Abstract
Background:
Advances in treatment have enabled patients with haemophilia A to live longer and therefore may be subjected to comorbidities associated with ageing, in addition to disease-associated morbidities. There have been few reports to date on efficacy and safety of treatment specifically in patients with severe haemophilia A and comorbidities.
Objective:
To explore the efficacy and safety of damoctocog alfa pegol prophylaxis in patients with severe haemophilia A aged ⩾40 years with comorbidities of interest.
Design:
A post hoc analysis of data from the phase 2/3 PROTECT VIII study and its extension.
Methods:
Bleeding and safety outcomes were analysed in a subgroup of patients aged ⩾40 years with ⩾1 comorbidity receiving damoctocog alfa pegol (BAY 94-9027; Jivi®) prophylaxis.
Results:
Thirty-four patients with severe haemophilia A were included in this analysis, with a mean age of 49.4 years at time of enrolment. The most prevalent comorbidities were hepatitis C (n = 33; chronic, n = 23), hepatitis B (n = 8) and hypertension (n = 11). Four patients had human immunodeficiency virus. All received damoctocog alfa pegol prophylaxis for the entire study [median (range) time in study = 3.9 (1.0–6.9) years]. During the main study and extension, median total annualised bleeding rates (ABRs) (Q1; Q3) were 2.1 (0.0; 5.8) and 2.2 (0.6; 6.0), respectively; median joint ABRs were 1.9 (0.0; 4.4) and 1.6 (0.0; 4.0), respectively. Mean adherence with prophylaxis schedule was greater than 95% throughout the study. No deaths or thrombotic events were reported.
Conclusion:
Efficacy, safety and adherence of damoctocog alfa pegol were confirmed in patients aged ⩾40 years with haemophilia A and one or more comorbidities, with data for up to 7 years supporting its use as a long-term treatment option in this group.
Plain language summary
Advances in treatment mean that people with haemophilia A are now living longer and, as a result, may have additional medical conditions that occur with ageing. We aimed to investigate the efficacy and safety of the long-acting replacement factor VIII damoctocog alfa pegol in people with severe haemophilia A who had additional medical conditions. To do this, we investigated the recorded information about patients aged 40 years of age or older who had been treated with damoctocog alfa pegol in a previously completed clinical trial. We found that the treatment was well-tolerated; no deaths or thrombotic events (undesirable clotting events) were reported. Treatment was efficacious in reducing bleeding in this group of patients. The findings support the use of damoctocog alfa pegol as a long-term treatment for older patients with haemophilia A and coexisting conditions.
This is a visual representation of the abstract.
Introduction
Prophylaxis with factor (F) VIII has become the gold standard for haemophilia A management, reducing both bleeds and arthropathy.1,2 Owing to the improvement of treatment over the last decade, patients with haemophilia A are now living longer and, consequently, are subject to many of the comorbidities associated with ageing of the general population, including hypertension,3,4 obesity, diabetes and cardiovascular diseases, in addition to the typical disease-associated morbidities.5,6
With older age, in addition to haemophilia-associated morbidities such as chronic and debilitating arthropathy with increased risk of falls and fractures,3,7 patients face late consequences of underlying concomitant diseases. These can include increasing risks of cancer, chronic liver disease or worsening of existing liver disease due to hepatitis B or C virus (HBV/HCV) and human immunodeficiency virus (HIV) as well as long-term effects of HIV and HCV treatment, 8 haematuria due to urologic diseases, 9 renal disease due to hypertension and long-term use of non-steroidal anti-inflammatory drugs, 10 and cardiovascular diseases due to atherosclerosis. 11 These additional risks pose new challenges for haemophilia treaters. Furthermore, older patients with haemophilia may have a reduced ability to self-infuse due to poor venous access, poor vision, reduced dexterity or dementia; these factors could represent additional barriers to treatment adherence.12,13 In addition, patients with comorbidities also have an increased likelihood of taking concomitant medications, with some cardiovascular medications interfering with haemostasis and potentially increasing bleeding tendency. 5
The Age-related DeVelopments and Comorbidities in Haemophilia (ADVANCE) Working Group, a panel of experienced haemophilia treaters from across Europe, is working to raise awareness of the management of age-related diseases that affect the older haemophilia population (e.g. cardiovascular disease, cancer, dementia). 14 A cross-sectional study by the ADVANCE Working Group has shown that cardiovascular disease is prevalent in people with severe haemophilia aged 40 years and above.6,15
To explore the efficacy and safety of damoctocog alfa pegol prophylaxis in patients with severe haemophilia A aged ⩾40 years with comorbidities of interest, we carried out a post hoc analysis of data from the phase 2/3 PROTECT VIII study and its extension. PROTECT VIII has previously demonstrated the efficacy and safety of damoctocog alfa pegol prophylaxis for the prevention of bleeding episodes in adults and adolescents with severe haemophilia A for ⩾5 years at dose intervals of up to every 7 days. 16 Here, we report results from PROTECT VIII and its extension study showing the efficacy and safety of damoctocog alfa pegol prophylaxis in this population of patients with comorbidities, aged 40 years and older.
Methods
Patients
In this post hoc analysis, patients were included if they were aged ⩾40 years at study entry, were treated with any prophylaxis regimen during both the main study and its extension, and had a medical history of the following comorbidities of interest, as noted by the investigator at baseline and according to the primary system organ class and preferred terms from Medical Dictionary for Regulatory Activities (MedDRA) version 22.1: infections (HIV with CD4+ lymphocyte count greater than 200/mm3, hepatitis B, hepatitis C); metabolism and nutrition disorders (hyperlipidaemia, diabetes, obesity); or vascular (hypertension) and cardiac disorders. Some patients had more than one comorbidity of interest and were included in more than one group.
Study design
The design, patient eligibility, treatment schedule and statistical analyses for PROTECT VIII (NCT01580293), a phase 2/3 partially randomised, open-label trial evaluating the efficacy and safety of damoctocog alfa pegol, and its extension study have been previously described.16–18 Of the 134 males aged 12–65 years with severe haemophilia A (FVIII < 1%) who were recruited into the main study, 104 patients received damoctocog alfa pegol prophylaxis in both the 36-week main study and the optional extension. During the main study, patients were randomly assigned to one of the three regimens based on their bleeding tendency during a 10-week run-in period. During the extension, patients could continue damoctocog alfa pegol on the same regimen as used in the main study [30–40 IU/kg twice weekly (2 × W), 45–60 IU/kg damoctocog alfa pegol every 5 days (E5D) or 60 IU/kg every 7 days (E7D)], or the regimen and dosage could be adapted to individual needs within these prespecified groupings, as determined by the prescribing physician. This post hoc study is of patients who received prophylaxis during the main and extension studies aged ⩾40 years and with one or more comorbidity of interest.
Efficacy and safety assessments
Throughout the extension study, patients were closely monitored at visits every 6 months for the incidence of adverse events (AEs), which were documented in terms of type, severity and relationship to study drug. Visits were more frequent during the 36-week main study period. Serum and urine samples were collected at baseline, at the end of the main study and throughout the extension study to measure levels of hepatic (alanine aminotransferase [ALT], aspartate transaminase [AST]) and renal (serum creatinine, creatinine clearance by calculation) biomarkers.
Bleeding events and administered infusions were recorded by patients using an electronic patient diary. Annualised bleeding rates (ABRs) were calculated for all patients and by comorbidity type (infections, metabolism and nutritional disorders, vascular and/or cardiac disorders) prestudy, during weeks 10 to 36 of the main study and during the extension study. Patients were also assessed for arthropathy and the development of new target joints, defined as ⩾3 spontaneous bleeds in the same joint during a 6-month period, 19 and resolution of existing target joints, defined as ⩽2 bleeds in the same joint in a 12-month period. 19
Statistical analyses
Statistical analysis was performed using SAS software 9.2. Summary statistics were calculated for continuous data, and frequencies were calculated for categorical data. No formal statistics were performed, and the analysis was descriptive. Final data, after completion of the study, were used in this post hoc analysis. Bleeding outcomes (ABR) were analysed for the total population and by regimen. Patients who switched regimen during the extension study were included for analysis in the variable frequency group.
Results
Patients
Of the 104 patients treated with damoctocog alfa pegol prophylaxis during the PROTECT VIII main study and its extension, a total of 35 (34%) patients were aged 40 years and above at study entry. Of the patients in this age group, 34 presented with one or more comorbidity of interest and were included in the analysis (Table 1).
Demographics and disease characteristics of patients aged ⩾40 years with ⩾1 comorbidity of interest from PROTECT VIII.
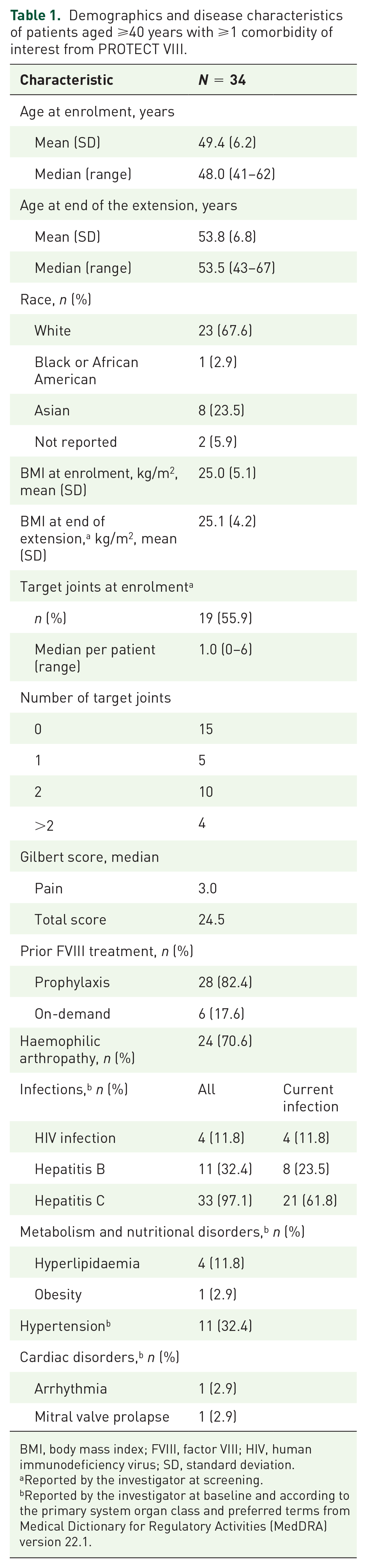
BMI, body mass index; FVIII, factor VIII; HIV, human immunodeficiency virus; SD, standard deviation.
Reported by the investigator at screening.
Reported by the investigator at baseline and according to the primary system organ class and preferred terms from Medical Dictionary for Regulatory Activities (MedDRA) version 22.1.
The mean [standard deviation (SD)] and median (range) ages of the 34 patients at enrolment were 49.4 (6.2) and 48.0 (41–62) years, respectively, and 53.8 (6.8) and 53.5 (43–67) years at the end of the extension, respectively. Mean body mass index (BMI) was 25.0 kg/m2. A large majority of the patients were White [n = 23 (67.6%)] and of non-Hispanic or Latino origin [n = 32 (94.1%)]. Eight (23.5%) patients identified as Asian and one (2.9%) as Black or African American descent.
Nineteen (55.9%) patients presented with a target joint at baseline (as reported by the investigator), ranging from one to six target joints, with a mean (median) of 1.5 (1.0) per patient. The majority of patients presented with two target joints (n = 10, 29.4%) or one target joint (n = 5, 14.7%) at baseline. Median (range) Gilbert score at baseline was 24.5 (8–51). The median (range) pain subscore was 3 (0–12).
Based on the medical history data, 33 (97.1%) patients had a previous history of viral infection [hepatitis B (n = 11; n = 8 current infection), hepatitis C (n = 33; of these, 23 patients had chronic hepatitis C) and HIV (n = 4)]. Five (14.7%) patients had a previous history of metabolism and nutritional disorders [hyperlipidaemia (n = 4), obesity (BMI > 30 kg/m2, n = 1)]. Eleven (32.4%) patients had hypertension, two of whom also had a separate cardiac disorder [arrhythmia (n = 1), mitral valve prolapse (n = 1)], according to the MedDRA definition. No other cardiac disorders were reported in the medical history. Arthropathy was reported in 24 (70.6%) patients.
Previous FVIII treatment is shown in Table 1. Prior to the main PROTECT VIII study, 28 of 34 (82.4%) patients were on regular FVIII prophylaxis and 6 of 34 (17.6%) were receiving on-demand treatment. Prophylaxis regimens received during PROTECT VIII are reported in Supplementary Appendix 1.
All 34 patients took at least one concomitant medication during the PROTECT VIII main study and extension. In total, 25 (73.5%) patients, including five patients with metabolism and nutrition disorders and all patients with vascular disorders (n = 11), received concomitant cardiovascular medications [vasoprotectives (n = 14), agents acting on the renin–angiotensin system (n = 13), cardiac therapy (n = 12), lipid modifying agents (n = 8), calcium channel blockers (n = 7), antihypertensives (n = 4), beta blocking agents (n = 4) and diuretics (n = 4)]. Twenty-four (70.6%) patients received concomitant anti-inflammatories or antirheumatics, and 25 (73.5%) patients in total received concomitant analgesics (Table 2).
Selected concomitant medications received by patients aged ⩾40 years with ⩾1 comorbidity of interest from PROTECT VIII.

Median (range) total time in study was 46.8 (12.0–82.8) months [3.9 (1.0–6.9) years]. Patients spent a median (range) of 8.4 (7.8–10.8) months [0.7 (0.65–0.9) years] in the main study, and 38.4 (3.6–74.4) months [3.2 (0.3–6.2) years] in the extension study.
Safety
AEs reported during the main and extension studies are presented in Table 3. Two (5.9%) patients had study drug-related AEs, classed as either mild (n = 2, two cases of palpitations in the same patient) or moderate (n = 1, arthralgia). Five (14.7%) patients reported serious adverse events (SAEs) during the main study, and none were drug related.
Adverse events during PROTECT main and extension studies in patients with comorbidities aged ⩾40 years (n = 34).
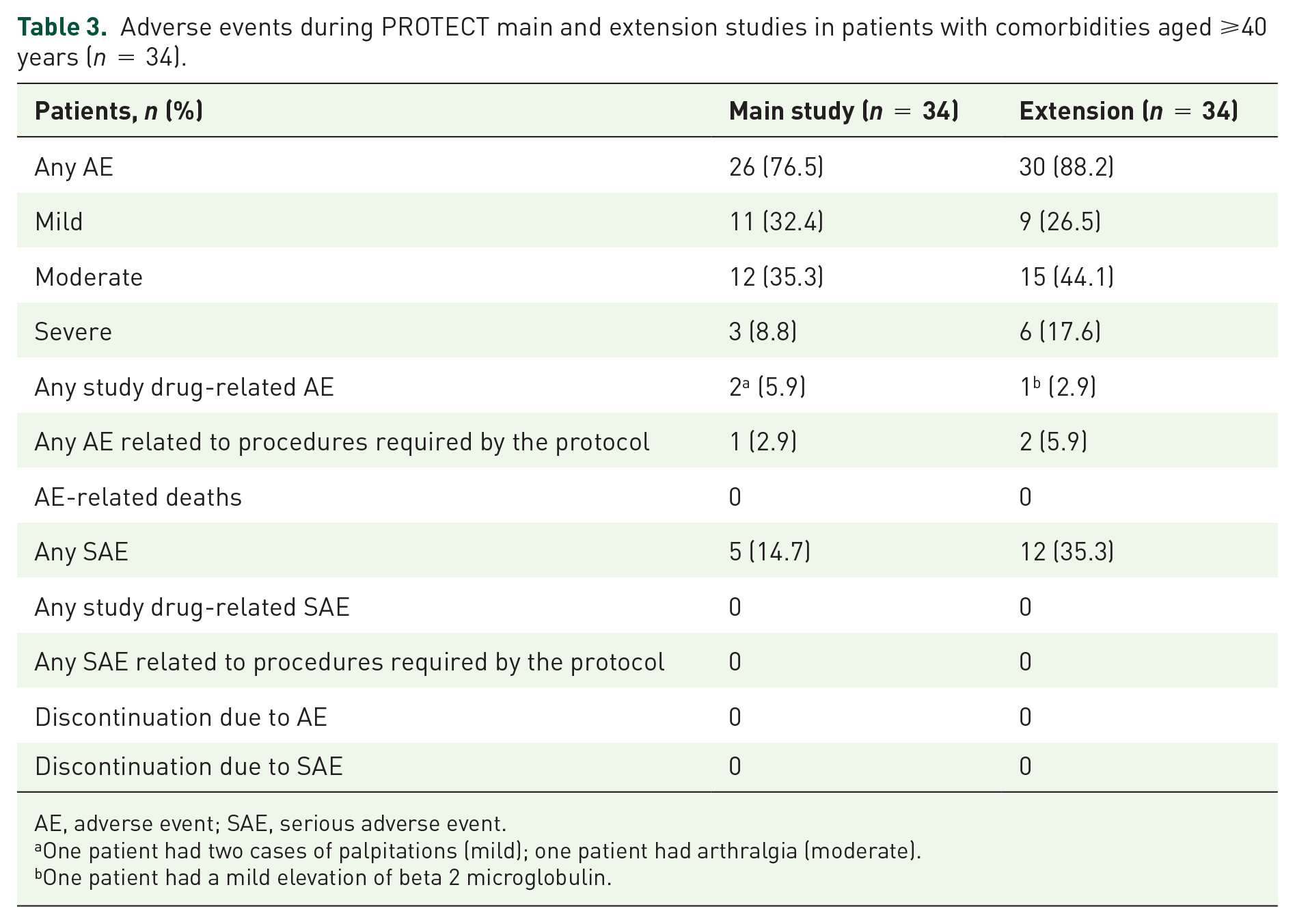
AE, adverse event; SAE, serious adverse event.
One patient had two cases of palpitations (mild); one patient had arthralgia (moderate).
One patient had a mild elevation of beta 2 microglobulin.
During the extension, 30 (88.2%) patients reported AEs, one (2.9%) of which was determined to be drug-related and classed as mild (increase in beta 2 microglobulin in urine). Twelve (35.3%) patients experienced a total of 20 SAEs during the extension study: none of these were considered drug-related and all resolved by the end of the study except for one case of pseudogout that was still ongoing.
No thrombotic or cardiovascular events were observed during the main or extension studies and no patient died during the main or the extension study. No discontinuations due to AEs or SAEs occurred in either the main or extension studies in this population.
There were no safety concerns relating to PEG during the main and extension studies. Of those with available data, median levels of serum creatinine, creatinine clearance, AST and ALT remained within the normal ranges, as provided by the central laboratory, at the final visit of the main study (n = 33) and at the final visit of the extension (n = 31) (Table 4).
Renal and liver biomarkers measurements by visit and change from baseline in patients with comorbidities aged ⩾40 years (n = 34).

AST, aspartate aminotransferase; ALT, alanine aminotransferase.
Normal ranges provided by central laboratory: creatinine clearance = 77–160 mL/min; serum creatinine = 0.5–1.3 mg/mL; AST = 11–36 U/L; ALT = 6–43 U/L.
n = 32.
Patient had high ALT and AST values at week 10, which had decreased by the end of the extension.
n = 30.
Patient with this high value had abnormal (high) ALT at screening, but a normal value at the end of the extension.
Bleeding outcomes
Total and joint ABRs for all patients aged ⩾40 years with ⩾1 comorbidity of interest
Prestudy, main study and extension study total and joint ABRs are presented in Figure 1. Prestudy median (Q1; Q3) total ABR for all 34 patients was 6.0 (0.0; 15.0). During the main study (weeks 10–36) and extension, median total ABRs were 2.1 (0.0; 5.8) and 2.2 (0.6; 6.0), respectively. Prestudy median (Q1; Q3) joint ABR for all 34 patients was 2.0 (0.0; 12.0). During the main study and extension, median joint ABRs were 1.9 (0.0; 4.4) and 1.6 (0.0; 4.0), respectively. Total and joint ABRs by prior FVIII treatment type are also shown in Figure 1.

Total and joint ABRs prestudy, during PROTECT VIII main study and extension in patients with comorbidities aged ⩾40 years (n = 34), and by prior FVIII treatment type.
Prestudy, main study and extension study total and joint ABRs stratified by comorbidity type are shown in Supplementary Table 1. There was little difference in the ABR between the comorbidity subgroups.
Target joints
All patients presenting with a target joint at baseline (n = 19) had ⩾1 resolved target joint by the end of the extension study. Of these, four target joints in four patients were unresolved. Seven patients developed a new target joint during the study, three of whom who did not have a target joint at baseline. Of these seven new target joints, four (57.1%) resolved during the last 12 months of the extension.
FVIII utilisation
During the main study, the median (range) prophylaxis dose per infusion was 48.41 (31.4–64.3) IU/kg, and median annual prophylaxis dose was 3245.51 (2707.3–3930.0) IU/kg. During the extension, the median (range) dose per prophylaxis infusion was 48.85 (30.9–65.3) IU/kg, and median annual prophylaxis dose was 3409.51 (2475.5–4841.8) IU/kg.
Adherence with prophylaxis schedule
Overall, mean adherence with prophylaxis schedule was greater than 95% throughout the study. In the main and extension studies, mean adherence (standard deviation) for 34 patients based on prophylaxis infusion count was 107% (7.01) and 99.7% (6.25), respectively. Adherence greater than 100% is suggestive of more injections given than prescribed. Mean adherence during the extension for patients analysed in the 2 × W (n = 5), E5D (n = 10), E7D (n = 5) and variable frequency (n = 12) groups was 97.0% (3.09), 103.5% (7.62), 97.1% (9.35) and 98.8% (2.60), respectively.
Discussion
To our knowledge, this is the first subanalysis dedicated to patients with severe haemophilia A and a history of comorbidities among pivotal trials of the extended half-life FVIII replacement products. Most comorbidities recorded were cardiovascular diseases (including hypertension, as may be anticipated from a study in this population) and infections, which are known to be burdens of haemophilia. Data from this subanalysis show that damoctocog alfa pegol prophylaxis was efficacious in this subgroup of ageing haemophilia A population without an increase in the yearly FVIII utilisation.
No SAEs related to the study drug, or other safety concerns, were observed in patients with cardiovascular comorbidities, and no cardiovascular events occurred during up to 7 years of study participation. Cardiovascular diseases are prevalent in ageing patients with haemophilia A, who have a higher prevalence of hypertension than the general population. 20 Atrial fibrillation occurs in 0.84% of patients with haemophilia, increasing to 3.4% in patients aged greater than 60 years, which is similar to that reported for the general population. 6 Almost one-quarter of the study population in the present study were taking concomitant cardiovascular medications, with two patients receiving antithrombotic therapy. However, among the total population of this study, median total and joint ABRs in the extension remained low, at 2.2 and 1.6, respectively. Furthermore, total and joint ABRs in the main study and the extension study in patients with vascular and/or cardiac disorders were also low (Supplementary Table 1). The ADVANCE Working Group is currently recruiting patients into a longitudinal, prospective analysis of the risk, incidence and management of cardiovascular disease in older patients with haemophilia A. The study will compare observed rates of fatal and non-fatal cardiovascular disease events with expected numbers based on cardiovascular risk prediction at baseline, as well as documenting the incidence of other comorbidities, haemophilia status and treatment. 14 As reported for the total PROTECT VIII population,16,18 damoctocog alfa pegol was well-tolerated in this subgroup throughout the main and extension periods in which: no thrombotic or cardiovascular events were observed; no treatment-related SAEs were observed; no discontinuations due to an AE occurred; and no patient died. Median levels of renal and liver biomarkers remained within normal ranges.
In this subgroup, damoctocog alfa pegol prophylaxis reduced total and maintained joint ABR from prestudy to the end of the extension, with the largest improvements observed in patients who were previously treated on-demand. ABRs in the subpopulation with comorbidities are comparable with those observed in the total PROTECT VIII population, in which efficacy of damoctocog alfa pegol was maintained over more than 5 years’ prophylaxis.16,18 In this study, ABR remained low in all prophylaxis groups during the extension period. Prolonged efficacy across different dose intervals means that patient outcomes can be improved using a preferred choice of prophylaxis regimen based on patients’ individual needs and circumstances.
Damoctocog alfa pegol prophylaxis resulted in the resolution of ⩾1 target joints in all patients assessed. Taken together, these efficacy data suggest that damoctocog alfa pegol effectively reduces bleeds in older patients with comorbidities and corroborates with those of the total PROTECT VIII population with no requirement for an increase in dose.17,18
Overall, levels of damoctocog alfa pegol utilisation did not increase substantially over time (from main study to extension study) and were comparable with those of the total PROTECT VIII population. Low FVIII utilisation may provide an additional benefit of reduced risk for complications in haemophilia A patients with comorbidities. In this subgroup, protection was achieved with administered doses that were at the lower end of the recommended narrow dose range for each infusion regimen.
Adherence to prophylaxis schedule was high (>95%) throughout the main and extension studies for all regimens, which is encouraging in this population who may find venous access problematic as a consequence of ageing and comorbidities. 12 Prior to PROTECT VIII, six patients included in this post hoc analysis were receiving on-demand treatment. The high overall adherence rate to a prophylaxis schedule may also suggest a smooth transition in patients switching from previous on-demand therapy to prophylaxis with damoctocog alfa pegol.
One of the strengths of PROTECT VIII was the flexibility of prophylaxis regimens, allowing for individualisation of dosing intervals. Prolonged dosing intervals can reduce treatment burden, which is particularly important for patients with comorbidities who may need to receive additional therapies, or those who have difficulties in administering the drug due to a decrease in manual dexterity.
The majority (88%) of patients were treated with extended interval prophylaxis (every 5 or 7 days) during the main study, and 50% chose either every 5- or 7-day prophylaxis regimen during the extension. In addition, the PROTECT VIII extension study has the longest individual patient follow-up among other extension studies in adolescents and adults with severe haemophilia A. 16 Such prolonged follow-ups provide important long-term efficacy and safety profiles.
There were some limitations to this analysis. The sample size (N = 34) should be considered when interpreting the results. In PROTECT VIII, comorbidity categories were defined by MedDRA definitions, and thus the available information on the patients’ specific comorbidities was limited. Of the total PROTECT VIII study population receiving prophylaxis during both the main and extension studies, 34% were aged ⩾40 years, and as all but one of these patients had a comorbidity of interest, no age-matched comparator group was available. The mean age at enrolment of patients in this study was close to 50 years old, which could be considered a relatively ‘young’ population in terms of CV complications, cancer and other diseases associated with ageing. In addition, the PROTECT VIII study enrolled patients with severe haemophilia A aged 12 to 65 years; hence, outcomes for elderly patients aged over 65 years could not be assessed in this post hoc analysis. 18
In conclusion, damoctocog alfa pegol is an efficacious and well-tolerated prophylactic treatment that does not pose additional risk of cardiovascular events, or renal and hepatic function deterioration, in patients with haemophilia A, aged ⩾40 years and who may have additional comorbidities. These data support the favourable benefit–risk profile of damoctocog alfa pegol and its use as a long-term treatment option for older patients with haemophilia A.
Supplemental Material
sj-docx-1-tah-10.1177_20406207231166779 – Supplemental material for Efficacy and safety of damoctocog alfa pegol prophylaxis in patients ≽40 years with severe haemophilia A and comorbidities: post hoc analysis from the PROTECT VIII study
Supplemental material, sj-docx-1-tah-10.1177_20406207231166779 for Efficacy and safety of damoctocog alfa pegol prophylaxis in patients ≽40 years with severe haemophilia A and comorbidities: post hoc analysis from the PROTECT VIII study by Mark T. Reding, Ingrid Pabinger, Pål Andrè Holme, Monika Maas Enriquez, Maria Elisa Mancuso, Shadan Lalezari, Wolfgang Miesbach, Giovanni Di Minno, Robert Klamroth and Cedric Hermans in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
