Abstract
Ikaros is the founding member of a family of zinc finger transcription factors whose function during early hematopoietic development is required for differentiation into the three major hematopoietic lineages. Ikaros deletions have been described in human malignancies, particularly precursor B-cell leukemia. Deletions of this transcription factor appear to mediate leukemogenesis, although the exact mechanism is unclear. This article reviews the structure and function of Ikaros proteins in chromatin remodeling and gene expression as well as the current knowledge of Ikaros deletions in human malignancies. A new proteomic platform, mass cytometry, is introduced which allows measurements of greater than 30 parameters at the single-cell level and should thus provide a greater level of detail to unravel the mechanistic consequences of Ikaros dysfunction in leukemia.
Introduction
Hematopoiesis involves the hierarchical organization of multipotential hematopoietic stem cells into erythroid, myeloid and lymphoid lineages. The steps that demarcate lineage commitment and maturation must be precisely regulated, a role fulfilled by the orchestration of multiple transcription factors.
A mechanistic understanding of these transcriptional programs is important in healthy hematopoiesis to serve as a reference for their deregulation, which is often causal, in disease pathogenesis. One critical regulator of these transcriptional programs is Ikaros (encoded by IKZF1), the founding member of a family of DNA binding proteins, required for the normal development of lymphocytes and other blood cell lineages with an important role in hematopoietic malignancies. During hematopoiesis, Ikaros functions either as a transcriptional activator or repressor by the recruitment of chromatin remodeling complexes.
Dysfunctional Ikaros proteins have gained notoriety for their link to hematologic malignancies such as acute lymphoblastic leukemia. In this review, I outline the discovery of this family of proteins and how their unique structure is linked to function. I explore the multitasking abilities of this protein and its oncogenic potential, unleashed through a variety of molecular changes.
Ikaros structure
Ikaros is a zinc finger protein with N-terminal DNA binding and C-terminal dimerization domains originally shown to target the lymphocyte-specific genes deoxynucleotidyltransferase, terminal (DNTT) and CD3δ [Georgopoulos et al. 1992].
The two zinc finger regions mediate different functional activities of the protein (Figure 1). The four amino terminal zinc fingers mediate sequence-specific binding to DNA at purine rich sequences with the TGGGAA consensus sequence [Molnár and Georgopoulos, 1994]. The two C-terminal zinc fingers are involved in protein–protein interactions with other Ikaros family members [Hahm et al. 1994]. Ikaros protein structure is increased in complexity by the presence of alternatively spliced isoforms. All Ikaros isoforms contain the first two coding exons as well as the last exon, but the number of internal exons varies (Figure 2). Initially, six distinct mRNAs were detected in mouse and subsequently in humans; currently there may be as many as 11 distinct isoforms in humans [Mullighan et al. 2008; Molnár et al. 1996; Hahm et al. 1994].
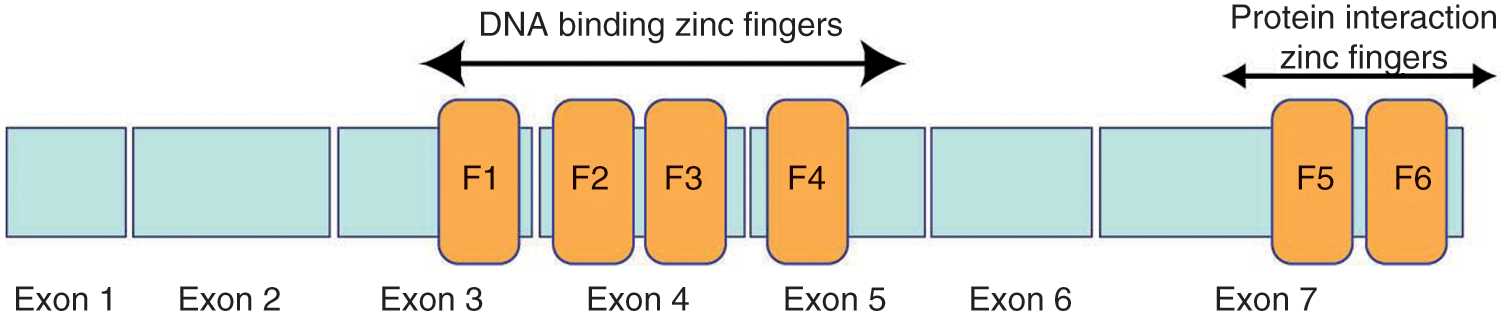
Two domains of zinc fingers that serve different functions characterize Ikaros proteins. The N-terminal zinc fingers (F1–F4) mediate DNA binding whereas the C-terminal zinc fingers are involved in protein–protein interactions.

Ikaros isoforms are the result of post-transcriptional splicing [Payne et al. 2001; Sun et al. 1996]. Isoforms lacking N-terminal zinc fingers do not bind DNA and act as dominant negatives. Isoforms lacking the C-terminal zinc fingers (ZnFs) are unstable.
DNA binding by Ikaros requires zinc fingers 2 and 3, with high-affinity binding requiring the additional presence of zinc finger 1 or 4 [Molnár and Georgopoulos, 1994]. Isoforms Ik1 and Ik2 are the most abundantly expressed isoforms and are present in early hematopoietic progenitors [Klug et al. 1998]. Isoforms incapable of high-affinity DNA binding are still capable of dimerizing with other members of the Ikaros family (see below) and act as dominant negatives (DNs) by sequestering them away from DNA [Sun et al. 1996].
Other proteins with homology to Ikaros have been identified resulting in a family of Ikaros transcription factors. Family members include Aiolos, Helios, Eos and Pegasus, all of which are capable of forming multimers with other members of the family. Similarly, the non-DNA-binding isoforms are also capable of inhibiting the function of these family members. There is some tissue specificity to these family members with Ikaros, Aiolos, and Helios enriched in hematopoietic tissues whereas Eos and Pegasus have more broad tissue expression. These related proteins bind similar DNA sequences but the consequence of dimers and multimers of these family members on transcription or epigenetic function is unknown.
Ikaros in chromatin remodeling and gene expression
Multiple studies support Ikaros having a dual function as a regulator of transcriptional silencing and activation. Current understanding supports a multifaceted role for Ikaros proteins in hematopoiesis that is stage and cell context specific.
Initial studies carried out in the murine system that supported a role for Ikaros in transcriptional silencing showed its colocalization to areas of transcriptionally inactive heterochromatin in both transformed pre-B-cell lines and cycling primary lymphocytes [Klug et al. 1998; Brown et al. 1997]. Specifically, Brown and colleagues used immunofluorescence in situ hybridization in cycling lymphocytes and showed the nuclear localization of Ikaros in discrete foci was closely associated with centromeric heterochromatin. These authors elegantly demonstrated a strong correlation between these foci and the location of transcriptionally repressed genes, including λ5, CD2, CD8α, and CD4 [Brown et al. 1997].
The observation that Ikaros proteins colocalize to areas of transcriptionally silenced genes was subsequently linked to their epigenetic regulation of gene expression when Ikaros and Aiolos were discovered to be complexed with chromatin remodeling machines. There are multiple chromatin remodeling complexes encoded by the human genome that mediate transitions between active and inactive states of chromatin and are thus essential for a cell to achieve the high level of control required for accessing the transcriptional program [Gao et al. 2009; Fry and Peterson, 2001]. In a T-lymphocyte model, a major fraction of Ikaros and Aiolos was found to be part of the Mi-2/nucleosome remodeling deacetylase (Mi-2/NuRD) chromatin remodeling complex primarily involved in gene silencing whereas a minor fraction (fivefold less) was found to be part of the SWI/SNF (related to yeast switch/sucrose) chromatin remodeling complex [Kim et al. 1999]. SWI/SNF chromatin-remodeling complexes include the Brahma (Brm/Smarca2) or Brahma-related gene 1 (Brg1/Smarca4) ATPases, which provide the energy to slide nucleosomes along the strands of DNA, allowing access to the transcriptional machinery resulting in gene activation.
Ikaros’ involvement in chromatin remodeling and transcriptional regulation is elegantly demonstrated in the erythroid developmental regulation of β-globin switching [O’Neill et al. 2000]. During red blood cell development, on chromosome 11, the five globin genes (ϵ, γG, γA, δ, β) are arranged and chronologically expressed in the 5’-3’ direction via transcriptional regulation by the upstream β-locus control region. During embryonic hematopoiesis, Ikaros is involved in the activation of γ-globulin expression in the yolk sac via direct interaction with GATA1 and participation in transcriptional elongation [Bottardi et al. 2011]. Yet, Ikaros is then involved in the silencing of γ-globin genes during the switch to the adult-type β-globulin expression during fetal development [Bottardi et al. 2009]. Loss of Ikaros activity during development of erythroid lineages results in decreased numbers of differentiating erythroid cells with concomitant decrease in transcription of key early erythroid transcription factors such as GATA1 FOG1, EKLF and EPOR [Dijon et al. 2008].
The location of Ikaros-Mi-2/NuRD and Ikaros-SWI/SNF chromatin remodeling complexes vary during the cell cycle. In resting, G0 T cells, both complexes were observed by immunofluorescence to be part of a distinct punctate nuclear structure, not part of heterochromatin. In G1 and S-phase T cells, Ikaros-Mi-2/NuRD complexes move to areas of heterochromatin where they fulfill their role in transcriptional silencing. Conversely, the Ikaros-SWI/SNF complexes form diffuse and speckled areas in activated T cells and localize to areas of euchromatin where they likely play a role in transcriptional activation [Kim et al. 1999]. The observation that Ikaros forms mutually exclusive and geographically distinct complexes with SWI/SNF and with Mi-2/NuRD suggests that a key aspect of Ikaros function is in chromatin remodeling and whether this is to repress or activate gene transcription is defined by the complex to which Ikaros associates (Figure 3).

Ikaros can act as a transcriptional repressor or activator based on cell type and developmental context. By its interactions with different chromatin-remodeling complexes, Ikaros mediates different transcriptional outcomes. HDAC, histone deacetylase.
The function of Ikaros proteins is also regulated by post-translational modification. Gómez-del-Arco and colleagues showed Ikaros hyper-phosphorylation during the S phase of the cell cycle. This modification precludes its DNA binding and permits progression through the cell cycle. During the G1–S transition, Ikaros is dephosphorylated and able to bind DNA causing cell cycle arrest [Gómez-del-Arco et al. 2004]. Thus, the function of Ikaros is regulated by association with chromatin remodeling machines, its location within the chromatin microenvironment, and post-translational modifications.
Ikaros: a master regulator of hematopoiesis
That Ikaros is a major determinant of hematopoietic lineage came from numerous studies in mice genetically engineered to address Ikaros function. In 1994, Georgopoulos and colleagues demonstrated dramatic defects in lymphopoiesis in mice engineered to express DN Ikaros. These mice displayed hypoplasia or absence of secondary lymphoid organs and absence of B- and T-cell precursors and mature lymphocytes [Georgopoulos et al. 1994]. In contrast to their lymphocyte population, mice expressing DN Ikaros had an expansion of myeloid and erythroid cells in the spleen. Corroborating these findings, Ikaros knockout mice (–/–) also lack early lymphocyte progenitors but retain a small population of clonal CD4+ cells [Wang et al. 1996].
Subsequent investigations in other hematopoietic cell types revealed Ik1 and Ik2 isoforms within the nucleus of long-term hematopoietic stem cells (HSCs) [Klug et al. 1998]. HSC differentiation coincides with the expression of the shorter Ikaros isoforms, Ik4 and Ik6, observed in pro-T and pro-B lymphocytes as well as in mature neutrophils. Functionally, hematopoietic stem cells from Ikaros–/– or Ikaros DN mice lack expression of Flk-2 and c-kit. The bone marrow from these mice displays about a third of the cellularity seen in normal bone marrow with a reduction in more committed erythroid and myeloid precursor cells [Nichogiannopoulou et al. 1999]. The consequence of Ikaros deregulation on hematopoietic stem cells and early lineage commitment was investigated further by Yoshida and colleagues using a reporter construct based on Ikaros regulatory elements in wild type or Ikaros–/– mice. Lymphoid-myeloid progenitors (LMP) in Ikaros–/– mice lack expression of Flt3, in agreement with earlier observations, and are unable to differentiate along the B-lymphocyte lineage, while retaining proficiency in generating some T lymphocytes and a full complement of myeloid cells. Ikaros also affects differentiation of the common myeloid progenitor (CMP) as shown by an increase in the myeloid-erythroid progenitors (MEP) at the expense of granulocyte-myeloid progenitors (GMP) in Ikaros–/– mice. However, in MEPs from the Ikaros–/– mice, the balance between erythroid and megakaryocyte populations are skewed in favor of more megakaryocytes [Yoshida et al. 2006]. These experiments demonstrate the multifaceted role of Ikaros in cell fate decisions in all lineages during early hematopoiesis.
Moving even further down the lineage commitment path, Ikaros also plays a role in more committed progenitors. Ikaros hypomorphic mice have an increase in neutrophil progenitors with a negative impact on their subsequent survival and migratory potential [Dumortier et al. 2003]. Additionally, after progenitors are committed to the B-cell lineage, expression of DN Ik6 results in a decrease in the number of CD19+ B cells in the bone marrow and spleen with concomitant decreases in PAX5, EBF and VhCμ mRNA. These observations demonstrate that Ikaros is also active in the later stages of B-cell development [Tonnelle et al. 2001]. Taken together, these studies have established a role for Ikaros in maintaining the homeostasis of hematopoietic cell fate decisions.
Ikaros in malignancy
Given the diverse roles of Ikaros in hematopoiesis and the growing list of key developmental genes that are co-regulated by Ikaros, it is not surprising that loss of Ikaros function has been observed in human hematopoietic malignancies. This loss of function can be mediated by several molecular mechanisms including deletions, either intragenic deletions or loss of the entire IKZF1 gene resulting in haploinsufficiency, or via imbalance in expression of Ikaros isoforms. Before Ikaros was identified in human leukemia, the development of T-cell lymphoblastic leukemia (T ALL) was observed in mice heterozygous for DN Ikaros proteins [Winandy et al. 1995]. However, in human disease, Ikaros mutations are rare in T-cell leukemia. Sun and colleagues described increased expression of non-DNA-binding isoforms of Ikaros (Figure 2) in half of 18 primary patient samples of T-cell leukemia. However, a subsequent study identified only one out of 25 primary T ALL samples that harbored nonfunctional Ikaros [Marçais et al. 2009; Sun et al. 1999a]. This is especially noteworthy given that T ALL is often found to have deregulation of Notch signaling and Ikaros is known to regulate the Notch promoter by silencing the Notch transcriptional program [Gómez-del-Arco et al. 2010; Jeannet et al. 2010].
In contrast to T ALL, alterations in Ikaros function are far more common in primary B progenitor ALL (B ALL). The first description of alterations in Ikaros function in a human disease was reported by Lei Sun and colleagues, who showed aberrant expression of Ikaros isoforms in infant B ALL. In a cohort of 12 primary diagnostic patient samples, non-DNA-binding isoforms of Ikaros were expressed in seven samples and were localized to the cytoplasm [Sun et al. 1999c]. In subsequent reports, expression of similar non-DNA binding isoforms of Ikaros was observed in larger independent cohorts of adult and pediatric B ALL samples [Nishii et al. 2002; Takanashi et al. 2002; Nakase et al. 2000; Olivero et al. 2000; Sun et al. 1999b]. The initial reports suggested the deregulation of Ikaros function occurred at the post-transcriptional level, with increased expression of DN isoform mRNA. However, genomic profiling of additional cohorts of pediatric leukemia identified copy number alterations at the IKZF1 locus [Yang et al. 2008; Kuiper et al. 2007]. These results were confirmed and further refined by Mullighan and colleagues using single nucleotide polymorphism (SNP) arrays on a cohort of Philadelphia chromosome positive B ALL. In 84% of the patients in this cohort, deletions of IKZF1 involving the entire gene or loss of the regions encoding exons 3–6 were observed and confirmed with resulting loss of expression of DNA binding Ikaros isoforms in both instances (Figure 2) [Mullighan et al. 2008].
Deletions in IKZF1 with consequent expression of DN protein isoforms found in B-cell precursor ALL correlate with an inherent resistance to current therapeutic strategies and increased risk of poor outcomes. Notably, such DN Ikaros proteins are present in about a quarter of high-risk pediatric B ALL cases [Mullighan et al. 2009a]. In these patients, expression of DN isoforms of Ikaros were associated with elevated minimal residual disease levels and higher cumulative incidence of relapse. The poor prognosis related to harboring an IKZF1 gene deletion in pediatric leukemia seems to persist despite uniform treatment regimens and, when coupled with minimal residual disease data, has been shown to very accurately predict patients who will relapse [Waanders et al. 2011; Kuiper et al. 2010]. Several groups have described the increased DN Ikaros isoforms in B ALL to coincide with expression of the Philadelphia chromosome, a translocation of chromosomes 9 and 22 resulting in constitutive activation of the ABL tyrosine kinase [Iacobucci et al. 2008; Mullighan et al. 2008; Olivero et al. 2000]. Additionally, Iacobucci and colleagues suggested that the increased expression of the DN isoform Ik6 in 49% of adult Philadelphia chromosome positive (Ph+) ALL cases is linked with resistance to targeted therapy with tyrosine kinase inhibitors [Iacobucci et al. 2008]. Deletions in IKZF1 confer poor prognosis in adult Ph+ ALL with shortened disease free survival (10 months versus 32 months) as well as increased cumulative incidence of relapse (69.1% versus 40.4%) [Martinelli et al. 2009]. In this genetic subtype of leukemia, the evolution of the disease, specifically the chronologic acquisition of genetic changes or their interaction, has yet to be uncovered.
Yet another example of Ikaros’ key role in leukemogenesis is in the pathogenesis of chronic myeloid leukemia (CML) upon transition to lymphoid blast crisis [Nakayama et al. 1999]. Examination of CML samples in chronic phase with matched samples in blast crisis revealed acquisition of IKZF1 deletions upon transition to lymphoid blast crisis [Mullighan et al. 2008]. Further in support of IKZF1 deletions mediating development of acute leukemia is the observation that loss of IKZF1 in the germline seems to confer risk of developing leukemia in children. Genome-wide association scan and SNP arrays identified germline SNPs within the IKZF1 locus (7p12.2) that were associated with an increased risk of developing childhood leukemia [Papaemmanuil et al. 2009; Treviño et al. 2009]. Taken together, these studies suggest that IKZF1 deletions are drivers of malignant transformation and not just coincident passenger mutations.
While Ikaros is important in the development of the erythroid and myeloid/granulocyte lineages of blood cells, its role in myeloid malignancies is only recently emerging, with few reports describing IKZF1 deletions in acute myeloid leukemia (AML). Yagi and colleagues described expression of Ik6 in seven of 10 cases of M4 or M5 AML. They provided some mechanistic insight by transfecting a murine cell line with Ik6 and showed increased expression of the antiapoptotic protein Bcl-xL [Yagi et al. 2002]. A recent study reported that in patients with history of myeloproliferative neoplasms, upon transformation to acute leukemia, the entire IKZF1 gene is deleted, leading to haploinsufficiency [Jäger et al. 2010]. This is in contrast to IKZF1 deletions in lymphoid neoplasms, which are often intragenic and result in the expression of only dominant negative proteins. However, both lesions result in a nonfunctional Ikaros protein but the mechanism involved in the loss of genetic material differs. The acquisition of Ikaros mutations upon transformation from more indolent forms of myeloproliferative neoplasms or chronic phase CML to their acute counterparts reinforces the notion that Ikaros deletions may further enhance deregulation of proliferation and survival pathways.
Ikaros, signal transduction and future directions
Despite the knowledge that dysfunctional Ikaros proteins are drivers of leukemogenesis, the precise mechanism by which this occurs is unclear. Data are emerging to support the association of Ikaros with deregulated signaling during normal lymphocyte developmental checkpoints leading to potentiated growth and survival pathways. Murine models of Ikaros–/– T cells have been shown to have a potentiated response to T-cell receptor signaling in a ligand-independent manner [Winandy et al. 1995]. Similar to this potentiated response to T-cell receptor signaling, in Ikaros-mutated pre-B-cell ALL, there appears to be increased sensitivity to proliferation signals mediated by the pre-B-cell receptor [Merkenschlager, 2010; Trageser et al. 2009; Nera et al. 2006]. During normal B-cell development, as pro-B cells rearrange their heavy chain, ligand-autonomous signaling via the pre-B cell receptor (pre-BCR) causes cells to cycle, expand and transition into the large pre-B-cell stage of development. In normal large pre-B cells, Ikaros plays a role in directing the transcriptional programs that limit this proliferation and drive differentiation, perhaps by silencing expression of pre-BCR component λ5 and downregulating pre-BCR signaling [Ma et al. 2008]. Within this developmental context, Ikaros acts as a tumor suppressor and deletions are permissive to cell replication. Experimental models of Ikzf1 mutated pre-B cells favor upregulation of pro-survival signaling pathways such as JAK-STAT, as well as increased expression of anti-apoptotic proteins such as Bcl-xL and Bcl-2 [Kano et al. 2008; Sezaki et al. 2000].
Given the established role of Ikaros in lymphocyte development and the emerging data linking dysfunctional Ikaros with cellular survival, a more precise mechanistic understanding of the role of Ikaros in leukemia may identify relevant therapeutic targets. In human leukemia, the myriad of genetic and molecular changes culminate in deregulated signaling of specific intracellular signaling pathways that provide a survival and proliferation advantage, such as JAK-STAT, phosphoinositide 3 kinase (PI3K), and Ras/Raf/mitogen-activated protein kinase (MAPK) [Oh et al. 2010; Rosen et al. 2010; Loh et al. 2009; Mullighan et al. 2009b; Kota et al. 2008]. Further, recent elegant work by Notta and colleagues and Anderson and colleagues has demonstrated the heterogeneous nature of cancer at diagnosis and the evolution of the disease with existence of cell subpopulations, each with differing malignant potential [Anderson et al. 2011; Notta et al. 2011]. This work further highlights the need to understand the mechanism of the malignant potential of each cell subpopulation so that therapy may be targeted not to one leukemia but the ‘many leukemias’ that exist at diagnosis, during therapy or at relapse [Notta et al. 2011]. Applying this concept to leukemia harboring dysfunctional Ikaros, it would therefore be informative to interrogate intracellular signaling in single cells to uncover the biological networks which mediate disease in these heterogeneous populations. Recently, multiparameter flow cytometry was utilized to measure changes in the phosphorylation status of intracellular signaling proteins in primary leukemic samples in response to an extracellular stimulus. Measurements of the evoked signaling within single cells was shown to be more informative to address a variety of clincial questions than measuring basal levels of signaling [Irish et al. 2010, 2008; Kornblau et al. 2010; Rosen et al. 2010; Kotecha et al. 2008]. A new technology has recently been developed using antibodies conjugated to transition metal element isotopes and a time of flight mass spectrometer that allows measurements of over 30 parameters on a single-cell basis [Ornatsky et al. 2010]. Application of this mass cytometry platform to normal human bone marrow and to primary B-cell ALL samples has characterized cell subpopulations based on their intracellular signaling responses at a level of detail not previously possible [Bendall et al. 2011; Davis et al. 2010]. This foundational mass cytometry study sets the stage for future work by which to characterize the changes in signaling networks consequent to loss of Ikaros function in primary leukemia.
Conclusions
Ikaros is a critical transcription factor required for the orderly development of all blood lineages. It serves as a prototype for the diverse functions fulfilled by transcription factors that depend upon cell type and developmental context. The participation of Ikaros in chromatin remodeling provides insight into the mechanisms by which it regulates gene expression outside of standard transcriptional programs in a coordinated fashion. Understanding this regulation may then shed light on which signaling pathways are deregulated as a consequence of alterations in Ikaros function. Overall, elucidation of Ikaros function should provide great insight into its role in normal hematopoietic regulation as well as for guiding therapeutic choices in malignancies characterized by loss of Ikaros function.
Footnotes
Acknowledgements
The author would like to thank W.J. Fantl for critical review and editing of this manuscript.
The author is supported by an Alex’s Lemonade Stand Young Investigator Award and the William Lawrence and Blanche Hughes Foundation.
The author declares no conflicts of interest in preparing this article.
