Abstract
Background
Adenovirus causes a number of diseases in human, and can cause serious infection in severely immunosuppressed individuals. Despite the seriousness of adenovirus infection, there is no definitely approved anti-adenoviral therapy. Many studies have shown that compounds derived from medicinal plants have antiviral activity. Therefore, this study evaluated in vitro anti-adenoviral activity of ethanol extract, fractions, and main phenolic compounds of pomegranate peel.
Methods
The ethanol extract of pomegranate peel was prepared with maceration method and fractionated by consecutive liquid/liquid partition. The cytotoxic and anti-adenovirus activities of the extract, fractions, and main phenolic compounds (ellagic acid, punicalagin and gallic acid) were evaluated on Hep-2 cell line using MTT assay. Inhibitory effect on adsorption and post-adsorption phases of the virus replication cycle was also evaluated.
Results
Pomegranate peel extract had a desirable effect against adenovirus with IC50 of 5.77 µg/mL and selectivity index of 49.9. Among the fractions and compounds, the
Conclusion
Pomegranate peel extract, especially its
Introduction
Adenoviruses (ADVs) are non-enveloped double-stranded DNA viruses that are of over 70 types (genotypes). Human ADVs are classified into seven species (A–G). Generally, human ADVs are not highly pathogenic, and are mostly associated with self-contained respiratory infections, hemorrhagic cystitis, and gastroenteritis, particularly in infants and young children. 1 In addition, ocular human ADV infections are among the leading causes of viral conjunctivitis. In immunocompromised patients, such as solid-organ transplant or hematopoietic stem cell transplant recipients, human ADV infections leaf to high morbidity and mortality. 2 In fact, human ADV is reportedly the most increased virus in transplant recipients, especially in pediatric units. 3
No effective therapy or definitely approved drug has yet been developed for ADV associated diseases. Cidofovir and ribavirin are occasionally used in clinics. Although no controlled clinical trial has yet been conducted on the two drugs, they appear to exhibit activity against ADV. 4 Two problems with cidofovir are severe central nervous system side effects and retinal toxicity.4,5 Because of the lack of effective anti-adenoviral drugs, it is necessary to develop alternative anti-adenoviral treatments.
Attempts are being made to develop various novel agents that may be effective against viruses, specifically human ADV. Naturally occurring anti-viral nutrients may be of special importance because they are widely available and may be used as a constituent of the diet to fight diseases, including ADV infection.
The pomegranate, botanically called
It has been observed that many plant polyphenols, such as ellagic acid, catechins, and chlorogenic, caffeic, and ferulic acids, act as antibacterial, antiviral, antioxidant, anti-inflammatory, and antineoplastic agents.10–12 In the present study, we prepared crude ethanol extract and four fractions of pomegranate peel, and then investigated the efficacy of these plant materials and three main phenolic compounds of pomegranate peel (ellagic acid, punicalagin and gallic acid) on human ADV.
Materials and methods
Chemicals and reagents
Ethyl alcohol,
Plant collection, extraction, and fractionation
Cell and virus
Hep-2 (human laryngeal epidermal carcinoma) cells were kindly provided by Pasteur Institute of Iran, Tehran, Iran. The cells were grown in DMEM supplemented with 10% of FBS, 100 µg/mL of streptomycin, 100 UI/mL of penicillin and 0.25 µg/mL amphotericin B at 37 °C with 5% CO2. The same medium containing 1.5% FBS was used for cytotoxicity and antiviral assays. ADV (type 5) was kindly provided by the Faculty of Health, Tehran University of Medical Sciences, Tehran, Iran. Virus stock was prepared by infection of 80% confluent monolayer Hep-2 cells in 75 cm2 culture flasks using DMEM with 1.5% FBS, at 37 °C in 5% CO2. Virus titer was determined by cytopathic effect (CPE) of ADV in Hep-2 cells and was expressed as the 50% tissue culture infective dose (TCID50) per mL.
Cytotoxicity assay
For cytotoxicity assays, 50% cytotoxic concentrations (CC50) of the plant materials were determined using the MTT assay.
14
Briefly, Hep-2 cells were seeded onto 96-well plates at a concentration of 8000 cells/well to a final volume of 100 µL per well. After incubation at 37 °C with 5% CO2 for 24 h, when the cell monolayer was 80% confluent, the cell culture medium of cells was aspirated and washed with phosphate-buffered saline (PBS). Cells were incubated with 200 µL/well of various concentrations of plant materials (in triplicate) and incubated at 37 °C with 5% CO2 for further four days. Cell viability was examined by ability of the cells to cleave the tetrazolium salt in the MTT assay using succinate dehydrogenase mitochondrial enzyme, which develops a formazan blue color product according to a procedure that has been described earlier.
14
Briefly, the supernatants were removed from the wells and 50 µL of an MTT solution (1 mg/mL in PBS) was added to each well. The plates were incubated for 4 h at 37 °C, and 100 µL of DMSO was added to the wells to dissolve the MTT crystals. The plates were shaken on a shaker for 15 min and the absorbance was read by an enzyme-linked immunosorbent assay reader (STATA FAX 2100, USA) at 570 nm wavelength. Data were calculated by the following formula and expressed as the percentage of toxicity: toxicity (%) = (100 − (
Antiviral assay
Antiviral activity of the plant materials was evaluated by testing their inhibitory activity using the MTT assay according to a procedure described previously. 12 Briefly, 100 µL (100 TCID50) of virus suspension was added to at least 80% of confluent HEp-2 cell monolayer in a 96-well plate and incubated at 37 °C for about 2 h to allow virus adsorption. Then, serial two-fold dilutions that had been prepared from non-toxic dose of the plant materials were added and the resulting solution was tested in triplicate. To prepare positive control, cells were infected with the same concentration of virus but without addition of plant materials. To prepare negative or cell control, only DMEM and 1.5% FBS were added to the cells. The plates were incubated at 37 °C in a humidified CO2 atmosphere for four days.
Cell viability was also determined using previously described MTT assay.
14
Data were calculated and expressed as the percentage of inhibition using the following formula: antiviral activity (%) = (
Mode of antiviral activity
To assess the mechanism of antiviral activity, the time-of-addition effect of the plant materials with better anti-adenoviral effects (crude extract,
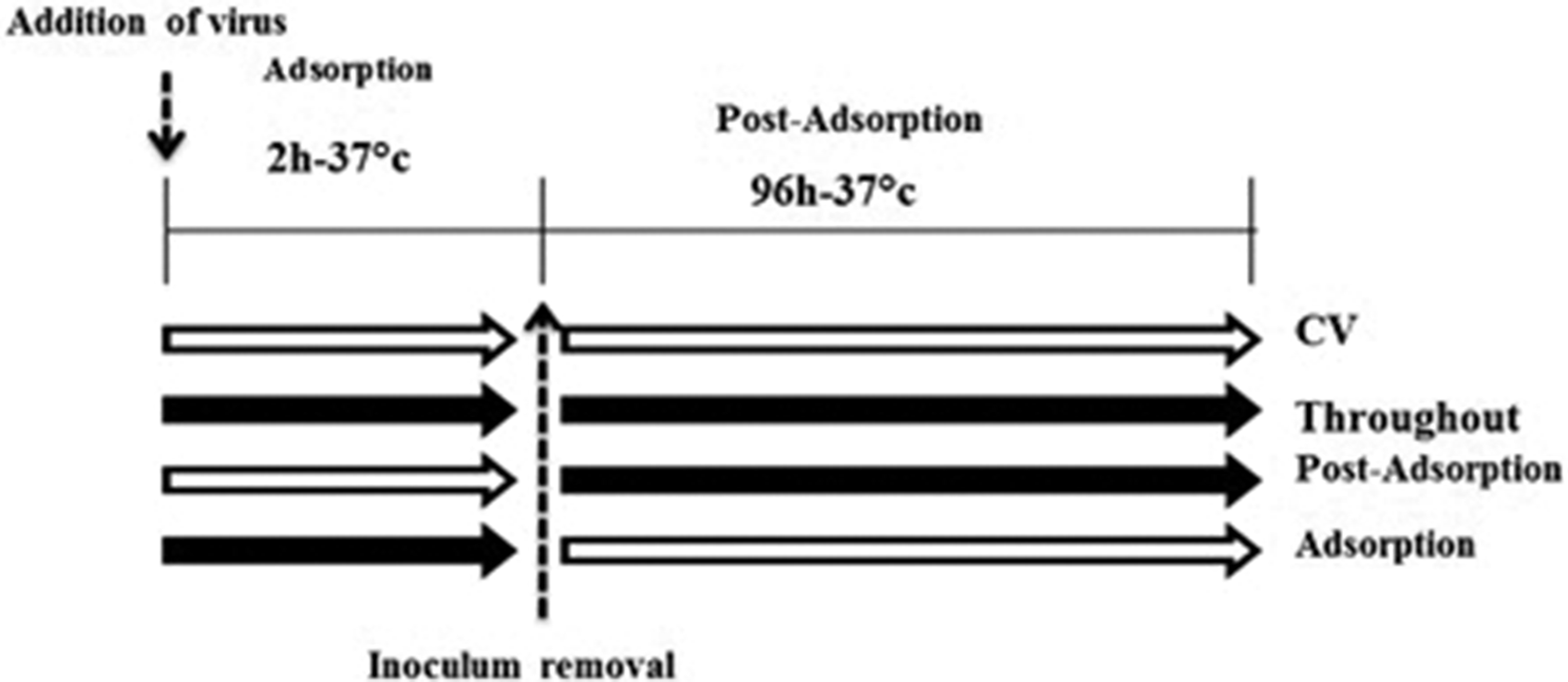
Scheme of addition of plant materials in the adsorption and post-adsorption phases of the virus. Open and black arrows indicate the absence and presence of plant materials, respectively.
Statistical analysis
All experiments were carried out in triplicate. The IC50 and CC50 were calculated using dose-response analyses and related models with probit procedure in the SPSS. Significance level (
Results
Cytotoxicity and antiviral assay
Cytotoxic activities of plant extracts, fractions, and components were evaluated using Hep-2 cell line. The CC50 of pomegranate peel crude extract on Hep-2 cells was 288.2 µg/mL. The ethyl acetate fraction showed the highest cytotoxic activity with a CC50 of 32.4 µg/mL. Other fractions including
Cell cytotoxicity, anti-adenoviral activity, and selectivity index of the crude extract, fractions, and main phenolic compounds of pomegranate peel.
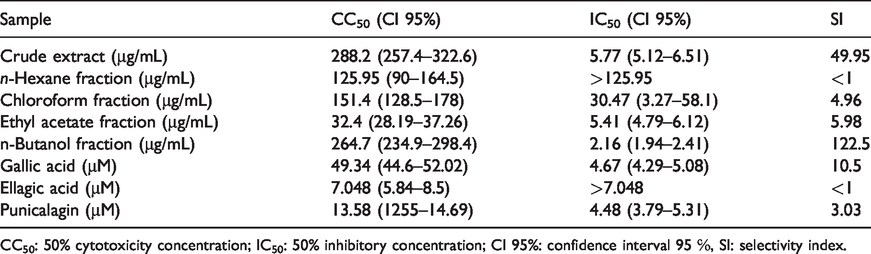
CC50: 50% cytotoxicity concentration; IC50: 50% inhibitory concentration; CI 95%: confidence interval 95 ℅, SI: selectivity index.
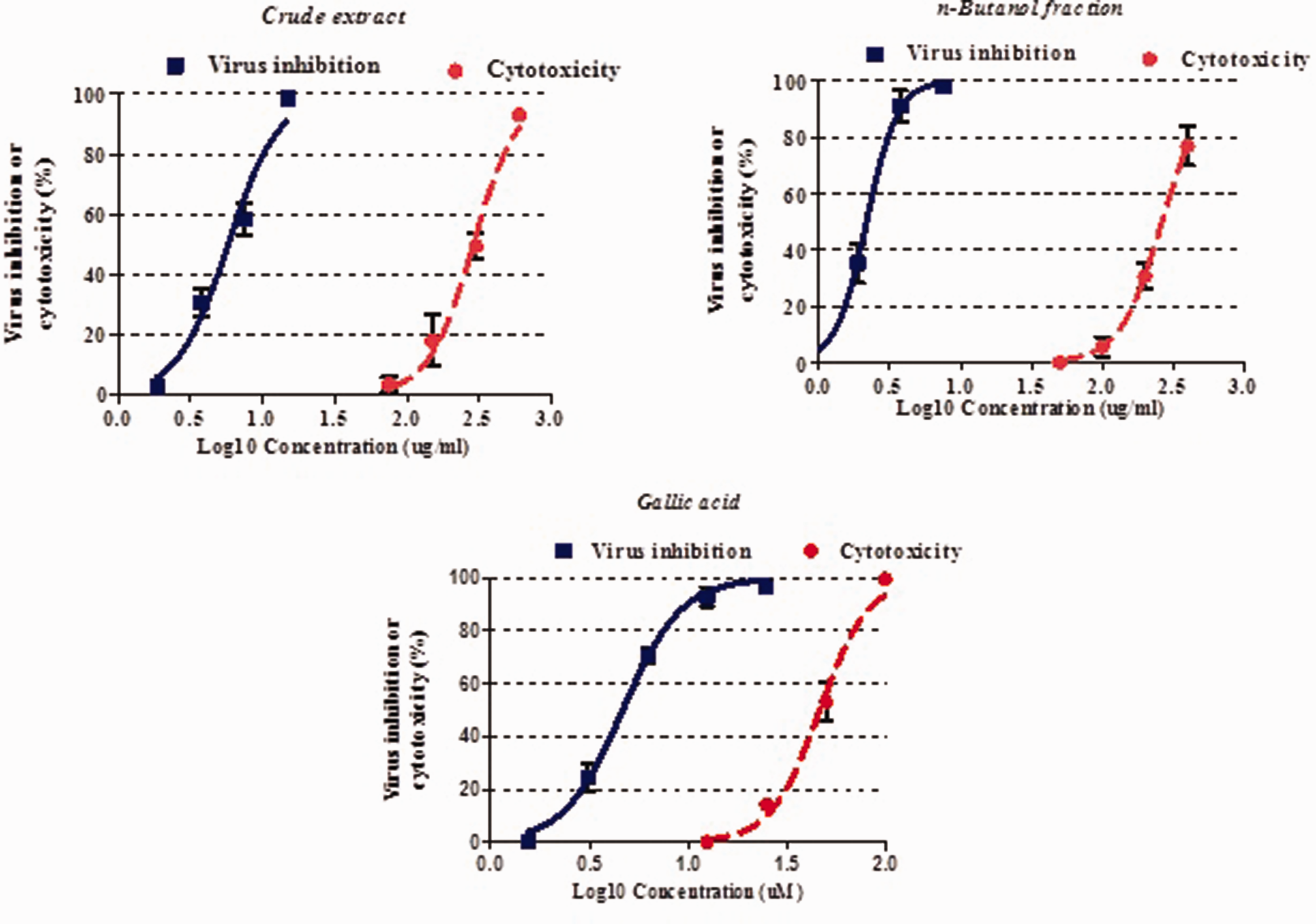
Cytotoxicity and anti-adenoviral activity of crude extract,
The antiviral activity showed that pomegranate peel extract had a desirable effect against ADV with an IC50 of 5.77 (CI 95%: 5.12–6.51) µg/mL, and SI of 49.9. Out of the fractions and compounds, the
Characterization of antiviral activity
Various experiments were carried out with one time IC90 of the plant materials with better anti-adenoviral effects to assess the mechanism of their antiviral action in the adsorption and post-adsorption phases of ADV replication (Figure 3). Our results showed that crude extract,
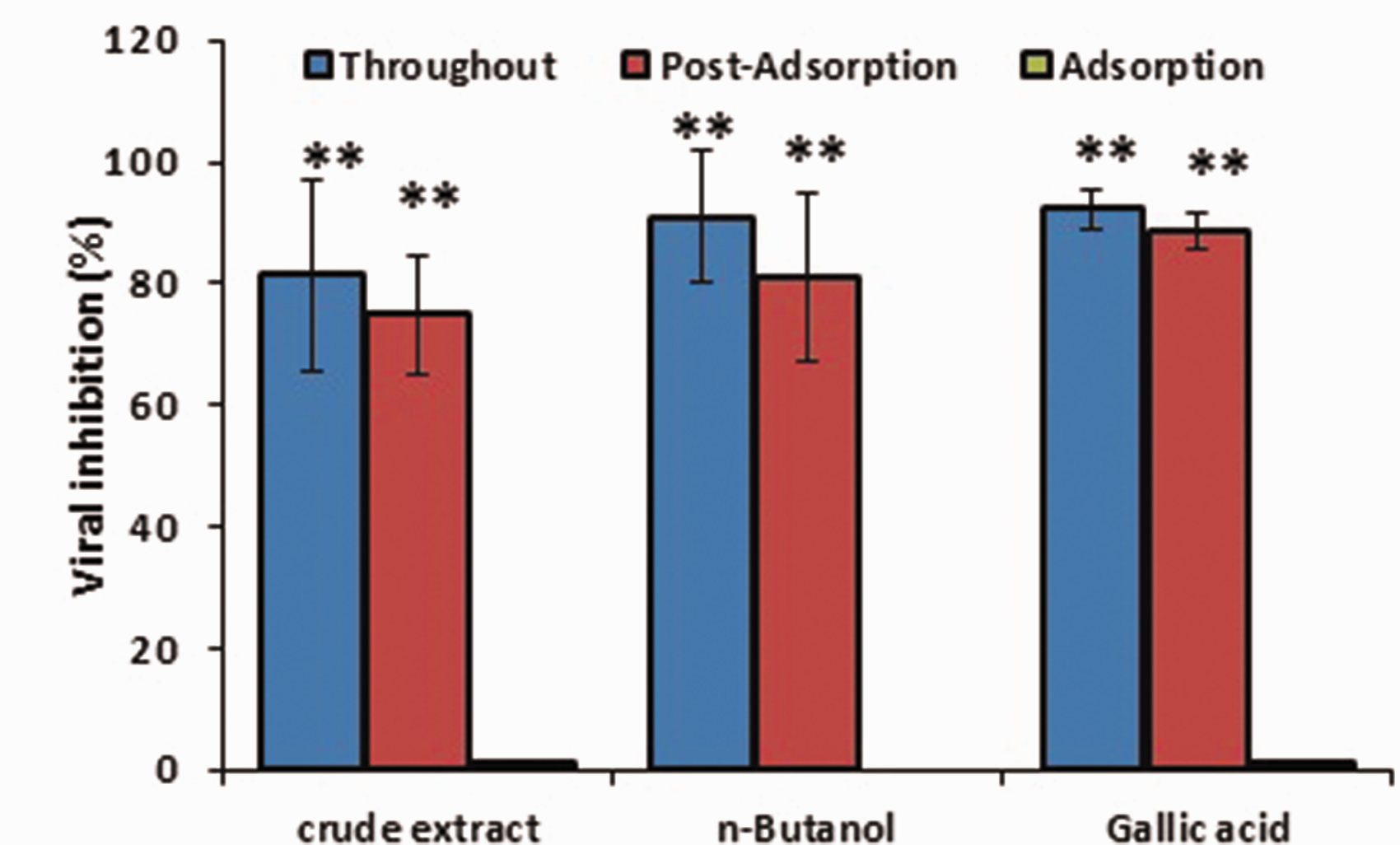
The effect of crude extract,
Discussion
In this study the inhibitory effect of crude extract, fractions, and main phenolic compounds of pomegranate peel on ADV in Hep-2 cell lines were investigated. Our results approved antiviral effect of pomegranate peel extract with IC50 of 5.77 µg/mL and SIs of 49.9. Among the fractions and compounds, the
As the IC50 of the herbal extract against infectious diseases is less than 100 µg/mL,
16
this extract and its
Fifteen percent of pomegranate peel contains phenolic compounds including 8% ellagic acid, 3% punicalagin, and 0.8% gallic acid. 17 According to previous studies, ellagic acid and gallic acid have anti-mutation, antiviral and antioxidant activities. 18 In addition, punicalagin has been reported to exhibit a wide range of biological activities including antiviral effects on human immunodeficiency virus, 19 influenza virus 20 and herpes simplex virus. 21
Because the anti-adenoviral effect of the pomegranate peel
Based on the results of this study, we suggest that in future studies, the amount of phenolic compounds in pomegranate peel fractions be evaluated using accurate methods such as high-performance liquid chromatography. Anti-adenoviral activity of punicalagin, combined with gallic acid, can also be evaluated to investigate its synergistic effect. We hope this study and supplemental research help to discover and produce effective compounds for the treatment of adenoviral diesis.
Various experiments were carried out with one time IC90 of the plant materials with better anti-adenoviral effects to assess the mechanism of their antiviral action in the adsorption and post-adsorption phases of ADV replication. Our results showed that crude extract,
Conclusion
Based on our results, pomegranate peel extract, especially its
Footnotes
Acknowledgments
Authors are thankful to the Director of Medical Plants Research Center and to the Deputy of Research and Technology of Shahrekord University of Medical Sciences, Shahrekord, Iran.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shahrekord University of Medical Science, Shahrekord, Iran (Grant No.: 2297).
