Abstract
Background:
Cord blood transplantation (CBT) has been reported as an acceptable option with comparable outcomes to conventional donors in adults with acute lymphoblastic leukemia (ALL). We aimed to analyze the long-term CBT outcomes and risk factors for early and long-term mortalities.
Methods:
Between 2006 and 2020, 112 patients (median age: 35 years; 62 Ph-negative ALL and 50 Ph-positive ALL) were treated with double CBT. Conditioning regimen consisted of total body irradiation (12 Gy) plus cytarabine (9.0 g/m2) plus fludarabine (150 mg/ m2), and graft-versus-host disease (GVHD) prophylaxis was attempted by administering tacrolimus plus mycophenolate mofetil.
Results:
The median time for neutrophil and platelet recovery was 25 days (range: 5–59 days) and 34 days (range: 7–185 days), respectively. The cumulative incidence of acute GVHD at 1 year was 43.8%, and the incidence of acute GVHD with grades III–IV was 8.9%. The overall cumulative incidence of chronic GVHD was 22.0%, and the incidence of moderate to severe chronic GVHD was 8.5%. After a median follow-up of 60.1 months (range: 5.7–181.3 months), the 5-year cumulative incidence of relapse (CIR) and nonrelapse mortality (NRM) were 15.9% and 28.5% (9.7% and 27.2% for CR1), respectively, and the 5-year overall survival (OS) was 57.9% (66.5% for CR1). In multivariate analysis of 88 patients receiving double CBT in CR1, delayed CR1 was related to high CIR, and age older than 40 years was associated with high NRM and early mortality. Unexpectedly, Ph-positive ALL with MRD had a higher NRM and early mortality than Ph-negative ALL and Ph-positive ALL without MRD subgroups, possibly due to delayed neutrophil and platelet recovery.
Conclusion:
Our data suggest that double CBT for adult ALL in CR1 has a greater benefit in younger patients and in patients with Ph-positive ALL without MRD or Ph-negative ALL.
Keywords
Introduction
Recent advances in immunochemotherapeutic agents have improved complete remission (CR) rates higher than 90% in adults with acute lymphoblastic leukemia (ALL), even in patients with relapsed or refractory ALL. However, many of them subsequently relapse, providing 35–40% of long-term disease-free survival (DFS).1–3 Allogeneic hematopoietic cell transplantation (HCT) from a human leukocyte antigen (HLA)-matched sibling donor (MSD) or matched unrelated donor (MUD) has been regarded as a primary post-remission treatment for adult ALL.4–6 However, a significant proportion of patients lack an HLA-matched donor, 7 leaving the need for alternative HCT from mismatched unrelated donor (MMUD), haploidentical donor (HID), or umbilical cord blood (CB) sources.8–12
Because the impact of donor sources on transplantation outcomes remains uncertain and may vary according to underlying disease and pre-HCT disease status, we previously analyzed the impact of donor type (MSD versus MUD versus MMUD versus CB) on long-term HCT outcomes, particularly focusing on the donor-type-specific difference in graft-versus-host disease (GVHD)-free/relapse-free survival (GRFS) in a large cohort of adults with ALL in first CR (CR1). With respect to GRFS, we found the benefit of CBT for adult ALL in CR1, which in turn may lead to an improved long-term quality of life. 13 Nevertheless, we must still consider delayed engraftment and early mortality of CBT due to lower cell counts compared to conventional donors. 14 In addition, our previous interim data on double CBT showed that patients with Philadelphia chromosome (Ph)-positive ALL had worse survival outcome than patients with Ph-negative ALL, probably due to delayed engraftment and high nonrelapse mortality (NRM). 15 Along with the subtype of ALL, the significance of immune-related parameters including HLA disparity,16–18 natural killer-cell alloreactivity,19,20 donor-specific antibodies (DSA),21,22 and noninherited maternal antigen (NIMA), 23 and several clinical factors such as age and infused CD34+ cell count were also analyzed in several studies.24,25 However, most parameters have not been proven to be influencing factors and some unclarities should be discussed.
In this study, we aimed to analyze risk factors affecting mortality and survival outcomes of double CBT in a larger number of patients with a longer follow-up period.
Patients and methods
Study population and treatment policy
From March 2006 to December 2020, 112 consecutive adults with ALL who received double CBT at a single, the largest institution in Korea were enrolled in this study. We have treated ALL patients with a uniform frontline chemotherapy and offered allogeneic HCT according to donor availability as a post-remission treatment during first CR. In relapsed or refractory ALL, allogeneic HCT was performed as soon as possible if CR was obtained after salvage therapy. We previously reported HCT outcomes according to donor type in adults with ALL in CR1, including our donor hierarchy. 13 Briefly, if an MSD is available, HCT is offered as a main post-remission therapy, irrespective of risk assessment. Patients who have no available MSD, especially those with high-risk features, are offered to receive MUD or 1-allele-MMUD transplantations. For patients without a suitable unrelated donor, CB was finally selected based on our center’s criteria. In the current study, CBT was performed in 88 patients during their CR1, 20 were in second CR (CR2, 9 of them were second transplantation), and the rest 4 were in non-CR (NR, 1 of them was second transplantation). First, we analyzed double CBT outcomes according to disease status in all patients. Then, we focused on patients treated with double CBT during CR1 for a detailed evaluation of risk factors. All patients provided written informed consent approved by the institutional review board of the Catholic University of Korea (KC17RESI0155). This study was conducted in accordance with the Declaration of Helsinki. The reporting of this study conforms to the STROBE statement (https://www.equator-network.org/).
Pre-HCT chemotherapy
As previously described,26–29 we applied modified hyper-CVAD, which consisted of cyclophosphamide, vincristine, daunorubicin, and dexamethasone (at each odd cycle). Then, patients received subsequent chemotherapy courses consisting of high-dose cytarabine and mitoxantrone (at each even cycle). Patients received up to four cycles in total, which was dependent on donor availability. In patients with Ph-positive ALL, tyrosine kinase inhibitors (imatinib or dasatinib) were added to the above chemotherapy regimens. Central nervous system prophylaxis was performed by intrathecal administration of triple agents (methotrexate, cytarabine, and hydrocortisone; six times in total). We defined patients who failed the first cycle of modified hyper-CVAD as primary refractory. For relapsed or primary refractory patients, the MEC regimen consisting of high-dose cytarabine, mitoxantrone, and etoposide has been used, and since 2016, blinatumomab has also been used. If a patient achieved CR after salvage therapies mentioned above, we defined it as delayed CR1.
Transplantation procedures
We routinely used double CBs for all patients because no single CB unit was adequate for the cell dose during the study period. For the selection of CB units, minimum HLA typing requirements followed the current practice of low-resolution typing for HLA-A and -B and high-resolution typing of HLA-DRB1 (class I at antigen level and class II at allele level). When several units were available, a 6/6 HLA-matched unit was preferred to a 5/6 HLA-matched and a 5/6 unit to a 4/6. At the same overall HLA compatibility level, the units with the higher total nucleated cell (TNC) and CD34+ cell counts were selected. HLA disparities between each unit and the recipient and between the two units were not necessarily at the same loci. Overall, we tried to match at a minimum of 4/6 loci and checked the TNC and CD34+ cell counts at least ⩾3 × 107 TNC/kg and ⩾1 × 105 CD34+ /kg. We did not routinely check donor-specific anti-HLA antibody or NIMA status. Conditioning regimen for double CBT consisted of TBI (12.0 Gy), fludarabine (150 mg/m2), and cytarabine (9.0 g/m2).13,15 Tacrolimus and mycophenolate mofetil were used for GVHD prophylaxis. The granulocyte-colony-stimulating factor was administered to all patients at a dose of 5 µg/kg/day subcutaneously from day 7 after transplantation until neutrophil recovery. Chimerism status was evaluated by short tandem repeat (STR) multiplex polymerase chain reaction (PCR) using an AmpFlSTR Identifier PCR Amplification kit (Applied Biosystems, Foster City, CA) according to the manufacturer’s instructions. GeneMapper ID Software Version 4.1 (Applied Biosystems, Foster City, CA) was used for automated genotyping and quantification of peak areas. Samples were collected at 1 and 3 months post-CBT, and complete donor chimerism was defined as the detection of >95% of donor cells (from one or both CB units). For cytomegalovirus (CMV) reactivation surveillance, we monitored real-time quantitative (RQ)-PCR for CMV DNA after neutrophil engraftment. If the CMV RQ-PCR level was higher than 1000 copies/ml (equal to >6500 IU/ml), preemptive therapy was conducted to prevent CMV disease.30,31 If residual leukemia was detected in the absence of GVHD at 3 months after double CBT, immunosuppressive agents were rapidly discontinued. For Ph-positive ALL, minimal residual disease (MRD) monitoring for the BCR-ABL1 transcript was evaluated centrally by RQ-PCR (4.5-log sensitivity) using bone marrow samples, as previously described.26–28 Based on MRD response to tyrosine kinase inhibitors before double CBT, imatinib (400 mg daily) or dasatinib (100 mg daily) were administered preemptively for 2 years to patients with detectable MRD after double CBT. Since we performed MRD testing for Ph-negative ALL from late 2020, these results were not included in this study.
Statistical analysis
The primary endpoints of this study included overall survival (OS), DFS, NRM, cumulative incidence of relapse (CIR), GRFS, and cumulative incidence of GVHD and CMV reactivation. Survival curves were plotted using the Kaplan–Meier method, and subgroups were compared by log-rank tests. Relapse, NRM, and GVHD were calculated using cumulative incidence estimates to accommodate the following competing events (death for relapse, relapse for NRM, and both death and relapse for GVHD), and subgroups were compared by Gray test. The prognostic significance of covariates affecting OS and DFS was determined by Cox proportional hazards regression model. The prognostic significance of covariates affecting cumulative incidences of relapse, NRM, and GVHD was determined using Fine-Gray proportional hazards regression for competing events. Multivariate analyses were performed using variables with a p value <0.10 in preliminary univariate analyses. All statistical analyses were performed using ‘R’ software version 4.0.3 (R Foundation for Statistical Computing, 2020). Statistical significance was set at p value <0.05.
Result
Patient characteristics
Baseline characteristics at the time of diagnosis and double CBT for all 112 patients (105 B-cell ALL and 7 T-cell ALL) are summarized in Table 1. The median patient age was 35 years (range: 18–62 years). Poor-risk karyotype was identified in 61 patients (54.5%), and among them, Ph was observed in 50 patients (44.6%). Double CBT was performed in 88 patients during CR1, while 24 were in ⩾CR2 [4 of them were in NR (3 Ph-positive ALL and 1 Ph-negative ALL)] who had a longer time interval from diagnosis to transplantation (6.5 versus 13.5 months, p < 0.001) and 10 were second transplantation. Median TNC count was 4.04 × 107 cells/kg, and 94 patients (80.6%) received ⩾3.0 × 107 TNC/kg (range: 0.33–5.85 × 107 TNC/kg). Median CD34+ cell count was 1.09 × 105 cells/kg, and 64 (64.2%) patients received ⩾1 × 105 cells/kg (range: 1.58–7.88 × 105 cells/kg).
Baseline characteristics of adult ALL patients treated with double CBT.

ALL, acute lymphoblastic leukemia; CBT, cord blood transplantation; CR, complete remission; HCT, hematopoietic cell transplantation; MRD, measurable residual disease; Ph, Philadelphia chromosome; TNC, total nucleated cells.
Engraftment, GVHD, and CMV reactivation
The cumulative incidence of neutrophil and platelet engraftment was 95.5% and 88.9%, and the median time for neutrophil and platelet recovery was 25 days (range: 5–59 days) and 34 days (range: 7–185 days), respectively. We observed that platelet count recovery was delayed in Ph-positive ALL compared to Ph-negative ALL (40 days versus 32 days, p = 0.055), while the neutrophil recovery was not different (26 days versus 25 days, p = 0.460). The chimerism status was evaluated in 99 (88.4%) patients at 1 month and in 92 (82.1%) at 3 months, respectively. Among them, complete donor chimerism was detected in 92.9% (92/99) at 1 month and 97.8% (90/92) of patients at 3 months, respectively. All patients with complete donor chimerism had a single unit predominance of >70% of donor cells. Except for 7 patients who died during aplasia, 49 developed acute GVHD (39 grade II, 7 grade III, and 3 grade IV), and the cumulative incidence of acute GVHD at 100-day was 41.1% and at 1 year was 43.8%. The cumulative incidence of acute GVHD with grades III–IV was 8.9% (Figure 1(a)). Except for patients who died within 100 days without chronic GVHD, 23 developed chronic GVHD (14 mild, 8 moderate, 1 severe), resulting in a 5-year cumulative incidence of 22.0%, and moderate to severe chronic GVHD was observed in 8.5% (Figure 1(b)). We experienced CMV reactivation in 91 (81.3%) patients, and 65 (71.4%) of them with high level of reactivation received preemptive antiviral therapy. The cumulative incidence of treated CMV reactivation was 58.4% (Figure 1(c)). However, there was no significant difference in OS according to CMV reactivation (54.7% versus 70.8%, p = 0.494; data not shown). Overall, 18 (16.1%) patients died within 100-day post-CBT – 11 were due to sepsis, 4 due to GVHD, 2 due to organ failure, and 1 due to brain hemorrhage (Figure 1(d)).

Transplantation complications and early death after double CBT. (a) Cumulative incidence of acute GVHD. (b) Cumulative incidence of chronic GVHD. (c) CMV reactivation. (d) Cumulative incidence of early death within 100 days. *Aplasia deaths without acute GVHD and early deaths or relapse within 100 days without chronic GVHD were treated as competing risks.
Survival outcomes of the entire cohort
Of a total of 112 patients, 17 relapsed at a median of 5.9 months (range: 0.1–20.5 months) after transplantation. The 5-year CIR was 12.7% (9.7% for patients in CR1% and 25.7% in CR2; p < 0.001; Figure 2(a)). Totally, 31 patients died of NRM at a median of 3.2 months (range: 0.3–26.6 months) after transplantation. The 5-year incidence of NRM was 28.5% [27.2% for patients in CR1, 40.0% in CR2, and 0.0% in NR (all died of leukemia); p = 0.218; Figure 2(b)]. After a median follow-up duration of 60.1 months (range: 5.7–181.3), 66 of the 112 patients remained alive. At the time of analysis, 46 patients had died (15 disease progression, 31 NRM). The primary causes of death were infections (n = 16), relapse (n = 15), GVHD (n = 10), organ failure (n = 4), and hemorrhage (n = 1). Five-year DFS and OS were 57.6% (63.1% for patients in CR1 and 34.3% in CR2) and 57.9% (66.5% for patients in CR1, 33.0% in CR2, and 0.0% in NR), respectively (Figure 2(c) and (d)). Five-year GRFS was 49.6% (55.5% for patients in CR1 and 24.0% in CR2; Figure 2(e)).

Survival outcomes of double CBT in the entire cohort. (a) CIR according to disease status. (b) NRM according to disease status. (c) DFS according to disease status. (d) OS according to disease status. (e) GRFS according to disease status.
Survival outcomes in CR1 subgroup
As the entire patient cohort comprised variable imbalances in disease status, previous allogeneic HCT history, and time to transplantation, we focused on patients receiving double CBT in CR1 to analyze adverse prognostic factors. In this subgroup of patients, OS (56.9% versus 74.0%) and NRM (34.8% versus 21.4%) were not significantly different between Ph-positive ALL and Ph-negative ALL (Figure 3(a) and (b)) and CIR (10.5% for Ph-positive ALL versus 9.0% for Ph-negative ALL, p = 0.718) was also similar. However, when we further analyzed considering both Ph-positivity and pre-HCT MRD status, Ph-positive ALL with MRD subgroup (n = 17) showed the worst OS and DFS (39.7% and 40.3%) compared to Ph-positive ALL without MRD (n = 22; 70.1% and 65.6%, p = 0.071 and p = 0.181, respectively) and Ph-negative ALL (n = 49; 74.0% and 69.6%, p = 0.010 and p = 0.017, respectively) subgroups (Figure 3(c) and (d)). In Figure 3(e) and (f), we identified that poor survival outcome was due to higher NRM of Ph-positive ALL with MRD subgroup compared to other subgroups (47.8% versus 24.8% and 21.4%, p = 0.047) rather than CIR (11.8% versus 9.4% versus 9.0%, p = 0.916). We identified early death rate within 100 days post-CBT was significantly higher in Ph-positive ALL with MRD subgroup than other subgroups (35.3% versus 9.1% versus 4.1%, p = 0.001). In multivariate analysis (Table 2), the early death rate was significantly higher in patients older than 40 years (HR = 22.6, 95% CI; 2.8–178.2, p = 0.003) and Ph-positive ALL with MRD subgroup (HR = 13.6, 95% CI; 2.9–64.3, p < 0.001), and the overall NRM was also higher in patients older than 40 years (HR = 6.72, 95% CI; 2.6–17.0, p < 0.001) and Ph-positive ALL with MRD subgroup (HR = 4.69, 95% CI; 1.9–11.2, p < 0.001). Delayed CR1 was the most adverse risk factor affecting CIR (HR = 9.59, 95% CI; 2.5–35.9, p < 0.001). For survival outcomes, all factors discussed above (old age, delayed CR, and Ph-positive ALL with MRD subgroup) were significant for both OS and DFS. Age older than 40 years (HR = 1.89, 95% CI; 1.1–3.2, p = 0.016) was associated with a poorer GRFS.

Survival outcomes in patients receiving double CBT in CR1 according to Ph-positivity and pre-CBT MRD status. (a) OS of patients with Ph-positive ALL and Ph-negative ALL. (b) NRM of patients with Ph-positive ALL and Ph-negative ALL. (c) OS according to Ph-positivity and MRD status. (d) DFS according to Ph-positivity and MRD status. (e) CIR according to Ph-positivity and MRD status. (f) NRM according to Ph-positivity and MRD status.
Uni- and multivariate analyses in adult ALL patients treated with double CBT in CR1.
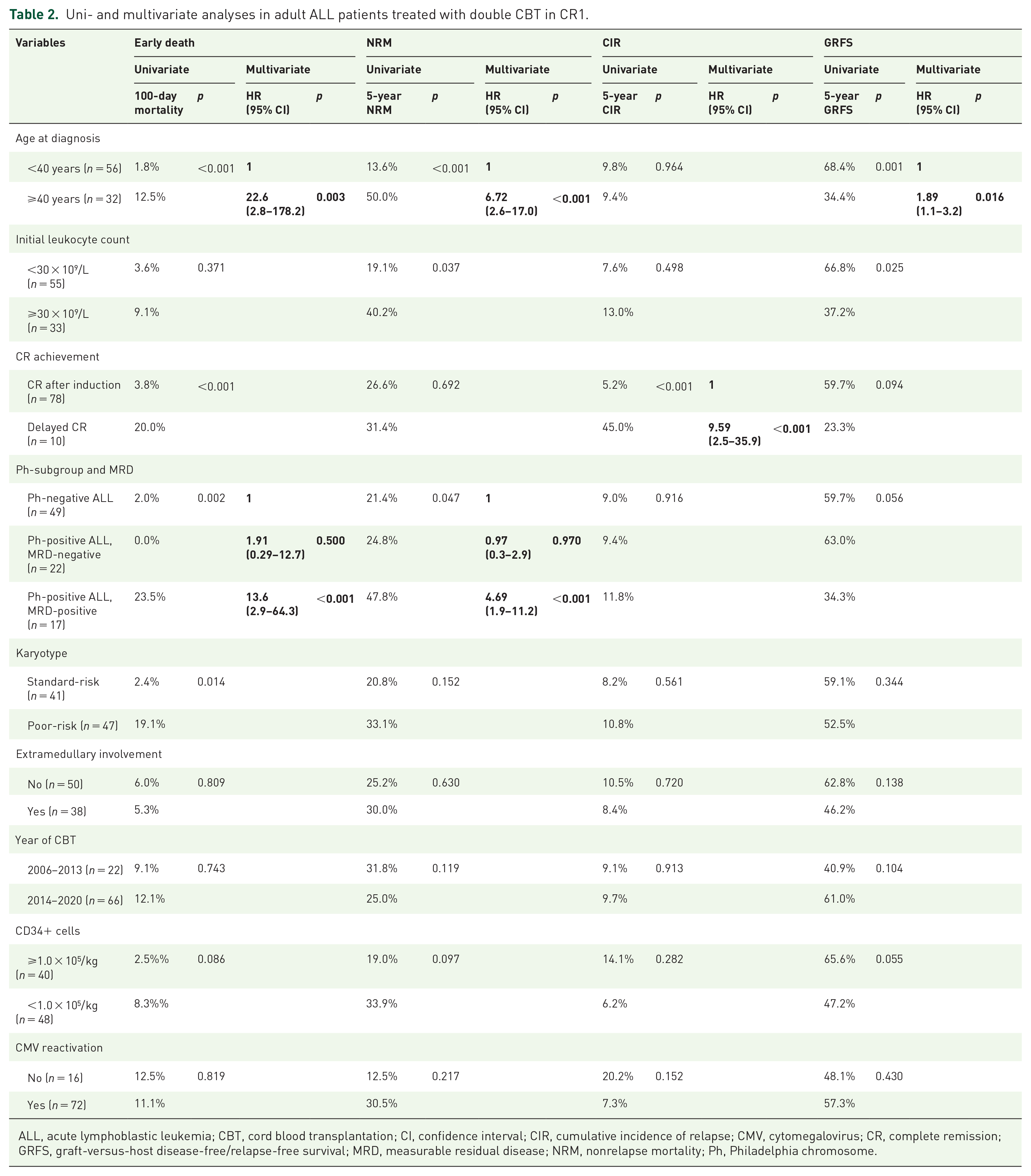
ALL, acute lymphoblastic leukemia; CBT, cord blood transplantation; CI, confidence interval; CIR, cumulative incidence of relapse; CMV, cytomegalovirus; CR, complete remission; GRFS, graft-versus-host disease-free/relapse-free survival; MRD, measurable residual disease; NRM, nonrelapse mortality; Ph, Philadelphia chromosome.
Discussion
CB is a readily available donor source for patients who do not have a suitable conventional donor, but we still have to consider a delayed engraftment and early fatality.13,14 We have been using double CBs as a primary alternative donor source for over a decade, and as our experience with its pros and cons has accumulated, we clearly felt the need to identify better CBT candidates. Our previous interim data on double CBT showed that patients with Ph-positive ALL had a poorer DFS than patients with Ph-negative ALL (37.7% versus 66.5%, p = 0.017), with trends of higher CIR and NRM. 15 However, in this study with a larger number of patients and more extended follow-up, Ph-positive ALL still showed poorer outcomes than Ph-negative ALL, but the difference was not statistically significant. However, when we further analyzed considering both Ph-positivity and MRD status before CBT, Ph-positive ALL with MRD subgroup showed significantly worse survival outcomes due to significantly higher NRM than Ph-negative ALL or Ph-positive ALL without MRD subgroups. In this Ph-positive ALL with MRD subgroup, the early death rate within 100 days post-CBT was exceptionally high, accounting for 75.0% of all NRM cases, resulting in a poorer OS than other subgroups. Overall, irrespective of disease subgroup, age older than 40 years was an important factor in high NRM and early mortality.
For both Ph-positive and Ph-negative ALL, MRD has been proven as one of the most important factors for predicting survival outcome after allogeneic HCT.27–29,32–34 Pre-transplantation positive MRD is generally supposed to be related to subsequent relapse rather than NRM.35,36 However, our data showed that NRM, especially early mortality, were significantly higher in Ph-positive ALL with MRD subgroup after double CBT. As a result, the long-term CIR was not substantially higher than Ph-positive ALL without MRD or Ph-negative ALL subgroups. These were unexpected findings and might be a coincidence. However, as a possible reason for frequent early deaths in Ph-positive ALL with MRD subgroup, we suggest that delayed neutrophil (29 days versus 24 days, p = 0.038) and platelet (42 days versus 37 days, p = 0.056) recovery may increase fatal infectious or hemorrhagic complications, possibly due to myeloablative conditioning regimen (Supplementary Table 1). A recent study found a higher incidence of bloodstream infection after CBT, which was more severe in patients with delayed neutrophil engraftment, TBI conditioning, and infection with multidrug resistance. They also showed that NR or advanced disease status before CBT was at high-risk of blood stream infection (37.9% versus 24.1%), although it was offset in the multivariate analysis. 37
Vice versa, many early deaths may offset possible candidates for subsequent relapse, resulting in a similar overall CIR between Ph-positive ALL with MRD subgroup and other subgroups in our study. Recently, Milano et al. examined the effect of MRD on mortality and relapse between the donor sources (CB versus MUD versus MMUD). 38 The authors showed that the risk of relapse was significantly lower in the CBT group than in either MUD or MMUD groups, while no significant difference in NRM was found between the three groups. Interestingly, pre-HCT MRD status had a significant impact on risks of death and relapse among patients receiving MUD- or MMUD-HCT but no impact among patients receiving CBT. These important results should be confirmed in future studies. Moreover, we need solutions to reduce early mortality in high-risk patients, such as older patients or those with MRD/advanced disease status, by modulating conditioning intensity or using other alternative donors such as HID.
CBT has a high-risk for CMV reactivation due to slow CMV-specific immune reconstitution. 39 CMV seropositivity is frequent in the Korean population, and post-CBT CMV reactivation is confirmed in many patients. 40 Our data showed that CMV reactivation was observed in 81.3% and about 70% of them were treated with antiviral agents. Because antiviral agents suppress BM function, active preemptive treatment of CMV in the early post-CBT period may be harmful to hematological stability after CBT. Nevertheless, survival outcomes were not significantly different according to CMV reactivation.
Several studies have reported that single CBT showed more frequent engraftment failure,12,41 while double CBT showed more frequent acute GVHD or extensive chronic GVHD.42–46 Along with the high incidence of GVHD, an enhanced GVL effect was also observed after double CBT, and one study found a significantly lower incidence of relapse with TBI-based conditioning regimen without antithymocyte globulin. 45 In this study, all patients were treated with double CBT after TBI-based myeloablative conditioning without antithymocyte globulin. Our data showed that the cumulative incidence of grade III-IV acute GVHD and moderate to severe chronic GVHD was less than 10% each, indicating a relatively lower incidence of clinically significant GVHD compared to other studies.42–46 In addition, most acute or chronic GVHD could be managed safely, and only a small number of GVHD-related deaths occurred. The reasons for the low incidence of grade III-IV acute GVHD in this study are not clear, but it is likely due to the relatively lower number of infused cells compared to other studies. Indeed, despite the use of double CBs for all patients in this study, the median number of TNC was 4.04 × 107/kg, 1.09 × 105/kg for CD34+ cells, and 5.66 × 106/kg for CD3+ cells, which were lower than those in other reports.43–46 Another possible cause is that the high early mortality within 100 days post-CBT may have partially masked the true incidence of acute GVHD.
Several comparative studies showed no significant difference in survival outcome between single CBT and double CBT if a single unit had sufficient cell dose and HLA compatibility.45–47 Although our previous interim data showed that a lower CD34+ cell number was associated with a higher NRM after double CBT, 15 it was not significant in this final analysis. This might be due to our appropriate CB selection criteria. However, we could not compare the results of double CBT with single CBT as no single CB unit met the minimum cell dose criteria during the study period in Korea.
In summary, our data suggest that double CBT for adult ALL in CR1 has a greater benefit in younger patients and in patients with Ph-positive ALL without MRD or Ph-negative ALL. However, delayed engraftment and high risk of early mortality are important issues to be addressed. In addition, our data opened a discussion point for Ph-positive ALL with MRD subgroup at high risk of overall NRM and early mortality. Our study has the limitation that it cannot fully address the effect of MRD status on CBT outcomes because MRD data were analyzed only for Ph-positive ALL. To address the importance of MRD status for both NRM and relapse in the CBT setting for adult ALL, our results should be further evaluated with thorough MRD monitoring.
Supplemental Material
sj-docx-1-tah-10.1177_20406207221076762 – Supplemental material for Durable outcomes of double cord blood transplantation in adults with acute lymphoblastic leukemia: high-risk features for early and long-term mortality
Supplemental material, sj-docx-1-tah-10.1177_20406207221076762 for Durable outcomes of double cord blood transplantation in adults with acute lymphoblastic leukemia: high-risk features for early and long-term mortality by Jae-Ho Yoon, Gi June Min, Sung-Soo Park, Silvia Park, Sung-Eun Lee, Byung-Sik Cho, Ki-Seong Eom, Yoo-Jin Kim, Hee-Je Kim, Chang-Ki Min, Seok-Goo Cho, Jong Wook Lee and Seok Lee in Therapeutic Advances in Hematology
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics,consent,and permissions
All patients were provided written informed consent. This research was permitted and conducted following the Institutional Review Board and Ethics Committee guidelines of the Catholic Medical Center (KC17RESI0155).
Consent for publication
Written informed consent was obtained at the stage of diagnosis in all patients. This manuscript does not contain any individual’s personal data.
Availability of data and materials
All data generated or analyzed during this study are included in this published article. Datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
