Abstract
Background:
The myeloproliferative neoplasm (MPN) is a heterogeneous group of clonal hyperplasia hematopoietic stem cell disorders, predominantly affecting middle-aged and elderly individuals, with a slow disease progression. With advancements in disease-related research, the survival rates of MPN patients have significantly improved. This research primarily focuses on cardiovascular disease mortality (CVM) and prognostic factors in MPN patients, aiming to provide clinicians with more comprehensive references.
Methods:
A total of 24,277 patients were included in the Surveillance, Epidemiology, and End Results (SEER) database. Cumulative mortality was assessed using a competing risk model, univariate and multivariate regression analysis of cardiovascular disease (CVD) mortality risk factors, and a comparison of standardized mortality ratio (SMR) and general population CVM.
Results:
Among the 24,277 patients included in this study, a total of 8841 deaths occurred during the follow-up period, with 2429 attributed to CVD. Notably, the risk of CVM was found to be significantly higher in patients with MPNs compared to the general population. Furthermore, this risk increased over time. CVD emerged as the predominant cause of death among individuals aged over 80 years and younger patients exhibited a significantly elevated SMR. Additionally, age, race, marital status, and insurance status were identified as independent prognostic factors for CVM.
Conclusion:
The incidence of cardiovascular events in patients with MPNs is significantly higher compared to the general population. Early screening and assessment of cardiac health should be implemented in MPN patients to prevent the occurrence of cardiovascular events and enhance their prognosis.
Plain language summary
Long-term risks of cardiovascular mortality in patients with MPNs
MPN is a group of hematopoietic stem cell disorders that primarily affects middle-aged and elderly individuals, with slow disease progression. This study focuses on cardiovascular disease mortality (CVM) and prognostic factors in MPN patients to provide clinicians with comprehensive references. A total of 24,277 patients from the SEER database were included in this analysis. Cumulative mortality was assessed using a competing risk model, univariate and multivariate regression analysis identified CVD mortality risk factors, and standardized mortality ratio (SMR) was compared to the general population’s CVM rate. Among these patients, 8,841 deaths occurred during follow-up, with 2,429 attributed to CVD. MPN patients had a significantly higher risk of CVM compared to the general population which increased over time. CVD emerged as the leading cause of death among individuals aged over 80 years while younger patients exhibited an elevated SMR. Age, race, marital status, and insurance status were identified as independent prognostic factors for CVM. The incidence of cardiovascular events is significantly higher in MPN patients compared to the general population; therefore early screening and assessment of cardiac health should be implemented to prevent such events and improve prognosis.
Keywords
Introduction
The Philadelphia chromosome-negative myeloproliferative neoplasms (MPNs) represent a malignant disorder arising from clonal hematopoietic stem cells and characterized by excessive proliferation of one or more lines of bone marrow, classified as primary myelofibrosis (PMF), polycythemia vera (PV), and essential thrombocythemia (ET). PMF clinically shows splenomegaly (often with giant spleen) and extramedullary hematopoiesis in multiple organs, which may change to bone marrow failure with the progression of fibrosis. 1 PV showed erythematous and purplish skin mucosa, splenomegaly, and vascular and neurological symptoms. 2 While ET favors spontaneous bleeding and potential thrombotic events, as well as splenomegaly, is observed in approximately half of the affected individuals. 3 Interconversion between ET, PV, and secondary myelofibrosis (SMF) can occur, with the possibility of progression to acute myelogenous leukemia (AML). 4 Notably, the risk of mortality significantly escalates following conversion to SMF/AML. 5
The prevalence of MPN is commonly observed among individuals aged 50–70, primarily affecting the middle-aged and elderly population, with a notable trend toward extended patient survival. 6 The elucidation of the mechanism underlying MPN has facilitated the development of novel therapeutic agents, thereby enhancing the overall prognosis for patients with MPN. 4 The reduction in primary tumor progression results in an extended life expectancy and an elevation in various nonprimary tumor-related mortality, with a particular emphasis on CVDs. The risk of cardiovascular disease mortality (CVM) in patients with tumors has been consistently demonstrated to be significantly elevated compared to the general population, as evidenced by numerous studies.7–9 However, to date, there is a dearth of literature addressing this topic in the context of MPN. Our study will facilitate timely implementation of preventive treatment and follow-up planning for MPN patients.
Methods
Data source and patient selection
The information about MPN patients was extracted from the Surveillance, Epidemiology, and End Results (SEER; Incidence—SEER Research Plus Data, 18 Registries, November 2020 Sub (2000–2018)). The data were publicly available and exempt from ethical approval for analysis.
We collected patients with ICD-O-3 histological codes 9950/3, 9961/3, and 9962/3 diagnosed with PV, PMF, and ET according to the third edition of the International Classification of Diseases for Oncology. The study adhered to the STROBE checklist for observational research. The exclusion criteria were as follows: (1) Not first primary malignant tumor; (2) No positive histology; (3) <1 year old; (4) Unknown cause of death; (5) Survival time was unknown or <1 month; (6) Unknown race; (7) Unknown marital status; (8) Unknown insurance status (Figure 1).

Selection of eligible patients.
Study variables and classification of cause of death
Study Variables: race (black, white, other (American Indian/AK Native, Asian/Pacific Islander)), sex (female, male), year of diagnosis (2000–2004, 2005–2009, 2010–2014, 2015–2018), age (<40, 40–59, 60–79, ⩾80), marital status (married, unmarried (divorced, separated, single (never married), unmarried or domestic partner and widowed)), insurance status (Yes, No), and survival time.
Classification of cause of death: death from MPN, death from cardiovascular, death from other cancers, and death from other noncancers. The primary endpoint of this study was CVM. Furthermore, in accordance with ICD-10, certain fatalities related to cardiovascular conditions were categorized as a composite variable encompassing diseases of the heart, cerebrovascular diseases, hypertension without heart disease, other diseases of arteries, arterioles, capillaries, aortic aneurysm and dissection, and atherosclerosis. Additionally, the subsequent 10 categories are classified as the predominant causes of mortality stemming from noncancerous origins: chronic obstructive pulmonary disease and allied conditions, accidents and adverse effects, Alzheimer’s (ICD-9 and 10 only), pneumonia and influenza, nephritis, nephrotic syndrome and nephrosis, diabetes mellitus, septicemia, other infectious, and parasitic diseases including HIV, symptoms, signs and ill-defined conditions, chronic liver disease, and cirrhosis.
Statistical analysis
We employed the competing risk model to evaluate the cumulative incidence of cause-specific mortality across patient variables for MPN, cardiovascular disease (CVD), other malignancies, and nonmalignant conditions. The standardized mortality ratio (SMR) was defined as the ratio of the number of deaths observed in our study to the number of deaths expected in the general population matched by age, sex, race, and year of diagnosis. We used precise methods to calculate a 95% confidence interval (95%CI) for the SMR. The absolute excess risk (AER) was calculated by subtracting the expected number of deaths from the number of deaths observed in the study by dividing the number of person-years at risk and then dividing the result by 10,000. The results were presented as the incidence rate per 10,000 person-years. Univariate and multivariate survival analyses were performed to identify independent predictors of CVD, and all analyses were performed using R software (version 4.2.2), where p-values <0.05 were considered statistically significant.
Results
Patient characteristics
In the study, a total of 24,277 patients were diagnosed with MPN from 2000 to 2018, including 10,409 (42.9%) for PV, 3,229 (13.3%) for PMF, and 10,639 (43.8%) for ET. The prevalence of this condition was higher among Caucasian males in PV and PMF compared to females in ET. According to age distribution at diagnosis: <40 years old, accounting for 8.0%; 40–59 years old, accounting for 29.0%; 60–79 years old, accounting for 47.0%; >80 years old, accounting for 16.0%. Among these patients, 8841 experienced death during the follow-up period after diagnosis, of which 2664 (30.1%) died from MPN, 2429 (27.5%) died from CVD, 777 (8.8%) died from other cancers, and 2971 (33.6%) died from other noncancer diseases. The main type of CVD was heart disease (74.2%), followed by cerebrovascular diseases (17.7%), hypertension without heart disease (4.1%), atherosclerosis (0.7%), aortic aneurysm, and dissection (0.9%), and other diseases of arteries, arterioles, capillaries (2.1%). Among other noncancer diseases besides CVD, chronic obstructive pulmonary disease, and allied conditions were the most common causes of death, followed by Accidents and Adverse Effects and Alzheimer’s (ICD-9 and 10 only). The proportion of cancer-related deaths gradually decreased with survival time in patients with ET and PV, while the proportion of noncancer-related deaths gradually increased, which contradicted the trend observed in ET patients (Table 1).
Baseline clinical characteristics of MPN patients.

COD, cause of death; ET, essential thrombocythemia; MPN, myeloproliferative neoplasm; PMF, primary myelofibrosis; PV, polycythemia vera.
American Indian/AK Native, Asian/Pacific Islander.
Divorced, separated, single (never married), unmarried or domestic partner, and widowed.
Cumulative mortality of CVD
When we analyzed causes of death in MPN patients, we found that at 200 months of follow-up, the cumulative mortality of CVD, other noncancer, MPN, and other cancers was 17.9%, 22.1%, 18.8%, and 6.1%, respectively (Figure 2(a)), with the highest incidence of other noncancer related deaths. However, over 50% of PMF patients succumbed to the primary disease within a follow-up period of 200 months (Figure 2(b)–(d)). The ease of transformation from PMF to AML may be attributed to the fact that compared to ET and PV, PMF exhibits a higher susceptibility. According to several studies, the transformation of PMF into AML is associated with an unfavorable prognosis.

Cumulative mortality for all causes of death. (a) for MPN, (b) for ET, (c) for PV and (d) for PMF.
We then categorized patients based on age, sex, race, marital status, and insurance status. Our analysis revealed that the risk associated with all these factors increased over time. Notably, male, other race, married, insured, and young patients exhibited a relatively lower risk of CVM (Figure 3). Further analysis of MPN patients stratified by age revealed that in individuals aged over 80 years, the risk of CVD-related mortality surpassed that attributed to other causes, emerging as the primary cause of death (Figure 4). Further analysis of patients with ET, PV, and PMF showed that CVD was also the main cause of death in PV patients over 80 years old, and CVD also played an important role in the cause of death in ET and PMF patients (Figures 5–7).
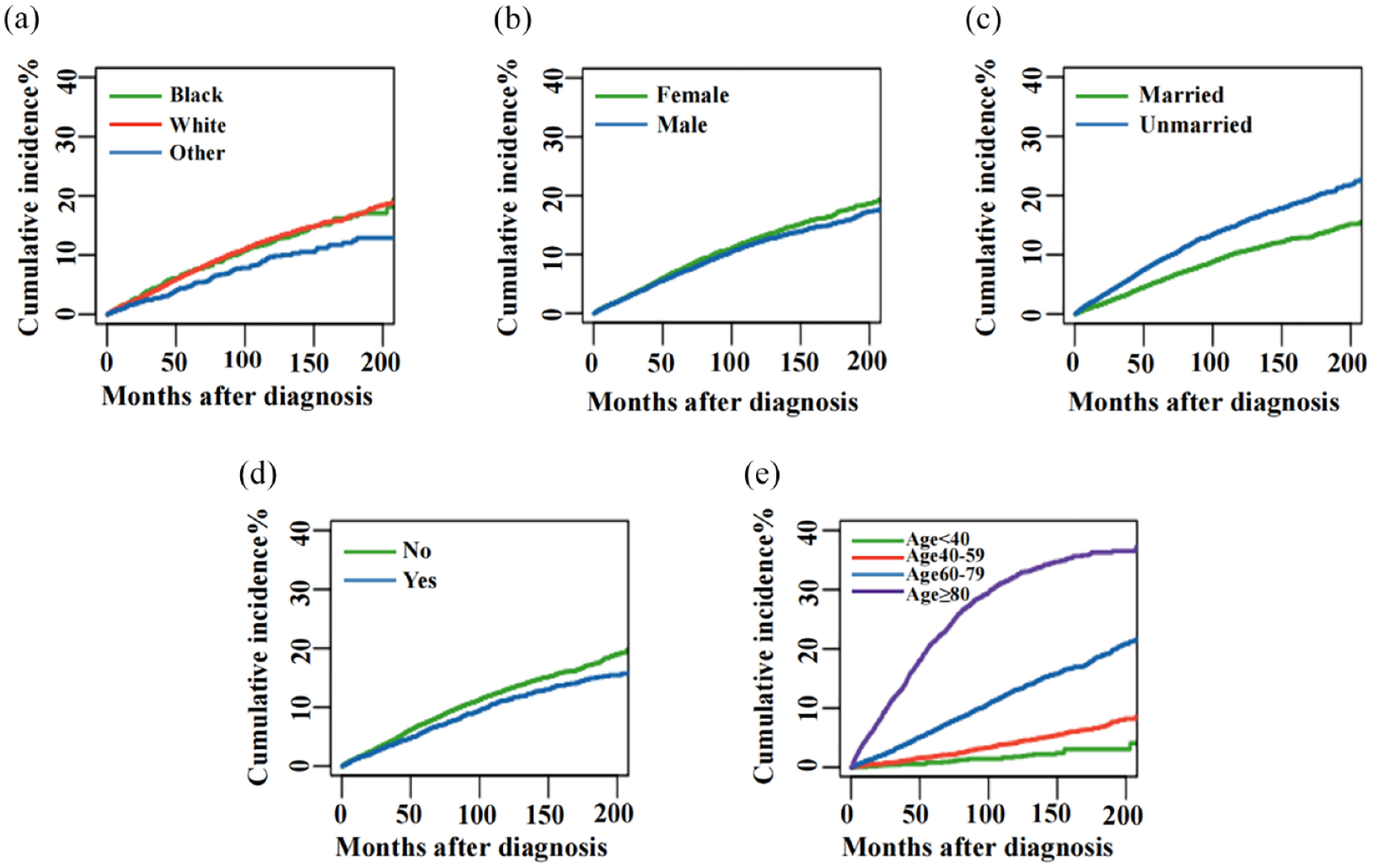
Cumulative mortality for CVM in MPN patients stratified by race (a), sex (b), marital status (c), insurance status (d), and age (e).

Cumulative mortality for all causes of death in MPN patients stratified by age at diagnosis: (a) age < 40; (b) age between 40 and 59; (c) age between 60 and 79; (d) age ⩾ 80.
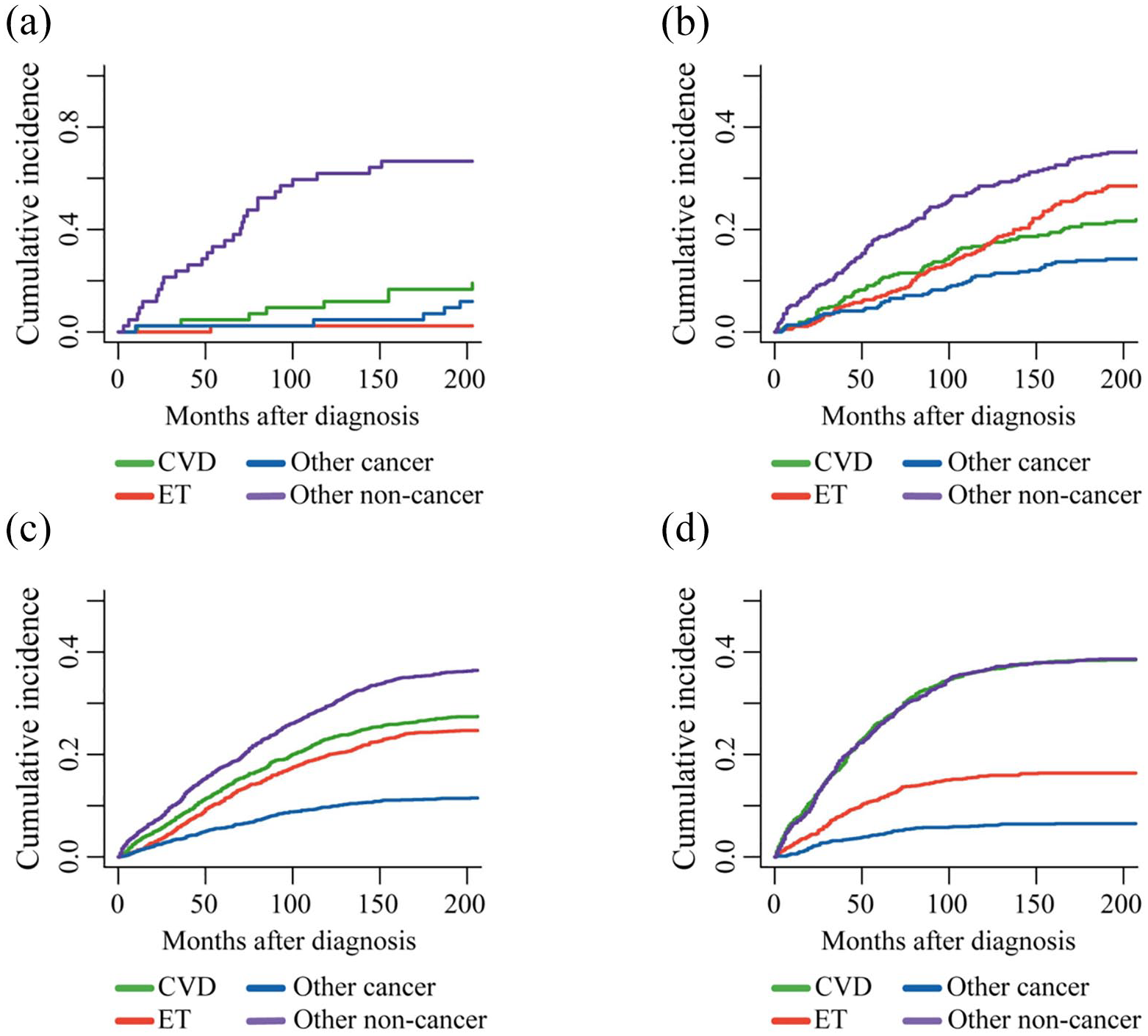
Cumulative mortality for all causes of death in ET patients stratified by age at diagnosis: (a) age < 40; (b) age between 40 and 59; (c) age between 60 and 79; and (d) age ⩾ 80.

Cumulative mortality for all causes of death in PV patients stratified by age at diagnosis: (a) age < 40; (b) age between 40 and 59; (c) age between 60 and 79; (d) age ⩾ 80.

Cumulative mortality for all causes of death in PMF patients stratified by age at diagnosis: (a) age < 40; (b) age between 40 and 59; (c) age between 60 and 79; (d) age ⩾ 80.
CVM and all-cause mortality compared to the general population
When comparing the risk of CVM in MPN patients with the general U.S. population, the SMR for CVM was 3.25 (95%CI, 3.14–3.35) and the AER was 1341.44. The subgroup analysis, stratified by clinical features, revealed significant increases in SMRs compared to the general U.S. population across all subgroups, including sex, age at diagnosis, race, marital status, insurance status, and year of diagnosis. Furthermore, our findings indicate a significantly higher SMR of CVM in patients diagnosed at a younger age, with a gradual decline in SMR as patients advance in age. For example, the SMR for CVM in patients younger than 40 years old diagnosed was 80.55 (95%CI, 50.11–133.62), compared to 2.42 (95%CI, 2.32–2.53) for CVM over 80 years old. The lowest SMR for CVM in MPN patients was 2.76 (95%CI, 2.62–2.90) at 1–5 years after diagnosis, and this SMR decreased during follow-up. Aneurysm and dissection (SMR, 3.94 (95%CI, 2.50–5.91)) had the highest SMR for the type of CVD death. This was followed by other diseases of arteries, arterioles, capillaries (SMR, 3.80 (95%CI, 3.04–4.70)), and cerebrovascular diseases (SMR, 3.45 (95%CI, 3.19–3.72); Table 2). Other major causes of noncancer-related death also saw increases in SMR, including chronic liver disease and cirrhosis (SMR, 6.97 (95%CI, 5.74–8.37)), diabetes mellitus (SMR, 4.64 (95%CI, 4.08–5.25)), septicemia (SMR, 4.50 (95%CI, 3.93–5.13)), and other infectious and parasitic diseases including HIV (SMR, 4.45 (95%CI, 3.66–5.35; Table 3)).
SMR of CVM among MPN according to baseline characteristics.

AER, absolute excess risk; CVD, cardiovascular disease; CVM, cardiovascular disease mortality; MPN, myeloproliferative neoplasm; SMR, standardized mortality ratio.
American Indian/AK Native, Asian/Pacific Islander.
Divorced, separated, single (never married), unmarried or domestic partner, and widowed.
SMR of other noncancer disease morbidity among MPN.

AER, absolute excess risk; MPN, myeloproliferative neoplasm; SMR, standardized mortality ratio.
Predictors of CVM
Cox regression analysis was conducted on MPN cases to evaluate the prognostic impact of clinical characteristics on cardiac-specific mortality (CSM), and the results are shown in Table 4. The results of the univariate analysis showed that race, age at diagnosis, marital status, and insurance status affected patients’ CSM. White race (HR: 1.503, 95%CI: 1.247–1.786) and black race (HR: 1.492, 95%CI: 1.247–1.786) had worse CSM than those of other races, and unmarried, uninsured patients also had worse CSM. Additionally, we observed a significant positive correlation between HR and age, as well as a significant increase in CSM. For example, HR (95%CI) in patients aged 40–59, 60–79, and >80 years were 2.497 (1.827–3.412), 9.988 (7.401–13.479), and 42.286 (31.274–57.174), respectively, compared with patients aged <40 years. The association between gender and CSM was observed in both ET and PV patients, wherein female ET patients exhibited a higher risk of CSM, while the opposite trend was observed in PMF. Multivariate Cox regression analysis showed that old age, unmarried, uninsured, black race, and white race were independent adverse prognostic factors for CSM in MPN patients.
Univariate and multivariate analysis for associations between patient characteristics and CVD in MPN patients.

AER, absolute excess risk; MPN, myeloproliferative neoplasm; SMR, standardized mortality ratio.
American Indian/AK Native, Asian/Pacific Islander.
Divorced, separated, single (never married), unmarried or domestic partner, and widowed.
Discussion
The analysis of the causes of death among MPN patients will enhance our understanding of the disease’s characteristics and bolster disease control and management. To our knowledge, this contemporary large population study is the first to examine post-MPN diagnosis mortality causes, particularly CVM risk factors, using data from the SEER database.
Patients with MPN often coexist with CVD, which is often associated with inflammation, pro-thrombotic and pro-fibrotic factors, and abnormal function of mutant leukocyte and platelet circulating clones in affected individuals. 10 In addition, other studies have found that gene mutations, such as the JAK2V617F and thrombopoietin receptor (MPL) mutation, promote endothelial-to-mesenchymal transformation under stress and endothelial dysfunction leads to CVD. 11 Because CVD is associated with a poor prognosis, we wanted to further explore the impact of CVD-related death on MPN patients.
By conducting an analysis of competing risks, we have determined that the primary disease does not emerge as the predominant cause of mortality among MPN patients. Instead, noncancer-related deaths exhibit the highest incidence rate, with CVD ranking third. However, upon examination of these three distinct diseases, discernible differences emerge. Patients with ET and PV exhibit a significantly heightened risk of arteriovenous thrombosis, as well as an increased susceptibility to CVD, attributable to the substantial elevation in blood cell count and the occurrence of augmented blood viscosity. The risk of CVD is significantly lower in patients with myelofibrosis (MF) compared to those with ET and PV, primarily due to the predisposition toward bone marrow failure. Additionally, there is a significantly higher likelihood of MF progressing into AML when compared to ET and PV. The primary disease is the predominant underlying factor leading to mortality in patients with MF. In the treatment, ET and PV patients often choose hydroxyurea and other cell-lowering drugs, 12 and use aspirin, clopidogrel, and other anti-plate drugs to prevent the formation of thrombosis.13,14 The JAK inhibitor Ruxolitinib was approved by the Food and Drug Administration (FDA) in 2011 and 2014 for the treatment of MF and PV,15–17 respectively, and can significantly improve outcomes in MPN patients. Novel JAK inhibitors, such as Jaktinib, are also being studied in patients who cannot tolerate Ruxolitinib or who are refractory to relapse.18,19 IFNα can also inhibit disease progression by inhibiting blood cell colony formation, inducing cytogenetic remission, and reducing the JAK2-V617F allele mutation load. Studies have shown that Ruxolitinib is a potent cardioprotective drug. 20 Multivariate competing risk analysis showed that the HR value of CVD decreased over time, with a 31.0% decrease in CVM among patients diagnosed between 2015 and 2018 compared to those diagnosed between 2000 and 2004 (HR 0.690, 95%CI 0.587–0.812).
As previously mentioned, MPN is frequently observed in individuals aged 50–70 years. Given the heightened susceptibility of this population to arteriosclerosis, dyslipidemia, diabetes, and other associated risk factors, the likelihood of CVD is significantly augmented compared to younger cohorts.21,22 Other studies have shown that with the increase of age and the occurrence of aging, the enhancement of oxidative susceptibility promotes the activation of key inflammatory transcription factors, and the normal gene regulation of aging cells leads to the intensification of pro-inflammatory state, causing various metabolic disorders. 23 For the MPN patients over 80 years old, CVD was the most common cause of death, and multivariate competing risk analysis showed that older patients were more likely to die of CVD (HR 42.916, 95%CI: 31.733–58.042). The SMR of MPN patients was 8.78 times higher than that of the general population. Especially in patients diagnosed at a young age, the SMR of CVM was very high and gradually decreased with the increase of age. The risk of CVM in MPN patients younger than 40 years old was 84.55 times higher than that in the general population (95%CI, 50.11–133.62).
We observed that race, marital status, and insurance status were significant independent predictors of risk factors for CVM. Specifically, the black population, and individuals without insurance exhibited higher HR and SMR. In many other types of tumors, black patients have been observed to exhibit a poorer prognosis and a higher risk of CVM, which may be attributed to the elevated incidence of CVD within this population.24,25 In addition, other studies have found that socioeconomic influences and black people have relatively low rates of cardiac rehabilitation. 26 Married and insured patients, who exhibit a relatively lower risk of CVM, may benefit from enhanced emotional and financial support, thereby facilitating early diagnosis, effective treatment, and improved prognosis.27,28
In terms of incidence, PV and PMF exhibit a higher prevalence among males, while ET demonstrates a greater predominance in females, aligning consistently with previous statistical findings. 29 The impact of gender on CVM risk is evident, with men exhibiting higher CVM risks compared to women in the context of multiple myeloma, bladder cancer, and esophageal cancer.30–32 This disparity may be attributed to lifestyle factors such as alcohol consumption and tobacco use. 33 In our study, there was no statistically significant disparity in CVM risk between genders; however, men with MPN exhibited a CVM risk 3.56 times higher than that of the general population. The risk of CVM in female MPN patients was three times higher compared to the general population, although it remained lower than that observed in male patients.
There are several limitations to our study. First, it should be noted that this study employed a retrospective analysis methodology, and despite rigorous subject screening, an inherent selection bias remains unavoidable. Additionally, the database lacks crucial patient information including weight, smoking status, blood lipids, comorbidity of diabetes, and gene mutation status. The absence of this information impedes our ability to conduct a comprehensive evaluation of treatment efficacy and disease outcomes, potentially introducing bias into our research. The findings of our study clearly demonstrate that CVD-related mortality holds significant importance as a leading cause of death among patients with MPNs. Considering the limitations of the public database, we anticipate future multicenter large-scale clinical studies to gather more comprehensive clinical information on MPN patients, aiming for improved precision in classification, management, and treatment strategies for this population.
Conclusion
Noncancer-related mortality emerged as a prominent cause of death among patients with MPN, while the risk of CVM exhibited a significant increase, particularly in association with advancing age. This underscores the importance of early CVD assessment and regular follow-up post-MPN diagnosis, necessitating further investigations into the underlying mechanisms linking CVD with MPN to facilitate long-term management strategies for affected individuals.
