Abstract
Primary mediastinal large B-cell lymphoma (PMBCL) is a rare hematologic malignancy with distinct clinical and immunopathological features. We report a case of a young male with disease refractory to multiple lines of therapy, including chimeric antigen receptor-T cells, who achieved his first complete remission after haploidentical bone marrow transplantation (haplo-BMT), following donor leukocyte infusions (DLIs) given concurrently with blinatumomab. While DLI has been used after T-replete haplo-BMT with post-transplant cyclophosphamide, there are no reports on its use for PMBCL. Similarly, blinatumomab is active against B-cell lymphomas, but literature is lacking in patients with PMBCL. Our experience illustrates that blinatumomab can be used concurrently with DLI in a haploidentical setting to achieve disease response in PMBCL. Despite our encouraging experience with this case, we would not recommend this approach outside of a clinical trial as blinatumomab may exacerbate the graft versus host disease risks of DLI, especially in a haploidentical setting. Evaluating this treatment combination in high-risk patients in the setting of a clinical trial may be meaningful.
Introduction
Primary mediastinal large B-cell lymphoma (PMBCL) is a rare hematologic malignancy representing only 2–3% of non-Hodgkin lymphomas (NHLs). 1 It has distinct clinical and immunopathological features intermediate to diffuse large B-cell (DLBCL) and Hodgkin lymphomas. It tends to occur in younger patients and is characterized by bulky, locally invasive disease that can infiltrate the lungs, pleura, chest wall and pericardium, often compromising the airway and venous blood flow. While prognosis is generally favorable, especially for younger individuals, those who relapse have a dramatically lower chance of survival. Autologous and, less frequently, allogeneic hematopoietic cell transplantations (HCTs) have been used for patients with relapsed/refractory disease with mixed results. 2 We report a case of a young adult male with disease refractory to multiple lines of therapy, including chimeric antigen receptor-T cells (CAR-T cells), who achieved his first complete remission after haploidentical bone marrow transplantation (BMT), following donor leukocyte infusions (DLIs) given concurrently with blinatumomab, a bispecific T-cell engaging antibody.
Case presentation
A 21-year-old male presented with left cervical swelling and pain over his clavicle. A computed tomography (CT) of his chest and abdomen was obtained, showing a large anterior mediastinal mass. Pathologic and immunohistochemical examination showed fibrotic tissue with nests of large CD19+, CD20+, CD23+, CD30+, CD45+, CD3–, CD5–, CD10–, CD15–, BCL6+, MUM1+, PAX5+, B-cells with loss of surface immunoglobulin expression and high expression of PD-L1 and a Ki-67 proliferative index of 90%, confirming the diagnosis of PMBCL, non-germinal center type. The cells were negative for rearrangement of MYC, BCL2 and BCL6 translocation by fluorescence in situ hybridization.
Upon diagnosis, he received six cycles of dose adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin and rituximab (DA-EPOCH-R). 3 An end of treatment positron emission tomography (PET)-CT scan demonstrated refractory disease, necessitating involved field irradiation. A follow-up PET-CT scan revealed extension of disease below the diaphragm [Figure 1(a)]. The patient then received salvage chemotherapy of ifosfamide, carboplatin, etoposide and rituximab, but his disease progressed. A cycle of rituximab, gemcitabine, navelbine and doxorubicin was given for debulking and pembrolizumab was initiated with an early response [Figure 1(b)]. However, his disease again demonstrated progression following eight cycles of pembrolizumab [Figure 1(c)]. A repeat biopsy demonstrated expression of CD30 in a diffuse and strong membranous fashion, leading to treatment with brentuximab vedotin, resulting in a partial response (PR) [Figure 1(d)]. He then received an infusion of CAR-T cells (tisagenlecleucel), but his disease progressed [Figure 1(e)]. Subsequent treatments included bendamustine and brentuximab followed by gemcitabine, cisplatin and dexamethasone, again yielding a very good PR [Figure 1(f)].
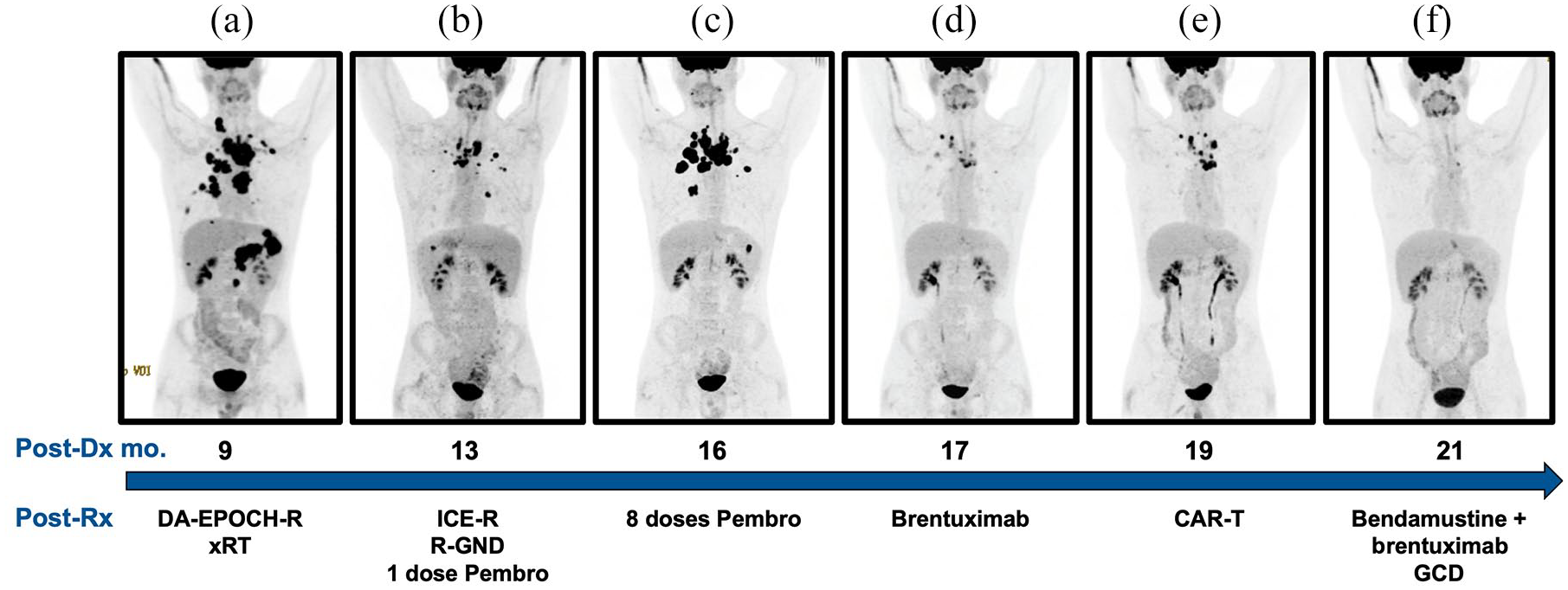
Responses to treatment before haploidentical bone marrow transplantation. Shown are positron emission tomography computed tomography images performed after each respective treatment (Post-Rx). Post-Dx mo. indicates months after initial diagnosis. (a) Extension of disease below the diaphragm following dose adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin and rituximab (DA-EPOCH-R) and involved field irradiation (xRT); (b) interim response after receiving ifosfamide, carboplatin, etoposide and rituximab (ICE-R), then navelbine and doxorubicin (R-GND) followed by first dose of pembrolizumab (Pembro); (c) progression of disease after eight doses of pembrolizumab; (d) partial response after brentuximab vedotin; (e) failure to respond to anti-CD19 chimeric antigen receptor-T (CAR-T) cell therapy; (f) very good partial response after bendamustine and brentuximab followed by gemcitabine, cisplatin and dexamethasone (GCD).
Having completed nine failed radiochemoimmunotherapy regimens, he consented to a T-replete haploidentical bone marrow transplantation (haplo-BMT) with his brother as the donor. He was conditioned with a myeloablative regimen of busulfan, fludarabine and melphalan with post-transplant cyclophosphamide (PT-CY).4–6 His post-BMT course was complicated by enterococcal bacteremia with meningitis on day +13, requiring 6 weeks of intravenous antibiotics. He showed no clinical signs of acute graft-versus-host disease (aGvHD) despite maintaining low tacrolimus levels (4–6 ng/ml) during the first month post-BMT. Early tacrolimus taper was started on day +33 with discontinuation on day +40. On day +44, a suprasternal chest wall mass emerged and a repeat biopsy on day +47 confirmed PMBCL. A PET-CT scan on day +49 demonstrated interval progression of disease [Figure 2(a)].

Responses to treatment after haploidentical bone marrow transplantation (haplo-BMT). Shown are positron emission tomography computed tomography images performed after each respective treatment (Post-BMT). Post-Dx mo. indicates months after initial diagnosis. (a) Disease progression after haplo-BMT; (b) mixed response with decreased mediastinal and chest wall FDG avidity but new focal subpleural avid areas, new mildly avid pulmonary nodules and a new focal lesion in the hepatic dome during second cycle of blinatumomab (Blina) and following donor leukocyte infusion (DLI) but before progression to grade III graft-versus-host disease (GvHD); (c) sustained tumor response with intense FDG uptake throughout the small and large colon compatible with intestinal GvHD while on tapering doses of prednisone (PRED) and cyclosporine A (CSA) ; (d) complete metabolic response with less intestinal fluorodeoxyglucose (FDG) uptake but evidence of esophangitis and gastritis after discontinuing immunosuppression; (e) disease recurrence after stopping blinatumomab and resolution of GvHD; (f) complete metabolic response.
Blinatumomab infusion was initiated on day +50 at a dose of 10 µg/m2 per day, escalated to 15 µg/m2 per day on day +52. While on blinatumomab (day +54), the patient received a haploidentical DLI from the same donor at a dose of 3 × 106 CD3+ cells/kg. His chest wall mass continued to expand with intensifying pain, so, 2 weeks later, he was given a second DLI infusion consisting of 1 × 107 CD3+ cells/kg (day +68). On day +78, the patient developed a stage I aGvHD skin rash, and, upon exam, his chest wall mass was noticeably smaller. aGvHD progressed to stage II skin on day +82 and he started topical steroids. A repeat PET-CT scan on day +85 showed a mixed response with decreased mediastinal and chest wall fluorodeoxyglucose (FDG) avidity but new focal subpleural avid areas, new mildly avid pulmonary nodules and a new focal lesion in the hepatic dome [Figure 2(b)]. On day +89, his aGvHD had progressed to stage III with vomiting, diarrhea and weight loss. Therefore, prednisone 2 mg/kg and cyclosporine were started. He developed pancytopenia and hypoalbunemia. He was readmitted to the hospital on day +92, as he was not tolerating oral medications and had developed significant diarrhea (stage III) with overall grade III aGvHD. His stool was negative for c-difficile and for other gastrointestinal (GI) pathogens. With progression of GvHD his chest mass and lymphadenopathy decreased. A repeat PET-CT scan on day +113 confirmed a sustained tumor response and revealed intense FDG uptake throughout the small and large colon compatible with intestinal GvHD [Figure 2(c)]. After initial improvement of skin and GI aGvHD symptoms cyclosporine was decreased in order to optimize graft-versus-lymphoma (GvL) effects. Drug levels were kept subtherapeutic (mean of 50 ng/ml) from day +110 until day +150, at which time it was discontinued. Likewise, prednisone was progressively tapered to 1 mg/kg by day +120 and discontinued on day +160. The patient’s pancytopenia and hypoalbunemia resolved. Moreover, no palpable masses or lymphadenopathy were appreciable by physical examination. A repeat PET-CT scan was performed on day +167, after four cycles of blinatumomab, which demostrated, for the first time since his diagnosis, a complete metabolic response of his PMBCL with less intestinal FDG uptake due to aGvHD [Figure 2(d)]. Skin GvHD worsened and prednisone was restarted at 20 mg daily on day +197. On +218 the patient noted increasing lympadenopthy and repeat PET-CT confirmed extensive relapse of his PMBCL [Figure 2(e)]. An additional 4 week course of blinatumomab was then given between days +219 and +246 at double the previous dose of 30 µg/m2 per day but without evidence of clinical improvement as an enlarging chest wall mass and lymphadenopathy was evident. The patient was transferred to hospice care where all prescribed medications including prednisone were discontinued. A month later, as his overall condition was improving, he discharged himself from hospice while seeking care at another institution. A PET scan was done on day +293, which, remarkably, again demonstrated complete resolution of disease [Figure 2(f)]. He is now day +386 following his haplo-BMT with no evidence of disease recurrence. He is receiving prednisone and ruxolitinib for chronic GvHD with skin involvement, contractures, ocular manifestations and pulmonary involvement.
Discussion
DA-EPOCH-R has been used frequently as front-line treatment of PMBCL, but, due to the rarity of the disease, there is no consensus on optimal consolidation or treatment for relapsed/refractory disease. Many of the agents our patient received have been used previously for relapsed/refractory PMBCL.3,7,8 A study from Italy evaluated pembrolizumab in 17 patients with relapsed/refractory PMBCL and found that two achieved a complete response (CR) and five a PR. 9 The same investigator reported on the use of brentuximab in 15 patients with PMBCL and found that only two patients achieved a PR, as was the case with our patient. 10 In contrast to our patient who failed to respond to anti-CD19 CAR-T cell therapy, there is a single recent report of a patient with PMBCL who achieved a CR. 11 It is also important to note that Food and Drug Administration approval for tisagenlecleucel is for DLBCL and not PMBCL; however, it was decided to treat our patient due to lack of other promising options.
Autologous HCT has traditionally been used as salvage therapy for NHL, although allogeneic HCTs have been utilized by some centers. Our patient did not undergo an autologous HCT as he did not have chemotherapy sensitive disease. A recent four-institution retrospective analysis reviewed the outcomes of 28 patients with relapsed/refractory PMBCL who received allogeneic HCT (54% matched sibling, 29% matched unrelated and 18% mismatched unrelated or cord blood). 2 Eighty percent of patients were sensitive to pre-transplant therapy and 86% received reduced intensity conditioning. The 5-year progression-free and overall survivals were 34% and 45%, respectively. None of the patients received prior PD-1 blockade or CAR-T cells and none received haploidentical HCT. Our case, therefore, is unique in that our patient previously received PD-1 blockade, CAR-T cells followed by a haplo-BMT. Despite having been treated with a PD-1 inhibitor before his transplant, receiving myeloablative conditioning and having his tacrolimus levels purposefully maintained in the low therapeutic range and discontinued by day +40, the patient did not develop aGvHD. Recent studies have documented that PT-CY lowers the risk of GvHD in patients that have received checkpoint inhibitors. 12
DLI has been used after T-replete haplo-BMT with PT-CY against relapsed leukemia and NHL, but to our knowledge there are no reports employing DLI for PMBCL. 13 Blinatumomab has shown activity against other B-cell lymphomas, but there are no published reports in patients with PMBCL. 14 The most effective dose of blinatumomab for NHL is 4-fold higher (60 µg/m2 per day) than what our patient received as his dose was restricted due to ongoing peripheral neuropathy and persistent headaches. There is a single case report on concomitant use of DLI with blinatumomab following a matched unrelated donor transplant for leukemia but no reports in haploidentical HCT. 15 While our experience illustrates that blinatumomab can be used concurrently with DLI in a haploidentical setting, the relative contribution of DLI versus blinatumomab in achieving a metabolic CR was not clear. Our patient relapsed after discontinuing blinatumomab; however, he had also restarted prednisone because of worsening GvHD symptoms therefore GvL activity was likely suppressed. His chest wall mass and lymphadenopathy continued to increase even during the fifth cycle of blinatumomab, despite receiving a higher dose. One cannot rule out rule out CD19 antigen escape since a repeat biopsy was not performed. Abruptly discontinuing prednisone during hospice led to worsening of chronic GvHD while evidently inducing another remission.
Despite our encouraging experience with this case, we would not recommend this strategy outside a clinical trial as blinatumomab may exacerbate the GvHD risks of DLI especially in a haploidentical setting. Formally studying this treatment combination in high-risk patients may be meaningful.
Footnotes
Acknowledgements
We most importantly would like to express gratitude to our patient for allowing us to report his case. The authors would also like to thank the inpatient and outpatient nursing and other staff on the pediatric HCTT unit at Banner University Medical Center in Tucson, AZ for their outstanding care of our patient.
Author contributions
J.S. reviewed the clinical records and wrote the manuscript. A.K. edited the manuscript and treated the patient. N.A.S. managed the patient and edited the manuscript. E.K. co-wrote the manuscript and managed the patient during his cell therapies and transplantation.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics and Informed Consent Statement
The data in this case report falls under our University of Arizona IRB board, under protocol number 2007806899A001. Written consent was not obtained, as this board waived the consent requirement for the following reasons: the use or disclosure of protected health information involves no more than minimal risk to the individuals; the research could not practicably be conducted without the alteration or waiver; the research could not practicably be conducted without access to and use of the protected health information; there is an adequate plan to protect the identifiers from improper use and disclosure; there is an adequate plan to destroy the identifiers at the earliest opportunity consistent with conduct of the research, unless there is a health or research justification for retaining the identifiers, or such retention is otherwise required by law; and there are adequate written assurances that the protected health information will not be reused or disclosed to any other person or entity, except as required by law, for authorized oversight of the research project, or for other research for which the use or disclosure of protected health information would be permitted by this subpart.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
