Abstract
While noncanonic xanthine nucleotides XMP/dXMP play an important role in balancing and maintaining intracellular purine nucleotide pool as well as in potential mutagenesis, surprisingly, acyclic nucleoside phosphonates bearing a xanthine nucleobase have not been studied so far for their antiviral properties. Herein, we report the synthesis of a series of xanthine-based acyclic nucleoside phosphonates and evaluation of their activity against a wide range of DNA and RNA viruses. Two acyclic nucleoside phosphonates within the series, namely 9-[2-(phosphonomethoxy)ethyl]xanthine (PMEX) and 9-[3-hydroxy-2-(phosphonomethoxy)propyl]xanthine (HPMPX), were shown to possess activity against several human herpesviruses. The most potent compound was PMEX, a xanthine analogue of adefovir (PMEA). PMEX exhibited a single digit µM activity against VZV (EC50 = 2.6 µM, TK+ Oka strain) and HCMV (EC50 = 8.5 µM, Davis strain), while its hexadecyloxypropyl monoester derivative was active against HSV-1 and HSV-2 (EC50 values between 1.8 and 4.0 µM). In contrast to acyclovir, PMEX remained active against the TK– VZV 07–1 strain with EC50 = 4.58 µM. PMEX was suggested to act as an inhibitor of viral DNA polymerase and represents the first reported xanthine-based acyclic nucleoside phosphonate with potent antiviral properties.
Introduction
The concentration and ratio of purine nucleotides and deoxynucleotides in the nucleotide pool is highly regulated in order to maintain the proper function and genetic stability of mammalian cells. 1 Imbalances in (deoxy)nucleotide pool may have mutagenic consequences 2 and may lead to various diseases, such as combined immunodeficiency (loss of purine nucleoside phosphorylase (PNP) function), 3 hyperuricemia (loss of hypoxanthine-guanine phosphoribosyltransferase (HGPRT) function) 4 or cancer (uncontrollable activity of inosine-5′-monophosphate dehydrogenase, IMPDH). 5
Xanthosine monophosphate (XMP,
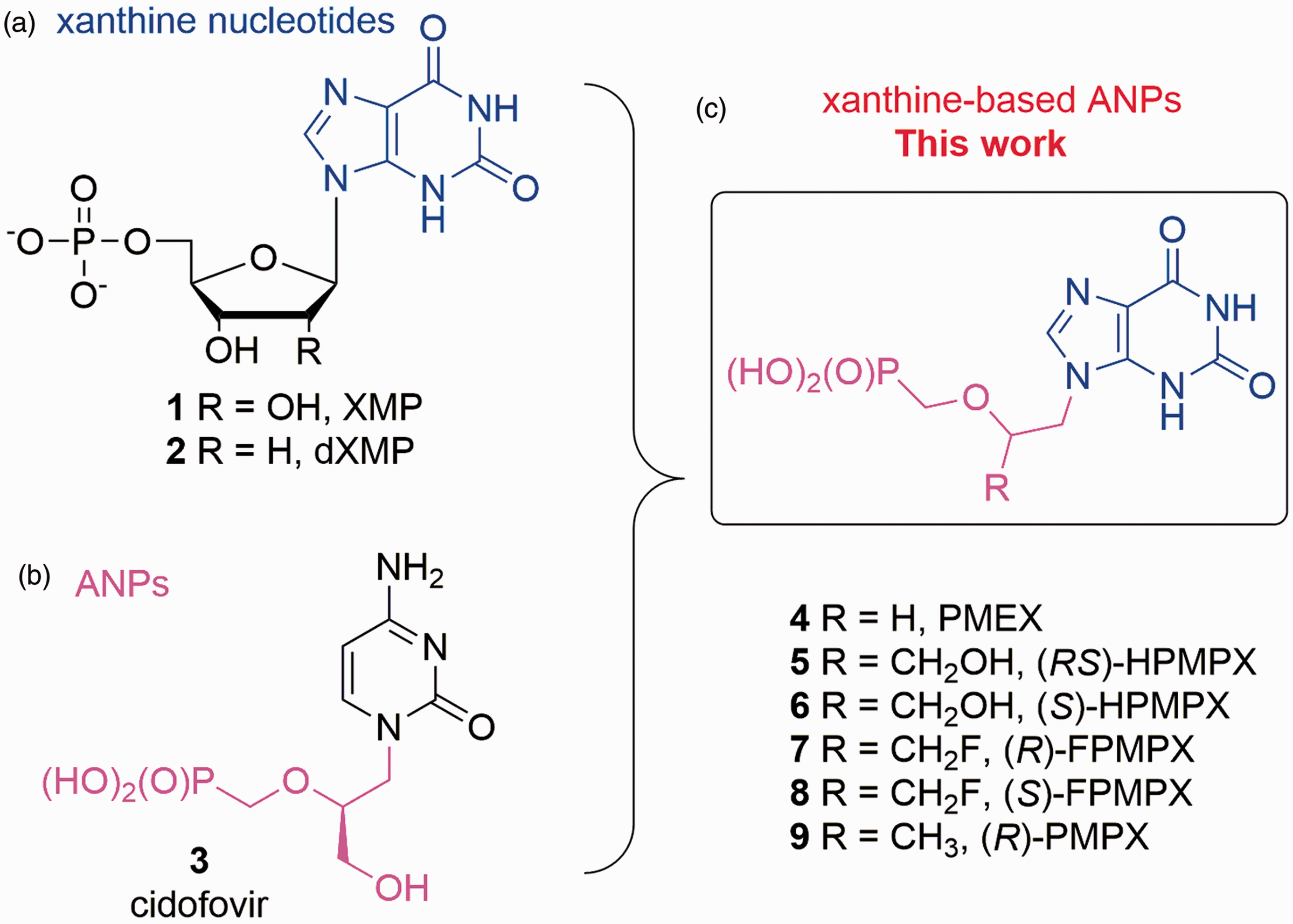
(a) Xanthine-based nucleotides; (b) cidofovir (an example of acyclic nucleoside phosphonate, ANP); (c) target xanthine-based ANPs.
In contrast, the corresponding deoxyribonucleotide analogues, dXMP (
Under cell physiological homeostasis, the concentration and ratio of potentially mutagenic nucleotide intermediates, such as (d)IDP/(d)ITP/(d)XTP, is maintained by housekeeping enzymes, 14 especially those from nudix family such as ITPases/XTPpases,15,16 NUDT 16 or ITPA, 17 that can hydrolyze corresponding nucleoside di- or triphosphates. The main function of housekeeping enzymes is to prevent or minimize the incorporation of noncanonical nucleotides into DNA/RNA. Unfortunately, the literature on housekeeping enzymes hydrolyzing dXDP/dXTP has been quite rare up to date.
Herpesviruses
18
are DNA-containing enveloped viruses from large Herpesviridae family and include herpes simplex virus (HSV), varicella-zoster virus (VZV), cytomegalovirus (CMV), and Epstein-Barr virus (EBV). Although current anti-herpetic therapy uses powerful antivirotics such as nucleoside analogues (acyclovir (ACV), penciclovir, vidarabine, and ganciclovir (GCV)), acyclic nucleoside phosphonate (ANP) cidofovir (CDV) (
ANPs,
25
mimics of natural nucleotides (avoiding the first phosphorylation step), represent a potent group of antiviral agents. ANPs are converted inside the cells to their diphosphates (ANPpp) that target DNA polymerase – viral and/or cellular.
19
These nucleoside triphosphate analogues act as competitive inhibitors and/or alternative substrates of the respective enzymes, in the later case leading to termination of DNA chain elongation.
19
Although some ANPs derived from xanthine were studied before as potential antiviral agents (namely the 9-[3-fluoro-2-(phosphonomethoxy)propyl] derivative, FPMPX),
26
the general lack of interest in such compounds was probably caused by their relatively complicated synthesis, since simple alkylation of xanthine base was expected to give a mixture of several regioisomers as well as polyalkylated products. Recently, we have reported
27
a simple and high-yielding synthesis of xanthine ANPs exploiting the MW-assisted hydrolysis of the corresponding 2,6-dichloropurine derivatives. Here, we report the synthesis and antiviral evaluation of a series of xanthine-based ANPs (compounds
Chemistry
The synthesis of 9-[2-(phosphonomethoxy)ethyl]xanthine (PMEX,
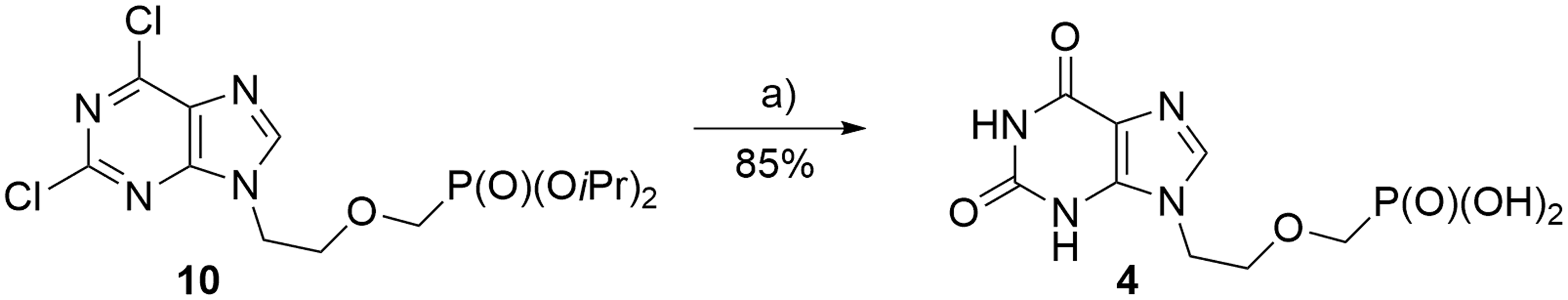
Preparation of PMEX (
For the synthesis of other target ANPs, compounds
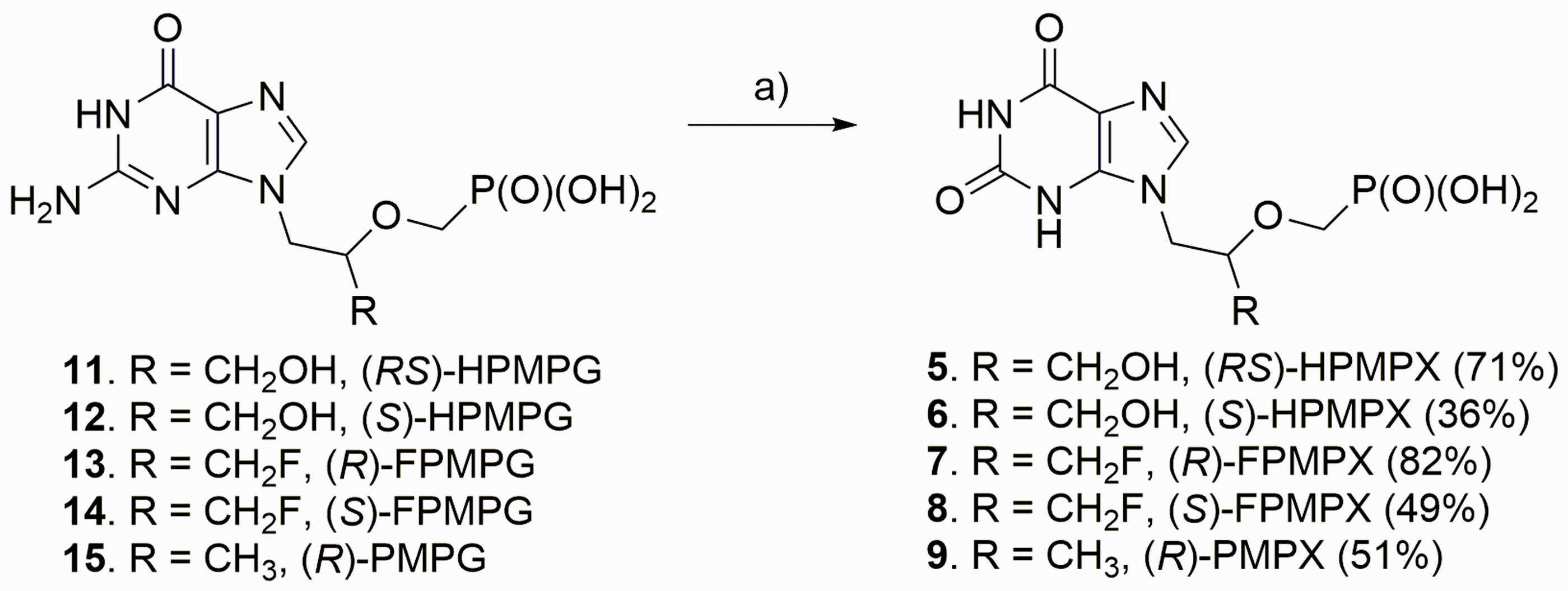
Preparation of xanthine-based ANPs
Since PMEX (
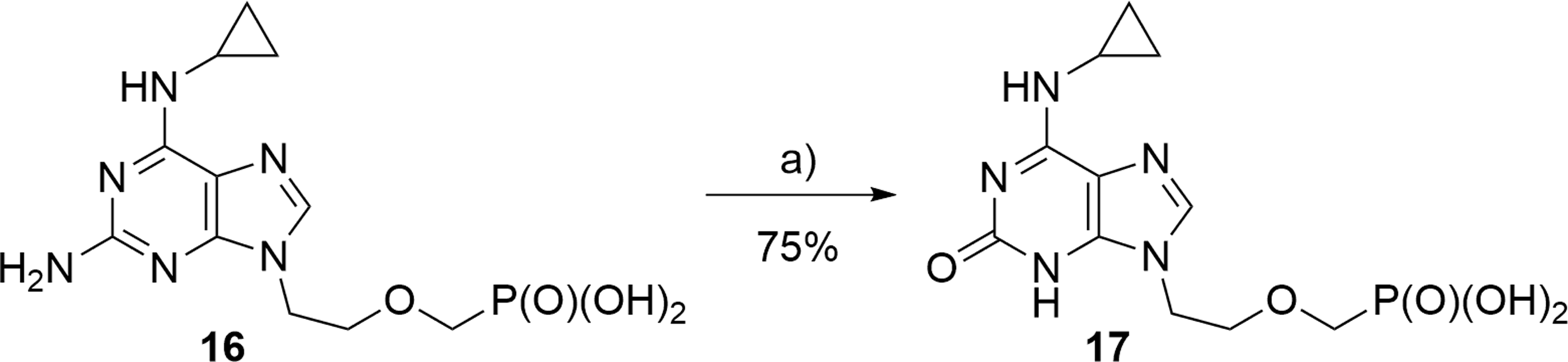
Synthesis of compound
Next, PMEX hexadecyloxypropyl (HDP) monoester
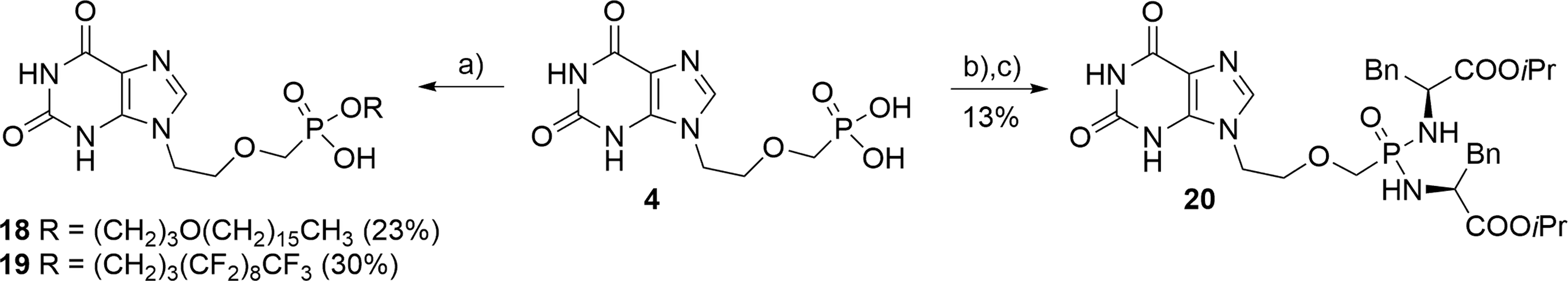
Synthesis of PMEX prodrugs
In order to confirm the expected mode of action of PMEX (
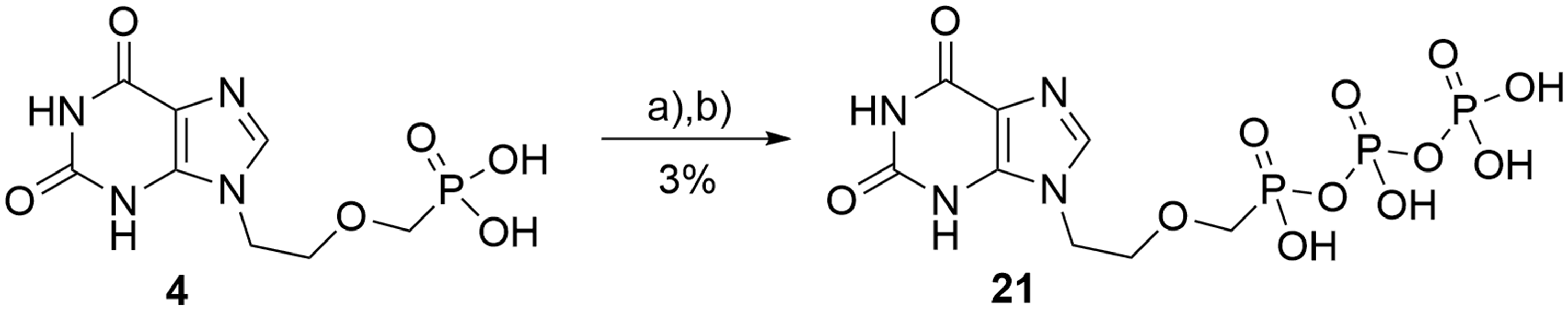
Synthesis of PMEX diphosphate
Biology
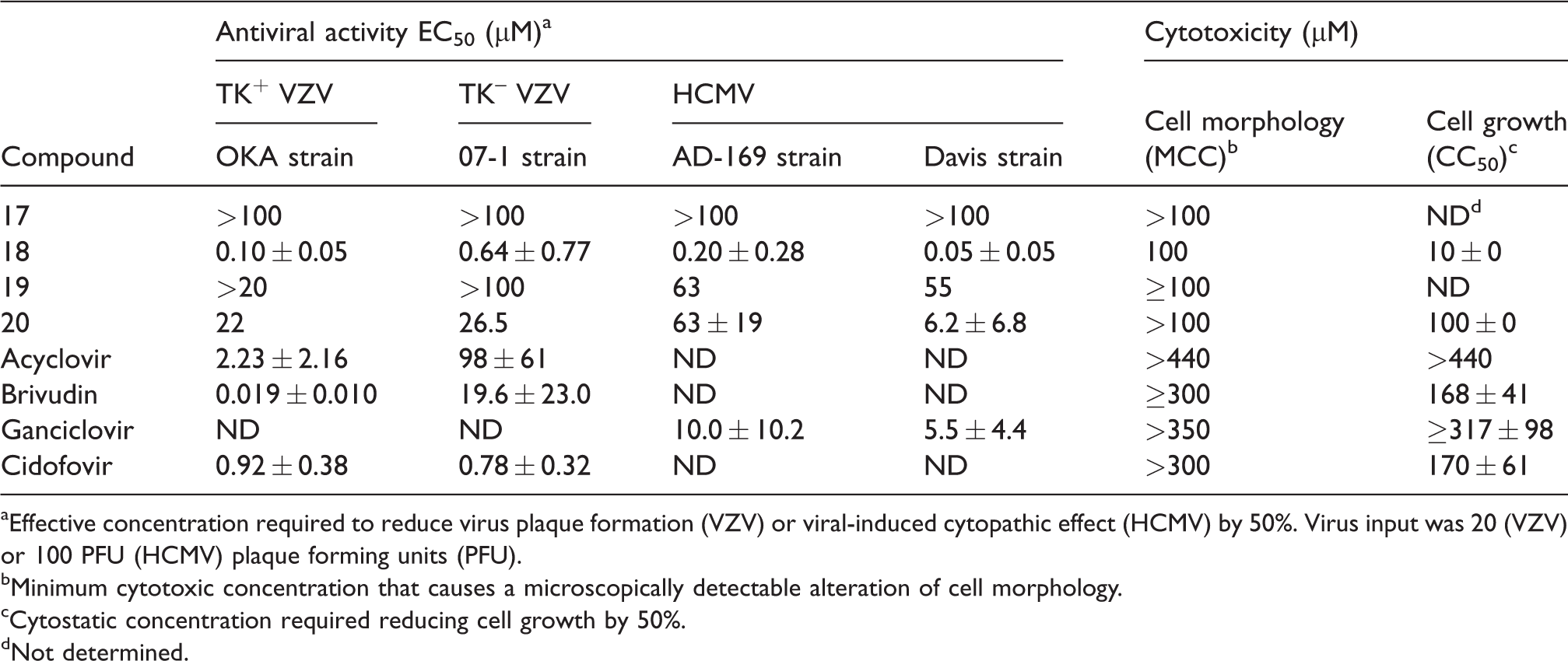
The synthesized xanthine-based ANPs (compounds
Activity of compounds
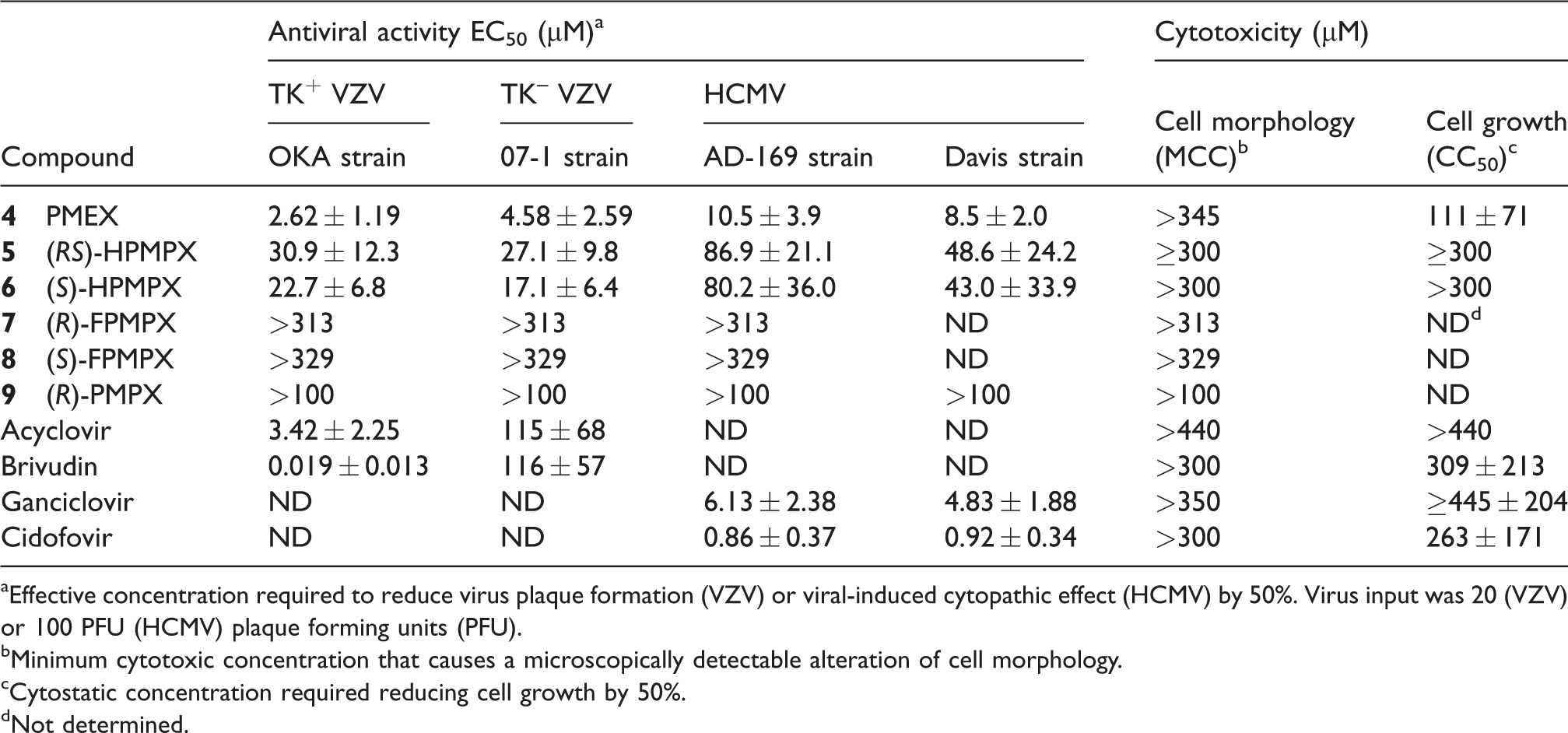
aEffective concentration required to reduce virus plaque formation (VZV) or viral-induced cytopathic effect (HCMV) by 50%. Virus input was 20 (VZV) or 100 PFU (HCMV) plaque forming units (PFU).
bMinimum cytotoxic concentration that causes a microscopically detectable alteration of cell morphology.
cCytostatic concentration required reducing cell growth by 50%.
dNot determined.
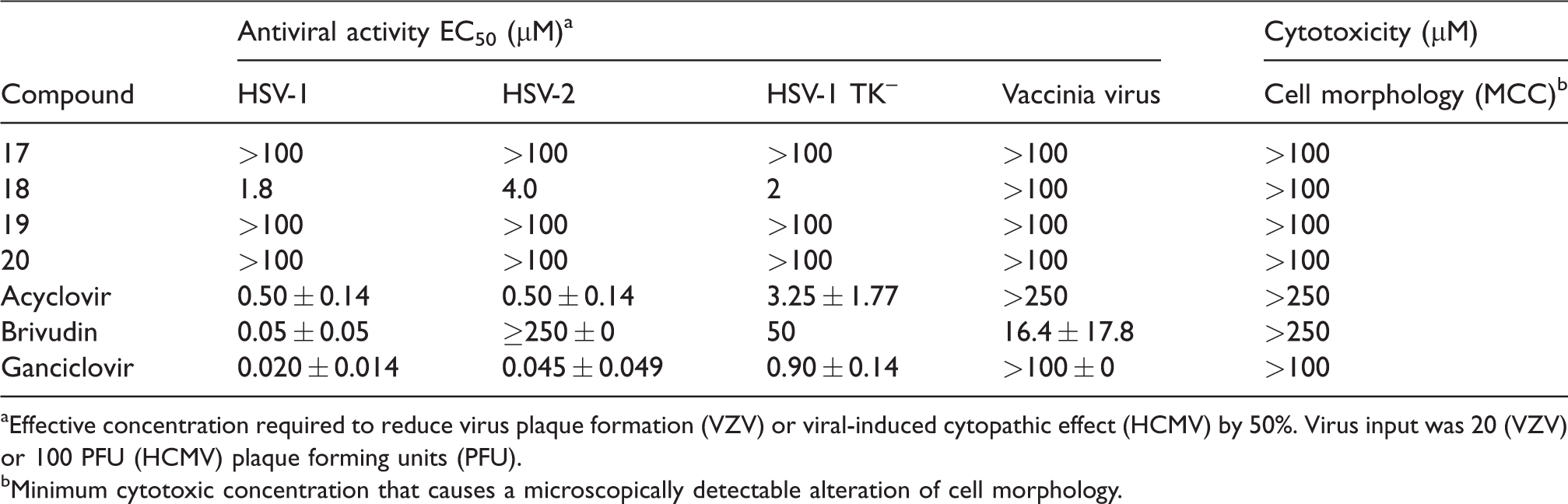
Activity of compounds
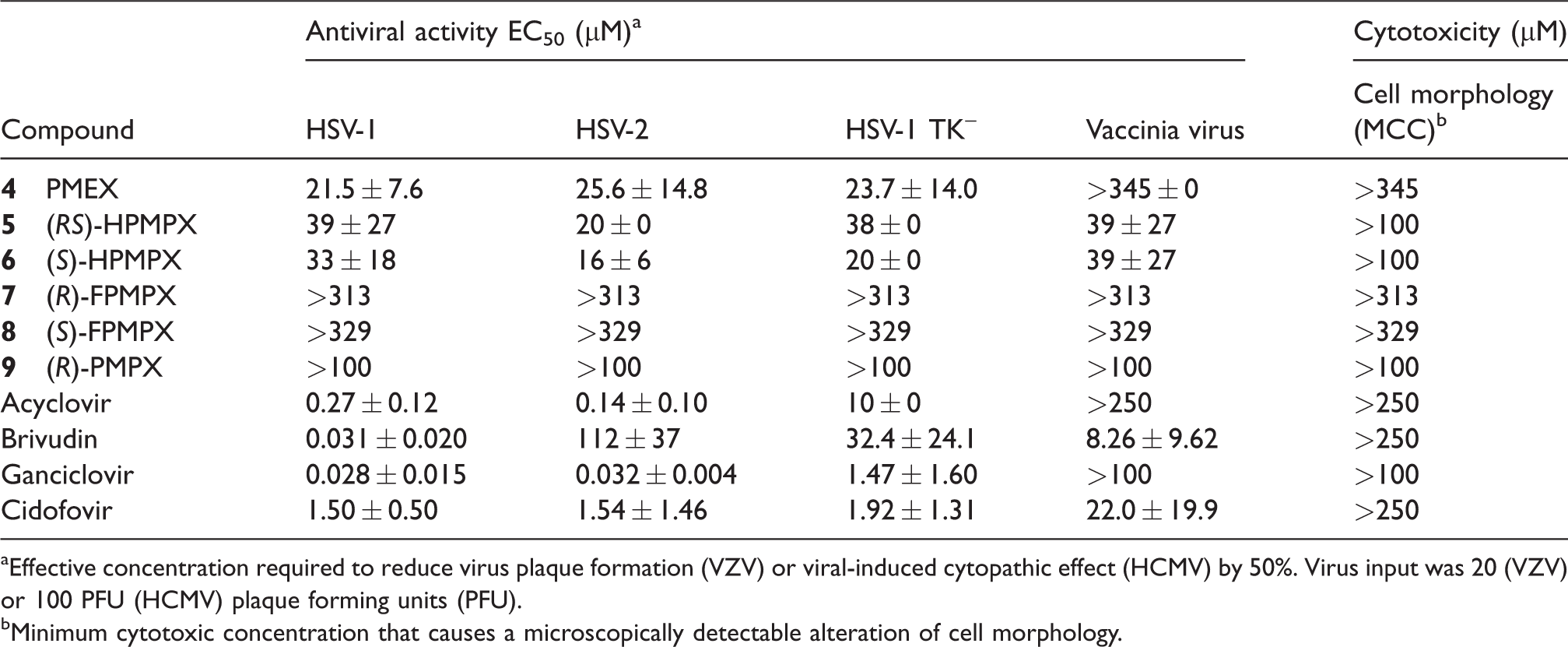
aEffective concentration required to reduce virus plaque formation (VZV) or viral-induced cytopathic effect (HCMV) by 50%. Virus input was 20 (VZV) or 100 PFU (HCMV) plaque forming units (PFU).
bMinimum cytotoxic concentration that causes a microscopically detectable alteration of cell morphology.
ANPs, including PMEX, are polar compounds, showing severely limited bioavailability. To increase the likelihood of good cell wall permeability, several different prodrug approaches were tested for PMEX (Tables 3 and 4). Compound
Evaluation of prodrug compounds
aEffective concentration required to reduce virus plaque formation (VZV) or viral-induced cytopathic effect (HCMV) by 50%. Virus input was 20 (VZV) or 100 PFU (HCMV) plaque forming units (PFU).
bMinimum cytotoxic concentration that causes a microscopically detectable alteration of cell morphology.
cCytostatic concentration required reducing cell growth by 50%.
dNot determined.
Evaluation of prodrug compounds
aEffective concentration required to reduce virus plaque formation (VZV) or viral-induced cytopathic effect (HCMV) by 50%. Virus input was 20 (VZV) or 100 PFU (HCMV) plaque forming units (PFU).
bMinimum cytotoxic concentration that causes a microscopically detectable alteration of cell morphology.
In order to determine whether the DNA polymerase was the actual target of action of PMEX (
Activity of compound
The inhibitory activity of the diphosphate form of PMEX (PMEXpp,
Inhibition of viral and cellular DNA polymerases (pol) by compound

a50% inhibitory concentration or compound concentration required to inhibit the polymerase-catalyzed DNA synthesis by 50%.
bEnzyme reaction in the presence of calf thymus DNA and radiolabeled [3H]dGTP or [3H]dTTP.
cNot determined.
Experimental part
Methods
Starting compounds and other chemicals were purchased from commercial suppliers or prepared according to the published procedures. Solvents were dried by standard procedures. Solvents were evaporated at 40 °C/2 kPa. Analytical TLC was performed on plates of Kieselgel 60 F 254 (Merck). Column chromatography was performed on silica gel 230–400 mesh, 60 Å (Merck). Reverse phase HPLC separation was performed on a Waters Delta 600 instrument with a Waters 486 Tunable Absorbance Detector using column Phenomenex Gemini C18 (10 μm, 250 × 21.2 mm, flow 10 ml/min preparative column). NMR spectra were recorded on Bruker Avance 500 (1H at 500 MHz, 13 C at 125.8 MHz, 31 P at 202.4 MHz,) spectrometer with TMS or 1,4-dioxane (3.75 ppm for 1H, 67.19 ppm for 13 C NMR) as internal standard or referenced to the residual solvent signal. HR MS spectra were taken on a LTQ Orbitrap XL spectrometer. The purity of the tested compounds was determined by HPLC (H2O-CH3CN, linear gradient) and was higher than 95%.
Method A. General procedure for diazotization/2-hydroxy-dediazoniation of guanine-based starting compounds
Guanine-based phosphonate (0.5 mmol) was dissolved in 80% AcOH (20 mL) and excess of isoamylnitrite (2.0 mL) was added. The reaction mixture was stirred at 20 °C for 16 h. Volatiles were evaporated, and the residue was co-evaporated with water (3 × 10 mL) and evaporated to dryness. The crude product was dissolved in a small amount of water and purified by preparative HPLC in 0.1 M TEAB buffer using gradient water/methanol (from 98/2 to 20/80).
((2–(2,6-Dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)ethoxy)methyl)phosphonic acid (4), PMEX
Synthesis of compound 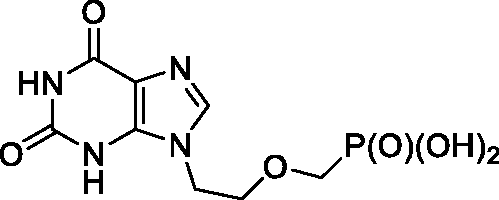
(RS)-(((1–(2,6-Dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)-3-hydroxypropan-2-yl)oxy)methyl)phosphonic acid (5), (RS)-HPMPX
Treatment of 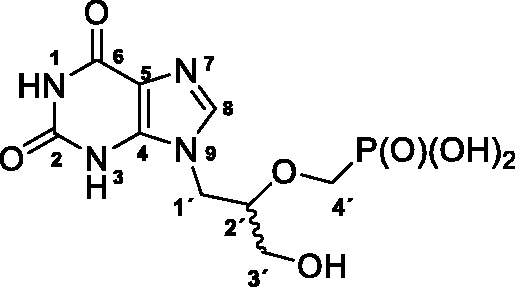
(S)-(((1–(2,6-Dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)-3-hydroxypropan-2-yl)oxy)methyl) phosphonic acid (6), (S)-HPMPX
Treatment of 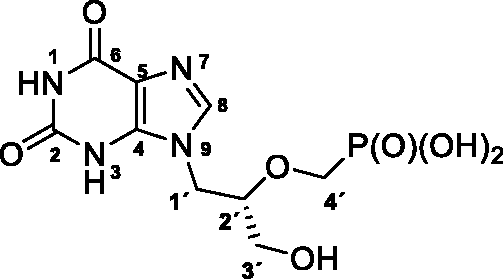
(R)-(((1–(2,6-Dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)-3-fluoropropan-2-yl)oxy)methyl)phosphonic acid (7), (R)-FPMPX
Treatment of 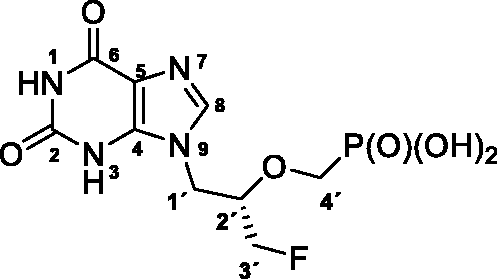
(S)-(((1–(2,6-Dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)-3-fluoropropan-2-yl)oxy)methyl)phosphonic acid (8), (S)-FPMPX
Treatment of
(R)-(((1–(2,6-Dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)propan-2-yl)oxy)methyl)phosphonic acid (9), (R)-PMPX
Treatment of 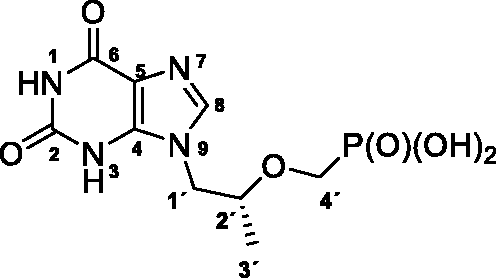
((2–(6-(Cyclopropylamino)-2-oxo-2,3-dihydro-9H-purin-9-yl)ethoxy)methyl)phosphonic acid (17)
Treatment of 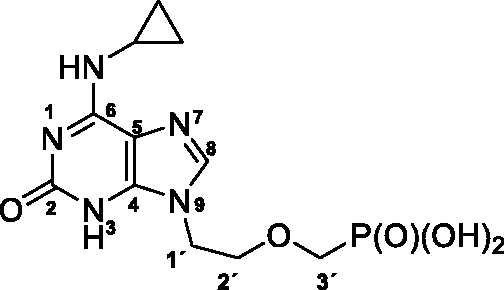
3-(Hexadecyloxy)propyl hydrogen ((2–(2,6-dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)ethoxy)methyl)phosphonate (18)
A suspension of 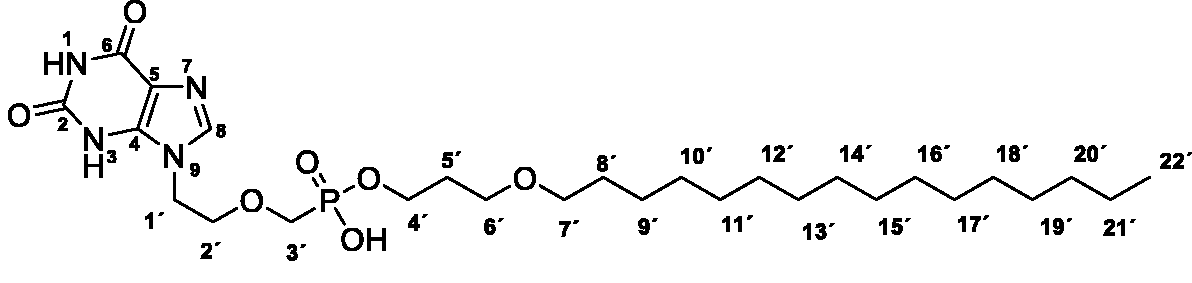
4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,11-Heptadecafluoroundecyl hydrogen ((2–(2,6-dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)ethoxy)methyl)phosphonate (19)
A suspension of 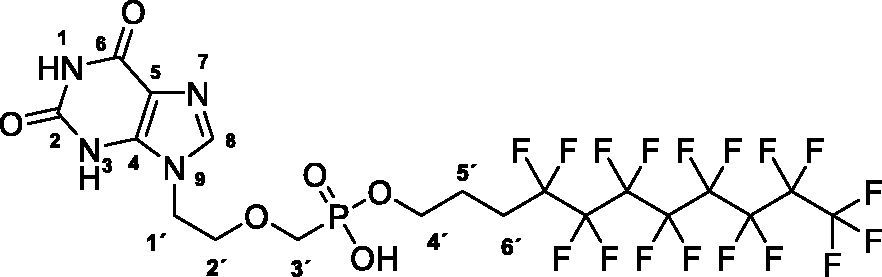
Bis(L-phenylalanine isopropyl ester) ((2–(2,6-dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)ethoxy)methyl)phosphonate (20)
TMSBr (330 µL) was added to the mixture of 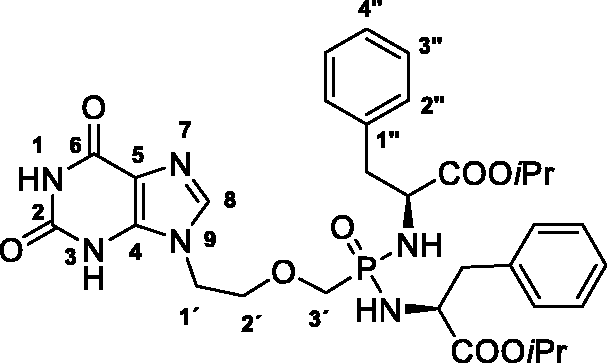
((2–(2,6-Dioxo-1,2,3,6-tetrahydro-9H-purin-9-yl)ethoxy)methyl)phosphonic diphosphoric anhydride (21)
PMEX morpholidate: Morpholine (0.35 mL, 4.0 mmol) was added to a mixture of 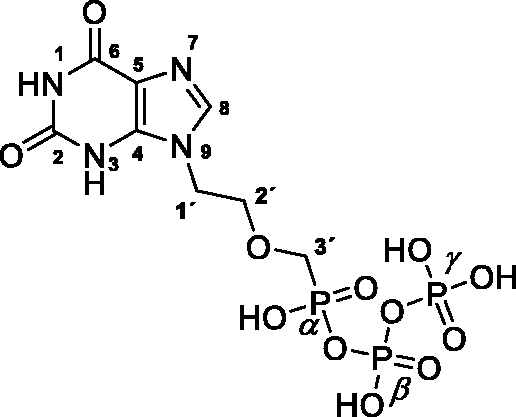
Pyrophosphate coupling: Prepared morpholidate (0.2 mmol) was carefully dried over P2O5 and treated with (NHBu3)2H2P2O7 (0.5 M solution in DMF, 3 ml) at room temperature for 48 h. The product was precipitated with diethyl ether (10 ml) and the solid was washed with diethyl ether (10 ml). The precipitated product was dissolved in 0.05 M TEAB (4 ml) and purified on a column packed with POROS® 50 HQ (50 ml) with use of a gradient of TEAB in water (0.05–0.5 M). The product was co-evaporated several times with water and converted into a sodium salt form (Dowex 50 in Na+ cycle). Lyophylisation afforded
Biological assays
The compounds were evaluated against different herpesviruses, including herpes simplex virus type 1 (HSV-1) strain KOS, thymidine kinase-deficient (TK–) HSV-1 KOS strain resistant to ACV (ACVr), herpes simplex virus type 2 (HSV-2) strain G, VZV strain Oka, TK– VZV strain 07–1, human cytomegalovirus (HCMV) strains AD-169 and Davis as well as vaccinia virus, adeno virus-2, vesicular stomatitis virus, para-influenza-3 virus, reovirus-1, Sindbis virus, Coxsackie virus B4, Punta Toro virus, RSV, FIPV and influenza A virus subtypes H1N1 (A/PR/8), H3N2 (A/HK/7/87) and influenza B virus (B/HK/5/72). The antiviral assays were based on inhibition of virus-induced cytopathicity or plaque formation in human embryonic lung (HEL) fibroblasts, African green monkey kidney cells (Vero), human epithelial cervix carcinoma cells (HeLa), Crandell-Rees feline kidney cells (CRFK), or Madin Darby canine kidney cells (MDCK). Confluent cell cultures in microtiter 96-well plates were inoculated with 100 CCID50 of virus (1 CCID50 being the virus dose to infect 50% of the cell cultures) or with 20 PFU, and the cell cultures were incubated in the presence of varying concentrations of the test compounds. Viral cytopathicity or plaque formation (VZV) was recorded as soon as it reached completion in the control virus-infected cell cultures that were not treated with the test compounds. Antiviral activity was expressed as the EC50 or compound concentration required reducing virus-induced cytopathicity or viral plaque formation by 50%.
Compound
Cytotoxicity of the tested compounds was expressed as the minimum cytotoxic concentration or the compound concentration that caused a microscopically detectable alteration of cell morphology. Alternatively, the cytostatic activity of the test compounds was measured based on the inhibition of cell growth. HEL cells were seeded at a rate of 5 × 103 cells/well into 96-well microtiter plates and allowed to proliferate for 24 h. Then, medium containing different concentrations of the test compounds was added. After three days of incubation at 37°C, the cell number was determined with a Coulter counter. The cytostatic concentration was calculated as the CC50, or the compound concentration required reducing cell proliferation by 50% relative to the number of cells in the untreated controls.
The inhibitory effects of PMEXpp on human (α and β) and viral (VZV and HCMV) DNA polymerases were determined as previously described using activated calf thymus DNA, 100 µM of each of the three unlabeled dNTPs, and 0.5 µM of the rate limiting tritium-labeled dNTP, and serial dilutions of PMEXpp (
Conclusions
A series of novel ANPs bearing xanthine as a nucleobase was prepared and evaluated for their potential antiviral properties. Two synthetic approaches were exploited for the synthesis of the target compounds: (a) recently developed MW-assisted hydrolysis of 2,6-dichloropurine derivatives and (b) well-established diazotization/2-hydroxy-dediazoniation of the corresponding guanine analogues. All prepared ANPs were tested against a wide range of DNA and RNA viruses. Two compounds exhibited antiviral activity. PMEX (
Footnotes
Acknowledgements
This study is a part of the research project RVO61388963 of the Institute of Organic Chemistry and Biochemistry, Czech Academy of Sciences. The authors thank the Mass Spectrometry team (Dr. Josef Cvačka) for measurement of HRMS spectra. The authors wish to express their gratitude to Leentje Persoons, Ellen De Waegenaere, Mrs Lizette van Berckelaer Mrs Bianca Stals, Mrs, Kirsten Lepaige, Mr Niels Willems and Mrs. Nathalie Van Winkel for excellent technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following finacial support for the research, authorship, and/or publication of this article: This article received funding from the Institute of Organic Chemistry and Biochemistry, Czech Academy of Sciences (RVO61388963).
