Abstract
Background:
Patients with Philadelphia-negative chronic myeloproliferative neoplasms (MPN) typically incur high rates of infections and both drugs and comorbidities may modulate infection risk.
Objectives:
The present study aims to assess the effect of immunosuppressive agents on clinical outcomes of MPN patients affected by the coronavirus disease 2019 (COVID-19).
Design:
This is an observational study.
Methods:
We specifically searched and analyzed MPN patients collected by EPICOVIDEHA online registry, which includes individuals with hematological malignancies diagnosed with COVID-19 since February 2020.
Results:
Overall, 398 patients with MPN were observed for a median of 76 days [interquartile range (IQR): 19–197] after detection of SARS-CoV2 infection. Median age was 69 years (IQR: 58–77) and 183 individuals (46%) had myelofibrosis (MF). Overall, 121 patients (30%) of the whole cohort received immunosuppressive therapies including steroids, immunomodulatory drugs, or JAK inhibitors. Hospitalization and consecutive admission to intensive care unit was required in 216 (54%) and 53 patients (13%), respectively. Risk factors for hospital admission were identified by multivariable logistic regression and include exposure to immunosuppressive therapies [odds ratio (OR): 2.186; 95% confidence interval (CI): 1.357–3.519], age ⩾70 years, and comorbidities. The fatality rate was 22% overall and the risk of death was independently increased by age ⩾70 years [hazard ratio (HR): 2.191; 95% CI: 1.363–3.521], previous comorbidities, and exposure to immunosuppressive therapies before the infection (HR: 2.143; 95% CI: 1.363–3.521).
Conclusion:
COVID-19 infection led to a particularly dismal outcome in MPN patients receiving immunosuppressive agents or reporting multiple comorbidities. Therefore, specific preventive strategies need to be tailored for such individuals.
Plain language summary
Patients with Philadelphia-negative chronic myeloproliferative neoplasms (MPN) incur high rates of infections during the course of their disease.
The present study was aimed at assessing which patient characteristics predicted a worse outcome of SARS-COV-2 infection in individuals with MPN.
To pursue this objective, the researchers analyzed the data collected by EPICOVIDEHA, an international online registry, which includes individuals with hematological malignancies diagnosed with COVID-19 since February 2020.
The database provided clinical data of 398 patients with MPN incurring COVID-19:
Patients were mostly elderly (median age was 69 years);
Forty-six percent of them were affected by myelofibrosis, which is the most severe MPN;
Moreover, 32% were receiving immunosuppressive therapies (JAK inhibitors, such as ruxolitinib, steroids, or immunomodulatory IMID drugs, such as thalidomide) before COVID-19.
Hospitalization was required in 54% of the patients, and the risk of being hospitalized for severe COVID-19 was independently predicted by
Older age;
Comorbidities;
Exposure to immunosuppressive therapies.
Overall, 22% of MPN patients deceased soon after COVID-19 and the risk of death was independently increased over twofold by
Older age;
Comorbidities;
Exposure to immunosuppressive therapies before the infection.
In conclusion, COVID-19 infection led to a particularly dismal outcome in MPN patients receiving immunosuppressive agents, including JAK inhibitors, or reporting multiple comorbidities. Therefore, specific preventive strategies need to be tailored for such individuals.
Keywords
Introduction
Coronavirus disease 2019 (COVID-19) is a public health emergency of international concern since February 2020. It is caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), a rapidly spreading novel coronavirus which principally causes pneumonia and respiratory failure, alongside damage to several organs by inflammatory and pro-thrombotic pathways.1,2 Age and comorbidity were reported to independently predict the fatality rate (FR) in COVID-19, and individuals affected by baseline hematological malignancies (HM) were reported to incur twofold higher mortality compared with the general population.3,4
Philadelphia-negative chronic myeloproliferative neoplasms (MPN) represent one-third of myeloid malignancies and, due to its indolent course, prevalence is high (50 per 100,000 inhabitants). 5 MPN is characterized by thrombocytosis, leukocytosis, and erythrocytosis, but they may also present with or develop cytopenias, that associate with adverse clinical outcomes. Arterial and venous thromboses are the most common complications of MPNs, due to intrinsic abnormalities of endothelial cells as well as altered platelet and leukocyte function, which may be exacerbated by infections. Of significance, the risk of viral infections is threefold higher in MPN compared with matched controls.6,7 Based on the above clinical features, MPN patients with COVID-19 are expected to have an increased risk for severe outcomes.
The present study aimed to assess clinical outcomes of COVID-19 in patients with MPN. More specifically, we aimed at testing the impact of previous therapies for MPN onto SARS-CoV-2 infection outcomes and to identify the more frail subgroups potentially requiring intensified preventive measures (e.g. application of preventive monoclonal antibodies).
Methods
In February 2020, an international cooperative registry, named EPICOVIDEHA, was initiated by the Scientific Working Group Infection in Hematology of the European Hematology Association (EHA). 8 EPICOVIDEHA is a multicenter, noninterventional, observational study approved by the local Institutional Review Board and Ethics Committee of the Fondazione Policlinico Universitario Agostino Gemelli–IRCCS, Università Cattolica del Sacro Cuore of Rome, Italy (Study ID: 3226). The corresponding local ethics committee of each participating institution may approve additionally the EPICOVIDEHA study when applicable. EPICOVIDEHA is registered at http://www.clinicaltrials.gov, with the identifier NCT 04733729.
Researchers from different countries were invited to retrospectively review all episodes of COVID-19 disease occurring in patients with blood cancers identified at their institutions from February 2020 onward. The EPICOVIDEHA electronic case report form is available at www.clinicalsurveys.net (EFS Summer 2021, TIVIAN GmbH, Cologne, Germany).
The anonymized electronic clinical report form is the single access for each participating institution and is structured in different thematic pages, as follows: (1) identification, (2) demographics, (3) underlying diseases, (4) HM, (5) COVID-19, and (6) outcome.
Cases could be registered according to the following inclusion criteria: (1) age equal to or greater than 18 years, (2) history of active HM at any stage within the last 5 years before COVID-19, and (3) report of SARS-CoV-2 positive test documented by real-time reverse transcriptase polymerase chain reaction (RT-PCR) diagnostic panels.
Qualitative demographic and clinical data were described by frequencies and percentages, while medians and interquartile ranges (IQR) were used to summarize quantitative variables. Chi-square test, Fisher’s exact test, and Mann–Whitney’s U test were used for comparisons, as appropriate. Logistic and Cox multivariable regressions allowed to identify predictors of the outcomes.
SPSS software has been employed for the descriptive and inferential statistical analyses (SPSS, version 25.0, Chicago, IL, USA).
The reporting of this study conforms to the STROBE statement (www.equator-network.org/reporting-guidelines/strobe/).
Results
Overall, 398 MPN patients with COVID-19 were registered in EPICOVIDEHA from February 2020 to March 2022 (Table 1): 113 individuals (28.4%) with essential thrombocythemia (ET), 183 (46.0%) with myelofibrosis (MF), and 102 (25.6%) with polycythemia vera (PV). Thirty individuals were newly diagnosed MPN concomitantly or within 1 month from diagnosis of SARS-CoV-2 infection. Overall, 148 patients (37.2%) had received hydroxyurea as the most recent treatment before COVID-19, while 111 patients (27.9%) had received JAK inhibitors. More specifically, 92 MF patients (50.3%) and 17 PV patients (16.7%) were receiving JAK inhibitors, while 10 patients were receiving other therapies with an immunosuppressive potential, namely immunomodulating agents (IMID), such as thalidomide or lenalidomide, or steroids. Overall, 121 patients (who did had not underwent transplantation) received therapies with an immunosuppressive potential: this corresponds to 30.4% of the overall cohort and 55.2% of the MF subgroup. Moreover, 8.3% of the overall cohort (33 patients) were refractory or resistant to ongoing therapies: the rate was lower in ET (1.8%), while it was higher in MF (13.7%). Overall, seven patients (1.7%) had received allogeneic hematopoietic stem cell transplant (HSCT): three of them were transplanted more than 6 months before COVID-19.
Baseline characteristics and outcomes according to the underlying malignancy.

COVID-19, coronavirus disease 2019; IQR, interquartile range; n, number of patients; MPN, chronic myeloproliferative neoplasms.
Males were 58.3% of the whole cohort and median age at infection was 69 years (IQR: 58–77, range: 22–97). Moreover, in 61.1% of the patients, at least one comorbidity was reported, while in 10.1%, three or more comorbidities were listed in the medical history checking seven ever major comorbidities, namely chronic cardiovascular and pulmonary diseases, renal failure, diabetes, liver diseases, obesity, and concurrent neoplasms. In particular, 144 patients (36.2%) suffered from cardiovascular diseases (e.g. atrial fibrillation, arterial hypertension, obstructive arteriopathy), 52 from chronic pulmonary disease (13.1%), 59 from diabetes (14.9%), 21 from liver disease (5.3%), 27 from obesity (6.8%), 18 from renal impairment (e.g. serum creatinine above 2 mg/dl) (4.5%). Smoking history was reported in 41 patients (10.3%).
Overall, 76 (19.1%) individuals had received at least two prior vaccination doses before COVID-19 and 37 (9.3%) had received three doses. Messenger ribonucleic acid (mRNA) vaccines were administered to 70 (17.6%) patients.
In a small portion of patients (16.6%), asymptomatic SARS-CoV-2 infection was documented, while cough or dyspnea was reported by most of the individuals (53.3%). Both extrapulmonary and pulmonary symptoms were observed in 66 individuals (21.9%) and exclusively extrapulmonary symptoms in 65 (23.4%).
Overall, 216 patients (54.3%) were admitted to hospital and the median duration of hospital stay was 14 days (IQR: 6–22, range: 1–90). Univariable logistic regression identified MPN-related and unrelated clinical factors significantly related to the chance of hospitalization (Table 2). Among MPN-related factors, MF and exposure to immunosuppressive therapies were the most important variables, while among MPN-unrelated factors, age >70 years, cardiopathy, and number of comorbidities appeared to be linked to hospitalization. In multivariable regression analysis, exposure to immunosuppressive agents remained an independent predictor of the risk of hospital admission for MPN patients with COVID-19 [odds ratio (OR): 2.186; 95% confidence interval (CI): 1.357–3.519] and so was age >70 years (OR: 2.636; 95% CI: 1.683–4.129). However, prior exposure to ruxolitinib was not by itself an independent predictor of hospital admission.
Logistic regression analysis of hospital admission.
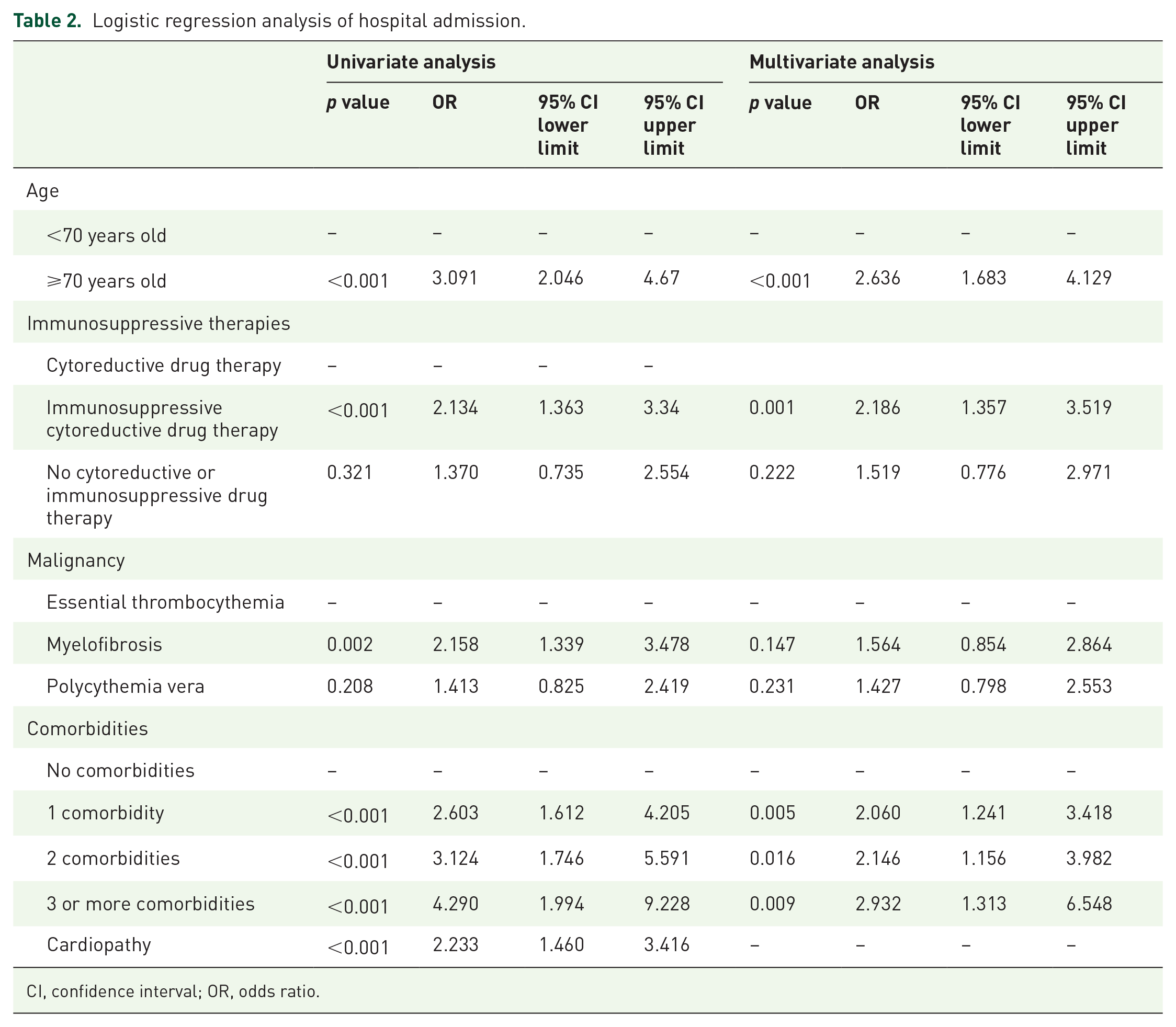
CI, confidence interval; OR, odds ratio.
Overall, 53 out of 216 hospitalized patients were admitted to intensive care unit (ICU) (24.5%) and mechanical ventilation was required for 29 patients (13.4%). The rate of admission to ICU was significantly higher in MF compared with the other MPN subtypes: 17.5% of the overall MF patients required ICU support versus 7.1% of patients with ET patients. Median length of the ICU stay was 10 days (IQR: 5–18).
MPN patients were followed for a median of 76 days (IQR: 19–197) and the reported mortality rate was 22.4%, which declined gradually over the time of the COVID-19 pandemic. During the first half of the year 2020, it was 38.9% (49 out of 126 patents), declining to 17.6% (27 out of 153) during the second half of 2020 subsequently further dropping down to 10.9% from 2021 on (p < 0.001) (Figures 1 and 2). Death occurred after a median of 14 days (IQR: 8–49) upon diagnosis of infection and was principally attributable to COVID-19 in 68 patients (76.4%) and contributable by COVID-19 in 10 (11.2%).

COVID-19 mortality rate in different time periods.

Survival curves after SARS-CoV-2 infection according to the period.
Multivariable Cox regression analysis revealed that age >70 years was an independent predictor of mortality [hazard ratio (HR): 2.191; 95% CI: 1.363–3.521], as well as comorbidity burden (Table 3). Mortality varied among MPN diseases from 13.3% in patients with ET to 31.1% in those with MF. Exposure to immunosuppressive agents before SARS-CoV-2 infection was an independent predictor of death (HR: 2.143; 95% CI: 1.369–3.354) (Table 3), as was the exposure to ruxolitinib (HR: 2.161; 95% CI: 1.381–3.354). A sub-analysis of 155 MF patients receiving active pharmacologic treatment for their disease before COVID-19 suggested that those exposed to immunosuppressive therapies might have an inferior survival compared with those receiving any other type of treatment (p = 0.059).
Cox regression analysis of fatality rate.
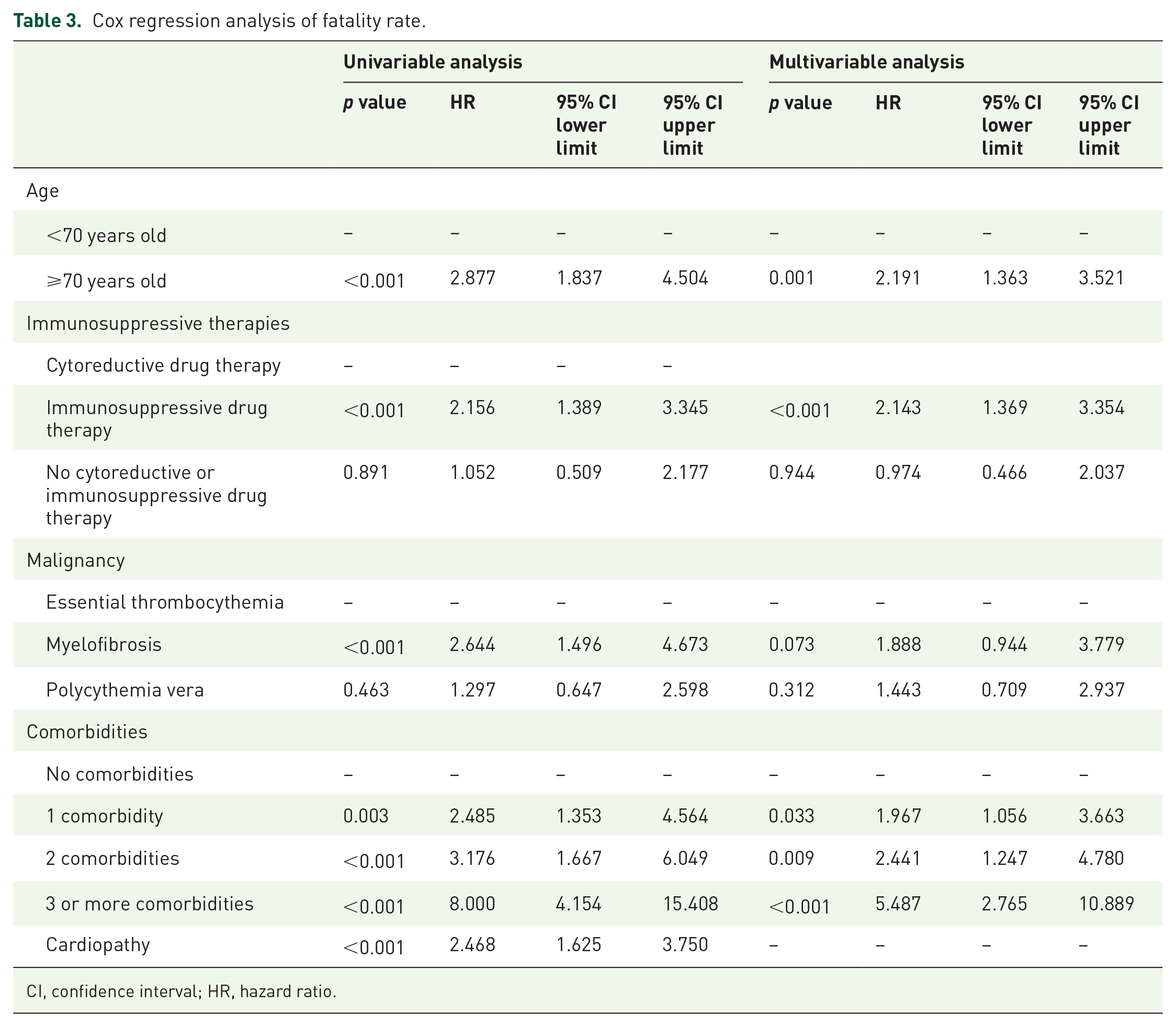
CI, confidence interval; HR, hazard ratio.
Multivariable Cox regression analysis of mortality reported similar results after including time from MPN diagnosis and COVID-19 infection wave (Table 4). Similarly, exposure to immunosuppressive drugs independently predicted a higher hospitalization rate irrespective of the infection wave and time from MPN diagnosis (Table 5).
Cox regression analysis of fatality rate including the pandemic wave and disease duration.

CI, confidence interval; COVID-19, coronavirus disease 2019; HR, hazard ratio.
Single comorbidities with statistically significant predictive value uniquely at univariate analysis included chronic cardiopathy (HR: 0.468; 95% CI: 1.625–3.750), chronic pulmonary disease (HR: 2.346; 95% CI: 1.438–3.829), diabetes mellitus (HR: 2.793; 95% CI: 1.758–4.438), liver disease (HR: 2.527; 95% CI: 1.217–5.247), and renal impairment (HR: 4.112; 95% CI: 2.178–7.763).
Logistic regression analysis of hospital admission including the pandemic wave and disease duration.

CI, confidence interval; COVID-19, coronavirus disease 2019; OR, odds ratio; HR, hazard ratio.
Statistically significant comorbidities only at univariate analysis: chronic cardiopathy (OR: 2.23; 95% CI: 1.460–3.416), chronic pulmonary disease (OR: 2.301; 95% CI: 1.218–4.347), diabetes mellitus (OR: 3.158; 95% CI: 1.671–5.968).
Discussion
MPN is a heterogeneous family of diseases particularly prone to thrombosis and bleeding, but also to infections and secondary neoplasms due to dysregulated innate and adaptive immunity. Spleen neoangiogenesis and chronic inflammation driven by MPN-specific mutations (e.g. JAK2 V617F) are hypothesized to be the underlying substrates for the above clinical manifestations.6,7,9–13 MPN phenotypes largely vary upon age at disease presentation, and therapies for MPN may specifically modulate the risk of thrombosis, infections, and secondary neoplasms. 14 Moreover, registry-based propensity-adjusted studies and two meta-analyses registered an increased risk of infections, mostly viral, in patients treated with the JAK2/2 inhibitor ruxolitinib.15–17 We, therefore, aimed to assess whether the exposure to ‘immunosuppressive’ therapies, namely JAK inhibitors, IMIDs, or steroids, might aggravate the clinical course of COVID-19 in MPN patients. As a consequence, we performed a specific query to the EPICOVIDEHA registry, which had previously reported very high admission rates to hospital (73.1%) and to ICU (18.1%) and mortality (31.2%) in the 3801 patients surveyed during the year 2020.8,18
As of 31 March 2022, overall, 398 MPN patients were reported by the EPICOVIDEHA survey: 46.0% of the patients suffered from MF, 30.4% were on immunosuppressive therapies (JAK inhibitors, steroids, or IMIDs), 47.2% were above 70 years of age, and 10.1% suffered from at least three comorbidities. Out of the overall MPN cohort, 54.3% were admitted to hospital, 13.3% were admitted to ICU, and 22.4% deceased. The outcomes of infection varied according to the underlying disease, MF being associated with the highest rates of hospitalization. 19 COVID-19 outcomes were worsened by previous exposure to immunosuppressive therapies before infection onset. Exposure to immunosuppressive agents independently increased the risk of both hospitalization (OR: 2.186; 95% CI: 1.357–3.519) and mortality (HR: 2.143; 95% CI: 1.369–3.354) and exposure to ruxolitinib was an independent predictor of mortality (HR: 2.161; 95% CI: 1.381–3.379).
JAK inhibitors have been reported to ameliorate systemic inflammation driven by SARS-CoV-2 and have even been proposed as potential therapeutic principles also for patients without MPN. 20 However, based on the broad immunosuppressive potential of JAK inhibitors (in particular, of ruxolitinib), longer exposure to ruxolitinib limits T-cell and humoral responses to BNT162b2 (Pfizer/BioNTech®) and mRNA-1273 (Moderna®) SARS-CoV-2 vaccines.21–29 Moreover, ruxolitinib discontinuation has recently reported to be associated with worse outcome in SARS-CoV-2-infected MPN patients. 19 At this first analysis of the EPICOVIDEHA MPN cohort, we could not ascertain whether worse outcomes were most attributable to interruption of immunosuppressive therapies; however, we planned to better address these issues in subsequent enquiries.
The COVID-19 vaccination campaign has led to a reduction in hospitalization rate and mortality, but risks are still high in patients with blood cancers. Further primary preventive strategies for frail patients include administration of fourth vaccination dose and long-life monoclonal agents, such as tixagevibam plus cilgavimab.29–31 While immune response to vaccination is still being ascertained in patients with HM and may vary according to the prevalent viral variant, long-life monoclonal agents in European Union are currently being restricted to patients undergoing cell therapy.31–33 This study enrolled many patients involved by the first SARS-CoV-2 waves and thus a very low percentage of patients had received at least one vaccine dose. The present report also suggests to devote intensified preventive measures to patients with MPN receiving immunosuppressive agents.
There are limitations of our study that must be acknowledged. First, MF patients are likely to be slightly overrepresented in our series, but we could not attribute this skewness to the higher severity of COVID-19 in MF patients. Second, as we included patients from different countries and centers, and diagnosed in different waves of the pandemic, a certain heterogeneity in our data is to be expected. In addition, only a small number of patients in this series were vaccinated: this is due to the fact that most data were collected during early waves, where vaccinations were not broadly available. Furthermore, SARS-CoV-2 vaccination was reported to be effective in patients with MPN; therefore, a few breakthrough infections are expected to occur. Another limitation of the study relates to data on outcome and survival, which must be interpreted carefully, since from the limited data forms, we could not ascertain any covariate that was correlated with JAK-inhibitor therapy and that might explain the inferior outcome. In particular, receiving immunosuppressive drugs may be related to advanced disease stage. Moreover, thromboembolic events after COVID-19 infection could not be accurately tracked by the study, which may hamper a definite analysis of the causes of death. Finally, abrupt interruption of, rather than prior exposure to, JAK inhibitors might explain the higher FR in hospitalized patients; however, this information was not required in the former data collection form and is currently not available.
For the above reasons, future data collection will be focused on drug–outcome relationships in a fully vaccinated population and will explore COVID-19 outcome in patients treated with JAK inhibitors, considering disease severity and therapy discontinuation among the covariates.
Conclusion
In conclusion, the present study confirmed dismal outcomes in the older MPN patients and in those with higher comorbidity burden, (i.e. cardiovascular or pulmonary disease or diabetes). In addition, to the best of our knowledge, this study is the first reporting that exposure to immunosuppressive agents before SARS-CoV-2 infection independently increased hospitalization rates and risk of death. Specific preventive strategies need, thus, to be tailored for these individuals at risk, including application of potentially protective preventive antibody cocktails as well as mindful tapering strategies in MPN patients pretreated with JAK inhibitors. Finally, therapeutic decisions in the future should also consider the abovementioned risk factors.
Footnotes
Acknowledgements
Toni Valkovič, Christian Bjørn Poulsen, Klára Piukovics, Monica Piedimonte, Irati Ormazabal-Vélez, Hans-Beier Ommen, Joseph Meletiadis, Maria Stamouli, Marina Machado, Austin Kulasekararaj, Carolina Garcia-Vidal, Nicola Fracchiolla, María Fernández-Galán, Noemí Fernández, Rita Fazi, Alessandro Busca, Marcio Nucci, Milan Navrátil, Carolina Miranda-Castillo, Nick De Jonge, Michelina Dargenio, Annarosa Cuccaro, Barbora Weinbergerová, Jens Van Praet, Zlate Stojanoski, Uluhan Sili, Guldane Cengiz Seval, Ikhwan Rinaldi, László Imre Pinczés, Jan Novák, Jorge Loureiro-Amigo, Lisset Lorenzo De La Peña, Tobias Lahmer, Jorge Labrador, Nina Khanna, María-Josefa Jiménez-Lorenzo, Anna Guidetti, Tomás-José González-López, Iker Falces-Romero, Maria Ilaria Del Principe, Cristina De Ramón, François Danion, Nicola Coppola, Maria Calbacho, Nathan Bahr, Natasha Ali, Osman Ilhan, Ramón García-Sanz, Josep Maria Ribera-Santasusana, Agostino Tafuri, Nicole García-Poutón, Sofia Zompi, Mariarita Sciumè.
