Abstract
Background
Recently, new direct-acting antivirals (DAAs) are known to eradicate chronic hepatitis C (CHC) virus infection and prevent the progression of liver fibrosis. Liver fibrosis may predispose to liver cirrhosis or hepatocellular carcinoma. We investigated the effect of DAAs on liver fibrosis using non-invasive methods, and evaluated the correlations of these methods.
Methods
We retrospectively analyzed 68 patients with CHC who were treated with DAAs and reached sustained virologic response at 12 weeks post-treatment from January 2016 to October 2018. The degree of liver fibrosis was assessed using serum biomarkers, such as AST-to-platelet ratio index (APRI) and fibrosis-4 (FIB-4) index. Liver stiffness was assessed using two-dimensional shear-wave elastography (2 D-SWE). The pre- and post-treatment serum biomarker levels and SWE findings were evaluated and compared.
Results
A total of 68 patients with CHC were enrolled. The median age was 58 years (52.3–73 years) and 37 patients (54.4%) were female. After treatment, the median APRI was decreased from 0.701 to 0.328 (P < 0.0001), and the median FIB-4 was decreased from 2.355 to 1.860 (P < 0.0001). The median kPa in 2 D-SWE significantly reduced from 6.85 to 5.66 (P = 0.013). APRI and FIB-4 were significantly correlated pre- and post-treatment; however, the correlation between the serum biomarkers and 2 D-SWE was partially significant.
Conclusion
The serum fibrosis biomarkers and liver stiffness on 2 D-SWE were shown to be improved after the treatment with DAAs. Further research including larger number of patients is needed to compare the efficacy of each evaluating method.
Introduction
The global prevalence of Hepatitis C virus (HCV) infection was estimated at 1.0% and the number of patients was estimated at 71 million in 2015. 1 Chronic HCV infection is known to be the leading cause of liver cirrhosis, hepatocellular carcinoma (HCC), and liver-related mortality. 2 The treatment of chronic HCV infection has been changed rapidly due to the new direct-acting antivirals (DAAs), which have superior efficacy in all genotypes and less adverse effects than the previous interferon (INF)-based treatment. 3 The sustained virologic response at 12 weeks post-treatment (SVR12) is defined as the absence of HCV RNA after 12 weeks of antiviral treatment. 4 Within the current decade, HCV can be eliminated in most patients with chronic HCV using DAAs, as assessed by the SVR12. 5 Its eradication reduces the incidence of cirrhosis and HCC, but these cannot be completely prevented in patients with advanced liver fibrosis. 6 , 7 The degree and extent of liver fibrosis is the predictor of disease progression and is related to the need and duration of the antiviral treatment, as well as the choice of treatment regimen.8–10 The SVR after interferon treatment improves the liver fibrosis and reduces all-cause mortality in patients with chronic HCV. 11 , 12 Therefore, the liver fibrosis status in patients cured by DAAs is important for surveillance and management. It has been suggested that liver fibrosis can be partially reversed after DAA therapy. This was confirmed by diminishment of the liver stiffness in transient elastography (TE) after treatment 13 and is probably due to the reduced inflammation and regression of the fibrosis. 14
The gold standard of measuring liver fibrosis is liver biopsy, but this test is known to have several disadvantages,15–17 such as invasiveness, inter-observer variability at pathologic evaluation, inadequate sampling size, and sampling variations. In addition, the dynamic process of liver fibrosis cannot be evaluated by biopsy. 18 Therefore, various non-invasive methods for evaluating liver fibrosis have been used. Non-invasive scoring systems based on laboratory blood tests, such as AST-to-platelet ratio index (APRI) and fibrosis-4 (FIB-4) score are widely used because they are easy to use in clinical practice and are easily reproducible [16]. In recent years, multiple studies have used TE to evaluate the liver fibrosis. 18 Both serum biomarkers and TE have shown equivalent performance to evaluate the liver fibrosis, but TE was superior in detecting cirrhosis. 19 , 20 Shear wave elastography (SWE) can be performed as an additional test during liver ultrasonography and does not require the purchase of additional machines like TE; however, its reproducibility has not been confirmed. 21
Even though it is of great importance to predict the progression of hepatitis C-related liver disease, real-world data evaluating fibrosis change after DAA treatment are limited. In the present study, we assessed the change in liver fibrosis after HCV treatment using various non-invasive measurements and evaluated the correlations of each method. We compared the change in the APRI, FIB-4, and liver stiffness value of SWE after DAA treatment.
Methods
We retrospectively analyzed 68 patients with chronic hepatitis C (CHC) who underwent DAA treatment from January 2016 to October 2018 at the Kyung Hee University Hospital, Gangdong, Seoul, Korea. HCV genotyping was performed in all patients. All patients underwent routine follow-ups at the outpatient clinic during the treatment, and none had failed to complete the treatment because of adverse events. Complete blood count, liver functional tests, and HCV RNA were evaluated before and 12 weeks after the end of treatment. The degree of liver fibrosis was assessed using the APRI and FIB-4, which were calculated using serum indirect markers. The APRI was calculated as AST level (IU/L)/Upper limit of norma ASTl (IU/L)/platelet counts (109/L) × 100, and the FIB-4 index was calculated as age (years) × AST (IU/L)/[platelet count (109/L) × ALT1/2 (IU/L)]. 22 In this study, the lowest and highest cutoff values of advanced fibrosis were set as APRI scores of 1 and 2, respectively. 23 For FIB-4, the lowest and highest cutoff values of advanced fibrosis were defined as FIB-4 scores of 1.45 and 3.25, respectively. 24 , 25 Liver stiffness was assessed using two-dimensional shear-wave elastography (2 D-SWE). The values were expressed in kilopascals (kPa) and were related to the validated MEAVIR fibrosis stages with the following cut-off values: ≤5.48 kPa = F0, 5.48–8.29 kPa = F1, 8.29–9.40 kPa = F2, 9.40–11.9 = F3, and ≥11.9 kPa = F4. SWE was performed by three abdominal radiologists using a C1-6 curved array probe of LOGIQ E9 (GE Healthcare, Chicago, IL, USA). Because this study was a retrospective analysis of the existing administrative and clinical data, informed consent was not required. This study was approved by the Institutional Review Board of the Kyung Hee University Hospital at Gangdong.
Statistical analysis
Statistical significance was defined as P < 0.05 and continuous variables were expressed as the medians (interquartile range or proportions). None of the variables satisfied the normality assumption as confirmed by the Shapiro-Wilk normality test. The Wilcoxon signed rank test was used to analyze the differences among continuous variables at pre-treatment and post-treatment, and the McNemar-Bowker test was used to determine the differences in the dichotomous dependent variables between the paired groups. The relationships between the variables were assessed using the Spearman’s correlation coefficients, which are often expressed as Rho. If the Rho value was 0, there was no correlation between the comparison groups, and the closer to 1, the stronger the correlation. All statistical analyses were performed with SPSS software version 24.0 (SPSS Inc., Chicago, IL, USA) and R version 3.5.1 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Patients
The baseline characteristics are shown in Table 1. The median age of the 68 patients was 58 (interquartile range, 52.3–73.0) years and 31 patients (45.6%) were male (Table 1). Eleven patients (16.2%) had consumed significant amounts of alcohol, and 14 patients (20.6%) were smokers. Twenty-one patients (30.9%) had hypertension and 12 (17.6%) had diabetes mellitus. None of the patients had a co-infection with hepatitis B virus (HBV) or human immunodeficiency virus (HIV), or a history of hepatocellular carcinoma. There were none post-liver transplant patients. Eighteen patients (26.4%) had compensated cirrhosis, and none had Child Pugh score C. Eight patients (11.8%) had received previous HCV treatment by pegylated interferon or interferon and ribavirin. Among the 48 patients with genotype 1 b, 29 received asunaprevir plus daclatasvir for 24 weeks, 15 received elbasvir plus grazoprevir for 12 weeks, 3 received ombitasvir plus paritaprevir plus ritonavir and dasabuvir for 12 weeks, and one received ledipasvir plus sofosbuvir for 12 weeks. All 18 patients with genotype 2a received sofosbuvir plus ribavirin, 14 of whom were treated for 12 weeks and 4 for 16 weeks. One patient with genotype 1a received ledipasvir plus sofosbuvir for 12 weeks and one patient with genotype 3 received daclatasvir plus sofosbuvir for 12 weeks.
Baseline patient characteristics.
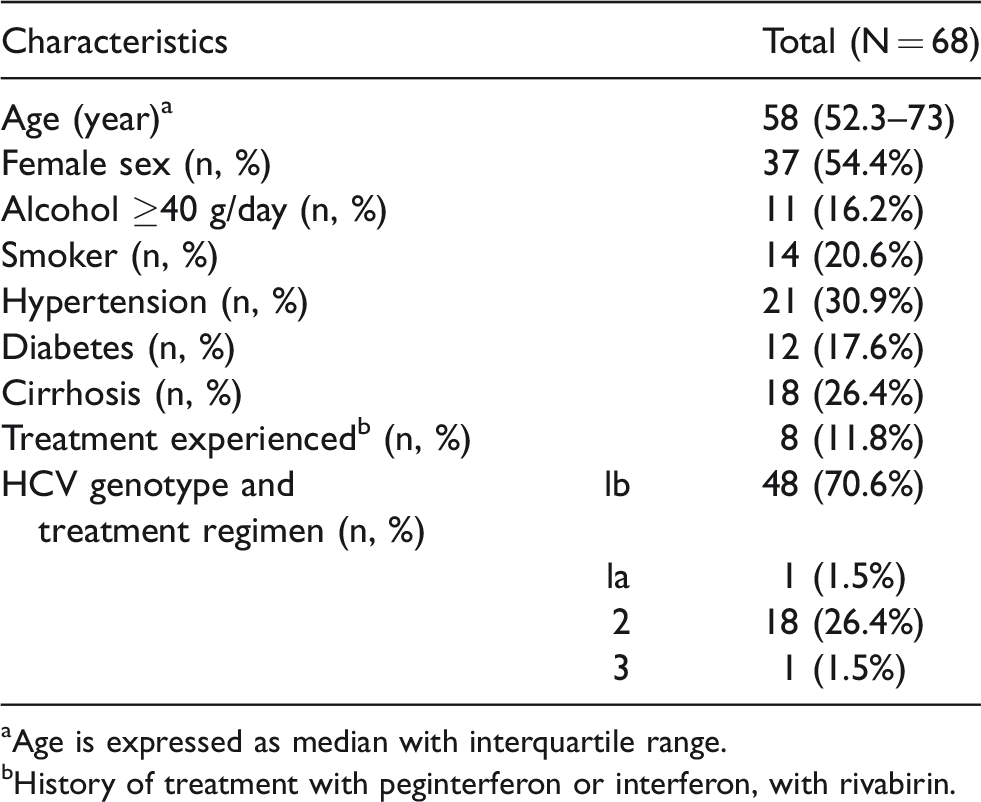
aAge is expressed as median with interquartile range.
bHistory of treatment with peginterferon or interferon, with rivabirin.
Changes of test results after DAA treatment
The laboratory test and SWE results of the patients before and after the HCV treatment are shown in Table 2. Platelet (PLT), Hemoglobin (Hb), and TB (total bilirubin) levels did not show any significant difference pre- and post-treatment. The levels of serum HCV RNA, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) significantly decreased after the HCV treatment. In detail, the serum median levels of AST had decreased from 53.5 (34.8–73.3) IU/L to 23.6 (18.0–29.3) IU/L after treatment, and the serum median levels of ALT had decreased from 52.5 (34.8–82.5) IU/L to 16 (11.0–22.3) IU/L. The median value of HCV RNA before treatment was 1910000 (396000–3600000) log10IU/mL and the HCV RNA measured after treatment was all negative. After the HCV treatment, the APRI was decreased from 0.701 (0.391–1.705) to 0.328 (0.208–0.497, P < 0.0001), and FIB-4 was decreased from 2.355 (1.436–5.095) to 1.860 (1.267–3.391, P < 0.0001). The distribution of calculated fibrosis was determined by the lowest and highest cutoff values of the APRI and FIB-4 and was classified to three groups. According to the APRI score, 22 patients were changed to a lower fibrosis group after the treatment and none were reassigned to a higher fibrosis group (P < 0.001). According to the FIB-4 score, 12 patients were reassigned to a lower fibrosis group after treatment and one patient was reassigned to a higher fibrosis group. (Table 3).
Laboratory and elastography results before and after HCV treatment.
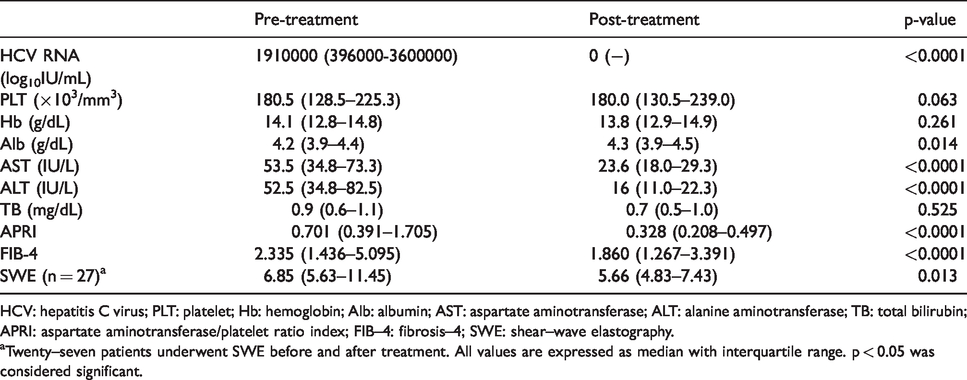
HCV: hepatitis C virus; PLT: platelet; Hb: hemoglobin; Alb: albumin; AST: aspartate aminotransferase; ALT: alanine aminotransferase; TB: total bilirubin; APRI: aspartate aminotransferase/platelet ratio index; FIB–4: fibrosis–4; SWE: shear–wave elastography.
aTwenty–seven patients underwent SWE before and after treatment. All values are expressed as median with interquartile range. p < 0.05 was considered significant.
Changes of fibrosis using APRI and FIB–4 scores after HCV treatment.
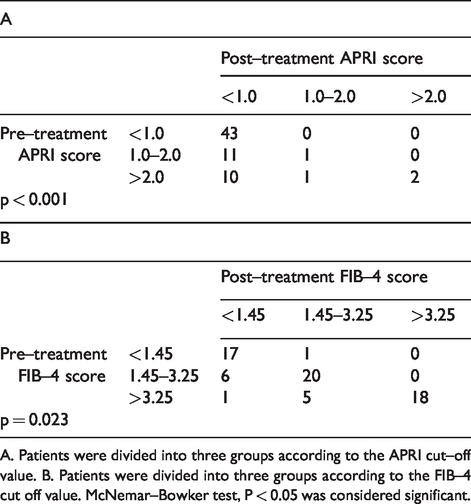
A. Patients were divided into three groups according to the APRI cut–off value. B. Patients were divided into three groups according to the FIB–4 cut off value. McNemar–Bowker test, P < 0.05 was considered significant.
Only 27 patients underwent SWE before and after treatment. The median kPa of SWE significantly reduced from 6.85 (5.63–11.45) to 5.66 (4.83–7.43, P = 0.013). Figure 1 shows the changes of liver stiffness using the calculated APRI and FIB-4, and the SWE findings after HCV treatment.
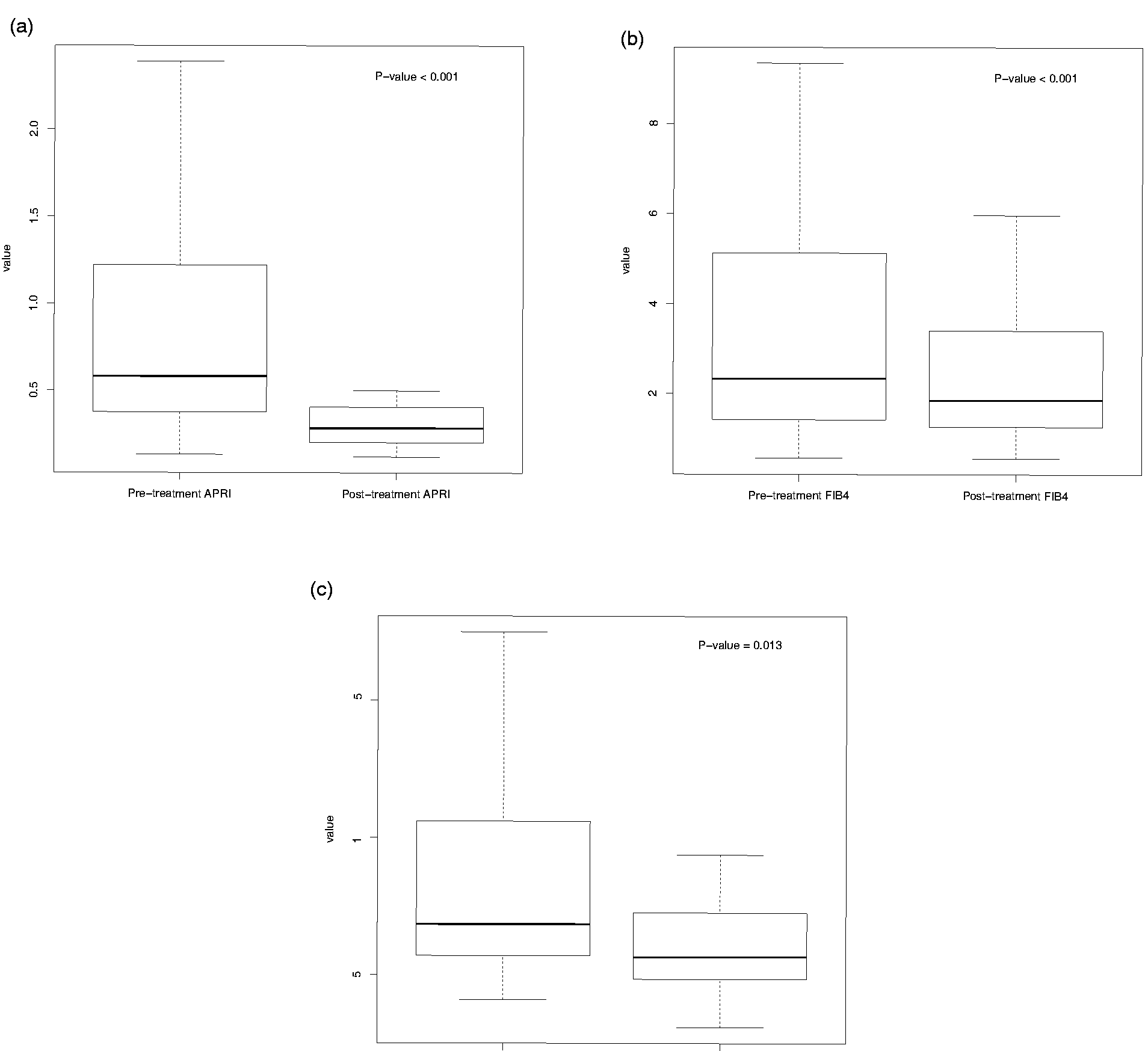
Changes in liver stiffness assessed by non-invasive methods. There were significant differences after the HCV treatment. (a) Changes of APRI, before and after treatment. (b) Changes of FIB-4, before and after treatment. (c) Changes of SWE, before and after treatment. The middle box represents the interquartile range of the study group, and the bottom and top of each box mean the first and third quartiles. The middle lines in the box represent the median values. The upper and lower whiskers represent the scores outside the middle 50%. APRI: aspartate aminotransferase/platelet ratio index; FIB-4: fibrosis-4; SWE: shear-wave elastography.
We assessed the correlation between the APRI, FIB-4, and SWE as indicators of the liver fibrosis before and after treatment (Figure 2). Regarding the correlation between the serum biomarkers, the pre-treatment APRI and pre-treatment FIB-4 showed a statistically significant correlation, as presented in Figure 2(a) (Rho = 0.84, p < 0.001), and the post-treatment correlation was also statistically significant, as presented in Figure 2(b) (Rho = 0.91, p < 0.001).
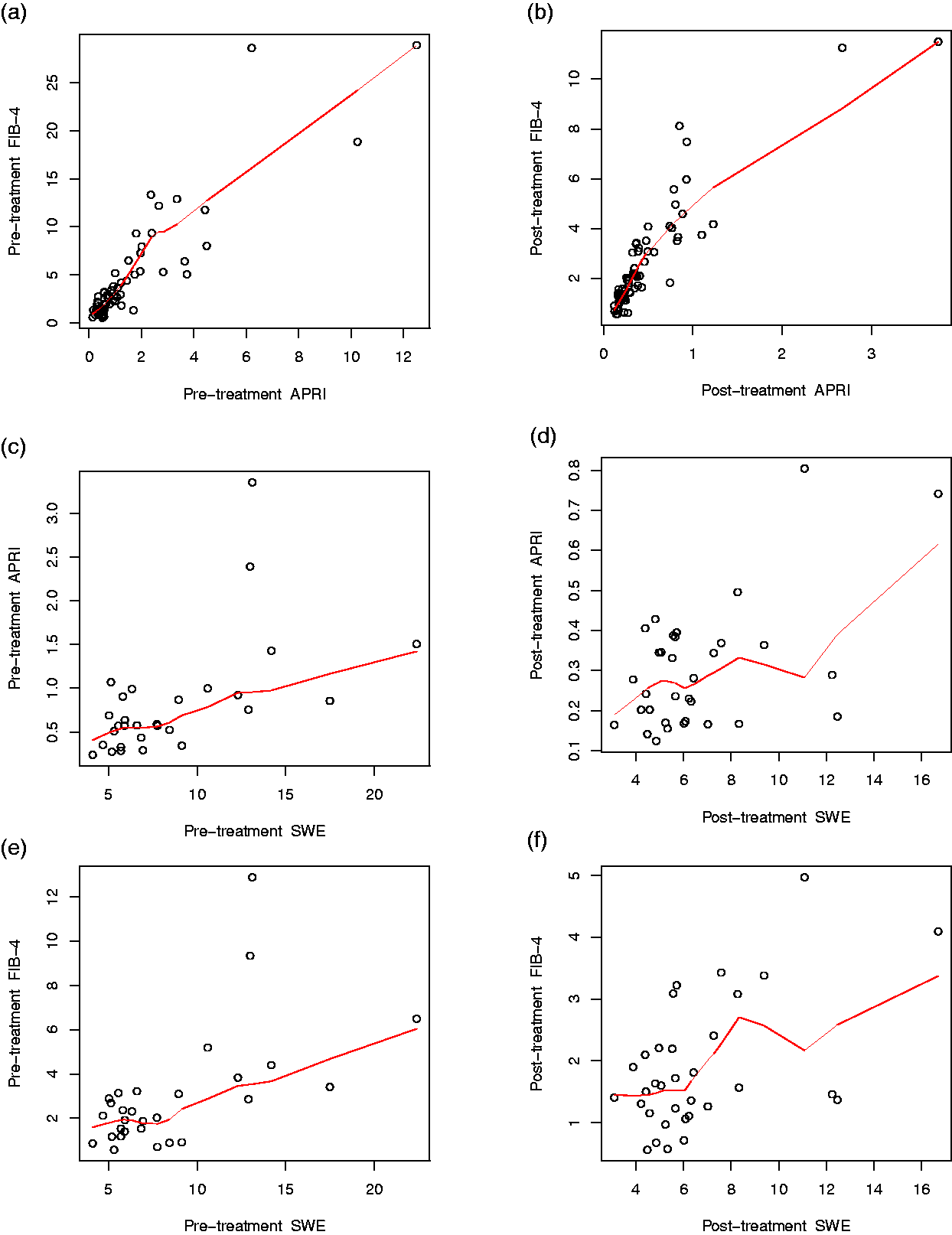
Correlation between the methods to check the fibrosis. (a) Correlation between APRI and FIB-4 before treatment. (b) Correlation between APRI and FIB-4 after treatment. (c) Correlation between APRI and SWE before treatment. (d) Correlation between APRI and SWE after treatment. (e) Correlation between SWE and FIB4 before treatment. (f) Correlation between SWE and FIB4 after treatment. The Rho and p value of each case are as follows: (a) Rho = 0.84, p < 0.001; (b) Rho = 0.91, p < 0.001; (c) Rho = 0.58, p = 0.0009; (d) Rho = 0.27, p = 0.132; (e) Rho = 0.55, p = 0.002; (f) Rho = 0.35, p = 0.048.
Regarding the correlation between the serum biomarkers and SWE, the pre-treatment APRI and SWE (Rho = 0.58, p = 0.0009) were significantly correlated; however, post-treatment APRI and SWE failed to show a significant correlation (Rho = 0.27, p = 0.132). Pre-treatment FIB-4 and SWE were significantly correlated (Rho = 0.55, p = 0.002), and post-treatment FIB-4 and SWE also showed a significant correlation (Rho = 0.35, p = 0.0048).
Discussion
CHC infection causes liver fibrosis, resulting in decompensated cirrhosis and liver cancer, leading to liver-related death. 26 Antiviral therapies can decrease the incidence rate of hepatocellular carcinoma, 27 but not completely eliminate the risk, even in patients who achieved SVR. The patients with an advanced fibrosis had a higher risk of HCC, and HCC surveillance using liver ultrasound with or without alpha-fetoprotein test twice per year are recommended for patients with an advanced fibrosis. 28
Previous studies have shown that the liver fibrosis was improved in patients who underwent interferon (IFN)-based HCV treatment. 11 The IFN therapy seemed to inhibit the hepatic hemostasis and profibrotic cytokines, such as bFGF and TGF-ß1, resulting in anti-inflammatory and antifibrotic effects. 29 Many studies have shown that patients with CHC who achieved SVR after the treatment with DAAs had improvement in terms of liver stiffness. 30 , 31 However, the real-world data using various methods to evaluate the change of fibrosis has not been well studied.
Repeated liver biopsy, before and after treatment, is practically difficult; therefore, non-invasive methods for liver fibrosis evaluation have been used. The non-invasive serum biomarkers and elastography are easy to use and elastography is superior in detecting cirrhosis [19,20]. The liver stiffness is influenced by the liver fibrosis; however, it is also influenced by the necro-inflammatory activity in the liver. 32 Therefore, the improvement of the liver stiffness reflects the improvement of both the liver fibrosis and the necroinflammation of the liver. As a result, it is difficult to accurately quantify the improvement in the liver fibrosis using the SWE or TE methods for measuring the liver stiffness. Factors such as the flare of transaminases, cholestasis, non-fasting, and liver steatosis are further limitations for accurately assessing the liver fibrosis. 33 Additionally, there has been little analysis on how the improvement after DAA treatment could be properly assessed and on the correlation between the methods used to evaluate the fibrosis.
In our study, the overall fibrosis-related test results were improved after DAA treatment. The APRI, FIB-4, and SWE findings showed significant improvement. The severity of the fibrosis as measured by the cutoff values of APRI and FIB-4 scores was also improved after HCV treatment (Table 3).
Only 27 patients underwent SWE before and after treatment; therefore, we assessed the correlation between the APRI, FIB-4, and SWE as indicators of liver fibrosis before and after treatment (Figure 2). The correlation between the serum biomarkers, APRI and FIB-4, was statistically significant both pre- and post-treatment (Figure 2(a) and (b), p < 0.001); thus, any of these biomarkers can be used for evaluation of the liver fibrosis. We also evaluated the correlation between the serum biomarkers and SWE. The pre-treatment APRI and SWE (Rho = 0.58, p = 0.0009) were significantly correlated; however, the post-treatment APRI and SWE failed to show a significant correlation (Rho = 0.27, p = 0.132). Conversely, the FIB-4 and SWE showed a statistically significant correlation both pre and post-treatment (Figure 2(d) and 2(e)).
The limitations of our study are the short duration of the follow-up and the small number of patients who had undergone SWE (27 of 68 patients) before and after treatment. Even after SVR12, fibrosis can be improved, so studies about fibrosis need to be over long period of time. Due to the small number of patients, there was difficulty to prove the exact correlation with other marks. And it is possible that the patients with a high AST level before treatment were included more and the degree of fibrosis of serum biomarkers may be exaggerated. In addition, this was a retrospective study, and we assessed several different regimens and did not include many patients in each regimen.
In conclusion, the improvement in the noninvasive fibrosis panels and elastography seems to be evident in patients after DAA treatment and each method showed a possibility of correlation. In future studies, it is necessary to observe the long-term effect of DAAs on liver fibrosis using various methods including transient elastrography and to establish proper tools that can monitor the change of liver fibrosis after DAA therapy. Furthermore, more studies are needed to further verify the effect of DAA treatment on the incidence of hepatocellular carcinoma.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
