Abstract
With the worldwide number of human immunodeficiency virus positive patients stagnant and the increasing emergence of viral strains resistant to current treatment, the development of novel anti-human immunodeficiency virus drug candidates is a perpetual quest of medicinal chemists. Herein, we report a novel group of diarylpyrimidines, non-nucleoside reverse transcriptase inhibitors, which represents an important class of current anti-human immunodeficiency virus therapy. Series of diarylpyrimidines containing o,o-difluorophenyl (A-arm), 4-cyanophenylamino (B-arm), and a small substituent (e.g. NH2, OMe) at positions 2, 4, and 6 of the pyrimidine ring were prepared. The A-arm was modified in the para position (F or OMe) and linked to the central pyrimidine core with a variable spacer (CO, O, NH). Antiviral activities of 20 compounds were measured against wild type human immunodeficiency virus-1 and mutant reverse transcriptase strains (K103N, Y181C) using a cytoprotection assay. To the most promising structural motives belong the o,o-difluoro-p-methoxy A-arm in position 4, and the amino group in position 6 of pyrimidine. Single digit nanomolar activities with no significant toxicity (CC50 > 17,000 nM) were found for compounds
Keywords
Introduction
Human immunodeficiency virus (HIV), which causes acquired immune deficiency syndrome (AIDS), is one of the greatest threats of the modern age, annually killing one million people worldwide according to the WHO 2016 report. 1 Numbers of AIDS patients are not decreasing in spite of billions of dollars being spent on public education, prevention, and treatment. In economically developed countries, current therapy allows the majority of treated patients to live with AIDS for >10 years, while most untreated patients die within two years of AIDS onset. 2 Additionally, AIDS treatment is particularly challenging due to mutations that yield new viral strains as well as a growing resistance to marketed drugs. 3 Continuing development of new treatments is therefore critical.
Etravirine (ETR)4–6 and rilpivirine (RPV)7,8 are highly potent, FDA approved non-nucleoside reverse transcriptase (RT) inhibitors (NNRTIs) belonging to the diarylpyrimidine (DAPY) class of drugs. (Figure 1).3,9–12 RT is an essential enzyme in the HIV life cycle and DAPY compounds bind to its allosteric hydrophobic site. DAPY analogues, containing a characteristic “horseshoe” or “U-shape” structure, 13 have been studied extensively during the last two decades for their high potency and relatively low cytotoxicity. Due to the rapid emergence of multi-drug resistant HIV strains (e.g. K103N and Y181C for NNRTIs), continuous effort has identified a number of new NNRTI drug candidates.13–15
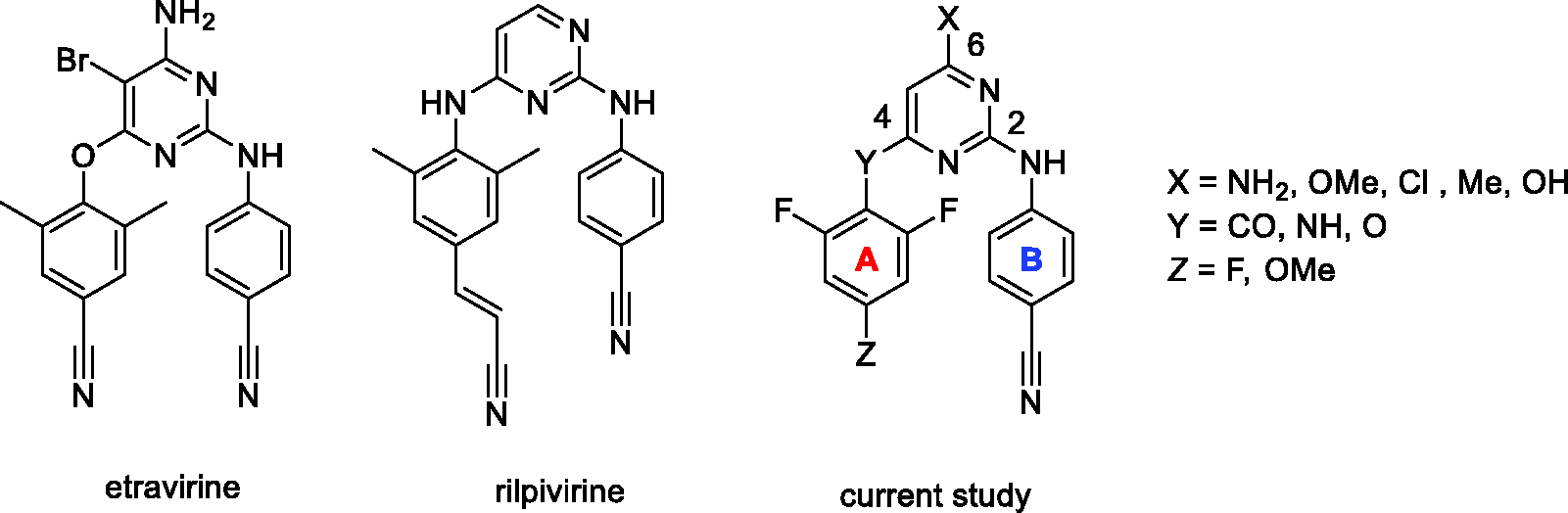
Structures of FDA approved NNRTIs etravirine (ETR) and rilpivirine (RPV), and a general structure of newly synthesized derivatives.
Various structure–activity relationship studies were performed focusing on characterization and optimization of DAPY structural motives important for high anti-HIV potency. 9 The general structure of DAPY analogues consists of a central heteroaryl core (usually pyrimidine) bearing two aryl rings connected using various linkers. Currently, several type of linkers (Y = CO, NH, O) between the pyrimidine core and A-arm are being part of highly potent NNRTIs connecting distinct substitutions on the A and B aromatic rings (Figure 1). Since different ortho substituents lead to the formation of atropoisomers, the A-arm typically contains one para and two identical ortho substituents. The B arm of choice in anti-HIV DAPY research is a 4-cyanophenylamino moiety, attached through position 2 of the pyrimidine ring, as in RPV and ETR.
Well-studied linkers connecting the A-arm to the pyrimidine core include oxygen (ETR) and nitrogen (RPV), which have no or limited capacity for further modifications. Introducing a carbonyl linker has opened new possibilities for expanding this key region of the DAPY structure.
The first published compounds contained an unmodified carbonyl linker, 16 which was later expanded by reacting the linker with hydrazine 17 or hydroxylamine 18 forming Schiff bases. Schiff bases with amines provided, after reduction of the imino double bond, (cyclopropylamino)methylenes 19 or (alkylamino)methylenes. 20 Further types of carbon-based linkers include halomethylene, 21 cyanomethylene, 22 and hydroxymethylene 23 linkers. Additionally, hydroxy(alkyl)methylene analogues were prepared by reacting alkylmagnesium compounds with the carbonyl group. 24 Recently, diatomic linkers for increased conformational flexibility have been described. 25
Our previous work, 26 as well as other published reports, 27 demonstrates that presence of ortho substituents on the A-arm is critical for anti-HIV activity. While RPV and ETR bear two methyl groups, a similar effect on antiviral activity was observed for o,o-difluoro 28 or o,o-dichloro 29 substitution.
Based on literature data, 29 an effective NNRTI DAPY inhibitor consists of a pyrimidine ring bearing a 4-cyanophenylamino arm in position 2 and an o,o-disubstituted phenyl arm connected to position 4 through a short linker.
The goal of our work was to develop novel compounds with increased potency, better resistance profiles, and enhanced drug-like properties to improve the efficacy of current anti-HIV therapy. Our current research focuses on a p-substituted o,o-difluoro A-arm, a standard 4-cyanophenylamino B-arm, preferentially a carbonyl linker, and a substitution at position 6 of the pyrimidine core.
Developing a new synthetic strategy for DAPY methanones in our group 26 gave promising compounds that led us to explore advanced analogues modified in positions 2, 4, and 6 of the pyrimidine ring. Such compounds, which combine an o,o-difluorophenyl A-arm with a carbonyl linker, have not been systematically studied through varying the substituents on the pyrimidine ring. Furthermore, compounds bearing nitrogen and oxygen linkers were also prepared.
Results and discussion
Chemistry
The synthetic procedure for preparation of carbonyl linked DAPYs developed in our group
26
was used to synthesize a 2,4,6-trisubstituted pyrimidine series. Positions 2, 4, and 6 were substituted with 4-cyanophenylamino, o,o-difluorophenylmethanone, and an amino group, respectively. Initially 2,4,6-trichloropyrimidine was reacted with o,o-difluorobenzaldehyde and sodium hydride using N,N′-dimethylbenzimidazolium iodide as a catalyst (Scheme 1).30,31 Two dichloropyrimidine regioisomers were isolated for each aldehyde with the carbonyl group either in the primarily desired position 4 (compounds
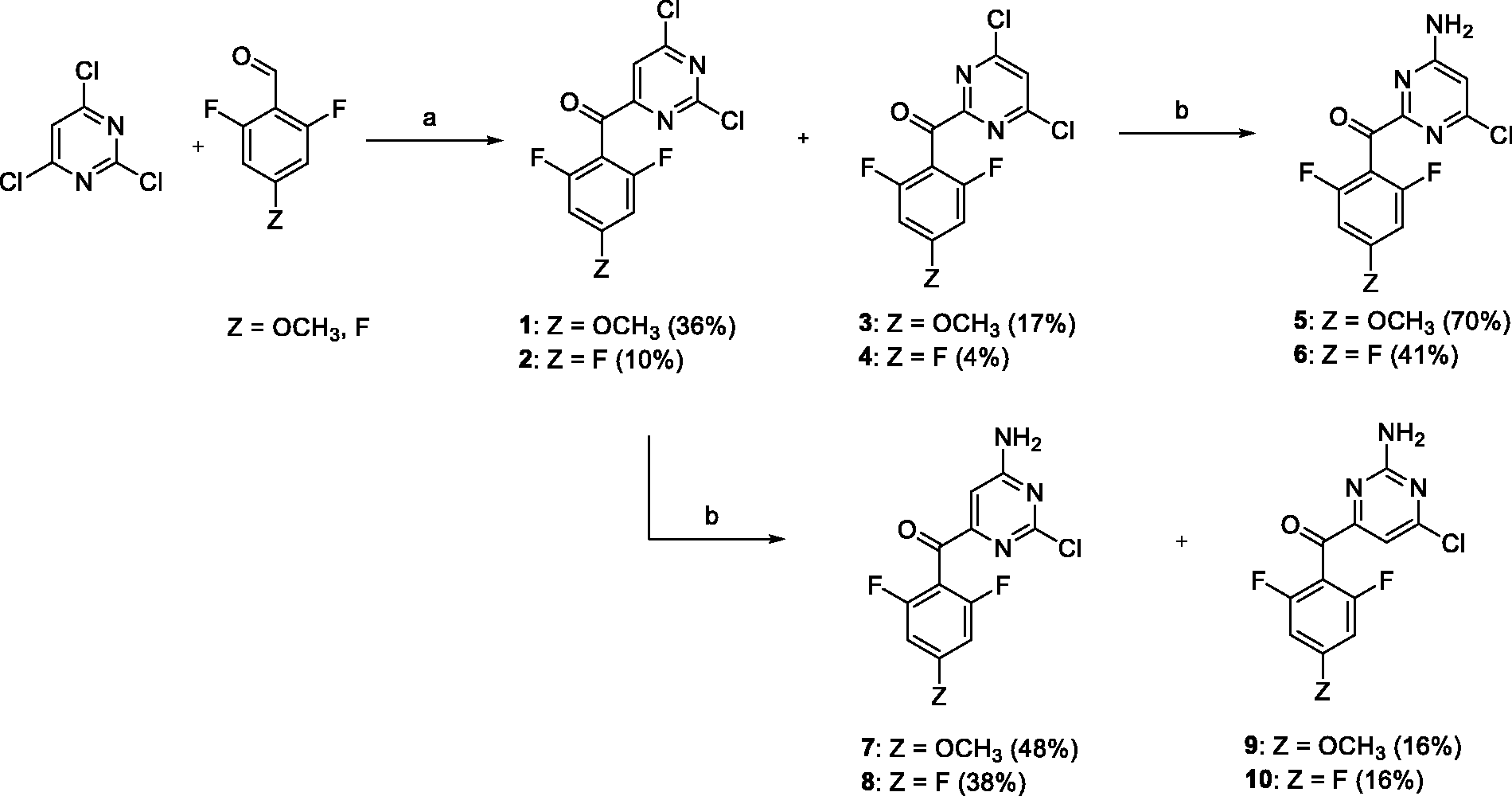
Conditions and reagents: (a) NaH, N,N′-dimethylbenzimidazolium iodide, dioxane, 60°C and (b) ethanolic ammonia (2.5 M), 25°C.
Each monochloropyrimidine derivative was treated with 4-aminobenzonitrile under Buchwald–Hartwig reaction conditions (Scheme 2).
33
Trisubstituted pyrimidine derivatives
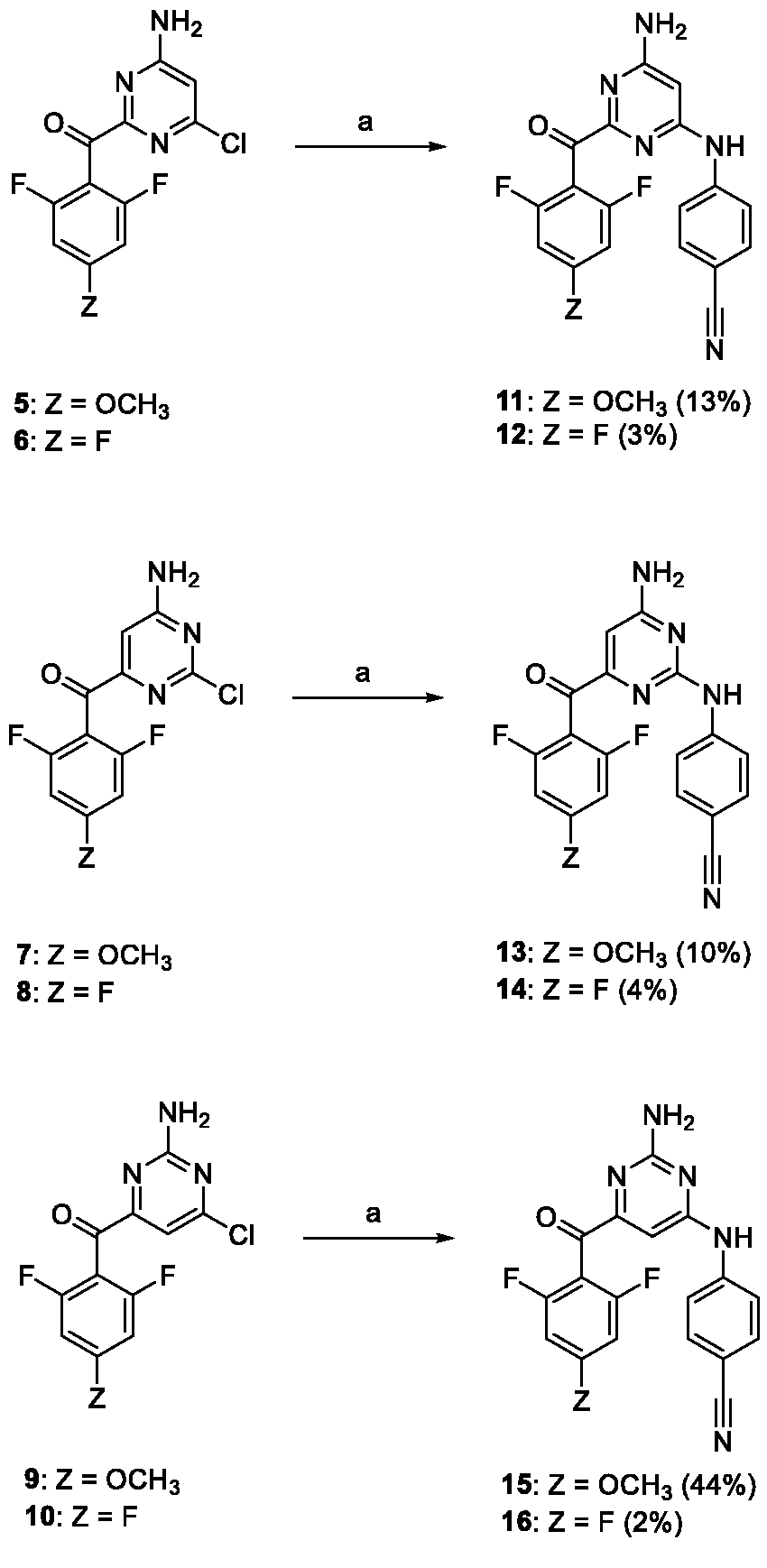
Conditions and reagents: (a) 4-aminobenzonitrile, Pd(OAc)2, XantPhos, Cs2CO3, dioxane, 100°C.
Compounds
Anti-HIV activity (wild type, EC50) and toxicity (CC50) in the MT-4 cell line (n = 3).
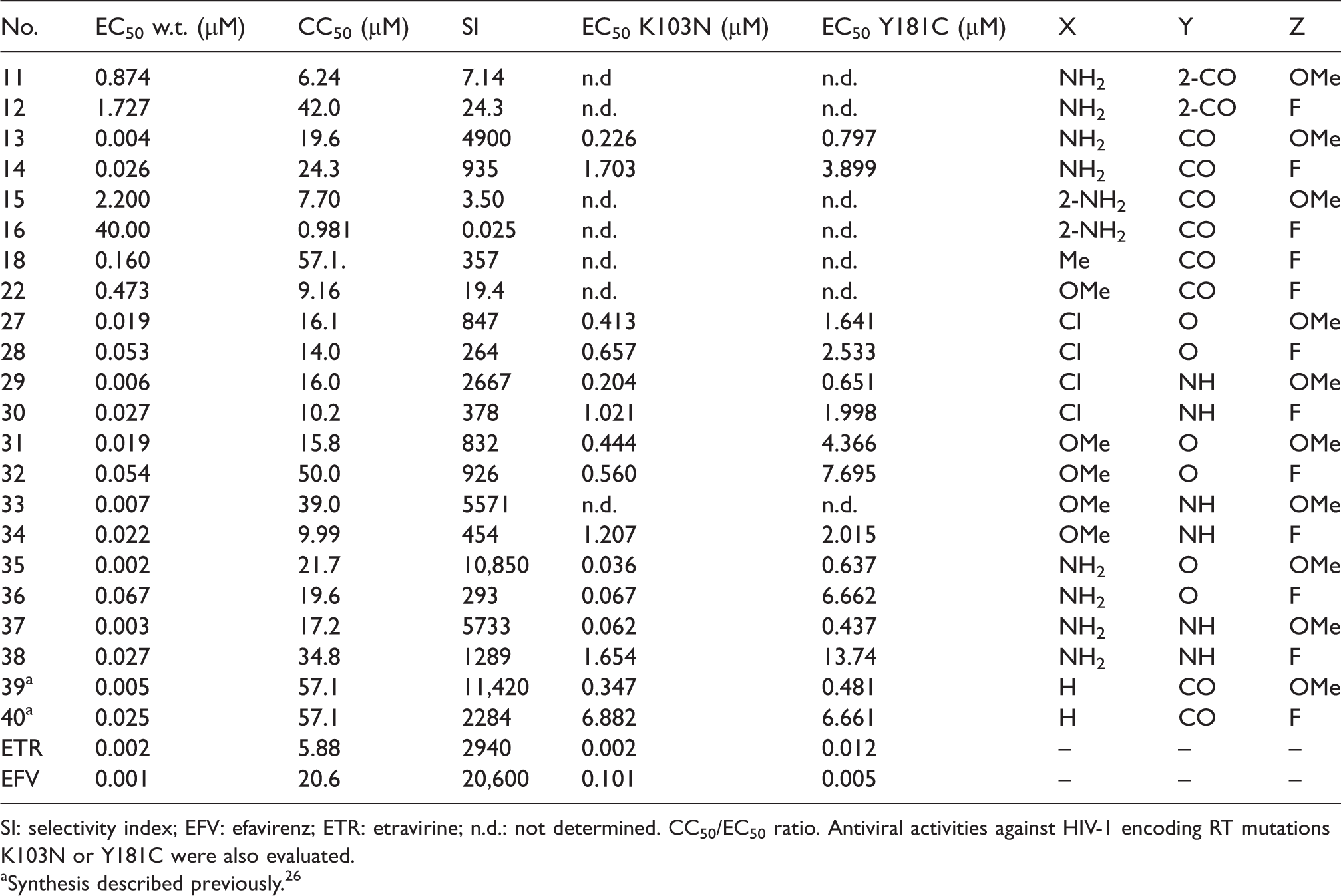
SI: selectivity index; EFV: efavirenz; ETR: etravirine; n.d.: not determined. CC50/EC50 ratio. Antiviral activities against HIV-1 encoding RT mutations K103N or Y181C were also evaluated.
Synthesis described previously. 26
Since derivative
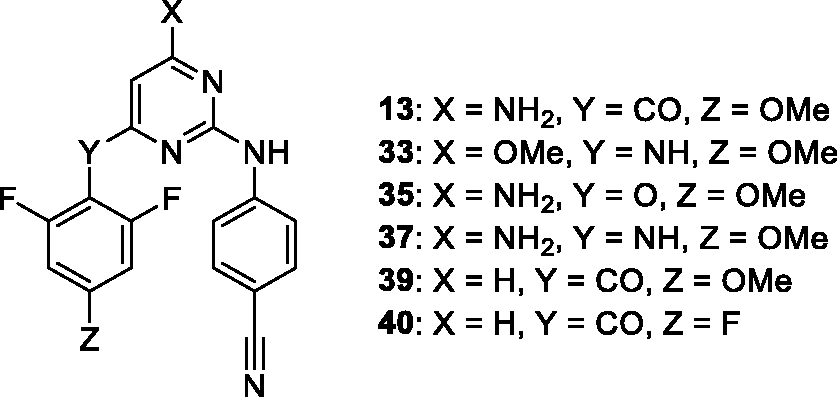
Compounds with low nanomolar anti-HIV activity (wild type). Synthesis of derivatives 39 and 40 was described previously. 26
As the first member of this series, 6-methyl (X = Me) derivative
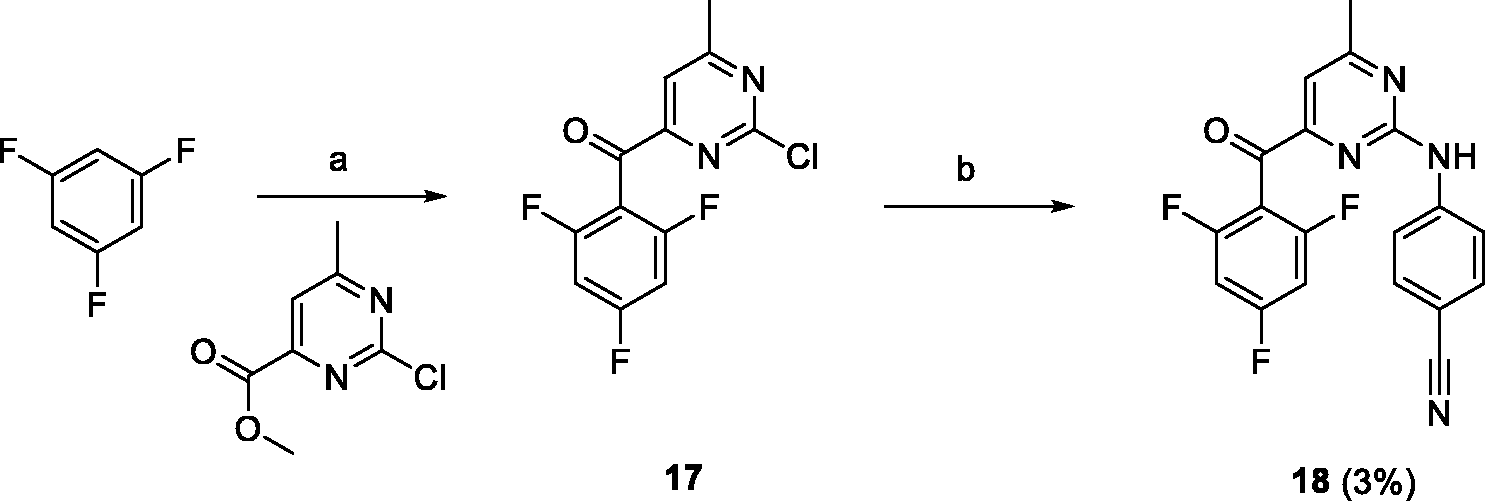
Conditions and reagents: (a) butyllithium, THF, –78°C; (b) 4-aminobenzonitrile, Pd(OAc)2, XantPhos, Cs2CO3, dioxane, 100°C.
To introduce a methoxy group to position 6 of pyrimidine, our procedure
26
was applied to 4,6-dichloro-2-methylthiopyrimidine (Scheme 4). The synthetic sequence consisted of 6-chloro substitution by sodium methoxide, methylsulfide oxidation followed by ammonolysis, and Buchwald coupling of the intermediate
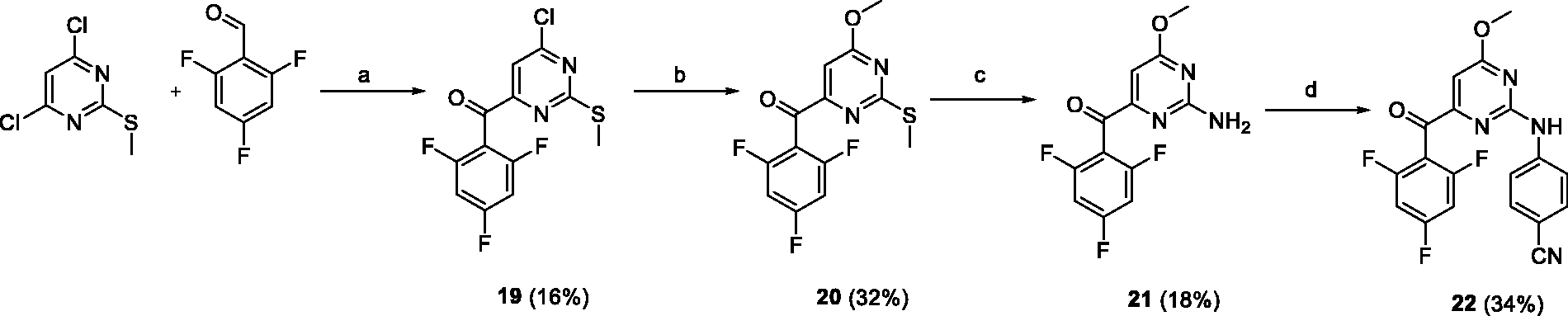
Conditions and reagents: (a) NaH, N,N′-dimethylbenzimidazolium iodide, dioxane, 60°C; (b) MeONa, MeOH, reflux; (c) i: mCPBA, CH2Cl2, 0°C, ii: ethanolic ammonia (2.5 M), 25°C; (d) 4-bromobenzonitrile, Pd(OAc)2, XantPhos, Cs2CO3, dioxane, 80°C.
Overall yields in the carbonyl-linked series were moderate to low, due to the formation of isomers, excessive reactivity of the chlorine atom, and other side reactions.
To compare the influence of the carbonyl linker on anti-HIV potency, another series of DAPY compounds with modified linkers was prepared. ETR and RPV, which have an oxygen and nitrogen linker, respectively, served as an inspiration.
Oxygen and nitrogen linked derivatives (
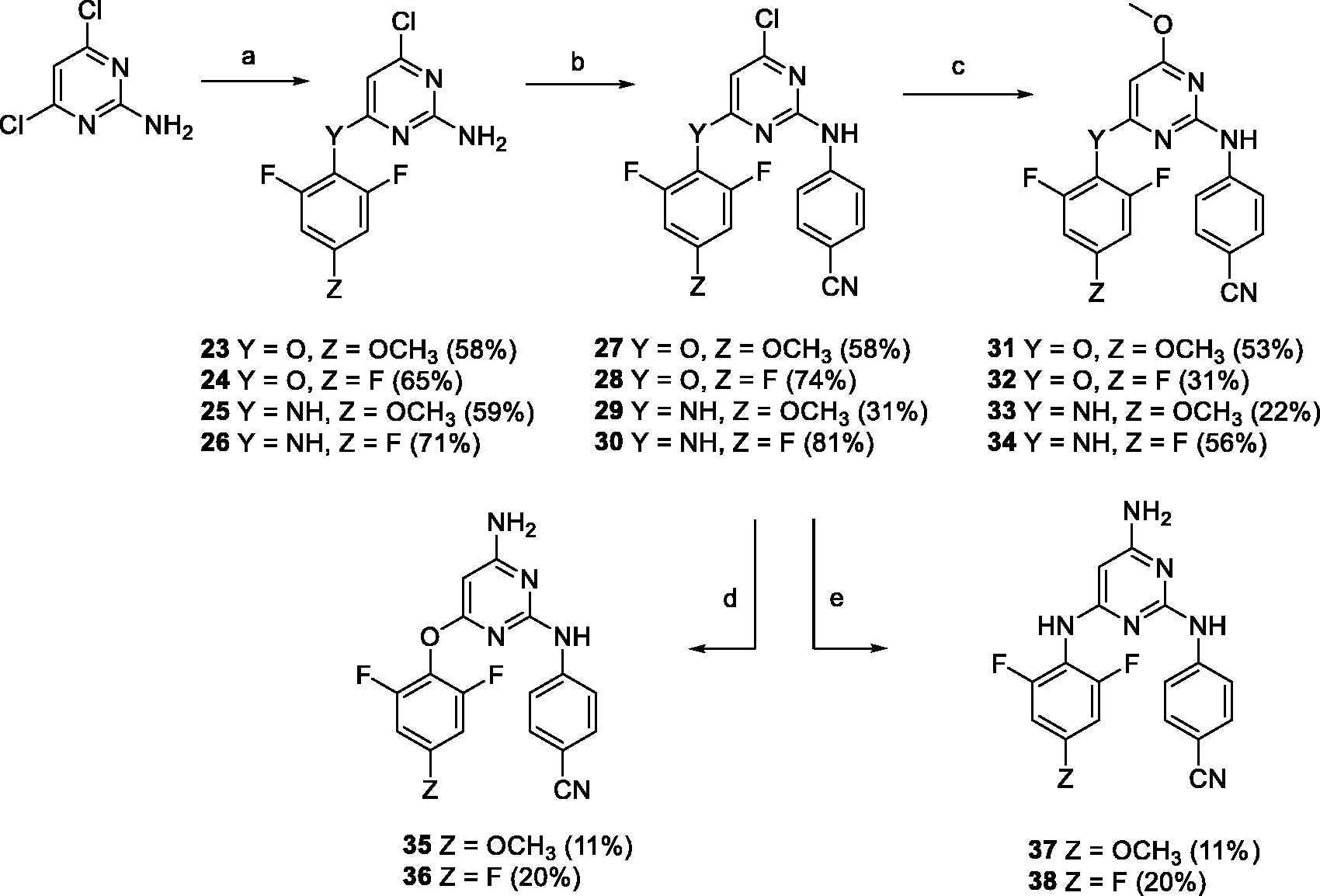
Conditions and reagents: (a) Y = NH, 2,4,6-trifluoroaniline or 2,6-difluoro-4-methoxyaniline, cat. HCl, dioxane, reflux; Y = O, 2,4,6-trifluorophenol or 2,6-difluoro-4-methoxyphenol, Cs2CO3, DMF, 80°C; (b) 4-bromobenzonitrile, Pd(OAc)2, XantPhos, Cs2CO3, dioxane, 80°C; (c) Y = NH, MeONa, MeOH, heating; Y = O, MeONa, MeOH, reflux; (d) i: 4-methoxybenzylamine, Pd(OAc)2, XantPhos, Cs2CO3, dioxane, 80°C; ii: CF3COOH, 25°C; (e) i: NaN3, DMF, MW, 130°C; ii: triphenylphosphine, THF, 25°C, then water, HCl (cat.).
NMR studies
Isomeric compounds bearing the carbonyl bridge in positions 2 and 4 exhibit very similar proton and carbon shifts in their respective NMR spectra. Due to the overall lack of protons in these molecules, unambiguous assignment of carbon signals and structure confirmation was complicated. Based on standard one-dimensional experiments combined with heteronuclear single quantum coherence (HSQC) and heteronuclear multiple bond correlation (HMBC) spectra, it was possible to assign the signals of aromatic substituents on the pyrimidine ring. However, distinguishing between substituent positions on the pyrimidine moiety in isomeric compounds
Biological activity
Entire series of compounds was tested for its in vitro anti-HIV activity (Table 1) using a previously described five-day multi-cycle assay that measures protection from virus-induced cytopathic effects in MT-4 cells acutely infected with HIV-1 (IIIB strain).38–40 Another evidence that studied compounds are specific inhibitors of HIV RT is a X-Ray co-crystal structure of compound
In the CO linker series, polarity of the X substituents directly impacts biological activity. The X = NH2 substituent (compound
This trend was not observed for the O-linked series (Z = F), where compounds with X = Cl (
The influence of the linker (in the Z = F series) is demonstrated for the compounds with X = OMe, where the measured activity for the CO, O, and NH linker is 473 nM (
All compounds with single digit nanomolar activities contain the electron donating p-methoxy group (Z = OMe). Compounds
Two compounds with an oxygen linker (Y = O), where Z = OMe, possessed slightly lower activity for the X = Cl (
Direct comparison of X = NH2 carbonyl-linked compounds
Selected derivatives were also tested against clinically relevant K103N and Y181C HIV RT mutants (Table 1). In all cases, the compounds were less potent against the mutants (36 nM–13.7 µM) than the wild type (2 nM–67 nM) for selected derivatives.
The selectivity index (SI) was calculated for all compounds to evaluate the toxicity–activity ratio (Table 1). The highest SI was found for compounds
In summary, three regioisomers of 2,4,6-trisubstituted pyrimidine were prepared with the A-arm connected via a CO linker. Supported by our previous research, as well as data available for marketed drugs, compounds
Additionally, a series of derivatives with the 2,4,6-trifluorophenyl A-arm (Z = F) was prepared and the influence of the linker (CO, O, NH) and the substitution in position 6 of the pyrimidine core (Cl, OMe, NH2, Me) were investigated. Fluorine analogues (Z = F) had anti-HIV activities ranging from 22 to 55 nM. These data correspond well with the X = H analogue
Compounds bearing the 4-methoxy-2,6-difluorophenyl A-arm showed an increase of approximately an order of magnitude in antiviral activity compared to the 2,4,6-trifluorophenyl series. The best X substituent on the pyrimidine core is the NH2 group with 4.4 nM, 2.9 nM, and 2.3 nM activities for the CO, NH, and O linkers, respectively. Again, demonstrating good agreement with our previously reported X = H analogue
Molecular modeling
Glide XP (Schrodinger 2015) was used to investigate the binding modes of compound
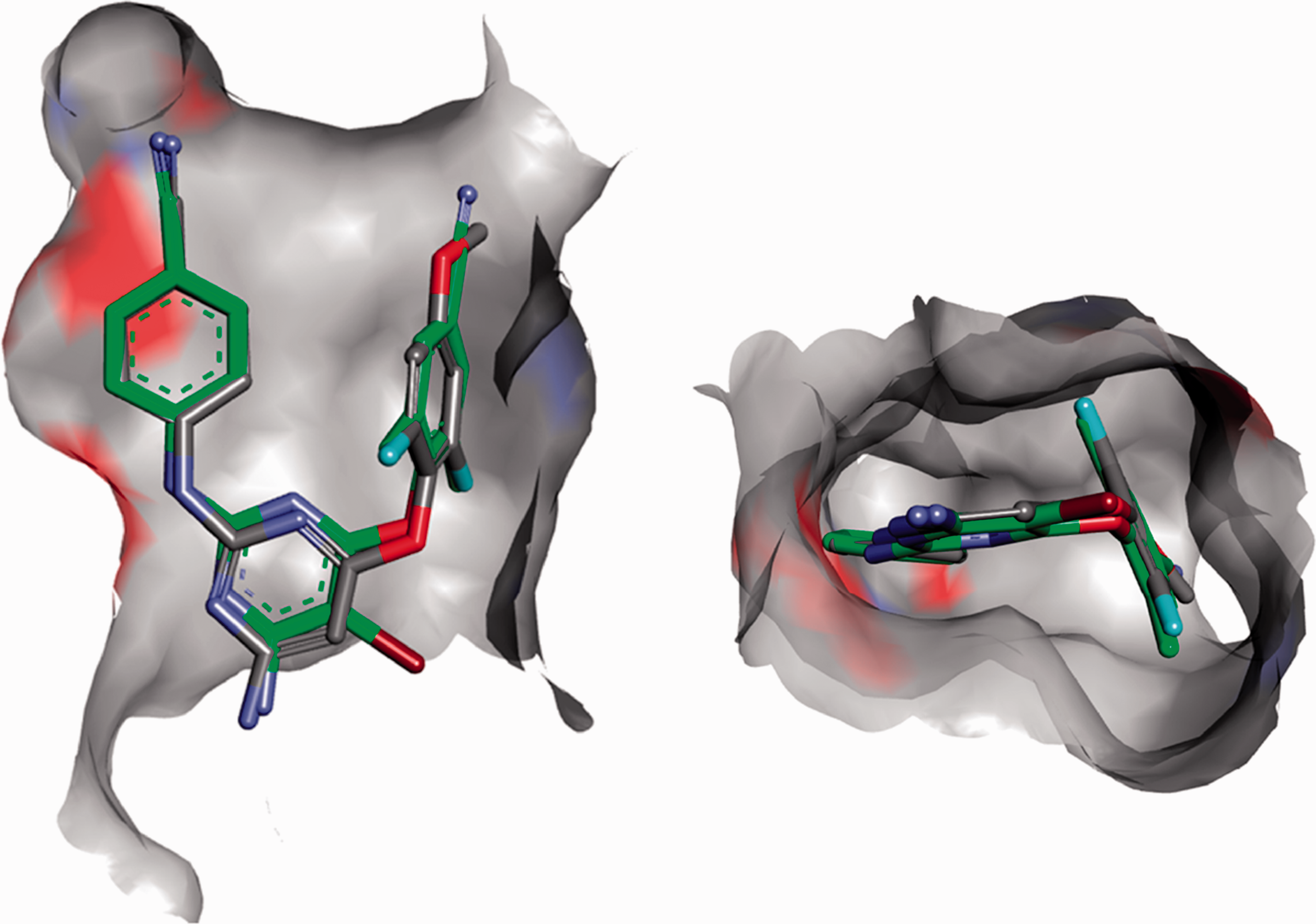
Docking of compound
Conclusions
A new series of trisubstituted DAPY analogues was prepared starting from commercially available trisubstituted pyrimidines and evaluated for their anti-HIV potency against wild type and two clinically relevant mutant strains (K103N and Y181C). Several inhibitors exerted single digit nanomolar potency.
Each compound consists of a small substituent in the position 6 of the central core (Cl, OMe, NH2, Me), 4-cyanophenylamino B-arm and p-substituted o,o-difluorophenyl A-arm connected to the central pyrimidine core through a variable linker (Y = CO, NH, O). Influence of the A-arm para substituent (F, OMe) was also investigated.
Our data show that 4-cyanophenylamino B-arm is indispensable for high antiviral activity and OMe is clearly the best para substituent of the A-arm. Influence of the C-6 substitution of the central core appears to vary based on the linker connecting A-arm to the pyrimidine core. In the case of CO linker, the biological activities dramatically decrease with decreasing polarity of the substituent; however, in the NH and O linker series it had only marginal effect.
Evaluation of the most suitable linker showed rather small impact on the resulting anti-HIV potency in the most active series of compounds. This is very interesting as only the CO linker has any space for further derivatization and it will be investigated in our further work, which will be especially aimed at improving activity against mutants.
Experimental
Chemistry
Chemical reagents and analytical grade solvents were used as received from commercial sources. 1H NMR and 13C NMR spectra were recorded on a Bruker Avance III NMR spectrometer at 600.1 MHz (for 1H) equipped with a 5-mm TCI cryoprobe head in DMSO-d6 (Aldrich, 99.8% D). Chemical shifts are reported in δ, residual solvent peaks (DMSO-d6 1H 2.5 ppm and 13C 39.5 ppm) were used as references. For details concerning the experimental techniques and methods, see the NMR studies section. The following abbreviations were used to describe peak patterns: Py: pyrimidine part of molecule; An: aromatic system bonded to amino group (B-arm); Ar: ortho-substituted aromatic system (A-arm); b: broad; s: singlet; d: doublet, t: triplet. The high-resolution mass spectra (HRMS) were measured on an LTQ Orbitrap XL spectrometer (Thermofisher Scientific) using ESI ionization or GC/TOF-MS GCT Premier (Waters) using EI ionization. Reaction progress was monitored either with thin-layer chromatography (TLC) on silica gel plates and UV visualization at 254 nm or with a Waters UPLC/MS system using a water–acetonitrile gradient (0.1% formic acid as modifier) on Waters BEH 1.7 μ C18 130 Å, 100 × 2.10 mm, flow 0.5 mL/min. Flash chromatography separations were performed on silica gel or C18 modified silica gel (300–400 mesh) using a Teledyne Isco system.
Microwave-assisted reactions were carried out in a CEM Discover (Explorer) microwave aparatus, with a 24-position system for 10-mL vessels sealed with Teflon septa. It was operated at a frequency of 2.45 GHz with continuous irradiation power from 0 to 300 W and IR monitored temperature. The solutions were steadily stirred during the reaction.
All final derivatives were lyofilized and thus isolated as amorphous solids. Derivatives General procedure A: The appropriate benzaldehyde (1 eq.) and N,N′-dimethylbenzimidazolium iodide (0.5 eq.) were dissolved in dry 1,4-dioxane under an argon atmosphere. 2,4,6-Trichloropyrimidine (1 eq.) was added followed by NaH (60% in mineral oil, 3.5 eq.). The reaction mixture was heated at 60°C for 16 h. The solution was added to a mixture of water (100 mL) and ethyl acetate (EtOAc) (100 mL). The layers were separated and the water layer was further extracted with EtOAc (2 × 100 mL). The combined organic phases were dried over sodium sulfate, filtered, and evaporated. The products were isolated by flash silica gel chromatography (gradient from hexane to EtOAc, 0–100%) followed by reverse phase flash chromatography (gradient from water to MeOH, 0–100%). General procedure B: Appropriate dichloro derivative (1 eq.) was dissolved in ethanolic ammonia (2.5 M, 50 eq.), and the reaction mixture was stirred at room temperature for 16 h. The solution was diluted with water (100 mL) and extracted with EtOAc (3 × 50 mL). The combined organic layers were collected, dried over sodium sulfate, filtered, and evaporated. The products were isolated by flash silica gel chromatography (gradient from hexane to EtOAc, 0–100%) followed by reverse phase flash chromatography (gradient from water to MeOH, 0–100%). General procedure C: Appropriate chloro derivative (1 eq.), aniline (1.1 eq.), palladium(II) acetate (0.1 eq.), 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (0.2 eq.) and cesium carbonate (2 eq.) were mixed in dry 1,4-dioxane under argon atmosphere. The reaction mixture was heated at 80°C for 3 h. After cooling to room temperature, the mixture was diluted with water (50 mL) and extracted with EtOAc (3 × 40 mL). The combined organic layers were dried over sodium sulfate, filtered, and evaporated. The product was isolated by flash silica gel chromatography (gradient from hexane to EtOAc, 0–100%) followed by reverse phase flash chromatography (gradient from water to MeOH, 0–100%). General procedure D: Appropriate amino derivative (1 eq.), 4-bromobenzonitrile (1.5 eq.), palladium(II) acetate (0.1 eq.), 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (0.2 eq.) and cesium carbonate (2 eq.) were mixed in dry 1,4-dioxane under argon atmosphere. The reaction mixture was heated at 80°C for 5 h. After cooling to room temperature, the mixture was diluted with water (50 mL) and extracted with EtOAc (3 × 40 mL). The combined organic layers were dried over sodium sulfate, filtered, and evaporated. The product was isolated by flash silica gel chromatography (gradient from hexane to EtOAc, 0–100%) followed by reverse phase flash chromatography (gradient from water to MeOH, 0–100%).
Preparation of (2,6-dichloropyrimidin-4-yl)(2,6-difluoro-4-methoxyphenyl)methanone (1) and (4,6-dichloropyrimidin-2-yl)(2,6-difluoro-4-methoxyphenyl)methanone (3)
Treatment of 2,4,6-trichloropyrimidine (5.26 g, 29 mmol) with 2,6-difluoro-4-methoxybenzaldehyde (4.50 g, 26 mmol) according to general procedure A gave
(
(
Preparation of (2,6-dichloropyrimidin-4-yl)(2,4,6-trifluorophenyl)methanone (2) and (4,6-dichloropyrimidin-2-yl)(2,4,6-trifluorophenyl)methanone (4)
Treatment of 2,4,6-trichloropyrimidine (4 g, 22 mmol) with 2,4,6-trifluorobenzaldehyde (3.52 g, 22 mmol) according to general procedure A gave
(
(
Preparation of (4-amino-6-chloropyrimidin-2-yl)(2,6-difluoro-4-methoxyphenyl)methanone (5)
Treatment of
(
Preparation of (4-amino-6-chloropyrimidin-2-yl)(2,4,6-trifluorophenyl)methanone (6)
Treatment of
(
Preparation of (6-amino-2-chloropyrimidin-4-yl)(2,6-difluoro-4-methoxyphenyl)methanone (7) and (2-amino-6-chloropyrimidin-4-yl)(2,6-difluoro-4-methoxyphenyl)methanone (9)
Treatment of
(
(
Preparation of (4-amino-2-chloropyrimidin-4-yl)(2,4,6-trifluorophenyl)methanone (8) and (2-amino-6-chloropyrimidin-4-yl)(2,4,6-trifluorophenyl)methanone (10)
Treatment of
(
(
Preparation of 4-((6-amino-2–(2,6-difluoro-4-methoxybenzoyl)pyrimidin-4-yl)amino)benzonitrile (11)
Treatment of
(
Preparation of 4-((6-amino-2–(2,4,6-trifluorobenzoyl)pyrimidin-4-yl)amino)benzonitrile (12)
Treatment of
(
Preparation of 4-((4-amino-6–(2,6-difluoro-4-methoxybenzoyl)pyrimidin-2-yl)amino)benzonitrile (13)
Treatment of
(
Preparation of 4-((4-amino-6–(2,4,6-trifluorobenzoyl)pyrimidin-2-yl)amino)benzonitrile (14)
Treatment of
(
Preparation of 4-((4-amino-6–(2,6-difluoro-4-methoxybenzoyl)pyrimidin-2-yl)amino)benzonitrile (15)
Treatment of
(
Preparation of 4-((2-amino-6–(2,4,6-trifluorobenzoyl)pyrimidin-4-yl)amino)benzonitrile (16)
Treatment of
(
Preparation of 4-((4-methyl-6–(2,4,6-trifluorobenzoyl)pyrimidin-2-yl)amino)benzonitrile (18)
A solution of butyllithium (1.6 M in hexanes, 6.1 mL, 9.8 mmol) was slowly added to a precooled (–78°C) solution of 1,3,5-trifluorobenzene (1 mL, 10 mmol) in dry THF (10 mL). The reaction mixture was stirred at –78°C for 2 h, after which a solution of methyl 2-chloro-6-methylpyrimidine-4-carboxylate (1 g, 5.4 mmol) in dry THF (10 mL) was slowly added and this mixture was slowly allowed to reach room temperature. The resulting solution was poured into EtOAc (100 mL) and water (100 mL) mixture, the organic layer was separated, washed with brine (100 mL), and dried over sodium sulfate. Crude intermediate
Treatment of crude
(
Preparation of (6-chloro-2-(methylthio)pyrimidin-4-yl)(2,4,6-trifluorophenyl)methanone (19)
Treatment of 4,6-dichloro-2-(methylthio)pyrimidine (1 g, 5 mmol) with 2,4,6-trifluorobenzaldehyde (0.8 g, 5 mmol) according to general procedure A gave
(
Preparation of (6-methoxy-2-(methylthio)pyrimidin-4-yl)(2,4,6-trifluorophenyl)methanone (20)
Compound
(
Preparation of (2-amino-6-methoxypyrimidin-4-yl)(2,4,6-trifluorophenyl)methanone (21)
A solution of
(
Preparation of 4-((4-methoxy-6–(2,4,6-trifluorobenzoyl)pyrimidin-2-yl)amino)benzonitrile (22)
Treatment of
(
Preparation of 4-chloro-6–(2,6-difluoro-4-methoxyphenoxy)pyrimidin-2-amine (23)
2-Amino-4,6-dichloropyrimidine (1.6 g, 9.8 mmol), 2,6-difluoro-4-methoxyphenol (1.72 g, 10.8 mmol), and cesium carbonate (3.2 g, 9.8 mmol) were dissolved in N,N-dimethylformamide (DMF) (40 mL), and the reaction mixture was heated at 80°C for 1 h. The solution was diluted with water (70 mL) and extracted with EtOAc (3 × 40 mL). The organic layers were collected, dried over magnesium sulfate, filtered, and evaporated under vacuum. Flash silica gel chromatography (gradient from cyclohexane to AcOEt, 0–40%) gave
(
Preparation of 4-chloro-6–(2,4,6-trifluorophenoxy)pyrimidin-2-amine (24)
2-Amino-4,6-dichloropyrimidine (2 g, 12 mmol), 2,4,6-trifluorophenol (1.95 g, 13 mmol), and cesium carbonate (5.9 g, 18 mmol) were dissolved in DMF (50 mL), and the reaction mixture was heated at 80°C for 4 h. The solution was diluted with water (70 mL) and extracted with EtOAc (3 × 50 mL). The organic layers were collected, dried over magnesium sulfate, filtered, and evaporated under vacuum. Flash reverse phase chromatography (gradient from water to MeOH, 0–100%) gave
(
Preparation of 6-chloro-N4-(2,6-difluoro-4-methoxyphenyl)pyrimidin-2,4-diamine (25)
2-Amino-4,6-dichloropyrimidine (500 mg, 3 mmol) and 2,6-difluoro-4-methoxyaniline (525 mg, 3.3 mmol) were dissolved in dioxane (40 mL) and a catalytic amount of HCl was added. The reaction mixture was heated to reflux for 3 h after which it was diluted with water (60 mL) and extracted with EtOAc (3 × 40 mL). The organic layers were collected, dried over magnesium sulfate, filtered, and evaporated under vacuum. Flash reverse phase chromatography (gradient from water to MeOH, 0–100%) gave
(
Preparation of 6-chloro-N4-(2,4,6-trifluorophenyl)pyrimidin-2,4-diamine (26)
2-Amino-4,6-dichloropyrimidine (2 g, 12 mmol) and 2,4,6-trifluoroaniline (1.94 g, 13 mmol) were dissolved in dioxane (50 mL) and a catalytic amount of HCl was added. The reaction mixture was heated to reflux for 16 h, diluted with water (80 mL), and extracted with EtOAc (3 × 50 mL). The organic layers were collected, dried over magnesium sulfate, filtered, and evaporated under vacuum. Flash silica gel chromatography (gradient from CHCl3 to MeOH, 0–15%) gave
(
Preparation of 4-((4-chloro-6–(2,6-difluoro-4-methoxyphenoxy)pyrimidin-2-yl)amino)benzonitrile (27)
Treatment of
(
Preparation of 4-((4-chloro-6–(2,4,6-trifluorophenoxy)pyrimidin-2-yl)amino)benzonitrile (28)
Treatment of
(
Preparation of 4-((4-chloro-6-((2,6-difluoro-4-methoxyphenyl)amino)pyrimidin-2-yl)amino)benzonitrile (29)
Treatment of
(
Preparation of 4-((4-chloro-6-((2,4,6-trifluorophenyl)amino)pyrimidin-2-yl)amino)benzonitrile (30)
Treatment of
(
Preparation of 4-((4–(2,6-difluoro-4-methoxyphenoxy)-6-methoxypyrimidin-2-yl)amino)benzonitrile (31)
Compound
(
Preparation of 4-((4-methoxy-6–(2,4,6-trifluorophenoxy)pyrimidin-2-yl)amino)benzonitrile (32)
Compound
(
Preparation of 4-((4-((2,6-difluoro-4-methoxyphenyl)amino)-6-methoxypyrimidin-2-yl)amino)benzonitrile (33)
Compound
(
Preparation of 4-((4-methoxy-6-((2,4,6-trifluorophenyl)amino)pyrimidin-2-yl)amino)benzonitrile (34)
Compound
(
Preparation of 4-((4-amino-6–(2,6-difluoro-4-methoxyphenoxy)pyrimidin-2-yl)amino)benzonitrile (35)
A mixture of
(
Preparation of 4-((4-amino-6–(2,4,6-trifluorophenoxy)pyrimidin-2-yl)amino)benzonitrile (36)
A mixture of
(
Preparation of 4-((4-amino-6-((2,6-difluoro-4-methoxyphenyl)amino)pyrimidin-2-yl)amino)benzonitrile (37)
Compound
(
Preparation of 4-((4-amino-6-((2,4,6-trifluorophenyl)amino)pyrimidin-2-yl)amino)benzonitrile (38)
Compound
(
MT-4 antiviral and cytotoxicity assay
Compounds were tested in a high-throughput 384-well assay format for their ability to inhibit the virus replication-induced cytopathic effect in MT-4 cell cultures acutely infected with HIV-1 (IIIB strain). Compounds were serially diluted (1:3) in DMSO on 384-well polypropylene plates and further diluted 200-fold into complete RPMI media (10% FBS, 1% P/S) using Biotek Micro Flow and Agilent ECHO acoustic dispenser. Each plate contained up to eight test compounds, with negative (no drug control) and 5 µM AZT positive controls. MT-4 cells were pre-infected with 10 µL of either RPMI (mock-infected) or a fresh 1:250 dilution of an HIV-1 (IIIB) concentrated virus stock. Infected and uninfected MT-4 cells were further diluted in complete RPMI media and added to each plate using a Micro-Flow dispenser. After five days of incubation in a humidified and temperature controlled incubator (37°C), Cell Titer Glo (Promega) was added to the assay plates to quantify the amount of luciferase. EC50 and CC50 values were defined as the compound concentration that causes a 50% decrease in luminescence signal and were calculated using a sigmoidal dose–response model to generate curve fits. 26
Footnotes
Acknowledgement
This study is a part of the research project RVO61388963 of the Institute of Organic Chemistry and Biochemistry, Czech Academy of Sciences. We would like to thank Amberlyn Peterson for language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the subvention for development of research organization (Institute of Organic Chemistry and Biochemistry of the Czech Academy of Sciences, RVO: 61388963).
