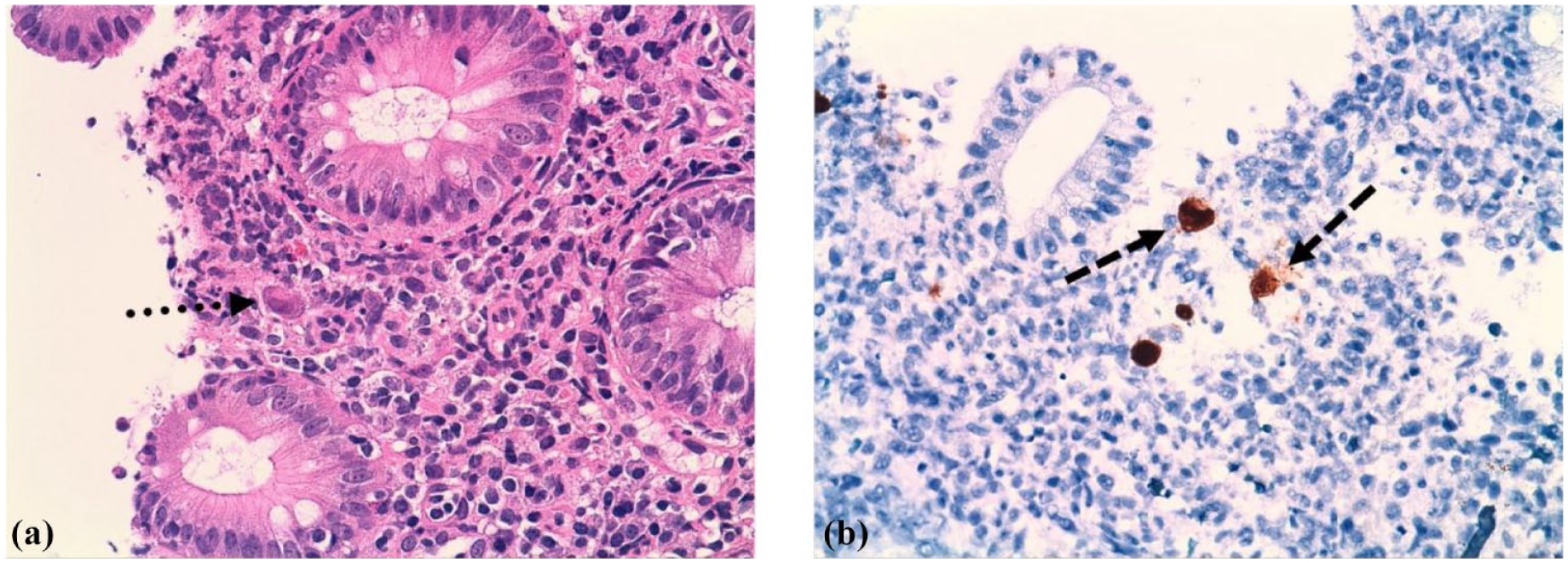
(a) Colorectal mucosa with increased lymphoplasmacytic inflammatory infiltrate in the lamina propria and cryptitis (hematoxylin and eosin staining, 40×). The presence of cytomegalovirus-infected stromal cells (dotted arrow). (b) Positive CMV-specific IHC staining (dashed arrows) (40×).
Dasatinib was suspended and the patient started valganciclovir 900 mg twice a day, which was maintained for 8 weeks. Iron deficiency anemia resolved, and a follow-up colonoscopy showed normal findings with negative results of tissue qPCR. After symptoms subsided and CMV treatment was completed, the patient was switched to nilotinib 300 mg twice a day because of previous toxicities related to dasatinib, maintaining MR4.0 until the present day.
Case 2
A 74-year-old male diagnosed with chronic phase CML in January 2014 with a high-risk Sokal score and low-risk EUTOS score was initially treated with dasatinib 100 mg once a day and achieved MR4.0 18 months later.
Subsequent blood tests revealed iron deficiency anemia with a progressive drop in hemoglobin levels from 16.3 to 12.2 g/dL from 2018 to 2019. Endoscopic studies showed Helicobacter pylori in gastric biopsies with diverticulosis and nonspecific inflammatory changes in the colonoscopy, so the patient was started on antibiotics and oral iron supplements. Since then, hemoglobin levels fluctuated between 12 and 13.5 g/dL with low ferritin. The patient tested positive for fecal occult blood test and once again underwent endoscopic studies, which showed peridiverticular colitis. IHC staining for CMV in colon biopsies was negative but a positive result for CMV was detected by qPCR in a stool sample (4617 copies/mL). Dasatinib was then suspended, and the patient was started on valganciclovir 900 mg twice a day. After 6 weeks of treatment, endoscopic studies drastically improved, and colon biopsy qPCR was negative. Currently, the patient remains on MR5.0 15 months after stopping dasatinib.
Case 3
A 70-year-old male diagnosed with chronic phase CML in 2002 with a low-risk Sokal score, intolerant to imatinib and nilotinib, started treatment with dasatinib 100 mg once a day in 2017 achieving MR4.5. In January 2022, the patient presented a progressive decrease in hemoglobin levels with an iron deficiency pattern of several months of evolution, requiring oral iron therapy with moderate effectiveness.
Colonoscopy findings were normal at first, but given the persistence of iron deficiency anemia, together with several positive fecal occult blood tests, studies were repeated in January 2023. The endoscopy revealed then a patchy, inflamed colonic mucosa, and a magnetic enteroresonance showed no abnormalities. While suspecting either inflammatory bowel disease or celiac disease, a third colonoscopy revealed a patchy mucosa with aphthous ulcers in the rectum and right colon. Biopsies were not taken because of poor preparation but a qPCR of ulcer exudate tested positive for CMV. Blood qPCR was also positive (498 copies/mL). In April 2023, dasatinib was interrupted, and the patient was started on valganciclovir 900 mg twice a day for 4 weeks, after which the anemia improved. CMV viral load in blood was not detectable and endoscopic findings were resolved. Since the patient had been under CML treatment for so long and had achieved an MR4.5, dasatinib was not resumed. The molecular monitoring shows stability with MR5.0 10 months after stopping TKI therapy.
Discussion
Dasatinib is a second-generation oral TKI designed to treat CML and Philadelphia-chromosome-positive acute lymphoblastic leukemia through potent inhibition of BCR::ABL1 rearrangement. 6 It is also a multi-kinase inhibitor with activity on c-KIT, SCR family kinases, PDGFR receptors, EPHA2, and ephrin receptor kinases, among others. 3 This broad spectrum of inhibitory activity in different kinases may be related to its most frequent side effects, such as cytopenias, pleural effusion, rash, pulmonary hypertension, immunosuppression, and bleeding. 6
Hemorrhagic complications have been described with the use of dasatinib. The mechanism of action may involve direct platelet dysfunction by impairment of arachidonic acid and epinephrine-induced aggregation. 7 Second, bleeding could be indirectly associated with immune alterations. Large granular lymphocytes (either cytotoxic CD8+ T or NK cells) have been found in pleural liquid and colon biopsies of patients receiving dasatinib and are thought to be cytotoxic to endothelial cells, thus contributing to mucosal bleeding. 8 Finally, dasatinib-mediated lymphocyte immunosuppression can induce reactivation of unexpected pathogens in myeloid malignancies, 9 such as CMV, a double-stranded DNA virus with a high prevalence worldwide. While CMV does not result in serious disease in immunocompetent hosts, reactivations are frequent in immunocompromised patients and may lead to systemic manifestations, increasing mortality. 10 Because dasatinib is eliminated in the feces, a local downregulation of the immune response may occur, 9 contributing to a higher risk of CMV reactivation in the gastrointestinal area and the development of hemorrhagic colitis.
Up to now, 10 confirmed cases of CMV-induced hemorrhagic colitis in patients with CML undergoing treatment with dasatinib have been reported.3–5,11–13 All of them developed hematochezia and/or diarrhea which were the first symptoms of suspicion for hemorrhagic colitis. By contrast, we present here three cases in which despite the absence of clinical digestive manifestations, the presence of iron deficiency anemia prompted the need to perform endoscopic studies. In our patients, colonoscopy findings included an edematous, friable, and patchy mucosa, diffuse erythema, aphthous erosions, spontaneous bleeding, and loss of vascularity all along the colonic frame, similar to those found in 70%–80% of patients with CMV colitis. 14
IHC staining in colon biopsies is the gold-standard method for CMV colitis diagnosis. 14 qPCR can be performed in blood and intestinal-derived samples (stool samples, exudates, or colon biopsies) and might aid in the diagnostic process. 14 IHC-confirmed CMV presence in all 10 cases reported up to now. Blood qPCR was performed in 3 out of 10 patients, resulting in positive in two, while intestinal or stool qPCR was not performed. Conversely, out of our two cases where IHC was performed one was positive and showed the highly specific “owl eye” CMV inclusions in colon biopsies. qPCR, either in intestinal ulcer exudate or stool, was performed in two of our cases, rendering a positive result in both. Finally, two patients had a blood qPCR test, resulting in positive in only one of them. It has been reported that blood qPCR is only positive in 50% of patients with biopsy-proven CMV colitis. 15 Despite viremia levels detected in blood or bowel show limited concordance,16,17 our experience suggests that qPCR performed in intestine-derived samples may be of help to detect colitis due to CMV reactivation.
Out of the 10 cases reported, six were manageable on an outpatient basis—two were treated with oral valganciclovir, and one did not require any treatment; information about the remaining three cases is lacking. The other four cases needed to be admitted to the hospital and required treatment with intravenous ganciclovir.3–5,11–13 By contrast, none of our patients required hospitalization and could be treated in an outpatient setting with oral valganciclovir plus iron supplements. Similar to most cases in the literature, all three cases presented here showed improvement in CMV infection symptoms after 4–8 weeks of treatment. Antiviral therapy was maintained until a new colonoscopy showed normalization of the colonic mucosa.
Although the treatment of CMV infection is best defined in the context of bone marrow transplantation, 18 the incidence, mortality, and morbidity rates are also high in non-transplant hematologic malignancies. 19 This suggests the need for establishing recommendations to guide an early treatment for CMV disease in all hematologic patients to prevent complications.
Considering bleeding complications while receiving dasatinib, in one study with 138 patients in all phases of CML, 30/32 (81%) bleeding episodes were in the gastrointestinal tract, particularly in patients with advanced phase CML and low platelet counts. Only 8/66 patients with chronic phase CML had gastrointestinal bleeding episodes. 20
Dasatinib was interrupted in 8 out of the 10 confirmed cases in the literature. Of these, two reintroduced dasatinib after symptoms subsided, while one changed to nilotinib, without subsequent CMV infection relapses. By contrast, the remaining two patients maintained dasatinib, both also showing no recurrence of colitis.
In our three cases, dasatinib was suspended during the hemorrhagic episode. After resolution, one patient switched to nilotinib because of previous toxicities with dasatinib, the other two did not restart dasatinib since they had received it for 8 and 5 years, respectively, and had reached a deep and durable MR, which they maintained until the present day. None of them have had subsequent CMV reactivations.
There is no consensus regarding the management of dasatinib adverse events and the need for anti-CMV therapy. Dose adjustment or temporary suspension of dasatinib until resolution is a safe option, especially in clinically stable patients who have reached a deep MR.21,22 In most cases, valganciclovir/ganciclovir showed improvement in symptoms, independent of dasatinib management.3,4,11 In other reports, patients only improved after dasatinib was stopped without the need for anti-CMV treatment. 13
The management of dasatinib and the need for anti-CMV therapy is therefore controversial given the lack of prospective studies and should be individualized.
Conclusion
CMV hemorrhagic colitis is a potential complication associated with dasatinib treatment and may show a variable clinical course, ranging from indolent to severe forms. The occurrence of iron deficiency anemia in patients treated with dasatinib may be the first sign of suspicion for CMV hemorrhagic colitis and should indicate endoscopic studies for confirmation. The maintenance or suspension of dasatinib during CMV hemorrhagic colitis is under debate as well as the need for anti-CMV therapy. This decision must be individualized, balancing potential risks and benefits according to the clinical status and disease state of the patient.
