Abstract
Background:
The phase II, exploratory, open-label Exploring Nilotinib BCR-ABL Effects (ENABL) study [ClinicalTrials.gov identifier: NCT00644878] assessed the impact of switching to nilotinib therapy in patients with chronic myeloid leukemia in chronic phase (CML-CP) who had a suboptimal molecular response with imatinib.
Methods:
Patients with CML-CP who had previously achieved a complete cytogenetic response (CCyR), but had a suboptimal molecular response, with frontline imatinib therapy (N = 18) were assigned to receive nilotinib 300 mg twice daily. The primary endpoint was the change in BCR-ABL1 transcript levels from baseline after 12 months; rates of major molecular response (MMR) and safety were also assessed.
Results:
At 3 months after switching to nilotinib, 10 of 17 (59%) evaluable patients had achieved MMR. At 12 months, 9 of 12 (75%) evaluable patients had achieved MMR, and the median BCR-ABL1 level among all patients remaining in the study was 0.020% on the International Scale (IS), equivalent to a 3.7-log reduction from the standardized IS baseline (primary endpoint). Adverse events (AEs) were typically grade 1/2 and manageable with dose interruptions. A total of three patients experienced serious study drug-related AEs, including pancreatitis, bradycardia, and vertigo. No deaths were reported.
Conclusions:
Overall, results from this exploratory study suggest that switching to nilotinib due to suboptimal molecular response with imatinib can result in improved molecular response for patients with CML-CP.
Introduction
Imatinib was the first BCR-ABL1 tyrosine kinase inhibitor (TKI) approved for the treatment of patients with newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia in chronic phase (CML-CP), as well as for patients with Ph+ CML in accelerated phase (AP) or blast crisis (BC) after failure of interferon-α therapy. For most patients with CML-CP, frontline therapy with imatinib is effective for inducing a complete cytogenetic response (CCyR) [O’Brien et al. 2003]. However, many patients treated with imatinib do not meet the molecular response targets designated by the National Comprehensive Cancer Network (NCCN) and the European LeukemiaNet (ELN) [National Comprehensive Cancer Network, 2016; Baccarani et al. 2013]. Failure to achieve an early molecular response at 3 months following initiation of TKI treatment, which is a criterion for considering a switch in therapy according to the NCCN guidelines [National Comprehensive Cancer Network, 2016], is associated with poorer clinical outcomes in patients with CML-CP [Marin et al. 2012]. In patients treated with TKI therapy for ⩾12 months, the ELN considers an optimal response as achievement of major molecular response (MMR), defined as BCR-ABL1 ⩽0.1% on the International Scale [IS (BCR-ABL1IS); equivalent to a 3-log reduction from the standardized IS baseline]. Achievement of MMR at 12 months is associated with favorable long-term outcomes and a lower rate of disease progression compared with lack of MMR at 12 months [Hughes et al. 2010]. The ELN considers patients with a response level equivalent to CCyR without MMR (BCR-ABL1IS >0.1–1%) at 12 months following initiation of frontline TKI therapy to be in a ‘warning zone,’ indicating that they may be at risk for treatment failure [Baccarani et al. 2013].
Among patients with CCyR at 12 months in the International Randomized Study of Interferon Versus STI571 (IRIS) trial of frontline imatinib, >30% did not achieve MMR at 12 months [Hughes et al. 2010]. For patients without MMR after ⩾12 months of frontline imatinib therapy, a change in treatment may lead to improved long-term outcomes. However, limited data are available regarding the impact of switching therapy for this subset of patients. There is also evidence that a rising BCR-ABL1 transcript level is an early sign of disease relapse and may predict emergence of BCR-ABL1 mutations and loss of response [Wang et al. 2006; Press et al. 2009]. Additional investigation is needed to determine whether switching therapy due to increasing BCR-ABL1 transcript levels leads to improved patient outcomes.
Nilotinib is a highly selective BCR-ABL1 TKI approved for the treatment of patients with newly diagnosed CML-CP or imatinib-resistant or imatinib-intolerant CML-CP or CML-AP. With 5 years of follow up in the Evaluating Nilotinib Efficacy and Safety in Clinical Trials–Newly Diagnosed Patients (ENESTnd) study, frontline nilotinib resulted in higher response rates and a lower risk of disease progression versus frontline imatinib for patients with Ph+ CML-CP [Hochhaus et al. 2016]. Frontline nilotinib has also been associated with a lower rate of discontinuation due to suboptimal response or treatment failure than frontline imatinib [Larson et al. 2012]. In addition, nilotinib has shown efficacy as second-line therapy in studies of patients with suboptimal response, treatment failure, resistance, or intolerance to imatinib [Hughes et al. 2014a, 2014b; Goh et al. 2011; Kantarjian et al. 2011]. With 24 months of follow up in a trial of second-line nilotinib in patients with imatinib resistance or intolerance, approximately 40% of patients achieved CCyR and 28% of patients achieved MMR (including 38% of those with complete hematologic response at baseline) [Kantarjian et al. 2011].
Given the superior potency of nilotinib compared with imatinib, we hypothesized that switching to nilotinib could result in improved outcomes for patients with suboptimal molecular response on imatinib. Although treatment guidelines have been updated over time to highlight the importance of molecular responses and monitoring, and to reflect the availability of multiple treatment options for patients with CML-CP, criteria for switching therapy due to failure to achieve MMR or rising BCR-ABL1 transcript levels have not been established. The Exploring Nilotinib BCR-ABL Effects (ENABL) study was designed to assess whether patients with CML-CP treated with imatinib who achieved CCyR and either did not achieve MMR (⩾1 year of imatinib treatment required) or experienced a >1-log increase in BCR-ABL1 transcript levels (regardless of the duration of imatinib treatment) could achieve further BCR-ABL1 reductions by switching to nilotinib.
Materials and methods
Study design and patients
ENABL was a phase II, exploratory, open-label, 12-center study conducted in the United States [ClinicalTrials.gov identifier: NCT00644878] [US National Institutes of Health, 2015]. Institutional review board or independent ethics committee approval was obtained at each participating study center. The study was conducted in accordance with the Declaration of Helsinki and local applicable laws and regulations. All patients provided written informed consent prior to their participation in the study. The original protocol intended to enroll 160 patients (80 patients per group). However, the study was terminated early due to a high screen failure rate leading to slow enrollment over approximately 3 years. Due to the small sample size, the planned analyses could not be performed.
Eligible male and female patients with Ph+ CML-CP were aged ⩾18 years, were receiving frontline therapy with imatinib, and had an Eastern Cooperative Oncology Group (ECOG) performance status of ⩽2. Prior to enrollment, all patients were required to have achieved CCyR and suboptimal molecular response [defined as either lack of MMR during treatment with imatinib for ⩾1 year (group 1) or a >1-log increase in BCR-ABL1 transcript level from the best response achieved, regardless of the duration of imatinib treatment (group 2), based on real-time quantitative polymerase chain reaction (RQ-PCR) assessments prior to study entry] while receiving imatinib ⩾400 mg daily. Patients in group 1 were stratified at screening as either plateau PCR (defined as ⩾2 PCR samples performed ⩾1 month apart with no >1-log changes in BCR-ABL1 transcript levels) or falling PCR (defined as ⩾2 PCR samples with >1-log cumulative reduction in BCR-ABL1 transcript levels). For enrollment in group 2, patients were required to have a rising PCR trend at screening, defined as a cumulative >1-log increase in BCR-ABL1 transcript levels with ⩾2 PCR samples performed within 6 months prior to screening. Patients with prior imatinib dose reductions were eligible for enrollment if the minimum imatinib dose received was 300 mg daily and the duration of the dose reduction was ⩽28 days; for enrollment in group 1, patients must have been receiving imatinib 400 mg daily for ⩾6 consecutive months prior to screening.
Patients with prior CML-AP or CML-BC, previously documented T315I mutations, or prior treatment with a TKI other than imatinib were excluded. Patients who achieved prior CCyR on imatinib and had a confirmed loss of a CCyR prior to study entry were also excluded. Other exclusion criteria included impaired gastrointestinal function that could alter study drug absorption, history of chronic pancreatitis or acute pancreatitis within 1 year of study, impaired cardiac function [including congenital or family history of long QT syndrome, Fridericia-corrected QT interval (QTcF) >450 mg on screening electrocardiogram (ECG), past or current ventricular or atrial tachyarrhythmias, bradycardia, myocardial infarction within 1 year of treatment initiation, or other clinically significant heart disease (e.g. congestive heart failure, uncontrolled hypertension, or unstable angina)], severe or uncontrolled medical conditions (e.g. uncontrolled diabetes), and acute or chronic liver disease or severe renal disease.
Study treatment
Study treatment for all patients was nilotinib 300 mg twice daily. Imatinib and nilotinib treatment were separated by a washout period of ⩾3 days. Patients were instructed to take nilotinib under fasting conditions (no food for ⩾2 h prior to and ⩾1 h after dosing). In patients who did not achieve an MMR with nilotinib 300 mg twice daily by month 6 after switch (with 1 month defined as one 28-day cycle), nilotinib dose escalation to 400 mg twice daily was permitted at any time after the end of month 6, per physician discretion. Dose escalation to nilotinib 400 mg twice daily was not allowed if patients were receiving a reduced dose of nilotinib 400 mg once daily or were off treatment due to drug-related adverse events (AEs), as per the ENABL dose reduction guidelines; however, once such patients were able to tolerate nilotinib 300 mg twice daily for ⩾2 weeks, dose escalation to nilotinib 400 mg twice daily was allowed. After dose escalation, patients remained on the new dose until the end of the study. Dose reduction guidelines were specified for certain grade 3/4 hematologic AEs and grade 2–4 nonhematologic AEs. Patients continued nilotinib treatment until end of study, disease progression, unsatisfactory therapeutic effect, intolerance, or study discontinuation.
Study assessments
The primary efficacy endpoint was the log change in BCR-ABL1 transcript levels from the standardized IS baseline after 12 months on nilotinib therapy. BCR-ABL1 transcript levels were assessed by peripheral blood RQ-PCR carried out in a central laboratory (Molecular MD, Portland, OR, USA). For patients in group 1, RQ-PCR analysis was performed at screening and every 3 months until the end of the study. For patients in group 2, RQ-PCR was performed at screening, monthly for the first 3 months, and every 3 months thereafter.
Secondary endpoints included rates of MMR and deeper reductions (⩾4-log and ⩾4.5-log) in BCR-ABL1 transcript levels from the standardized IS baseline at selected time points and time to best molecular response. Time to best molecular response was defined as the time from enrollment until maximum observed reduction in BCR-ABL1 transcript level.
Patients’ imatinib trough levels were measured prior to stopping imatinib. Nilotinib trough levels were measured at screening and days 1, 8, and 84 (end of month 3). Due to the small sample size and early termination of the trial, planned nilotinib trough level assessments at month 12 and end of study were removed from the amended study protocol.
Safety assessments included evaluation of AEs and serious AEs (SAEs), laboratory parameters (hematologic assessment, biochemical testing), ECG evaluation, and physical examinations, including vital signs. Lipid profiles (including high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, very low-density lipoprotein cholesterol, triglycerides, and total cholesterol) were monitored with all scheduled blood biochemistry assessments, including the screening visit, days 1 and 8, end of months 1, 2, and 3, every third month through month 24, and upon early discontinuation. AEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 3.0 [National Cancer Institute, 2006].
Statistical analysis
Efficacy variables were analyzed based on all patients who received ⩾1 dose of study drug, had ⩾1 postbaseline assessment, and were evaluable at the indicated time point. Safety analyses were based on the safety population, which included all enrolled patients who received ⩾1 dose of study drug. Because of the very small sample size, no statistical testing was performed; only descriptive summary statistics were used.
Results
Patients and treatment
A total of 62 patients entered screening. Of these, 44 patients failed screening due to unacceptable laboratory values (n = 16), unacceptable test procedure results (n = 14), not meeting diagnostic/severity criteria (n = 8), withdrawal of consent (n = 6), use of exclusionary medications/therapies (n = 1), or other reasons (n = 2). There were two patients who failed screening for more than one reason. Overall, 24 of the excluded patients did not have MMR at screening, including 15 patients with a <2-log reduction in BCR-ABL1 transcript levels from the standardized IS baseline (BCR-ABL1IS >1%) and 9 patients with a 2- to 3-log reduction (BCR-ABL1IS >0.1–1%).
A total of 18 patients were enrolled and included for analysis (17 patients in group 1 and 1 patient in group 2). Due to the small patient number in each group, all analyses were conducted based on the combined population (N = 18). There were three patients who had prior interferon treatment. Most patients (n = 13; 72.2%) had an ECOG performance status of 0 at screening; five patients (27.8%) had an ECOG performance status of 1. The median age of enrolled patients was 44 years (range, 29–72 years; Table 1). The median duration of prior imatinib treatment was 2.6 years (range, 1.2–9.4 years). Prior to switching from imatinib therapy, patients’ median imatinib trough serum concentration was 1937 ng/ml (range, 487–8990 ng/ml). A total of seven patients had BCR-ABL1IS >1% at screening: one patient without confirmation of CCyR by standard bone marrow cytogenetics at screening (a protocol violation) and six patients who met the study eligibility criteria of CCyR at screening based on fluorescence in situ hybridization or bone marrow test results.
Patient demographics and baseline characteristics.

CML, chronic myeloid leukemia.
Based on 15 patients with known date of diagnosis.
Log reduction relative to the standardized IS baseline.
Patients were treated with nilotinib for a median of 1.9 years (range, 0.2–3.5 years). There were two patients who had dose escalation to nilotinib 400 mg twice daily. Median nilotinib dose intensity was 596 mg/day (range, 259–703 mg/day). At month 3, the median nilotinib serum concentration was 1063 ng/ml (range, 94–2300 ng/ml). Due to low enrollment, the potential correlation between molecular response and imatinib or nilotinib trough levels could not be assessed. Overall, 14 patients were treated for ⩾6 cycles and 12 were treated for ⩾12 cycles. A total of 11 patients (61.1%) completed the study as per protocol, including the 2 patients with dose escalation to nilotinib 400 mg twice daily. A total of seven patients (38.9%) did not complete the study: of these, three patients discontinued from the study due to abnormal laboratory values, two due to AEs, and two due to protocol deviations.
Efficacy
The median BCR-ABL1IS level among all patients at study start was 0.472% (range, 0.107–7.1%), equivalent to a 2.326-log (range, 1.15–2.97-log) reduction from the standardized IS baseline. At 12 months after treatment switch (primary endpoint), the median BCR-ABL1IS level among evaluable patients (n = 12) was 0.022% (range, 0.0032–0.174%), equivalent to a 3.659-log (range, 2.76–4.50-log) reduction from the standardized IS baseline (Figure 1). Thus, BCR-ABL1IS transcript levels fell >1-log after 12 months of nilotinib. There was one patient who completed 45 months of treatment and had a 3.5-log reduction (BCR-ABL1IS = 0.032%) in BCR-ABL1 transcript levels at month 45. The median time to best molecular response was 13.8 months [95% confidence interval (CI), 4.2–23.1 months].

Median (range) log reduction in BCR-ABL1 transcript levels from the standardized IS baseline.
All patients maintained CCyR after switching to nilotinib, and all patients had a downward trend in BCR-ABL1IS levels on nilotinib (Figure 2). A total of three patients did not achieve MMR [all three of these patients discontinued within the first 3 months of the study due to AEs (types of AEs leading to discontinuation described below)]. One patient experienced a >1-log increase in BCR-ABL1IS at 6 months (from 0.003% at month 3 to 0.047% at month 6) and was subsequently found to have an E355G mutation. The patient remained in CCyR and MMR but discontinued from the study after 6 months due to abnormal liver function test results and rising cholesterol levels.

Trends in BCR-ABL1 transcript levels by patient (N = 18). All 11 patients who completed the study per protocol achieved MMR on study, including 10 patients who achieved confirmed MMR (in at least two consecutive assessments). Of the seven patients who discontinued early from the study, four achieved MMR on study (including two patients with confirmed MMR and two with unconfirmed MMR) and three did not achieve MMR on study.
Among evaluable patients, 10 of 17 (59%) achieved MMR at 3 months and nine of 12 (75%) achieved MMR at 12 months. Among the 10 patients with MMR at 3 months, five achieved a ⩾4-log reduction (BCR-ABL1IS ⩽0.01%; MR4) at 3 months or later in the study. Overall, six patients had a ⩾4-log reduction at any time during the study, and four achieved a ⩾4.5-log (BCR-ABL1IS ⩽0.0032%; MR4.5) reduction (Figure 3).
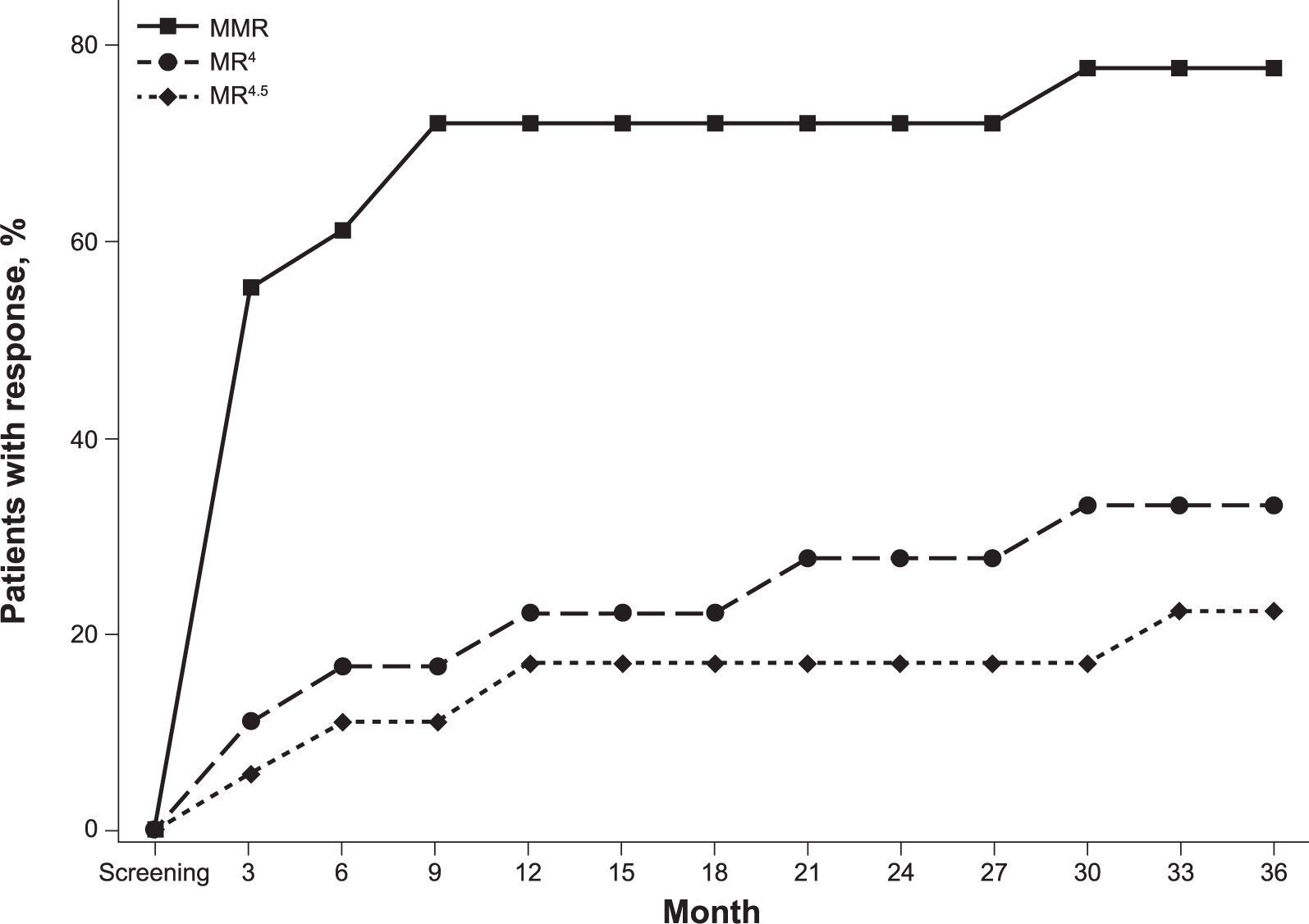
Cumulative incidence of MMR, MR4, and MR4.5 over time. The one patient who achieved MMR at the end of the study is not shown.
Safety and tolerability
AEs reported in ⩾20% of patients at any grade included fatigue in six patients (33.3%); arthralgia, headache, and rash in five patients each (27.8%); and cough, nausea, myalgia, sinusitis, and upper respiratory tract infection in four patients each (22.2%). The most common biochemical abnormalities were alanine aminotransferase (ALT) elevation in three patients (16.7%) and aspartate aminotransferase (AST) elevation, hyperbilirubinemia, hyperglycemia, hypercholesterolemia, and hypertriglyceridemia in two patients each (11.1%). Most AEs and laboratory abnormalities were grade 1 or 2 in severity; no grade 4 AEs or grade 4 hematologic abnormalities were reported. AEs suspected to be related to study treatment were reported in 16 patients (88.9%), including grade 3 events in five patients [27.8%: elevated ALT (n = 2), bradycardia (n = 1), rash (n = 1), and pruritic rash (n = 1)]. QTcF prolongation >60 mg from baseline was not observed in any patient and the maximum QTcF observed on study was 457 mg.
Most AEs reported with nilotinib therapy were manageable with brief dose interruptions. Overall, nine patients had dose reductions due to AEs that were attributed to nilotinib, including four patients with dose reductions due to a single type of AE (one each of anorexia, rash, pruritic rash, and hyperbilirubinemia) and five patients with dose reductions due to multiple types of AEs (myalgia and muscle fatigue; pancreatitis and nausea; speech impairment and vertigo; elevated ALT, elevated AST, pruritic rash, and rash; and elevated ALT, elevated AST, weight loss, and myalgia). There were three patients who discontinued due to abnormal laboratory results (grade 2–3 ALT elevations and grade 2 hyperbilirubinemia), and two patients discontinued due to nonhematologic AEs (grade 3 bradycardia and grade 3 pruritic rash). No deaths were reported.
Overall, 5 SAEs were reported in three patients (16.7%); 3 of these events (in two patients) were suspected to be related to study treatment, including one patient with grade 2 pancreatitis (reported twice) and one patient with grade 2 vertigo that required dose adjustment and grade 3 bradycardia (the only SAE that led to study discontinuation). The 2 SAEs not suspected to be related to study treatment included grade 3 pneumonia in the patient with pancreatitis and sinusitis in another patient. Pneumonia required hospitalization, and all other SAEs resolved after drug interruption or dose adjustment.
Discussion
Current management of CML involves targeting defined treatment goals and changing treatment when the goals are not met. Both the ELN and NCCN emphasize the importance of regular molecular monitoring and achieving molecular response milestones, such as early molecular response (BCR-ABL1IS ⩽10% at 3 or 6 months of frontline therapy, which can provide an indication of future outcomes) [Hughes et al. 2014c] and MMR, as vital components of CML disease management [National Comprehensive Cancer Network, 2016; Baccarani et al. 2013]. However, optimal management of patients who miss ELN- and NCCN-designated milestones remains under investigation. For example, although the ELN and NCCN both designate BCR-ABL1IS ⩽10% as a target response at 3 and 6 months [National Comprehensive Cancer Network, 2016; Baccarani et al. 2013], the long-term impact of switching therapy due to failure to achieve BCR-ABL1IS ⩽10% is unclear. Achievement of MMR is similarly known to be an important milestone; MMR is associated with longer duration of CCyR [Iacobucci et al. 2006; Cortes et al. 2005; Paschka et al. 2003; Press et al. 2007], higher rates of event-free survival [Hughes et al. 2010], and higher rates of progression-free survival [Press et al. 2006; Hehlmann et al. 2014]. However, NCCN guidelines do not recommend a switch in therapy due to lack of MMR in patients who have achieved CCyR, and although the ELN designates achievement of CCyR without MMR at 12 months as a ‘warning’ response level, there is no recommendation to switch treatment due to lack of MMR in these patients [National Comprehensive Cancer Network, 2016; Baccarani et al. 2013].
In the multicenter exploratory study of 18 patients reported here, the majority of patients who switched to nilotinib based on a suboptimal molecular response on imatinib therapy achieved MMR with nilotinib. Overall, nine of 12 (75%) evaluable patients achieved MMR at 12 months after switching to nilotinib; 10 of 17 (59%) achieved this response at 3 months. With a median follow up of approximately 2 years, there were no deaths reported among patients in ENABL; 88.9% of patients had AEs suspected to be related to study drug.
These results are consistent with those from previous studies that demonstrated clinical benefit from switching to nilotinib for patients with varying levels of suboptimal response or treatment failure on imatinib [Hughes et al. 2014a, 2014b]. In the ENESTnd extension study, patients from the imatinib arm of ENESTnd who had a suboptimal response or treatment failure (defined based on the 2009 ELN criteria) [Baccarani et al. 2009] with imatinib therapy were switched to nilotinib 400 mg twice daily; the majority (58%) of patients in that study achieved CCyR after switching to nilotinib, and approximately one-third (32%) achieved MMR by 12 months after switching to nilotinib [Hughes et al. 2014a].
In the ENEST Complete Molecular Response (ENESTcmr) study, patients with CCyR and detectable BCR-ABL1 after ⩾2 years on imatinib were randomized to continue imatinib or switch to nilotinib 400 mg twice daily [Hughes et al. 2014b]. Compared with patients in the imatinib arm, patients in the nilotinib arm of ENESTcmr achieved higher rates of MR4.5 (among patients without MR4.5 at study start; 20.8% versus 42.9%, respectively) by 2 years and undetectable BCR-ABL1 (among patients with detectable BCR-ABL1 at study start; 17.0% versus 31.7%, respectively) by 2 years [Hughes et al. 2014b].
The eligibility criteria and patient populations of ENABL and ENESTcmr differed in two key aspects. First, ENESTcmr required a longer duration of prior imatinib therapy than ENABL, and second, unlike ENABL, ENESTcmr included patients with MMR or deeper molecular responses at study entry. The narrower window of eligible molecular response levels for ENABL likely contributed to its slow enrollment and high screen failure rate. For example, among patients treated with frontline imatinib in ENESTnd, the cumulative rate of MMR after 2 years of treatment (the approximate median duration of prior imatinib therapy among patients in ENABL) was 44%, and with 1 additional year of imatinib therapy the rate of MMR increased to 53% [Hochhaus et al. 2016]; many of these patients who achieved MMR on imatinib may have been ineligible for ENABL but eligible for ENESTcmr.
Despite the differences in enrollment criteria, the results presented here from ENABL are consistent with those from ENESTcmr. In ENESTcmr, response improvements were particularly marked among those patients without MMR at the study start, which is the subset most comparable with the ENABL patient population; among patients in ENESTcmr without MMR at the study start, 29.2% and 3.6% of those in the nilotinib and imatinib arms, respectively, achieved MR4.5 by 2 years [Hughes et al. 2014b]. Together, results from both ENABL and ENESTcmr add support to the notion that patients with a suboptimal molecular response on imatinib can achieve further reductions in BCR-ABL1 transcript levels by switching to nilotinib. Data from the imatinib arm of ENESTcmr suggest that patients would be unlikely to achieve these BCR-ABL1 reductions with continued imatinib therapy [Hughes et al. 2014b].
While this study was limited by a high screen failure rate and low accrual, the rapid reduction in BCR-ABL1 transcript levels in most patients achieved in the initial months following TKI switch supports further investigation of the potential clinical benefits of switching to nilotinib for patients with less-than-optimal responses on long-term imatinib.
Overall, the findings of this study suggest that switching to nilotinib can induce deeper molecular responses in patients with CML-CP who achieve a suboptimal molecular response to imatinib. This is consistent with results from the larger, randomized ENESTcmr study, for which analysis is ongoing [Hughes et al. 2014b]. Additional clinical investigation and long-term follow up may help determine whether switching to nilotinib due to suboptimal molecular response to imatinib can result in improved long-term outcomes.
Footnotes
Acknowledgements
The authors thank Staci Heise, PhD, and Karen Kaluza, PhD (Articulate Science, Hamilton, NJ, USA), for medical editorial assistance with this manuscript. Financial support for medical editorial assistance was provided by Novartis Pharmaceuticals Corporation (East Hanover, NJ, USA).
Funding
This work was supported by Novartis Pharmaceuticals Corporation (East Hanover, NJ, USA).
Conflict of interest statement
Sikander Ailawadhi has no financial conflicts to disclose. Luke P. Akard has acted as a consultant for Ariad (Cambridge, MA, USA), Bristol-Myers Squibb (New York, NY, USA), Celgene (Summit, NJ, USA), and Novartis (East Hanover, NJ, USA); received research funding from Ariad (Cambridge, MA, USA), Bristol-Myers Squibb (New York, NY, USA), Novartis (East Hanover, NJ, USA), and Pfizer (New York, NY, USA); and attended a speaker’s bureau for Ariad (Cambridge, MA, USA), Bristol-Myers Squibb (New York, NY, USA), Celgene (Summit, NJ, USA), Millennium (Cambridge, MA, USA), and Novartis (East Hanover, NJ, USA). Carole B. Miller has acted as a consultant, received research funding and honoraria, and attended a speaker’s bureau for Novartis (East Hanover, NJ, USA) and received honoraria from and attended a speakers bureau for Bristol-Myers Squibb (New York, NY, USA). Anand Jillella has no financial conflicts to disclose. Daniel J. DeAngelo has served on a board of directors or advisory board committee for Novartis (East Hanover, NJ, USA), Bristol-Myers Squibb (New York, NY, USA), and Ariad (Cambridge, MA, USA). Solveig G. Ericson is an employee of and has equity ownership in Novartis Pharmaceuticals Corporation (East Hanover, NJ, USA). Felice Lin is an employee of Novartis Pharmaceuticals Corporation (East Hanover, NJ, USA). Ghulam Warsi has equity ownership in Novartis Pharmaceuticals Corporation (East Hanover, NJ, USA) and was an employee of Novartis Pharmaceuticals Corporation at the time the study was conducted. Jerald Radich has acted as a consultant for Ariad (Cambridge, MA, USA), Incyte (Wilmington, DE, USA), Novartis (East Hanover, NJ, USA), and Bristol-Myers Squibb (New York, NY, USA) and received research funding from Novartis (East Hanover, NJ, USA).
