Abstract
Background
Viruses are obligate parasites that depend on the cellular machinery of the host to regenerate and manufacture their proteins. Most antiviral drugs on the market today target viral proteins. However, the more recent strategies involve targeting the host cell proteins or pathways that mediate viral replication. This new approach would be effective for most viruses while minimizing drug resistance and toxicity.
Methods
Cytomegalovirus replication, latency, and immune response are mediated by the intermediate early protein 2, the main protein that determines the effectiveness of drugs in cytomegalovirus inhibition. This review explains how intermediate early protein 2 can modify the action of cyclosporin A, an immunosuppressive, and antiviral drug. It also links all the pathways mediated by cyclosporin A, cytomegalovirus replication, and its encoded proteins.
Results
Intermediate early protein 2 can influence the cellular cyclophilin A pathway, affecting cyclosporin A as a mediator of viral replication or anti-cytomegalovirus drug.
Conclusion
Cyclosporin A has a dual function in cytomegalovirus pathogenesis. It has the immunosuppressive effect that establishes virus replication through the inhibition of T-cell function. It also has an anti-cytomegalovirus effect mediated by intermediate early protein 2. Both of these functions involve cyclophilin A pathway.
Keywords
Introduction
Viruses, without their own replication machinery, depend on the host cell to proliferate. The viruses interact with host cells, activates enzymes and co-factors required for their replication. The response of the host cell thus reflects the pathogenic properties of the virus.1,2
In the treatment of viral diseases, the drugs primarily target special viral proteins. Current antiviral drugs include nucleotide analogs,3–7 neuraminidase enzyme and M2 channel,8,9 reverse transcriptase and protease inhibitors,10,11 and chemokine receptor 5 (CCR5) antagonist. 12 Many drugs targeting viral proteins are fraught with cytotoxicity and tend to cause drug resistance. 13 Although drugs that target cellular factors or pathways have the advantage of overcoming cellular barriers, their toxicities compromise their usefulness.14–17 For this reason, there is a need to discover alternative antiviral compounds with minimal side effects. 18
Immunosuppressed patients undergoing organ transplantation are susceptible to cytomegalovirus (CMV) infections that could cause rejections.19–21 Interestingly, two immunosuppressive drugs, mycophenolic acid and mizoribine, 22 have better anti-CMV activities than ganciclovir23–27 and allow for good graft survival.28–30 Some immunosuppressive drugs were developed to target specific protein kinases essential for virus replication. Among these are artesunate, a drug that inhibits cellular kinase signalling, NF-κB, and Sp1 proteins,31,32 and sirolimus and everolimus, rapamycin inhibitors targeting cellular signals.33–37 These drugs were shown to be active against ganciclovir- and foscarnet-resistant CMV strains.38,39
This review discusses the association between cellular pathways mediated by CMV and cyclosporin A (CsA) and demonstrates that CsA can be used both as an immunosuppressive drug in organ transplantation and as an anti-CMV agent.
Cytomegalovirus
CMV is a DNA virus of the herpesviridae family. 40 The highly species-specific human cytomegalovirus (HCMV) cannot be studied in animals. This results in the development of many animal models for the virus.41–46 Seroprevalence studies have shown that 30–97% of the population is seropositive for CMV.47–49 The immune system can easily control the virus from causing active infection and facilitating lifelong latency.50,51
Immunosuppressed transplant recipients, HIV patients, and the foetus are at risk of acquiring CMV infections.52–58 The high risk of infection in solid organ transplantation (SOT) recipients is associated with serostatus of the donor and recipient, type of transplanted organ, host immune status, and viral factors. In organ and tissue transplantation, CMV infections were mostly reported in the seropositive (D+)/CMV-seronegative (R–) recipients rather than in D–/R–recipients.57,59,60 It was also shown in hematopoietic cell transplant, the greatest risk of infection is when the donor is CMV seronegative (D–) and the recipient seropositive (R+). 61 In pregnant women, among factors that increase the risk of CMV infections are socioeconomic status, parasitic infections, CMV viral load and serostatus, age, non-Caucasian, education level, and close contact with young children, the prevalence of CMV infection is between 50 and 97%.62–66 Between 5 and 10% of babies born of mothers with primary CMV infection show neurological diseases at birth, making the prevalence of CMV-related disabilities higher than other childhood diseases.57,67–69 The risk of vertical transmission of CMV is higher in primary (33%) than that in non-primary CMV infection (1%), 70 but in both infections, symptomatic congenital CMV infections may also develop.71–75 Primary infection occurs in most seropositive women and in 1 to 4% of seronegative women. In the USA, 77% of congenital infections are acquired from non-primary CMV infection and only 22% from primary infections.76–78 Thus, 77% of congenital infection is acquired from non-primary CMV infection compared to 22% of primary infection in USA. 78 Regarding CMV disease outcomes, there is no evidence that the symptomatic outcomes in both primary and non-primary CMV infections are different.71,73,79 The greatest risk of CMV infection is during the period of fetal organogenesis, that is in the first and early second trimesters. Vertical transmissions mostly occur in the third trimester, and fetuses are mostly born healthy. There seems to be no difference in symptoms among babies born of pregnancies affected with primary and non-primary CMV infections. 80 Mortality is between 20 and 30% in symptomatic congenitally infected children,81,82 resulting from liver dysfunction, coagulation disorders, and secondary bacterial infections. 82 Complications of CMV congenital infections include central nerve system (CNS) diseases, such as meningoencephalitis, calcification, microcephaly, disruption of neuronal migration, germinal matrix cysts, ventriculomegaly, cellular hyperplasia, lethargy, hypotonia, seizure, and chorioretinitis.83,84 Complications are more severe if CMV infections occur at the early stage of pregnancy. 85 In some newborns, post-natal CMV infections cause hepatitis, neutropenia, thrombocytopenia, and premature and low birth weight. 86 Sensorineural hearing loss (SNHL) due to virus replication in the inner ear was reported in 15 to 25% of young kids.87–89 This disorder can develop early at birth (in 5.2% of symptomatic or asymptomatic neonates) or later in childhood (in 15.4% of children).52,87,88,90 In approximately 50% of infants, the congenital infection may manifest as the more severe and symptomatic cytomegalic inclusion disease (CID) or cytomegalic inclusion body disease (CIBD).67,68,91,92 Infants with CID can develop other CMV neurological diseases.84,93–97
CMV can reach the brain of foetuses. Brain CMV infections are associated with viral replication in the endothelial cells of the blood–brain barrier (BBB), 98 a process that facilitates virus-crossing of the brain parenchyma and access to astrocytes.99–102 Another way the virus enters the brain is by infecting the ependymal cells of choroids plexus, leading to dissemination of virus in the cerebrospinal fluid and subsequently infecting the brain parenchyma. 103 The virus can establish latent infections in myeloid cells that eventually infiltrate the brain and develop into microglia cells. 103 Destruction or injury to BBB endothelial cells can result in extravasation of monocytes and facilitate brain infection. 104
In the nervous tissues, CMV induces injuries by interfering with cell differentiation, morphogenesis and survival, controlling apoptosis mechanism, infecting neural stem cell and interfering with brain developmental process, and impacting the CMV replication on the endovascular system. 89 Viral replication interferes with brain development during migration of neocortical neurons to the cortical plate, causing disabilities.105–108
Cyclophilin A
Cyclophilins (CyPs) are a group of highly conserved cellular proteins,109–112 found ubiquitously in animal and plant tissues. These proteins have unique chemical structures consisting of 109 amino acids with. These amino acids contain variations in the interaction domain among proteins from various locations and of different functions. 113 At least 16 CyPs have been identified. 114 Although they differ in distribution in tissue and organ localization, their structures are similar to the same peptidyl-prolyl isomerase (PPIase) activities. 115 In humans, seven CyPs (40 kDa) have been identified, which are hCyPA, hCyPB, hCyPC, hCyPD, hCyPE, hCyP40, and hCyPNK.110,116 The cellular functions of CyPs have been observed in protein folding and trafficking.117,118
Cyclophilin A (CyPA), which can bind cyclosporin A (CsA), was first isolated from bovine thymocytes.115,119 CyPA resembles the high abundance CyPs in eukaryotic cells 119 with concentration ranging from 0.1 to 0.6% of total cellular proteins. 111 CyPA is encoded by Cyp18 gene in chromosome 7p11.2-p13 and consists of 165 amino acid with a molecular weight of 18 × 103 Daltons. 120 In humans, CyPA is composed of eight antiparallel β-barrel structure enclosed by two α-helices at each location. The hydrophobic core of CyPA contains one hydrophobic and seven aromatic residues within the drum of the molecule or the CsA-binding area. The CsA-binding site is composed of a loop region at Lys118 to His126 and four β-strands at B3 to B6 position.111,120
CyPA plays an important role in various cellular functions, including inflammation and apoptosis.121–129 This protein can stimulate the immune response and is produced by inflammatory cells such as endothelial cells, monocytes, vascular smooth muscle cells, and platelets.130–132 Extracellular CyPA, through interaction with cell membrane heparin receptor and CD147, also functions as a chemoattractant for monocytes, neutrophils, eosinophils, and T lymphocytes. 133 Like other CyPs, CyPA acts as a chaperone in the regulation of cellular protein111,120,125 and receptor expression and activities.134–136 This protein is also involved in several signalling pathways, such as the signal transduction pathway unique to T lymphocytes and the T cell-specific interleukin-2 tyrosine kinase (Itk).137–139
In the brain, CyPA is mostly localized in the neurons,140,141 where it plays a role in neuronal differentiation, embryo growth, and adult cortical plasticity.142,143 CyPA maintains the integrity and function of BBB and protects neurons during traumatic brain injury by blocking BBB permeability and the effects of ischemia and oxidative stress.144–146 In brain injuries, CyPA induces endothelial cell proliferation and migration as well as recruiting monocytes for the repair of brain blood vessels. 135 Moreover, CyPA induces extracellular signal-regulated kinases (ERK) and protein kinase B (PKB) or Akt signalling by binding to the cell surface receptor, CD147. This binding stimulates the expression of the anti-apoptotic protein Bcl-2147,148 and mediates neurogenesis in brain protection and repair.133,144
Cyclosporin A
Cyclosporin A (CsA), an 11-amino acid cyclic peptide (Figure 1), can be extracted from the fungus, Tolypocladium inflatum Gams. 149 It is an immunosuppressive drug primarily used for organ transplantation.150–152 This drug is also used for the treatment of renal, neurodegenerative, and autoimmune diseases153,154 and has been recommended for the treatment of rheumatoid arthritis, psoriasis, atopic dermatitis, and endogenous uveitis.155–158
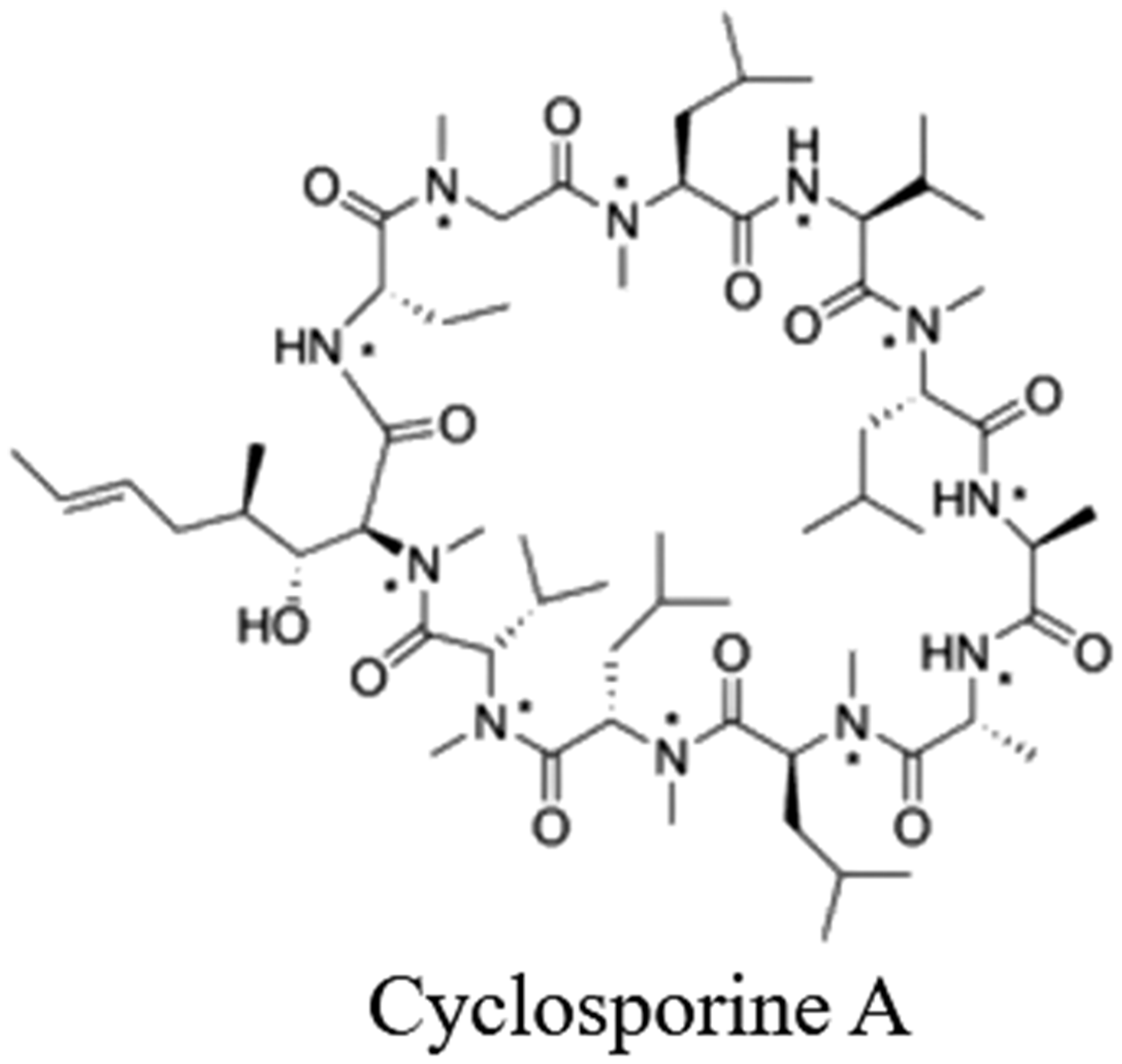
The chemical structure of cyclosporin A.
The immunosuppressive activity of CsA is the result of the formation of CsA-CyPA complex that has a high affinity for calcineurin, a cellular phosphatase mediating T-cell activation.120,159–161 The CsA–CyPA complex is located at the interface of calcineurin, between the catalytic and regulatory subunits, and controls phosphatase activity and biological functions.159,162,163 CsA also affects the expression of AP-1 and NF-κB,164–166 modulates the antigen-specific immune and Ca2+-independent responses, 167 and blocks the JNK and p38 MAPK signalling pathways.161,168–170 However, the use of CsA in the treatment of diseases has adverse effects. It causes abnormal glomerular filtration, nephrotoxicity, neurotoxicity, hepatoxicity, and cardiovascular disorders171,172 through calcineurin and other factors including endothelin and TGF-β1. 173 CsA, through T cells, can induce inflammation and apoptosis. This is mediated by JNK and p38 MAPK, the activators of T cell receptors, 161 ERK, and the transcription factor AP-1. 174 AP-1 controls cellular processes including differentiation, proliferation, and apoptosis. The activation of JNK and p38 MAPK also requires the involvement of the CsA-sensitive protein kinases, MKK6 and MKK7. 161
The majority of mitogen-activated protein kinases (MAPK), MAPK kinase (MAPKK), and MAPKK kinase (MAPKK-K)175–180 require MEKK1 to activate MKK7. MEKK1 mediates JNK pathway that modulates NF-κB activity, 181 JNK pathway signals, and NF-κB activity under CsA control.182–184 CsA mediates JNK signalling pathway through Rac1, a member of the Rho subfamily of small G-proteins, especially through its guanine nucleotide exchange factor (Vav1). Vav1 controls IL-2 expression.185–187 Other immunosuppressive agents such as FK506 and tacrolimus (calcineurin NFAT pathway inhibitor), use the same mechanism to block JNK and p38 pathways, and subsequently T-cell activation. Similarly, the immunosuppressive activity of CsA mediated by calcineurin blocks JNK and p38 activation.188–190 Unlike these immunosuppressive agents, calcineurin controls JNK while in complex with PKC-u.191–193
CsA derivatives
There are several CsA derivatives (Figure 2) that interact well with viruses and do not cause serious side effects.194–196 Among these derivatives, alisporivir (Debio-025) (Figure 2(a)), a non-immunosuppressive analog of CsA, was synthesized to contain sarcosine instead of d-methylalanine at position 3 of the PPIase domain and methyl-leucine instead of ethylvaline at position 4 of the calcineurin domain. These modifications have altered the PPIase activity and calcineurin-binding ability of the derivatives. 197 However, alisporivir still results in side effects including reversible hyperbilirubinemia and pancreatitis when used in combination with PEG-IFNa2a and ribavirin to treat hepatitis C virus (HCV) infection. 198
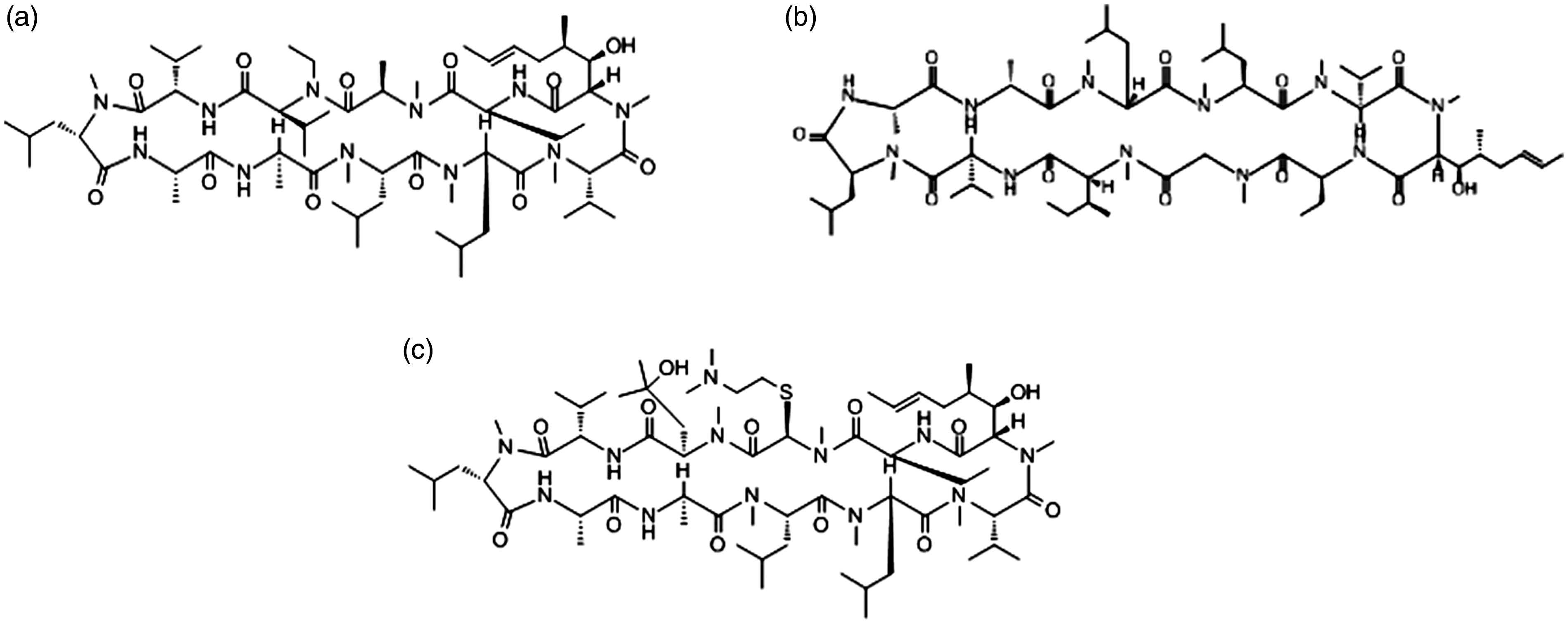
The chemical structure of cyclosporin A (CsA) derivatives. (a) Alisporivir (Debio-025), (b) NIM811 and (c) SCY-635.
NIM811 (Figure 2(b)), another non-immunosuppressive analog of CsA, is similar in structure to the parent molecule with one modification: methyl-leucine at position 4 being replaced by methyl-isoleucine. This derivative has antiviral effects 195 while exhibiting the same pharmacokinetic profile as CsA without nephrotoxicity.196,199
In 2010, SCY-635 (Figure 2(c)) was synthesized. 200 This CsA derivative inhibits HCV infection 201 without detectable inhibition of calcineurin phosphatase. Treatment with this drug is associated with transient increases in interferon α, λ1, and λ3, the cytokines responsible for the clearance of the viruses. 202
Another derivation of CsA, EDP-546, is metabolically stable with favorable pharmacokinetics that allows for less frequent and low therapeutic dosing of the drug. It is also an effective bilirubin transporter without affecting CYP450, a major protein in drug metabolism.203,204
Sanglifehrin A (SFA) (Figure 3) is a new immunosuppressive agent 205 isolated from the Streptomyces strain A92-308110. This protein interacts strongly with CyPA206–208 but not with calcineurin phosphatase. 209 It is also a potent inhibitor of mitochondrial permeability transition and heart reperfusion injury.210.
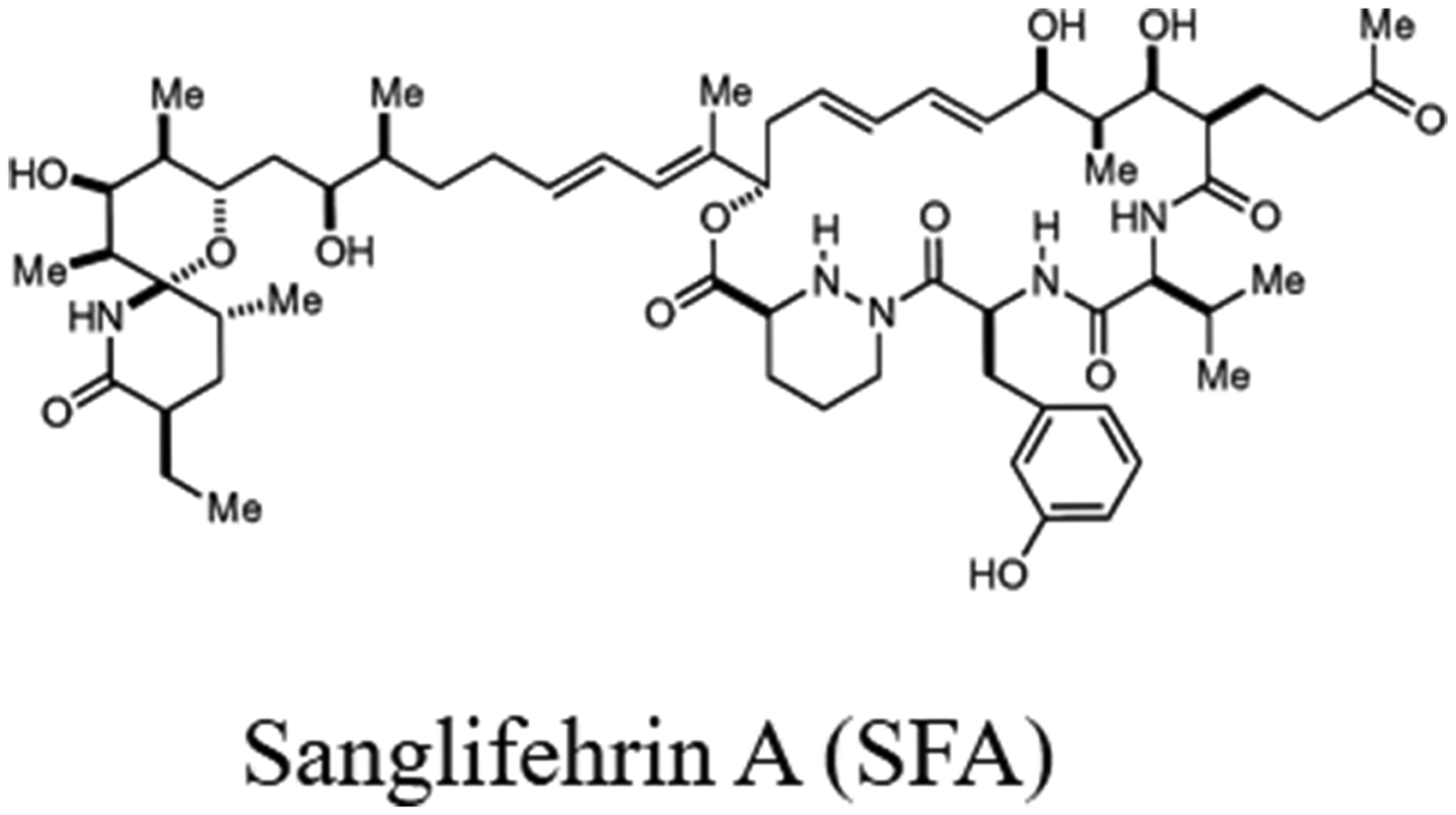
The chemical structure of sanglifehrin A (SFA).
CyPA and viral diseases
CyPA is an essential protein for the HCV and hepatitis B virus (HBV) replications. In HCV, CyPA interacts with several viral proteins including serine protease, NS3, viral RNA-dependent RNA polymerases, NS5A and NS5B, and cysteine protease, NS2.211–215 The interactions between CyPA with NS5A and NS5B stimulate viral genome replication,213,215–221 virion morphogenesis,222–224 and viral particles assembly through the NS5A-D3 domain.213,222,224,225 The replication of viruses is also mediated by other proteins including eEF1A and T-cell intracellular antigen 1 (TIA-1).226,227 eEF1A binds to the 3′-terminal stem-loop of flavivirus genomes and facilitates the synthesis of minus-strand RNA. 226 CsA can block CyPA-NS5 interaction213,228 and indirectly inhibit the genomic RNA amplification.220,225,229
The inhibitory effect of CsA and its derivative on HBV is associated with the distribution of mitochondrial transition pore and calcium signalling. 230 CyPA that can form a complex with CsA is important for virus entry by triggering the expression of HBV surface antigen (HBsAg) and mediating virus DNA replication and envelope protein secretion.231–237 The formation of the CsA–CyPA complex inhibits HCV and HBV through sodium taurocholate co-transporting polypeptide receptor.230,238
CyPA also mediates influenza virus replication through its association with the virus core. 239 In influenza viruses, CyPA interacts with M1 protein, resulting in the interference of virus nuclear translocation and preventing post-transcription of the viral genome. 240 Inhibition of influenza virus replication by CsA and its non-immunosuppressive derivatives involves both the CyPA-dependent and independent pathways.241,242 The CyPA-independent inhibition occurs through the RNA polymerase II. 243 The inhibition of virus replication can also be achieved with the non-immunosuppressive analogs of CsA such as SCY-635.125,241
Human immunodeficiency virus (HIV) is an RNA virus that causes acquired immunodeficiency syndrome (AIDS). The viral capsid undergoes morphological changes in the target cell cytosol. 244 This is mediated by cell factors,245,246 such as TRIM5 247 which forms the TRIM5-CyPA fusion complex, and TRIMCyp that blocks virus replication at the post-entry step. 248 TRIMCyp interacts with the retrovirus capsids, causing capsid disassembly and inhibits virus infection.247,249–251 The HIV-1 CA protein on the capsid surface facilitates interaction with cellular CyPA during infection and controls virus replication. CyPA also interacts with HIV-1 Gag polyprotein that mediates virus fusion, entry, uncoating,252–258 viral genome integration into host DNA,259–261 and modulates the immune response. 262 CsA and its analogs, Debio-025 and NIM811, interact with CyPA. This disrupts CyPA-binding loop located at the N-terminal of HIV-1 CA protein254–256,263,264 and inhibits HIV-1 infection in certain cell types.253,265,266
In severe acute respiratory syndrome (SARS) virus, CyPA binds to the SARS-CoV N protein 267 and mediates viral RNA synthesis.268–270 In most coronaviruses, CyPA also interacts with the non-structural viral protein 1 (Nsp1).
CyPA and CMV replication
Cell signals and CMV
CMV cellular entry and replication involve several factors including epidermal growth factor receptor (EGFR), integrin β1 and β2, and platelet-derived growth factor receptor-α (PDGFRα).271–273 Binding to these receptors initiates Ca+2 homeostasis, activation of phospholipase C and A2, the release of arachidonic acid and its metabolites, diacylglycerol that activates protein kinase C, and inositol triphosphate inducing calcium influx.274–280 The virus also binds to αvβ3 integrin, another receptor that mediates actin depolymerization, facilitating virus translocation to the nucleus, 281 and activating the Src pathway. The alteration in cytoskeleton arrangement is also associated with the activation of αvβ3 integrin and EGFR receptors that reduce the activity of RhoA GTPase.281,282 The virus activates the extracellular signal-regulated kinase (ERK) 1/2, also known as MAPK, through the ERK/MEK1/2 pathway. These interactions that occur through MKK3, MKK6, and MKK4 kinases are important for viral replication.283–285
HCMV replication requires the NF-κB to activate the major immediate early promoter (MIEP),286–288 which is responsible for the transcription of more than 150 genes in cells. The NF-κB specific promoter 1 protein (Sp1) can be activated by HCMV IE1-72 and IE2-86 either independently or synergistically.289–291 MIEP has a special site for the cell transcription factor that either stimulates or represses transcription of NF-κB, CREB/ATF, AP1, 292 Ying Yang1 (YY1), and Ets-2 repressor factors (ERF).293–298 The repression of transcription occurs through factors including those involved in the post-translational modification of histones299,300 and TNF.301,302 In HCMV-permissive cells, MIEP associates with acetylated histone H4. In peripheral blood monocytes, MIEP associates with heterochromatin protein 1 (HP1), a chromosomal protein implicated in gene silencing.303–306 The HCMV remains latent in monocytes that serve as carriers for the virus. Although the mechanism of reactivation of HCMV is still not known, it has been suggested that the host complex facilitates chromatin transcription (FACT) binds to MIEP. The inhibition of this complex inhibits virus activation. 307
HCMV alters the expression of cell cycle regulatory proteins, the cyclin-dependent kinases.308,309 The infected cells are arrested at the G1 phase310,311 of the cell cycle, during which macromolecules needed for cell growth and virus replication are synthesized. Both HCMV IE1-72 and IE2-86 can transactivate p53 promoter and induce p53 accumulation. However, HCMV IE1-72 represses p53 transactivation activity. 312 The outcomes of these interactions include stimulation of DNA synthesis, cell cycle arrest, and inhibition of apoptosis. 312 The virus also responds to alterations in cell growth and activation of host genes by interacting with histone deacetylases (HDACs) and retinoblastoma (Rb) tumor suppressor proteins via the LxCxE-dependent pathway.313–315 The binding of Rb protein to the E2F transcription factor partially controls cell growth in the HCMV-infected cells and other pocket proteins, such as p107. Cell growth arrest occurs when Rb protein represses the transcriptional activity of E2F transcription factors.316,317 The binding of E2F transcription factor to p107 causes the release and activation of E/CDK 2 kinases,318,319 allowing for the cell cycle progression from G1 to S phase.
HCMV increases the expression of tumor suppressor factor p53 in infected cell.312,320,321 Both HCMV IE1-72 and IE2-86 bind to p53, although only HCMV IE2-86 can block its function.312,322,323 HCMV can inhibit the apoptosis pathways through several strategies. Both HCMV IE1-72 and IE2-86 stimulate the anti-apoptotic PI3-K/Akt signalling pathway and delay the onset of p53 activity.324,325 Also, the CMV genes, UL36, a viral inhibitor of caspase-8-induced apoptosis (vICA) and the UL37 gene encoding viral mitochondria-localized inhibitors of apoptosis (vMIA) affect permeabilization of the mitochondrial outer membrane, preventing the release of cytochrome c necessary for the activation of the pro-apoptotic caspase-3 pathway. 326 The adaptation and survival of the virus in host cells are further enhanced by its major tegument protein, pp150 protein. This protein binds to cyclin A2, causing cyclin A2-dependent phosphorylation and avoids the CDK-mediated inhibition of viral replication.311,327–329
CMV contains the UL146 and UL147 genes encoding for the CXC chemokines, vCXCL1 and vCXCL2. The virus uses these cytokines to activate CXCR1 and CXCR2, and preferentially attract neutrophils that can serve as virus carriers.330,331 The virus also possesses the UL 76 gene, which is an inducer of IL8 expression. 332 HCMV genome contains the G-protein-coupled receptors (GPCRs), UL33, UL78, US27, and US28.333–336 The GPCRs prevents leukocytes from recognizing HCMV-infected cells and blocks the effect of leukocytes on neighbouring cells.335,337–340 US28, the best characterized among the GPCRs, stimulates migration of HCMV-infected cell to vascular injury sites leading to atherosclerosis and restenosis.336,341
Host cells infected with HCMV express type 1 interferons (IFNs), IFN-stimulated genes, and proinflammatory cytokines, 342 which are components of the innate cellular response that require the transcriptional activity of interferon regulatory factor 3 (IRF-3). 343 As a defence mechanism, the HCMV inhibits production of IFN by blocking the IFNα-stimulated responses and disrupting the IFNα signal transduction pathway, decreasing the expression of JAK1 and p48344–346 or the multiple IFN-responsive genes.347–349 HCMV, particularly at late infection stage, also inhibits NF-κB signaling responsible for the production of pro-inflammatory cytokines, IL-6, CCL5, and TNFα.350,351
Cyclosporin A, CyPA, and CMV
CsA is an immunosuppressive agent22,352 that can potentiate CMV infection.103,353–355 The immunosuppressive activity of CsA is via CsA–CyPA interaction with calcineurin.159,160 Most of the cellular pathways mediated by CyPA are sensitive to CsA.356–358 These cellular pathways are fully controlled by CMV during viral infection and replication.287,288,291,292,359 Additionally, they share signals such as NF-κB, MAPK such as MKK7 and MKK6, ERK1/2, P53, and JNK. The involvement of CyPA in the activation and replication of CMV 360 was demonstrated in the mouse model through the silencing of the CyPA mRNA. In fact, CyPA is required for HCMV lytic infection, latency establishment, and reactivation. 361 It also regulates IE protein and lytic genes expressions in HCMV replication cycle. The utilization of CyPA by CMV342 may deplete its concentration in organs and compromise cellular protein folding, differentiation, and functions. 361 If the nervous tissues are affected, the infection may cause CNS-associated abnormalities. These phenomena suggest that CyPA could be a potential therapeutic target in the treatment of HCMV infection. Although CsA is a common immunosuppressive drug used during organ transplantations and mediates HCMV reactivation in vivo (CsA–CyPA complex), this response may result from overall immunosuppression. 353 When CMV enters the cell, it stimulates the secretion of type I IFN as well as proinflammatory cytokines such as CyPA362–364 through both NFκB and IRF3 transcriptions. 365 This is critical in CMV treatment.366,367 Both responses are controlled by CMV IE86 protein. 368 The other immune response to CMV is carried out by Toll-like receptor-2 (TLR2) stimulated by glycoproteins 364 and CD147. 369 Evidence from previous studies suggests133,370 that CD147 operates as a legend for CyPA. 362 CD147 upregulates through CMV infection and is involved in inflammation similar to CMV.371,372 The interaction was established between CMV and ectodomain of CD147. 373 When CMV enters the cell, it stimulates CyPA production that interacts with CD147 to mediate virus replication and early immune response such as ISG15 gene, IFNB1 signalling to block CMV reinfection. 374 Additionally, the CD147–CyPA activates NFκB through ERK1/2 pathway which is important for both monocytes and macrophages. Modulation of the cellular microenvironment is necessary and was approved for CMV IL-10 previously. 375 CMV successfully downregulates CD147 by encoding MiR-U25-1-5p encoded by US 24-US26. 374 This microRNA is necessary for cellular modulation. It downregulates the CD147 through special coding area in its UTR sequence. This leads to reduced activation of both IFNB1 and IFN-related gene ISG15, NFκB, IL-6, and TNF-α induced above. This inhibits immune response mediated by CD147. 374 This inhibition is also mediated by CsA as it blocks CyPA function and subsequently its interaction with CD147. 374
Many viruses successfully replicate by inducing oxidative stress,376–378 while CypA is important to mediate their replication.220,229,379 Xiao et al.’s study 372 demonstrated that CMV infection induces a substantial level of oxidative stress. 380 This oxidative stress stimulates CyPA production. CyPA plays a role in activating the p38/MAPK pathway. The P38/MAPK pathway is necessary to activate the NFκB transcription factor Elk-1, Sap-1, ATF-2, CREB, CHOP, and Max,381–386 mediating viral and cellular gene expression to enhance the virus replication.387–390 Silencing of CyPA inhibits virus gene expression. 372 CsA can affect IE1 expression and CyPA activity but not CyPA expression. 372 CsA as CyPA target drug inhibited the CyPA-mediated P38/MAPK pathway. Additionally, the antioxidant compound can also control and limit virus replication. 372 The ability of CyPA to mediate inflammatory response was reported by van de Berg et al. 380 CMV can also mediate the inflammatory response through CyPA. 372
CMV stimulates CyPA that mediates the activation of both the p38/MAPK and ER1/2 pathways. This mediates the viral replication372,374 through the expression of IE1 and IE2. 391 HCMV, through its UL4 promoter, activates both p38/MAPK and MAPK/ERK pathways. 392 In addition, both IE1 and IE2 proteins of CMV mediate the viral gene expression387,393–395 as well as the stimulation of cellular expression factors such as Tef-1, Sp1, c-Jun, JunB, ATF-2, CREB, histone acetyltransferase CREB-binding protein (CBP)-associated factor (P/CAF), and p53.322,396–399 Both IE1 and IE2 also play a role in controlling gene activation factor to mediate viral replication and its response to CsA as an antiviral drug.
Cyclosporin A as an immunosuppressive and anti-CMV drug
IE2 resembles the most abundant IE proteins that mediate virus gene expression and reproduction. The ability of IE2 to overcome CsA inhibition by CyPA can stimulate viral gene expressions. In addition, both chemokine-like receptor proteins, US3 and US6, interact with HL-AC and HL-AG and affect the expression of MHC class 1 antigen (Figure 4),400,401 enabling viruses to evade the immune response. 402 CMV gene expressions occur in sequence, with the expression of early (E) genes occurring first, followed by IE genes. The IE2 expression will eventually decline, allowing for the inhibition of CyPA by CsA through the formation of the CsA–CyPA complex. This complex inhibits NF-κB, and thus virus transcription.403,404 Since the early (E) gene expression will also cascade, no late (L) gene encoding structural protein will be expressed. This leads to the blockage of virus replication and production of virus progeny. Therefore, CsA may be activated by CMV replication 405 while functioning as an antiviral drug by inhibiting virus replication.
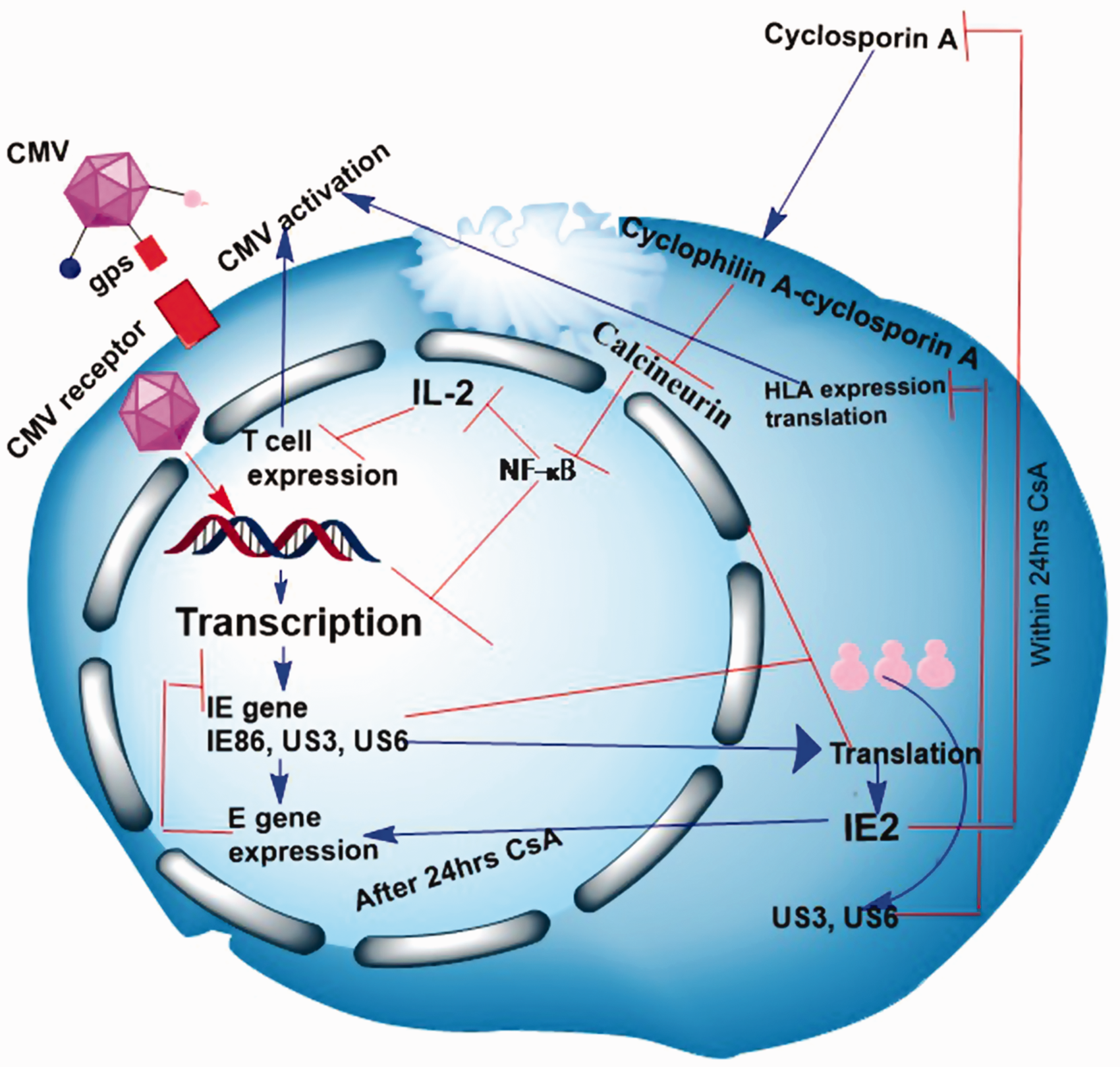
Interactions between CsA and CMV. The pathway shows the immunosuppressive property of CsA. The drug activates CMV infection within 24 h and shows antiviral properties after repeated doses. Adapted with permission from Kawasaki et al.366
Conclusion
CyPA is vital for HCMV lytic infection, latency establishment, and reactivation. This protein can serve as a target in the treatment of HCMV infection. The current review describes the anti-CMV activity of CsA, an immunosuppressive drug commonly used in organ transplantations. However, this drug is associated with HCMV reactivation and toxicities. The immunosuppressive activity of the drug is achieved through the formation of the CsA–CyPA complex that suppresses organ rejection by inhibiting the calcium/calmodulin-dependent and calcinerin. 353 The IE2 protein is the major IE protein, encoded by the CMV IE gene UL122, which mediates the CMV lytic cycle. 394 IE2 expression depends on which proteins or cellular signals it interacts with during the cascade of events, is core to the determination of whether CsA activates or suppresses virus proliferation. Since viral infections induce expression of inflammatory cytokines380,406,407 and thrive under immunosuppression, the inhibitory effect of CsA is most effective in early stages of the viral replication cycle, when the immune and inflammatory responses are still limited. This review with previously published data shows that CsA has anti-CMV activity.360,361 The use of CsA in combination with other drugs approved for the treatment of CMV infections may also prevent the development of unwanted toxicities and drug resistance.
Footnotes
Author’s contributions
All authors have read and approved the final version of the article.
Acknowledgements
The authors would like to express their gratitude to PerkinElmer Inc (PKI) Company to support free version ChemDraw professional 17.1
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Ministry of Higher Education Malaysia FRGS Grant No: 5524642, 5524931 and Universiti Putra Malaysia Grant No: 9428700. The authors are grateful to Taiz University and Ministry of Higher Education and Scientific research (MoHESR), Yemen for the financial support of Ashwaq’s scholarship.
