Abstract
Idiopathic multicentric Castleman disease (iMCD) is a rare and cytokine storm-driven inflammatory disorder. The exact cause of iMCD is still unknown, although several hypotheses have been proposed. However, regardless of the underlying cause, the ultimate result is the activation of the inflammatory pathway, which can lead to damage in multiple organs. Currently, there have been several reports highlighting the intricate link between coronavirus disease 2019 (COVID-19) and iMCD. To better understand the impact of COVID-19-induced immune storm on iMCD, we conducted a multicenter retrospective study in three hospitals in China. A total of 28 patients with iMCD were included, among whom 25 had confirmed COVID-19 infection, and we presented 4 cases that showed different disease progression after the infection of COVID-19, including 2 who did not receive any treatment for Castleman disease before. Our findings underscore the necessity of carefully monitoring iMCD patients with COVID-19 and promptly intervening to address any changes in their condition. Besides, this study also summarized the shared cytokines between COVID-19 and iMCD. Recent studies have shown promising results in treating severe COVID-19 and iMCD using tocilizumab, an interleukin-6 receptor antagonist. Therefore, it suggests that other potential cytokine storm therapy targets that have been effective in COVID-19 may also be explored for the treatment of iMCD.
Introduction
Castleman disease (CD) describes a group of heterogeneous lymphoproliferative disorders that share a common histopathological features on lymph node biopsy but exhibit significant variation in clinical presentation, treatment, and prognosis. 1 Idiopathic multicentric CD (iMCD) is a cytokine-driven subtype of CD that involves multiple lymph nodes and other tissues, but no infection with Human Immunodeficiency Virus (HIV), Human Herpes Virus 8 (HHV8), or Kaposi’s sarcoma-associated Herpesvirus (KSHV). Although the exact cause of iMCD remains not fully understood, it ultimately leads to the activation of the inflammatory pathway. 2 Multiple processes that are characterized by increased cytokine release could potentially give rise to iMCD. Recent RNA sequencing has revealed that the upregulation of IL-6, IL-2, plasma cell differentiation (XBP1) follicular dentritic cell (FDC) marker (CXCL13, clusterin), fibroblastic reticular cell cytokine (CCL21), angiogenesis factor (VEGF), and mammalian target of rapamycin C1 (mTORC1) pathway genes may play an important role in the progenesis of iMCD. 3
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative virus of the currently ongoing coronavirus disease 2019 (COVID-19), has given rise to an outbreak of a global pandemic. 4 With the termination of the draconian zero-COVID policy in mainland of China, a new wave of COVID-19 infections spread rapidly in China at the end of 2022. Emerging studies have shown that during SARS-CoV-2 infection, innate immune-mediated cytokine storm, including inflammatory molecules such as interleukin-1beta (IL-1β), IL-2, IL-6, IL-7, IL-8, tumor necrosis factor alpha (TNF-α), CCL2, macrophage inflammatory protein 1α (MIP-1α), and CXCL10 (inducible protein 10, IP10), attributed to severe COVID-19 cases.5–7 Currently, there have been several case reports that presented new diagnosis of CD after the infection of SARS-CoV-2 in sporadic cases, highlighting the intricate link between COVID-19 and iMCD.8–10 However, there have been no systematic reports, with sufficient sample capacity, focusing on CD patients in the COVID-19 wave and emphasizing the connection between these two inflammation-centered diseases. Therefore, to better understand of the impact of COVID-19-induced immune storms on the natural progression of iMCD, we firstly conducted a multicenter retrospective study in China.
Methods
Study population
We conducted this observational, retrospective study, including patients diagnosed with CD between October 2022 and February 2023, who had at least one hospitalization record at one of the three hospitals during the time interval from three hospitals in China (The First Affiliated Hospital of Zhejiang University, The Affiliated Jinhua Hospital of Wenzhou Medical University, and The Second Hospital of Shaoxing). Patients were eligible for inclusion if they had a confirmed diagnosis of MCD according to the criteria proposed by the China Castleman Disease Network (CCDN).11,12
The study was designed and conducted in accordance with the Declaration of Helsinki. Patients and parents included in this study consented to the use of their data for research purposes and medical ethic committee consent was obtained.
Definition
MCD was defined as the involvement of multiple lymph node regions (enlarged lymph nodes ⩾1 cm in short-axis diameter in ⩾2 lymph node stations) with pathological features consistent with CD features. Patients with diseases that might present with ‘Castleman-like’ lymph node pathological features, such as HIV infection, Epstein-Barr virus-lymphoproliferative disorders, POEMS syndrome, connective tissue diseases, lymphomas, plasmacytomas and follicular dendritic cell (FDC) sarcoma, were excluded from this study. The MCD patients enrolled in this study were further classified into HHV-8-associated MCD and HHV-8-negative MCD groups. HHV-8 status was confirmed by blood PCR or latency-associated nuclear antigen staining by immunohistochemistry. For HHV-8-negative MCD, patients who fulfilled both major criteria and at least 2 of 11 minor criteria proposed by the CCDN and did not meet any of the exclusion criteria were diagnosed with iMCD. iMCD patients were further classified into iMCD-not otherwise specified (iMCD-NOS) and iMCD-TAFRO groups. TAFRO was an acronym for thrombocytopenia (T), anasarca (A), fever (F), reticulin fibrosis of bone marrow (R), and organomegaly (O). 13 Moreover, based on the severity of the disease, iMCD patients were classified as having severe or mild/moderate/nonsevere disease according to the CCDN definition 14 : severe iMCD should have at least two of the five following criteria: Easten Cooperative Oncology Group (ECOG) ⩾2, stage IV renal dysfunction, anasarca, hemoglobin ⩽80 g/L, or pulmonary involvement/interstitial pneumonitis with dyspnea.
Treatment included anti-IL-6 therapy (Siltuximab/Tocilizumab), Rituximab, immunosuppressive agents, and chemotherapy. Response to therapy was clarified as: complete response (CR) with no residual tumorous disease and no associated clinical or biological abnormality; partial response (PR) with regression of at least 50% of the mass and significant improvement of associated symptoms; stable disease (SD) with stable mass and persistence of associated symptoms; progressive disease (PD) with enlargement of the mass or worsening of the associated symptoms. 13
Statistics
The data cut-off for this study was June 2023. The baseline and demographic characteristics were provided as descriptive statistics, including CD subtypes, clinical features, and treatment options. For continuous data, medians and ranges were presented. For categorical data, frequencies and percentages were presented. Statistical analyses were performed on IBM SPSS Statistics (Version 25.0). IBM SPSS Statistics (Version 25.0) is a statistical analysis software developed and distributed by IBM Corporation. The corporation’s headquarters is located in Armonk, New York, United States.
Results
Characteristics of patients are summarized in Table 1. A total of 28 patients with iMCD were included, with an average age of 55.7 years (range: 16–73 years), and 75% (21/28) being male. According to the CCDN criteria, patients consisted of 75% iMCD (21/28), 17.9% iMCD-TAFRO (thrombocytopenia, ascites, fever, reticulin fibrosis and organomegaly) (5/28) and 7.1% iMCD-POEMS (polyneuropathy, organomegaly, endocrinopathy, monoclonal plasma cell disorder and skin changes) (2/28), and the pathological categorization of HV accounted for 21.4% (6/28), and PC accounted for 67.9% (19/28). Among the 28 patients involved, 14 patients (50%) were classified as severe patients at time of diagnosis. Time intervals between symptom onset and diagnosis were commonly observed, of which, 25% (7/28) patients and 10.7% (3/28) patients were diagnosed ⩾12 and ⩾24 months, respectively, after the initial onset of symptoms. The maximum time interval to diagnosis was 84 months.
Characteristics of patients included in the analysis.

COVID-19, coronavirus disease 2019; CR, complete response; iMCD-NOS, idiopathic multicentric Castleman disease-not otherwise specified; PD, progressive disease; PR, partial response; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; SD, stable disease; PC, plasma cell; HV, hyaline vascular.
Prior to COVID-19 infection, except for 2 patients who elected not to receive treatment for personal reasons, the other 26 patients had received various treatment regimens [Figure 1(a)]. In all regimens, Rituximab (R)/lenalidomide plus dexamethasone (RD)/RD plus cyclophosphamide (RCD)/RD plus bortezomib (RVD) were used in 19 patients (73.1%), followed by Rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP)/R-CHOP-like regimens (10/26, 38.5%), anti-IL-6 therapy (siltuximab/tocilizumab) (8/26, 30.8%), bortezomib plus dexamethasone (VD)/VD plus cyclophosphamide (VCD)/Rituximab plus VCD (7/26, 27.0%), cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP)/CHOP-like (4/26, 15.4%), thalidomide, cyclophosphamide, and prednisone (TCP)/thalidomide, cyclophosphamide, and dexamethasone (TCD)/thalidomide plus and dexamethasone (3/26, 11.5%), single-agent glucocorticoids (1/26, 3.8%), and the Rituximab plus lenalidomide (R square) regimen (1/26, 3.8%) [Figure 1(a)]. By November 2022, 5 patients achieved CR, 11 patients achieved PR, 9 patients stayed SD, 2 patients were evaluated with PD, and 1 patient died of disease progression.

(a) Percentage distribution of patients with different treatment options. (b) Schematic of laboratory findings in patient 1, 3, 4 after the infection of COVID-19. (c) The cross-connection of cytokine storm between COVID-19 and iMCD.
Among the enrolled patients, 25 patients were confirmed to have infection of COVID-19 with at least one documented positive PCR test for SARS-CoV-2 in their nasopharyngeal samples between October 2022 and February 2023. Out of the total 25 patients, 10 individuals (9 with iMCD and 1 with iMCD-TAFRO) reported asymptomatic disease, 9 patients (4 with iMCD, 4 with iMCD-TAFRO, and 1 with iMCD-POEMS) reported having mild disease. Additionally, six patients with iMCD experienced moderate disease, which required hospitalization but did not necessitate ventilation or intensive care. The common symptoms included fever/chills (11/25, 44%), cough (5/25, 20%), loss of taste or smell (2/25, 8%), shortness of breath/difficulty breathing (1/25, 4%). One patient experienced a delay in CD treatment for a period of 1 month, but later resumed the treatment. Additionally, another patient opted to switch from R-based chemotherapy to R monotherapy and continued with this single-agent treatment regimen thereafter.
We observed disease progression in four iMCD patients (Table 2), and one of which two cases were observed with liver dysfunction following COVID-19 infection.
Clinical features of the four patients that exhibited disease progression following COVID-19.
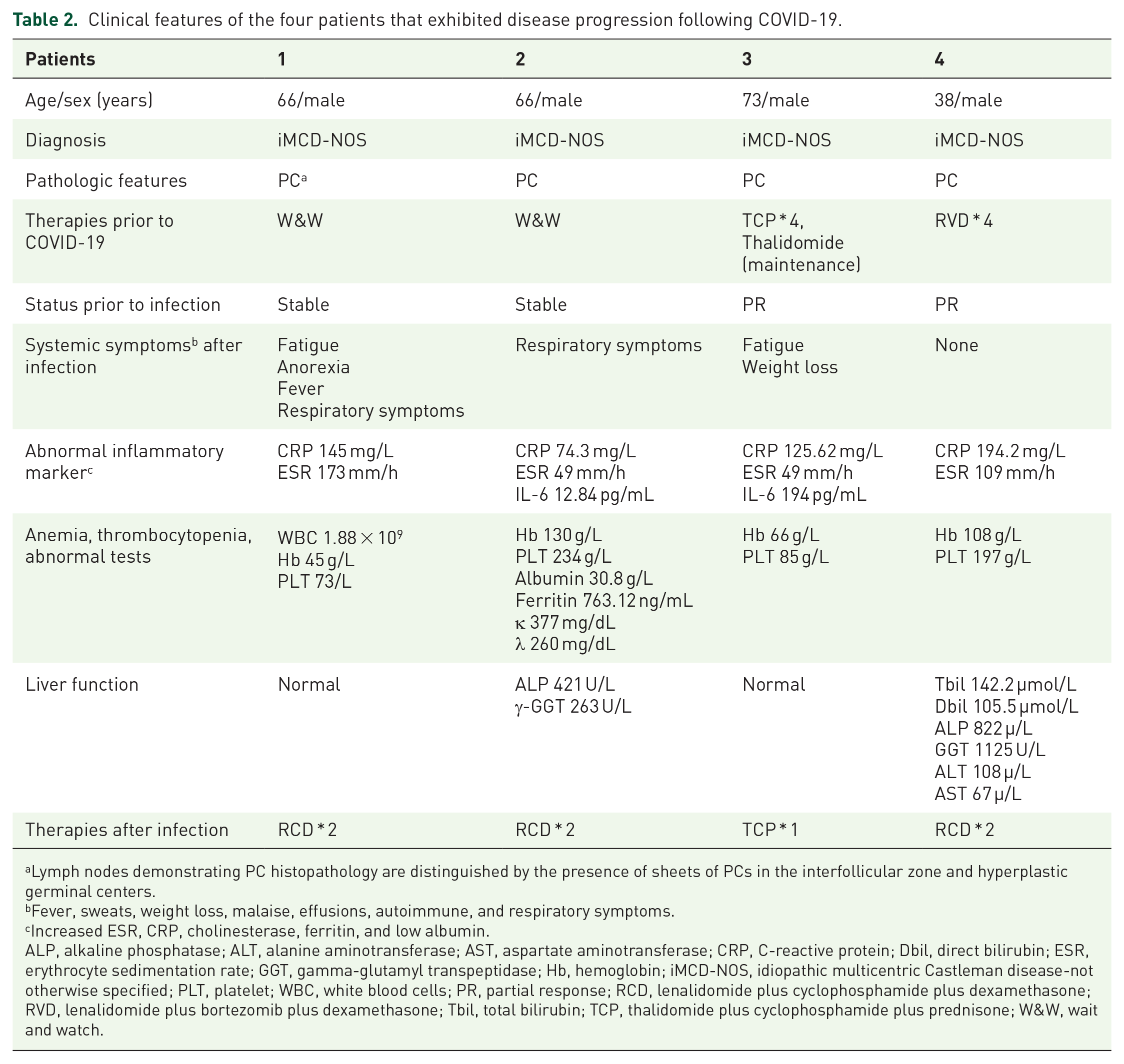
Lymph nodes demonstrating PC histopathology are distinguished by the presence of sheets of PCs in the interfollicular zone and hyperplastic germinal centers.
Fever, sweats, weight loss, malaise, effusions, autoimmune, and respiratory symptoms.
Increased ESR, CRP, cholinesterase, ferritin, and low albumin.
ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CRP, C-reactive protein; Dbil, direct bilirubin; ESR, erythrocyte sedimentation rate; GGT, gamma-glutamyl transpeptidase; Hb, hemoglobin; iMCD-NOS, idiopathic multicentric Castleman disease-not otherwise specified; PLT, platelet; WBC, white blood cells; PR, partial response; RCD, lenalidomide plus cyclophosphamide plus dexamethasone; RVD, lenalidomide plus bortezomib plus dexamethasone; Tbil, total bilirubin; TCP, thalidomide plus cyclophosphamide plus prednisone; W&W, wait and watch.
Patient 1 and patient 2 were diagnosed with iMCD 2 and 1 years ago, respectively. They had not received any treatment for their personal choice prior to COVID-19 infection. Patient 1 was re-admitted to hospital 3 weeks after COVID infection due to abnormal blood test indicating high inflammation: WBC 1.88 × 109/L, hemoglobin (Hb) 45 g/L, platelet (PLT) 73 × 109/L, C-reactive protein (CRP) 145 mg/L, and erythrocyte sedimentation rate (ESR) 173 mm/h [Figure 1(b)]. Patient 2 was hospitalized for the presentation of seizure and shortness of breath, and chest CT scan showed strikingly increased pleural and peritoneal effusion. Both inflammatory markers (CRP 74.3 mg/L and ESR 49 mm/h) and polyclonal immunoglobulins (Kappa 377 mg/dL, Lambda 260 mg/dL) confirmed the progression of CD. Up to date, both patients achieved PR status after two cycles of RCD regimen (Rituximab, cyclophosphamide, dexamethasone) (Table 2).
The other two patients (Patient 3–4) were undergoing regular treatment for CD before contracting COVID-19. Patient 3, a 73-year-old iMCD patient achieved CR after receiving four cycles of TCP regimen (thalidomide, cyclophosphamide, and dexamethasone) and maintenance treatment with thalidomide for 2 years. However, even after testing negative for COVID-19 2 weeks later, the patient continued to exhibit symptoms of fever and fatigue, and was discovered to have a high level of inflammation (CRP 125.62 mg/L, ESR 49 mm/h, IL-6 194 pg/mL), and decreased blood count (Hb 66 g/L, PLT 85 × 109/L) [Figure 1(b)]. He received TCP regimen again and both clinical features and laboratory tests gradually improved. Patient 4, a 38-year-old male, was diagnosed with iMCD-PC with liver cirrhosis 5 months ago. He had received four cycles of RVD regimens (Rituximab, bortezomib, cyclophosphamide) and achieved a PR status with normal liver function. However, he was readmitted to hospital again for painless progressive jaundice 2 weeks after recovering from COVID-19. His liver-function tests indicated obstructive jaundice (total bilirubin 142.2 µmol/L, direct bilirubin 105.5 µmol/L, alkaline phosphatase 822 µ/L, gamma-glutamyl transpeptidase 1125 U/L), and he also presented with high levels of inflammatory markers including CRP (194.2 mg/L) and ESR (109 mm/h) [Figure 1(b)]. After two cycles of the RCD regimen, a PR was achieved.
Discussion
There have been several reports emphasizing the complex connection between COVID-19 and iMCD. Mosleh et al. documented the case of a formerly healthy adolescent who developed iMCD-TAFRO 6 weeks after contracting COVID-19. 8 Additionally, Andreas Kessler et al. and Kazuhiro et al. reported instances of iMCD-NOS and iMCD-TAFRO, respectively, occurring in previously healthy young adults shortly after mRNA SARS-CoV-2 vaccination.9,10 David and their team presented findings from their follow-up cohort, which included 10 (14.5%) CD patients who had tested positive for nucleic acid, with the majority of patients exhibiting mild symptoms and no evidence of disease progression. 15 In our study, similar mild symptoms during the acute infection of COVID-19 were observed. However, within the 25 infectious cohort, 4 patients with a prior diagnosis of iMCD experienced disease progression after SARS-CoV-2 infection. Interestingly, their symptoms did not manifest immediately after infection, but appeared gradually about 2 weeks after recovery. This suggests that the virus may have triggered the onset and progression of iMCD through activation of cytokines and inflammatory pathways.
The cytokine storm is a critical factor in the pathogenesis of both COVID-19 and iMCD. Various studies have shown increased levels of inflammatory cytokines, including IL-1β, IL-2, IL-6, IL-10, interferon-gamma (IFN-γ), TNF-α, IFN-γ-IP10, granulocyte macrophage-colony stimulating factor, and monocyte chemoattractant protein-1, which are positively correlated with disease severity.16,17 Interestingly, both COVID-19 and iMCD share some cytokines, including IL-1β, IL-2, IL-6, IL-8, IFN-γ, and TNF-α.18,19 Recently, the lymph node transcriptome of iMCD also revealed that increased transcription of complement-related genes in lymphadenopathy and complement activation may act as a possible mediator of the inflammatory manifestations of iMCD. 3 Pathway analysis also revealed a key C3–CXCL4–CXCL13–CXCL9–CXCL10 cytokine axis that may play an important role in the process. 20 We attempted to summarize the cytokine profiles that have been proved associated with both CD and COVID-19 infection [Figure 1(c)]. Previous studies have shown that the use of tocilizumab, an IL-6 receptor antagonist, has shown promise in treating both severe COVID-19 and iMCD.21,22 This to some extent provided insights into the etiological research of CD, and also suggests that other potential cytokine storm therapy targets that have been effective in COVID-19 may also be explored for the treatment of iMCD.
In summary, our study represents the first investigation in China into the experience and natural history of CD patients during the COVID-19 pandemic. We innovatively highlight the significance of the link between the advancement of iMCD and SARS-CoV-2 infection. It is worth noting that in some of our cases, the progression of iMCD did not occur in parallel with an elevated cytokine/inflammatory state, suggesting alternative mechanisms may be involved. Additionally, the exact mechanism underlying liver function impairment in iMCD following SARS-CoV-2 infection remains unclear. Nevertheless, our findings underscore the necessity of carefully monitoring iMCD patients with COVID-19 and promptly intervening to address any changes in their condition.
