Abstract
18F-fluorodeoxyglucose positron emission tomography with (FDG-PET) has a well-established role in the pre- and post-treatment staging of Hodgkin lymphoma (HL), however its use as a predictive therapeutic tool via responded-adapted therapy continues to evolve. There have been a multitude of retrospective and noncontrolled clinical studies showing that early (or interim) FDG-PET is highly prognostic in HL, particularly in the advanced-stage setting. Response-adapted treatment approaches in HL are attempting to diminish toxicity for low-risk patients by minimizing therapy, and conversely, intensify treatment for high-risk patients. Results from phase III noninferiority studies in early-stage HL with negative interim FDG-PET that randomized patients to chemotherapy alone
Keywords
Introduction
The majority of Hodgkin lymphoma (HL) patients achieve complete remission and cure with current treatment paradigms. However, a minority of patients will experience relapse and death [Canellos et al. 2014]. In addition, given the young age at diagnosis and overall high survival rates, serious acute and long-term treatment-related toxicities remain a concern including second malignancies, arterial disease, and negative impact on quality of life [Eichenauer et al. 2014; Yeh and Diller, 2012; Hodgson, 2011; Greaves et al. 2014; Khimani et al. 2013]. There remains an unmet need for predictive tools to help guide individualized treatment decisions for patients. This includes the identification of high-risk HL patients where more intensive therapy may be indicated, and conversely, the attenuation of treatment in lower risk patients in an attempt to decrease acute toxicity and late effects.
Functional imaging with 18F-fluorodeoxyglucose positron emission tomography with (FDG-PET) noncontrast computerized tomography (CT) has become a standard tool together with contrast-enhanced CT scan for the initial staging and re-assessment of HL [Evens and Kostakoglu, 2014; Kostakoglu and Evens, 2014]. FDG-PET scans have been shown to more accurately identify correct pretreatment stage when compared with contrast-enhanced CT (CECT), also causing upstaging to a more advanced stage [Hutchings et al. 2006a, Isasi et al. 2005]. The role of post-treatment FDG-PET has also been evaluated extensively to distinguish viable metabolically-active tumor from fibrotic or necrotic tissue in residual masses. However, a number of questions remain regarding the potential value of FDG-PET as a predictive tool in HL. This review focuses on the reproducibility and interpretation of FDG-PET, studies incorporating ‘early’ response-adapted FDG-PET, and the use of FDG-PET in the setting of relapsed or refractory HL. Other papers delineating the role of FDG-PET in the staging and post-treatment surveillance of HL patients has been reviewed elsewhere [Kostakoglu and Evens, 2014].
Interpretation and reproducibility of FDG-PET
The nonspecific nature of low-to-moderate grade residual uptake within a tumor mass during therapy limits the specificity of FDG-PET readings. The imaging subcommittee of the International Harmonization Project in Lymphoma in 2007 was the first initiative for standardization of FDG-PET interpretation following treatment [Cheson et al. 2007]. The resultant criteria stipulated that FDG-uptake greater than that of the mediastinal blood pool in residual masses greater than or equal to 2 cm was considered positive for residual lymphoma. Of note, these criteria were not recommended for application in interim FDG-PET interpretation and were based upon a retrospective study of 54 diffuse large B-cell lymphoma (DLBCL) patients, which were not validated in HL patients [Juweid et al. 2005]. Subsequent efforts to develop a more specific interpretation method has yielded the Deauville 5-point scale reading system (5PS) (Table 1).
Deauville 5-point scale criteria for evaluation of interim positron emission tomography.

Recommended scoring of positive interim FDG-PET for early-stage Hodgkin lymphoma (score 3–5); recommended scoring for positive FDG-PET for advanced-stage Hodgkin lymphoma (score 4–5).
The Deauville 5PS allows for more accurate measurement of response by using a categorical scoring system with a continuous variable. It also allows for different thresholds for positive and negative tests to assess chemotherapy sensitivity
The improved prognostic value of FDG-PET was confirmed in a study showing that NPV was high with all of the criteria, but using a high threshold for positive interim FDG-PET led to increased PPV (Table 2) [LeRoux et al. 2011]. Using the Deauville 5PS increased the PPV from 19% to 45%. Interim FDG-PET correlated strongest with progression-free survival (PFS) using 5PS criteria (
Observational studies of interim PET (nonadaptive) in Hodgkin lymphoma.
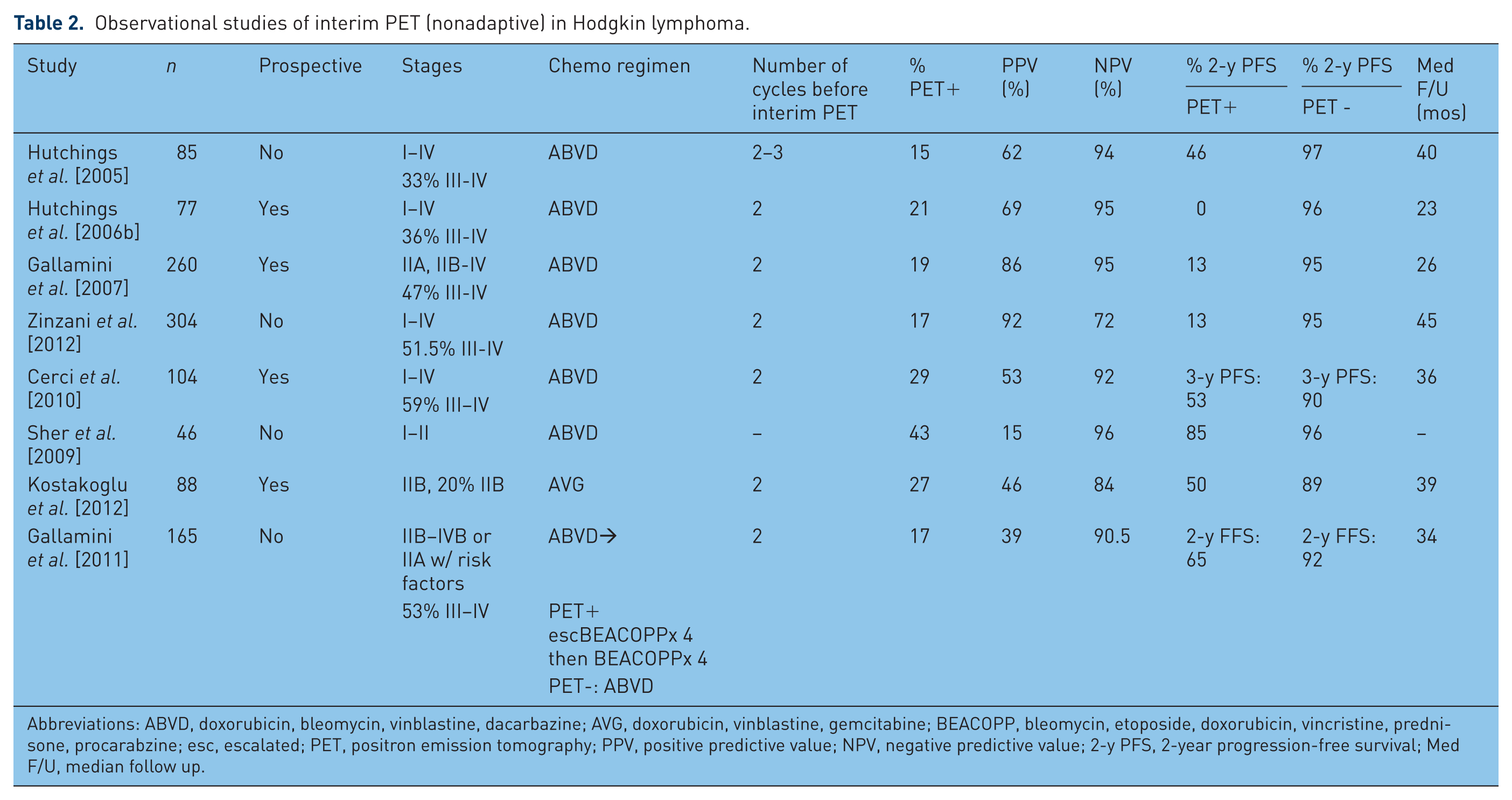
Abbreviations: ABVD, doxorubicin, bleomycin, vinblastine, dacarbazine; AVG, doxorubicin, vinblastine, gemcitabine; BEACOPP, bleomycin, etoposide, doxorubicin, vincristine, prednisone, procarabzine; esc, escalated; PET, positron emission tomography; PPV, positive predictive value; NPV, negative predictive value; 2-y PFS, 2-year progression-free survival; Med F/U, median follow up.
Early-stage HL
Current treatment recommendations for early-stage HL patients with favorable risk involves combined modality treatment, usually consisting of 2–3 cycles of ABVD followed by 20–30 Gy of involved field radiotherapy (IFRT) or involved node RT (INRT). For unfavorable risk profile early-stage HL, recommended therapy is four cycles of chemotherapy followed by 30 Gy of IFRT/INRT. Chemotherapy alone given for four to six cycles is an alternative treatment option in early-stage HL [Hay and Meyer, 2014]. Four published randomized clinical trials compared combined modality therapy (CMT)
Observational/prospective studies (nonresponse adapted)
There have been comparatively less early-stage response adapted analyses compared with advanced stage HL (Table 2). Initial reports of interim FDG-PET for early-stage HL demonstrated a consistently high NPV and low-moderate PPV in relation to treatment outcome. There may be a high number of false-positive FDG-PET scans with the high incidence of inflammatory processes in HL.
A retrospective analysis of 85 HL patients with interim PET after two or three cycles of ABVD showed that FDG-PET had less robust progostication for early-stage
Efficacy of therapy is also a factor that can impact the predictive value of FDG-PET. A total of 88 patients with early-stage nonbulky HL were prospectively studied after treatment with a nonstandard regimen of doxorubicin, vinblastine and gemcitabine (AVG). Two-year PFS rates were 88% and 54% for PET-2-negative and PET-2-positive patients, respectively (
Most interim FDG-PET prognostic analyses in early-stage HL have been in nonbulky patients. A recent analysis examined 121 consecutive early-stage HL patients with 30% patients having bulky disease (89% of which was mediastinal) [Pophali et al. 2014]. Interim FDG-PET was negative in 83% of bulky patients
Phase II FDG-PET response-adapted studies
There have been a paucity of phase II studies reported utilizing response-adapted interim FDG-PET for early-stage HL. As previously noted, LeRoux and colleagues studied early- and advanced-stage HL patients treated with a response-adapted strategy PET-4 after ABVDx 4 [LeRoux et al. 2011]. The NPV and PPV with PET-4 for 2-year PFS were 95% and 16%, respectively (
Prospective noncontrolled response-adapted studies in Hodgkin lymphoma.

Abbreviations: ABVD, doxorubicin, bleomycin, vinblastine, dacarbazine; BEACOPP, bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone; esc, escalated; PPV, positive predictive value; NPV, negative predictive value; PET, positron emission tomography;
2-year PFS, 2-year progression-free survival; Med F/U, median follow up.
Phase III response-adapted trials
A primary hypothesis of randomized studies in early-stage HL have been that acute disease control rates would be similar (noninferior) among patients with negative interim FDG-PET who receive radiotherapy

Clinical trial designs of completed and ongoing phase III randomized studies of response-adapted therapy for adult early-stage HL. (A) EORTC/LYSA/FIL H10F study. *None of the following present: (a) large mediastinal mass; (b) age ⩾50 years; (c) high ESR; or (d) four or more areas. EORTC/LYSA/FIL H10U study. *Any of the following present: (a) large mediastinal mass; (b) age ⩾50 years; (c) high ESR; or (d) four or more areas. (B) UK-led RAPID study. All PET-3 + patients received a fourth cycle of ABVD followed by 30 Gy of involved field radiotherapy. (C) GHSG HD16 favorable trial. *None of the following present: (a) large mediastinal mass; (b) extranodal disease; (c) high ESR; or (d) three or more areas. GHSG HD17 unfavorable trial. *Any of the following present: (a) large mediastinal mass; (b) extranodal disease; (c) high ESR; or (d) three or more areas. High ESR for all of above defined as: >50 mm without B symptoms or ESR <30 mm with B symptoms.
In H10F, approximately 190 patients were randomized to each arm of the study. The PET-2-negative rate was 86%. At interim analysis, one event had occurred in the INRT arm
Another objective of the H10F/U studies were to determine if intensification of therapy from ABVD therapy to escalated BEACOPP could improve outcomes for interim FDG-PET-2-positive patients [Raemaekers, 2015]. Preliminary data showed that a total of 361 patients had a positive interim FDG-PET-2, of which 188 received standard ABVD and 142 received escalated BEACOPP. Intention-to-treat analysis of ABVD
Results from the United Kingdom National Cancer Research Institute RAPID trial have recently been published (Table 4) [Radford et al. 2015]. This also was a phase III noninferiority randomized study that enrolled 602 patients with stage I/II nonbulky HL. All patients received three cycles of ABVD followed by FDG-PET (PET-3). Negative PET-3 was defined as Deauville 5PS 1–2. Patients with positive PET-3 went on to receive a fourth cycle of ABVD and IFRT, while PET-3-negative patients were randomized to IFRT
Randomized phase III response-adapted studies in adult early-stage (I–II) Hodgkin lymphoma.
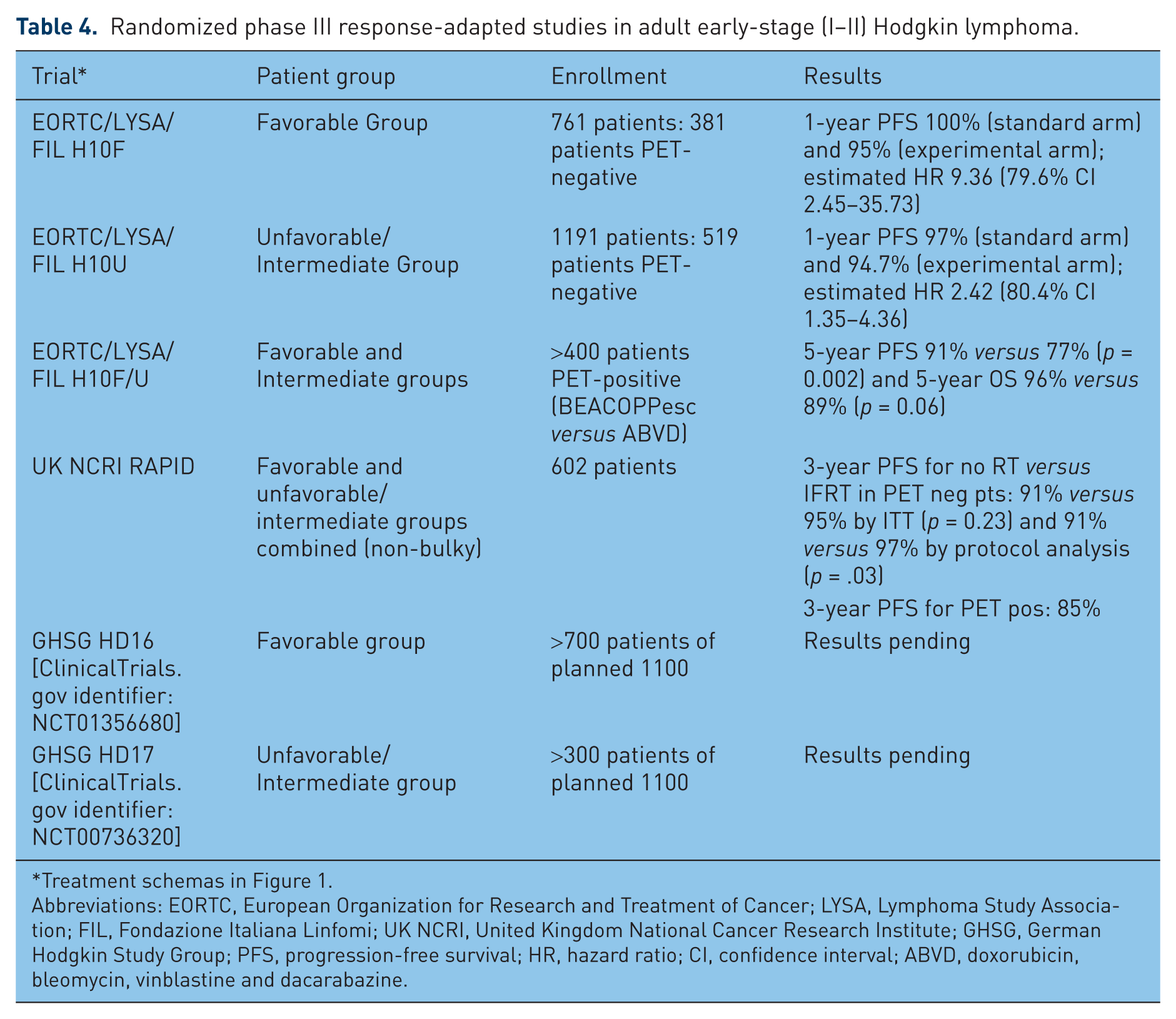
Treatment schemas in Figure 1.
Abbreviations: EORTC, European Organization for Research and Treatment of Cancer; LYSA, Lymphoma Study Association; FIL, Fondazione Italiana Linfomi; UK NCRI, United Kingdom National Cancer Research Institute; GHSG, German Hodgkin Study Group; PFS, progression-free survival; HR, hazard ratio; CI, confidence interval; ABVD, doxorubicin, bleomycin, vinblastine and dacarabazine.
How do we reconcile these ‘negative’ study outcomes given the preliminary data showing that interim FDG-PET in HL was prognostic for favorable outcomes for patients with a negative early scan? This lies in part in the difference between ‘prognostic’ and ‘predictive’ factors. A
Phase III clinical studies ongoing
HD16 and HD17 are GHSG-led noninferiority trials examining response-adapted therapy of favorable and unfavorable HL, respectively (Figure 2). Similar to the EORTC H10 study, these studies are randomizing patients to non-PET based standard therapy

Clinical trial designs of completed and ongoing phase III randomized studies of response-adapted therapy for adult advanced-stage HL. (A) GHSG HD18 includes patients with ‘high-risk’ stage II (*either of the following: large mediastinal mass or extranodal involvement) and stage III and IV disease. (B) The AHL2011 study includes patients with stage IIB and stage III and IV disease. (C) The Response Adapted Therapeutic Hodgkin Lymphoma (RATHL) study included patients with high-risk stage II disease (*with bulk or ⩾3 involved sites) and stage III and IV disease. Abbreviations: HL, Hodgkin lymphoma; Std, standard; Exp, experimental; esc, escalated; GHSG, German Hodgkin Study Group.
Advanced stage HL
Standard treatment for advanced-stage HL typically involves chemotherapy alone for six to eight cycles of therapy. The most common chemotherapy regimen recommended remains ABVD, however some authorities advocate utilization of the more intensive regimen, BEACOPP. The study of interim FDG-PET in advanced-stage HL has focused more on the identification of high-risk patients whereby therapy may be altered or intensified in order to improve outcomes.
Observational/prospective studies (nonresponse adapated)
The sensitivity and specificity of interim FDG-PET appear to be better for advanced-stage compared with early-stage HL (Table 2) [Cerci et al. 2010; Gallamini et al. 2006, 2007; Hutchings et al. 2005, 2006b; Terasawa et al. 2009; Zinzani et al. 2006, 2012]. A positive interim FDG-PET in advanced-stage HL has often been defined as a Deauville score of 4 or 5. Using this criteria with a higher cutpoint, the PPV will be improved (i.e. fewer false positives) so that a response-adapted strategy may identify high-risk patients with positive interim FDG-PET who may benefit from escalated treatment.
In a widely cited 2007 study, Gallamini and colleagues reported on the prognostic value of interim FDG-PET for newly-diagnosed advanced-stage HL. Among 260 patients, 190 had advanced stage disease; patients had FDG-PET after two cycles of ABVD (PET-2) [Gallamini et al. 2007] with or without IFRT following chemotherapy (Table 2). The 2 year PFS for PET-2-positive patients was 13%
A more recent retrospective study of 304 newly-diagnosed ABVD-treated HL patients examined the association of PET-2 with complete response (CR). A positive PET-2 was associated with continuous CR of 25%, while 92% of PET-2-negative patients achieved continuous CR at median of 31 months [Zinzani et al. 2012]. Another prospective study of 104 patients with HL showed a 3-year EFS of 55% for PET-2-positive patients
Phase II FDG-PET response-adapted studies
Investigators studied 160 patients with early-stage unfavorable or advanced-stage HL who were treated with ABVD and had therapy intensified to BEACOPP if PET-2 was positive; PET-2-negative patients remained on ABVD (Table 3) [Gallamini et al. 2011]. The 2-year FFS for PET-2-negative patients was 95%, while PET-2-positive patients had 2-year FFS of 62% with intensified therapy. PET-2 status was the only prognostic factor associated with FFS on multivariate analysis (
Building upon this concept, several groups have performed studies integrating PET-adapted therapeutic strategies. Le Roux and colleagues examined a cohort of 54 patients with early unfavorable or advanced-stage HL treated following a PET-adapted strategy following four initial cycles of ABVD [LeRoux et al. 2011]. Among 31 patients with positive PET-4, 6 patients had treatment failure (19%), while 7 of 59 with a negative PET-4 (12%) had treatment failure. These results yielded a high NPV of 96%, but low PPV of 16%. The low PPV could be possibly explained by the combined criteria based on CT and PET results, incomparability of criteria used for interim FDG-PET interpretation, and relatively late timing for FDG-PET.
The Haifa study group has prospectively examined a cohort of 124 advanced-stage HL patients using interim FDG-PET (response-adapted) and IPS score for an adaptive treatment strategy [Dann et al. 2007, 2012] (Table 3). Patients with IPS score 0–2 (low risk) and 3–7 (high risk) were treated with two cycles of baseline or escalated BEACOPP, respectively. An interim Gallium-67 or FDG-PET was done to determine subsequent therapy, with continuation of escalated BEACOPP if imaging was positive or de-escalation to baseline BEACOPP with negative imaging. Ten-year PFS and OS were 87% and 88%, respectively, with median follow up of 89 months. For patients with positive interim FDG-PET, 10-year PFS was 83%; 10-year PFS was 93% for patients with negative interim FDG-PET. Avigdor and colleagues studied 45 patients with new diagnosis of HL stages IIB-IVB and IPS of at least 3 who were treated with two courses of escalated BEACOPP (Table 3) [Avigdor et al. 2010]. Interim FDG-PET and contrast-enhanced CT were used to determine response and treatment arms. By IHP criteria, patients in CR or PR had de-escalation with ABVD for four cycles, while patients with less than a PR proceeded to autologous stem cell transplant (SCT). A total of 44 patients were in CR or PR and 70% had a negative PET-2, while 30% had a positive PET-2. Patients with negative PET-2 and positive PET-2 had CR rates of 97% and 69%, respectively, which yielded a PPV of 45% and NPV of 87%. Four-year PFS was 87% for PET-2-negative patients and 53% for PET-2-positive patients, which was statistically different (
A retrospective study reported at ASH 2014 examined the prognostic role of baseline FDG-PET with interim PET-2 with ABVD in untreated advanced HL [Cimino et al. 2014]. Among 162 patients analyzed with both PET and CECT, 57 patients were found to have extranodal (EN) disease. Baseline FDG-PET identified 27 EN sites of involvement missed by CECT, whereas CECT picked up 5 EN sites missed by PET-0 (25 EN sites were seen on both modalities). Univariate and multivariate analysis showed EN disease and positive PET-2 as the only significant variables on EFS with HRs of 3.9 [95% confidence interval (CI) 1.62–9.36,
The US Intergroup S0816 is a phase II study that has reported preliminary results of 371 advanced-stage HL patients treated with ABVD as noted in Table 5 [Press et al. 2013]. Patients with interim negative PET-2 continued with four further cycles of ABVD, while PET-2-positive patients had therapy intensified to escalated BEACOPP for six cycles. PET-2 was negative in 82% and 2-year PFS was 78% for these patients. The 2-year PFS was 61% for PET-2-positive patients, which appeared improved compared with historical controls. Notably, HIV positive patients were included, but had therapy changed to baseline BEACOPP with positive PET-2. Of the 13 HIV positive patients, 11 were PET-2-negative. Twelve out of 13 patients were progression-free and all 13 remained alive. In addition, the Children’s Oncology Group (COG) has completed accrual to a phase II study with similar design to S0816, but utilizing ABVE-PC as the chemotherapy backbone, CT-defined interim analyses, and with the assignment of patients to one of two consolidation regimens based on response to induction therapy [ClinicalTrials.gov identifier: NCT01026220]. Patients with CT-defined rapid early response (RER) received two further cycles of ABVE-PC, and slow early responders had therapy intensified to ifosfamide and vinorelbine. An additional endpoint of this study is the investigation of FDG-PET after one cycle of induction ABVE-PC to examine comparability with RER. Results are awaited from this study.
Prospective response-adapted studies in advanced stage (III/IV) HL.

Abbreviations: ABVD, doxorubicin, bleomycin, vinblastine, dacarbazine; AVD, doxorubicin, vinblastine, dacarbazine; ABVE-PC: doxorubicin, bleomycin, vincristine, etoposide, prednisone, cyclophosphamide; IV, ifosfamide, vinorelbine; BEACOPP, bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone; BEACOPP-14, 14-day cycle; esc, escalated; IGEV, ifosfamide, gemcitabine, etoposide, vinorelbine, prednisolone; PET, positron emission tomography; PETpos, positive PET scan; PETneg, negative PET scan; RT, radiotherapy;
ORR, overall response rate; CR, complete response; PFS, progression-free survival; OS, overall survival; IPS, international prognostic score; SWOG, Southwestern Oncology Group; COG, Children Oncology Group; UK NCRI, United Kingdom National Cancer Research Institute; RATHL, response adapted therapy for Hodgkin lymphoma; GITIL, Gruppo Italiano Terapie Innovative nei Linfomi; GHSG, German Hodgkin Study Group; LYSA, The Lymphoma Study Association.
The Italian GITIL0607 study has registered 730 patients treated with ABVD and subsequent response-adapted therapy (Table 5) [Gallamini et al. 2013]. A total of 82% of patients had negative PET-2 and proceeded to six total cycles of ABVD; PET-2-positive patients (18%) were randomized to escalated-BEACOPP for four cycles or standard BEACOPP for four cycles with or without rituximab. In the second interim analysis, 2-year PFS was 81% for all patients, with PET-2-negative and PET-2-positive PFS being 85% and 61%, respectively. Final results of this study are awaited. The HD0801 study enrolled 520 patients with advanced-stage HL in a response-adapted study with early transition to salvage therapy with SCT [Zinzani et al. 2013]. Patients with negative interim FDG-PET received six cycles of ABVD and were randomized to either radiation therapy to bulky mediastinal masses or no radiation therapy. Patients with positive interim FDG-PET received salvage therapy with IGEV (ifosfamide, gemcitabine, etoposide, vinorelbine and prednisolone); this was followed by autologous SCT with BEAM conditioning if FDG-PET post-IGEV was negative (58%). The 42% of patients with positive PET after IGEV had a tandem autologous SCT or autologous SCT followed by allogeneic SCT if a donor match was available. Two-year PFS and OS were 76% and 99% for interim PET-negative patients after two cycles of ABVD, while for PET-positive patients, the PFS and OS were 64% and 86%, respectively.
The Israeli H2 study has recruited 180 patients with advanced stage HL [Dann et al. 2013]. Patients with IPS 0–2 were treated initially with two cycles of ABVD, while patients in IPS 3–7 group were treated with two cycles of escalated BEACOPP. Negative PET-2 patients proceeded to four additional cycles of ABVD, while PET-2-positive patients received four cycles of escalated BEACOPP. After interim FDG-PET, 85% of patients were PET-2-negative and 15% positive. PET-2 was negative in 88% of the IPS 0–2 group and 80% of IPS 3–7 group. Furthermore, in the IPS 3–7 group, therapy was de-escalated in 89% of patients and 13% of the whole group had progression at 3 years. At a median follow up of 26 months, the 3-year PFS was 85%.
Phase III response-adapted trials
The ongoing GHSG HD18 trial is using an initial two initial cycles of escalated BEACOPP followed by FDG-PET assessment [Borchmann et al. 2012]. PET-2-negative patients were randomized to two
Preliminary results have been recently reported from a large randomized phase III study led by the UK NCRI in advanced-stage HL called the RATHL (Response-Adjusted Therapy for Hodgkin Lymphoma) study [Johnson et al. 2015]. Among 1214 enrolled advanced-stage HL patients, all patients received two cycles of ABVD followed by PET-2; 84% of patients had a negative PET-2 defined as Deauville scores of 4 or 5. PET-2-negative patients were randomized to continued ABVD or doxorubicin, vinblastine, and dacarbazine (AVD). PET-2-positive patients had their therapy intensified (i.e. BEACOPP-14). With a median follow-up of 32 months, PFS at 3 years appeared the same for ABVD 85%
Newer imaging techniques
Combined CT and PET analyses
In an effort to improve positive predictive value of FDG-PET imaging, several studies combined PET and CT results to further separate favorable and unfavorable patient outcomes. In a study of 88 patients with stage I and II non-bulky HL incorporating IHP and Deauville 5PS criteria, the percentage of decrease in the sum of products of perpendicular diameters of masses after two cycles strongly correlated with 2-year PFS [Kostakoglu et al. 2012]. Analysis of PET-2 with CECT-2 data suggested improvement in prediction of 2-year PFS
Metabolically defined and quantifiable tumor volumes
Although FDG-PET has proved useful for therapy monitoring in HL patients, the false positivity due to post-therapy inflammatory processes have raised concerns about its effective use in interim PET-adapted strategies. In an effort to increase both the interpretation accuracy and reproducibility, various quantitative methods have been proposed [Casasnovas et al. 2011; Lin et al. 2007; Weber, 2007]. In HL, a maximum standardized uptake value (SUVmax) cut-off of 4.0 has reportedly provided the best joint sensitivity and specificity for the prediction of progression after two cycles of chemotherapy [Hutchings et al. 2006b]. It is important to note that these criteria have not been prospectively validated and also the SUV cutoff as a prognostic indicator may be different for defining response very early during therapy compared to later time points.
Other proposed PET parameters that include functional tumor volume parameters, for example, metabolically active tumor volume (MTV) and total lesion glycolysis (TLG) are currently evolving and have not yet been standardized, thus, not widely used. MTV incorporates the size-dependent thresholding to determine MTV on the basis of SUVmax obtained within a volume of interest that represents the tumor biology. Baseline quantitative PET parameters can be used as prognostic factors and may have better predictive value than conventional clinical prognostic factors, that is, in HL patients.
In an analysis of early-stage HL analysis, pretreatment MTV and SUVmax did not correlate with outcome, however change in MTV between interim and baseline studies was associated with median PFS (
New imaging biomarkers
New imaging biomarkers include measures of heterogeneity, which is emerging as an important factor in imaging analyses [Hatt et al. 2010]. Assessment of tumor proliferative activity may provide a critical tool for individualized treatment. The 3′-deoxy-3′-18 F-fluorothymidine (FLT) is the most extensively investigated functional imaging probe for measurement of cancer cell proliferative capacity [Bading and Shields, 2008]. The role of FLT-PET will depend in part in its ability to predict early response during treatment, rather than determining the extent of disease involvement at staging. The clinical utility of FLT as an early response surrogate to date has been demonstrated in preliminary clinical studies in non-HL [Bading and Shields, 2008].
There are a number of limitations to these early functional imaging studies. Methodologically, there are ongoing challenges associated with tumor segmentation algorithms. Consequently, currently used methods are often being used without the needed validation or optimization. Imaging parameters such as scanner resolution, reconstruction algorithms, filtering, tumor-to-background ratio, and image noise impacts the accuracy and precision of tumor delineation methods. This implies that technical PET parameters and the tumor delineation methods require standardization and calibration of each scanner for reproducible and accurate volume determinations. Collectively, despite promising preliminary results, the prognostic and predictive value of functional tumor volume remains to be further investigated with standardized, prospective, multicenter studies to determine the extent that these new imaging modalities may play in the management of HL.
Novel therapeutic agents
Brentuximab vedotin (BV) is an antibody drug conjugate with significant activity in patients with HL. The prognostic impact of interim FDG-PET (using Deauville 5PS > 3 as positive) was examined in a study using single-agent BV for relapsed/refractory HL [Kahraman et al. 2014]. After receiving a median of 3 doses BV, 67% were interim PET-positive. One year PFS rate for interim PET-negative patients was 100% compared with 38% for interim PET-positive patients (
Conclusions
FDG-PET has an established role in the pretreatment staging of HL, but its use as a predictive therapeutic tool with use as responded-adapted therapy continues to evolve. As reviewed, there have been multiple studies showing that interim FDG-PET is highly prognostic in HL, particularly in the advanced-stage setting. Prospective studies have evaluated the utility of interim FDG-PET for response-adapted treatment approaches in an attempt to diminish toxicity by minimizing therapy for low-risk patients and to potentially improve outcomes by intensifying treatment for high-risk HL patients. Initial studies in early-stage HL examining negative interim FDG-PET showed a continued small improvement in PFS for patients who received radiation therapy (i.e. similar to the pre-PET era). It is possible that late survival advantages may emerge with longer follow up (especially OS). Preliminary reports of data escalating therapy for positive interim FDG-PET in early-stage HL and for de-escalation of therapy for negative interim FDG-PET in advanced stage HL showed that outcomes were improved. Maturation and longer follow up of these data are needed. Furthermore, continued refinement and optimization of response-adapted therapy is needed, including in the context of targeted therapeutic agents. In addition, a number of new and novel techniques of functional imaging, such as metabolic tumor volume and tumor proliferation using FLT and integrated PET/MRI, are being explored in order to enhance staging, characterization, and prognostication in HL.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
