Abstract
Following the first report on the nucleoside phosphoramidate (ProTide) prodrug approach in 1990 by Chris McGuigan, the extensive investigation of ProTide technology has begun in many laboratories. Designed with aim to overcome limitations and the key resistance mechanisms associated with nucleoside analogues used in the clinic (poor cellular uptake, poor conversion to the 5′-monophosphate form), the ProTide approach has been successfully applied to a vast number of nucleoside analogues with antiviral and anticancer activity. ProTides consist of a 5′-nucleoside monophosphate in which the two hydroxyl groups are masked with an amino acid ester and an aryloxy component which once in the cell is enzymatically metabolized to deliver free 5′-monophosphate, which is further transformed to the active 5′-triphosphate form of the nucleoside analogue. In this review, the seminal contribution of Chris McGuigan’s research to this field is presented. His technology proved to be extremely successful in drug discovery and has led to two Food and Drug Administration-approved antiviral agents.
Introduction
Nucleoside analogues
Viral infections represent a major problem to human society. Viruses are often difficult to eradicate due to the fact that they are easily spread, and are able to use the host biochemical pathways to replicate. Therefore, targeting viral machineries often presents the challenging task of reducing the viral load in the human cell without damaging it.
One of the most successful approaches to fight viral infections is using nucleoside analogues (NAs). 1 NAs are synthetic compounds that exhibit structural similarities to natural nucleosides. In the cell, they can undergo the same physiological processes as the endogenous nucleosides from the uptake to the metabolism, so that in their phosphate forms they can act on cellular functions. Mono-, di-, and triphosphorylated nucleosides are therefore the active forms of these drugs. These compounds act by interfering with viral enzymes as competitive inhibitors of their natural substrates as well as by being incorporated into newly synthesized viral DNA and RNA strands. Their incorporation into nucleic acids may induce either the termination of chain elongation or the accumulation of mutations in the viral genome. 2 Through these mechanisms NAs interfere with the viral genome replication and thereby work as antiviral drugs. However, human enzymes can also recognize NAs, which act as antimetabolites. Antimetabolites can have toxic effects on cells, such as halting cell growth and division and/or inducing apoptotic processes. Consequently, these NAs are specifically used as chemotherapy for cancer. NAs have been in clinical use for almost 50 years and have become cornerstones of the treatment for patients with viral infections or cancer conditions. 3
In the antiviral arena, NAs are commonly used in the therapy of human immunodeficiency virus (HIV), hepatitis B and C viruses (HBV and HCV), herpes simplex virus (HSV), cytomegalovirus (CMV), and varicella zoster virus (VZV) infections. These agents are generally safe and well tolerated as they are recognized by the viral, but not human polymerases in DNA replication. The NAs used to treat HIV infections are often referred to as nucleoside reverse transcriptase inhibitors (NRTIs), a viral DNA polymerase essential for HIV replication. However, they have activity against both DNA-dependent and RNA-dependent DNA polymerases. They inhibit viral replication by several mechanisms, either by competitive inhibition of the viral polymerase or by DNA chain termination. Many of the antiviral NAs either are missing or are blocked at the 3′-hydroxyl group, which results in failure of elongation of the nascent DNA molecule. Other antiviral NAs are negative enantiomers (L-forms) of the natural (D-forms) nucleosides and interfere with replication, partially because of steric hindrance when they are taken up by the viral polymerase or added to the DNA molecule.
There are currently 14 approved cyclic NAs
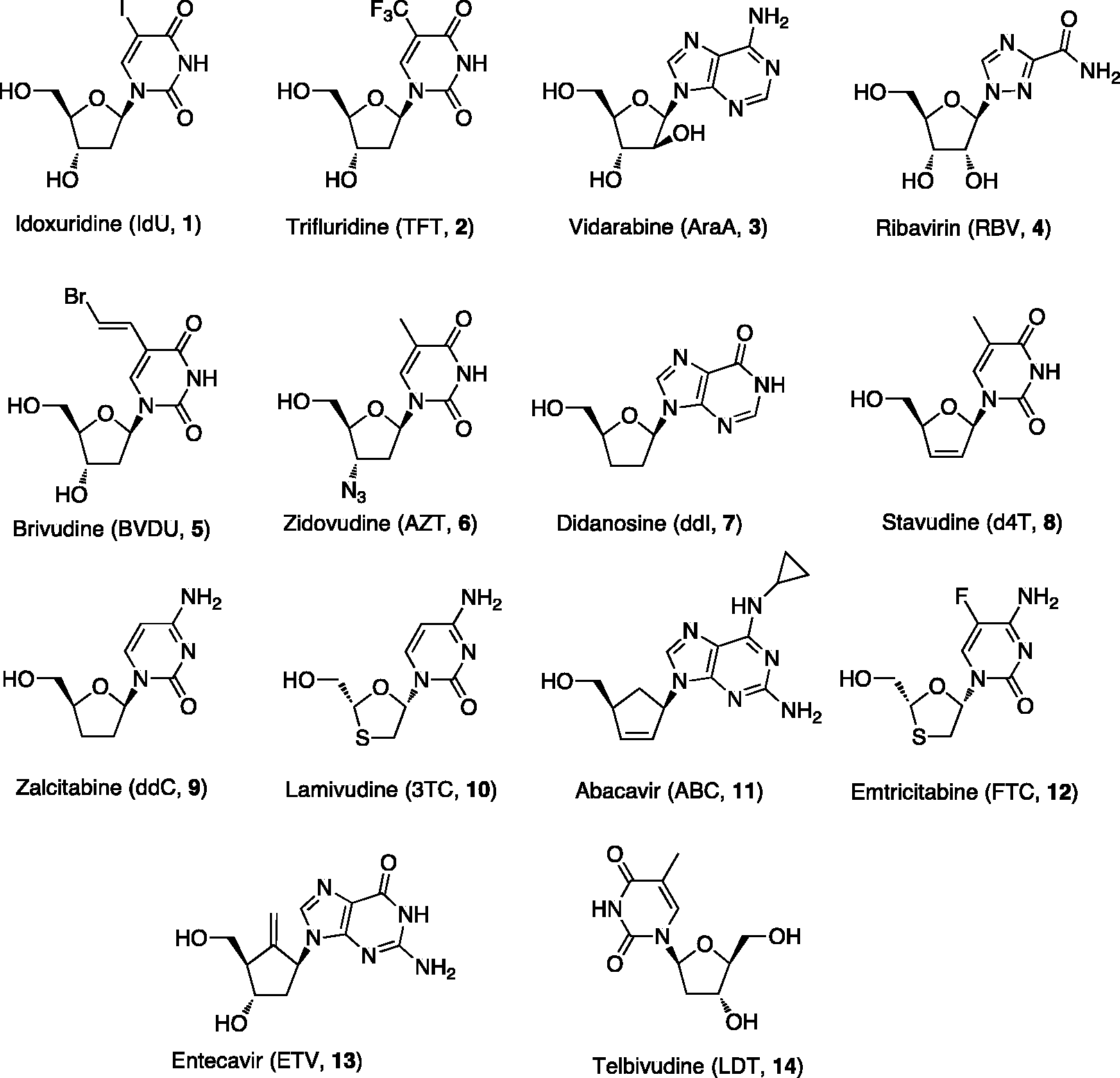
Cyclic NAs in clinical use as antiviral agents.
Vidarabine (AraA,
The combination of pegylated interferon-α with ribavirin (RBV,
Several other NAs were described as antiretroviral agents and were later approved and marketed for antiviral therapy. Among them there are brivudine (BVDU,
The clinical development of a second generation of antiviral NAs, including at present six approved compounds (Figure 2), started 35 years ago with the discovery of the acyclic NA ACV
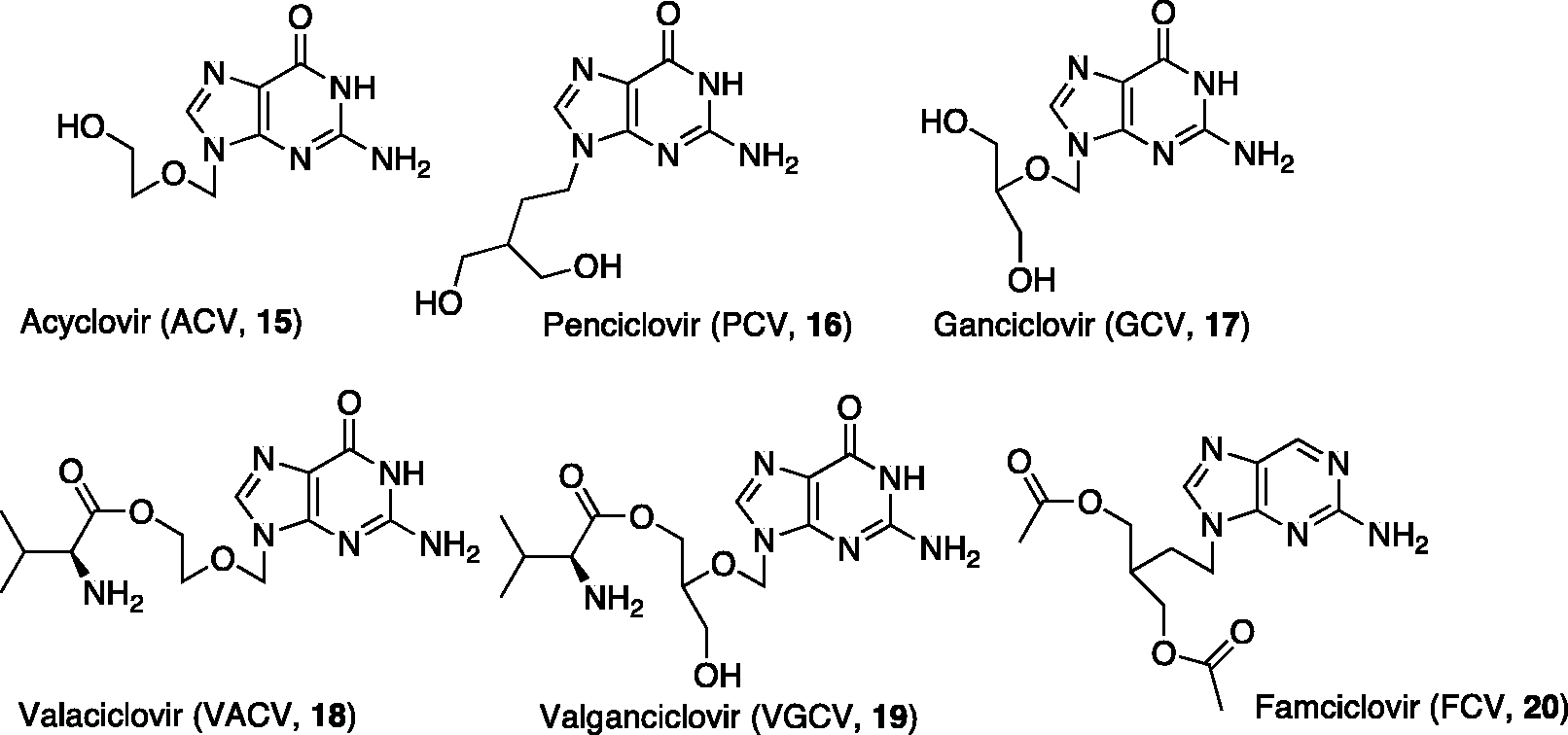
Acyclic NAs in clinical use as antiviral agents.
In order to exert their antiviral activity, NAs have to be phosphorylated (in vivo) via three consecutive phosphorylations with the first one being usually the rate-limiting step. 23 However, if the first phosphorylation of the nucleoside to its 5′-monophosphate cannot take place all these drugs are inactive. This happens when the virus either does not induce a specific kinase or has developed resistance to the compound through mutations in this enzyme while the human cell fails to secure phosphorylation. Thus, to overcome this issue and improve therapeutic properties, nucleosides with a phosphate group already present in the structure have been targeted. The idea of replacing the phosphate group by an isosteric and isoelectronic phosphonate moiety was also investigated leading to the discovery of nucleoside phosphonate analogues (NPs). 24 Since the CH2–P bond, unlike CO–P bond, is not susceptible to esterase and phosphatase hydrolysis, the resulting phosphonate compounds proved to be chemically and enzymatically more stable than the phosphate analogues. NPs are classified into major groups: cyclic nucleoside phosphonates (CNPs) and acyclic nucleoside phosphonates (ANPs). 25 CNPs are natural-like NAs as they contain a nucleobase and a sugar moiety. Compared to the large number of the ANPs described in the literature, only a few examples of CNPs with some antiviral activity have been reported.24,26 This scarcity of examples is due the fact that in general CNPs are characterized by weak (if none) antiviral activity, which is generally explained by their poor substrate properties for cellular and viral kinases. For this reason until now, none of these compounds have reached the clinic.
On the contrary, ANPs have acquired a prominent therapeutic position. 25 They exhibit a broad spectrum of antiviral activities, particularly against DNA viruses and retroviruses, which are ascribed to their ability to undergo intracellular phosphorylation to the diphosphate forms and to be incorporated in the growing nucleic acid strand. The common structural attribute of ANPs is a nucleobase attached to an aliphatic side chain containing a phosphonomethyl residue. A methylene bridge between the phosphonate moiety and the rest of the molecule excludes the possibility of enzymatic dephosphorylation; absence of the glycosidic bond in the structure of ANPs further increases their resistance to chemical and biological degradation. Flexibility in the acyclic chain is assumed to enable these compounds to adopt a conformation suitable for interaction with active sites of different enzymes involved in DNA replication.
This new family of marketed antiviral drugs, includes cidofovir (CDV,
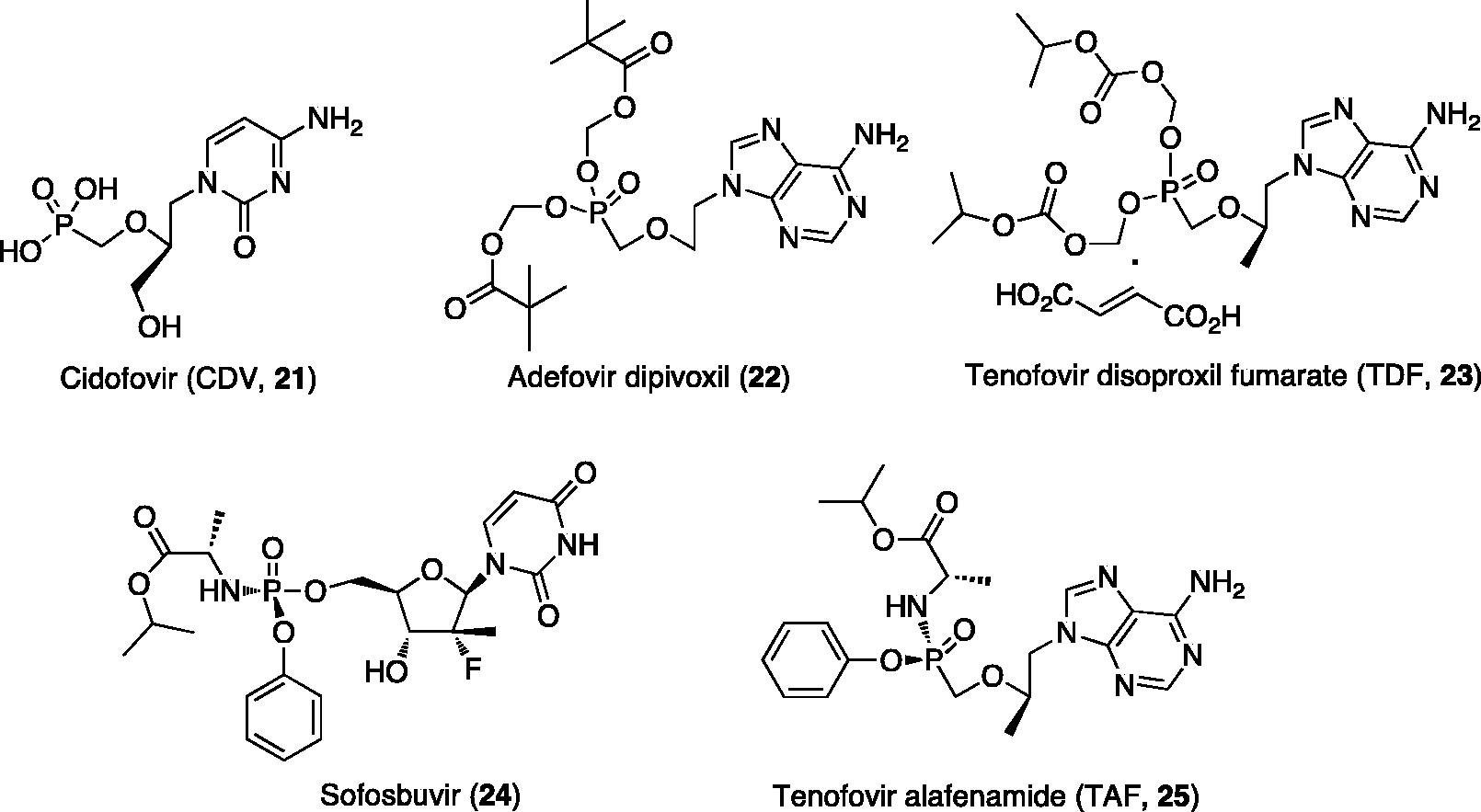
Nucleotide analogues in clinical use as antiviral agents.
Nucleoside phosphate and phosphonate prodrugs
NAs as hydrophilic molecules do not rapidly penetrate cell membranes by non-facilitated diffusion. Instead, they permeate the cell by carrier-mediated endocytosis, 28 which is an active or facilitated transport mechanism that requires energy and a specific receptor or protein on the cell surface. Unfortunately, carrier-mediated transport often requires very close structural resemblance to natural products.
As previously mentioned, nucleoside 5′-monophosphates or 5′-phosphonates bypass the slow first phosphorylation step performed by viral kinases. However, these two classes of compounds are subject to poor cell penetration as a consequence of the negative charges in the phosphate and phosphonate groups, at physiological pH. Similarly to NAs, they require active transportation to enter the cells, and might present a risk of being deactivated in vivo by several cellular enzymes. In addition, they are not ideal for oral administration, an extremely desirable requisite for the treatment of chronic diseases. To overcome these limitations, several prodrug structures of biologically active phosphate and phosphonate analogues have been developed.29–37 The rationale behind the design of such agents is to achieve temporary blockade of the free phosphonic functional group until their systemic absorption and delivery, allowing the in vivo release of the active drug only once at the target site. Such compounds have increased lipophilicity and as such are capable of altering cell and tissue distribution/elimination patterns of the parent drug. 38 Passive transcellular absorption is the most general route for absorption of lipophilic molecules. Many prodrug approaches have been utilized to overcome the limitations of phosphate- and phosphonate-containing drugs. Some of these approaches are still under development and until now no clinical investigations as antiviral agents of compounds, belonging to these classes of prodrugs, have been reported. Among them, there are aryl and phenyl esters, 39 cyclosaligenyl esters (CycloSal), 40 bis-S-acylthioethyl esters (Bis-SATE), 41 and peptidomimetic prodrugs.42–44 Nevertheless, other approaches have been more successful and include prodrugs that have reached the clinic for antiviral therapy. An example of such a well-investigated class of prodrugs is represented by the alkoxyalkyl ester prodrugs of ANPs, designed by Hostetler group. 45 These include brincidofovir (CMX001, hexadecyloxypropyl-CDV), an experimental antiviral drug in clinical development by Chimerix for the treatment of CMV, adenovirus, smallpox, and Ebola virus infections 46 and CMX157 (hexadecyloxypropyl-tenofovir) another novel lipid ANP that has completed a Phase I clinical trial in healthy volunteers, demonstrating a favourable safety, tolerability, and drug distribution profile. 47 ContraVir Pharmaceuticals is planning further clinical development of this compound against HBV and HIV.
Cyclic 1-aryl-1,3-propanyl ester prodrugs (HepDirect), are another example of phosphate prodrugs effective as antiviral agents. This class of prodrugs features pradefovir, the 3-chlorophenyl HepDirect prodrug of adefovir,
48
which has been advanced to human clinical trials for hepatitis B infection therapy. The clinical development of pradefovir, as an oral prodrug for chronic HBV infection, although discontinued in USA and Europe, is still progressing in China by Chiva Pharmaceutical.
49
Currently, IDX184, a (SATE)-phosphoramidate diester prodrug of 2′-
Two examples of prodrug approaches (Figure 3) applied to ANPs represented by the acyloxy and alkoxycarbonyl esters, were very effective and led to compounds that entered clinical trial studies and further obtained Food and Drug Administration (FDA) approval. Such compounds currently marketed for antiviral therapy are: adefovir dipivoxil
Finally, the ProTide approach, invented almost 25 years ago by Chris McGuigan has recently been proven very successful in the intracellular delivery of nucleoside monophosphate into the cell, improving the activity of the parent drug.36,60,61 During these 25 years, the ProTide technology was applied to a vast array of nucleosides and these studies have paved the way to the discovery of sofosbuvir (phosphoramidate of 2′-α-C-fluoro-2′-β-C-methyl uridine,
Given the tremendous importance of phosphor(n)amidate prodrugs in the antiviral arena and beyond, after the approval of sofosbuvir
Aryloxyphosphor(n)amidate prodrugs (ProTides)
McGuigan and his team at Cardiff University researched design of novel chemically protected phosphate prodrug groups or motifs, which later became known as “ProTide” technology. A ProTide (pronucleotide) is a nucleoside aryl phosphate or phosphonate masked with an amino acid ester promoiety linked via P–N bond (Figure 4). Such a prodrug is able to enter the cell via facilitated passive diffusion through the cell membrane and when cleaved, it delivers the nucleoside monophosphate or monophosphonate releasing the two masking groups. The amino acid motif is normally selected from a range of natural and unnatural amino acids, although usually
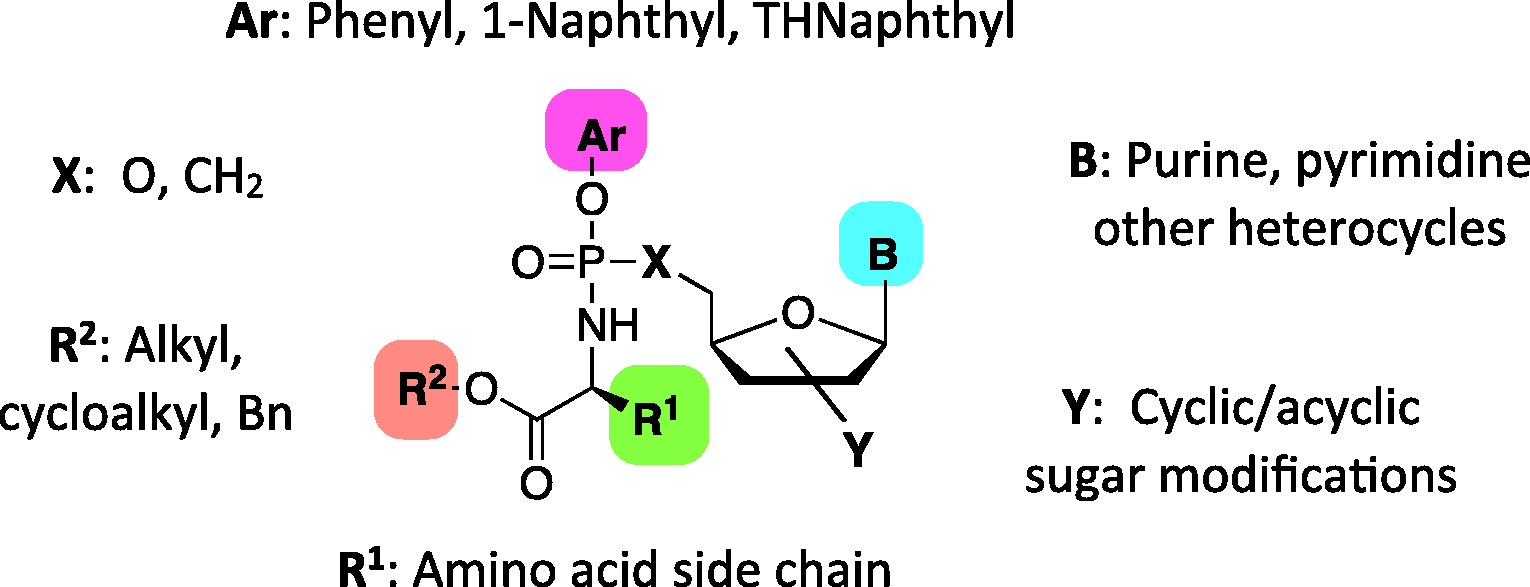
General structure of ProTide scaffold.
The ProTide approach as a strategy to circumvent an impeded 5′-monophosphate formation was extensively applied also to anticancer NAs.66,68–80 An Edinburgh-based clinical-stage pharmaceutical company NuCana, is currently pioneering this technology in the oncology setting in collaboration with our laboratories.
The established position of the ProTide approach in the antiviral and anticancer nucleotide prodrug field provided a foundation for its further expansion into additional research area and/or non-nucleoside type compounds. These include phosphoramidates of carbohydrates such as
Synthetic methods
Aryloxyphosphoramidates
There are three methods for the preparation of aryloxyphosphoramidates as highlighted in the retrosynthetic approaches presented in Scheme 1. These procedures differ in the way the phosphoramidate moiety is introduced at the 5′-hydroxyl group of a NA. Method A is based on a coupling reaction of a NA with a diarylphosphite to form a NA-5′-monoaryl-
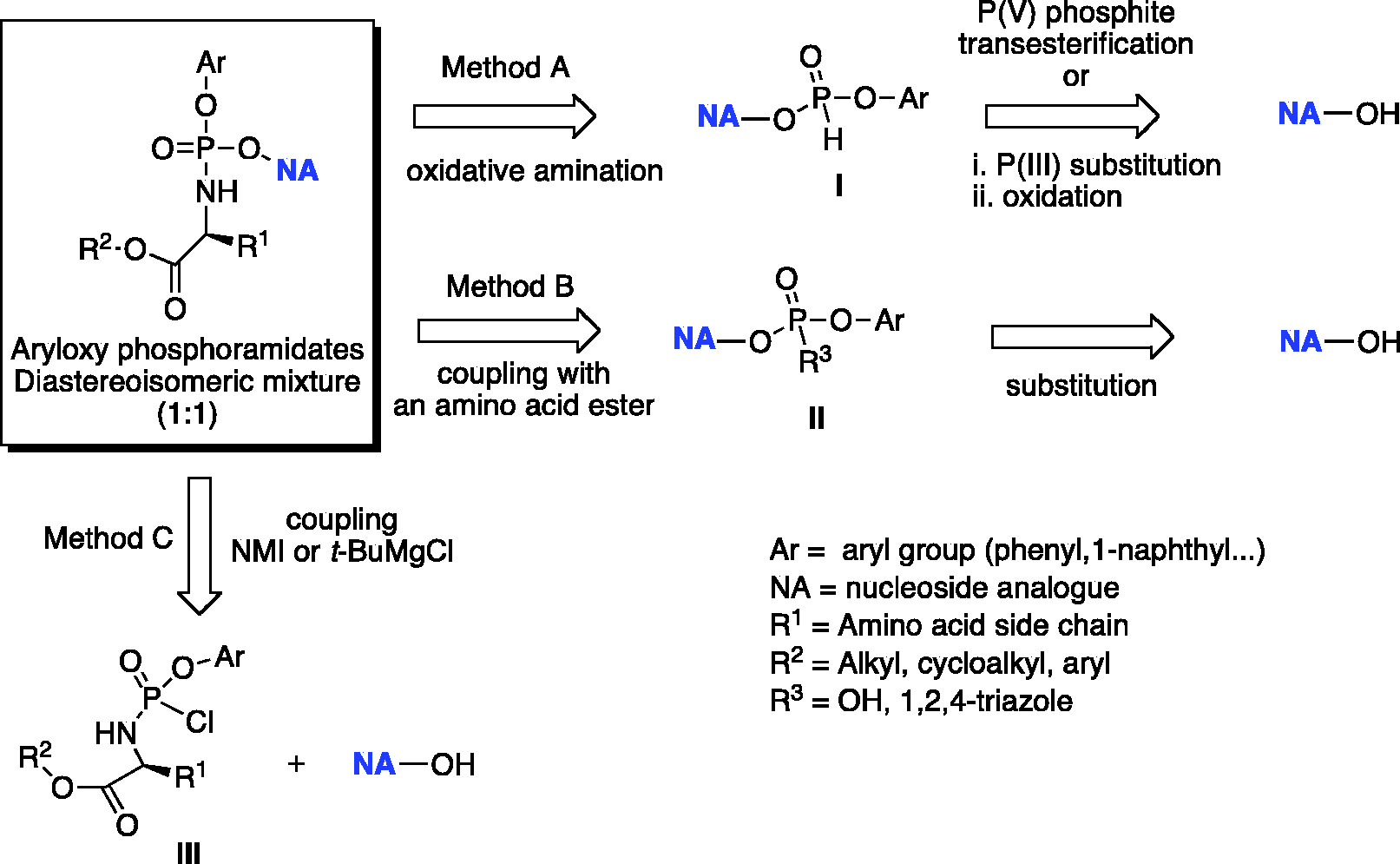
General retrosynthetic approaches for the conventional synthesis of aryloxyphosphoramidates.
The Grignard reagent is not selective thus when used for the coupling reaction, the formation of undesired 3′-phosphoramidate and 3′,5′-bis-phosphoramidate is usually observed. This methodology suffers some limitations such as the need of extensive purification from a complex mixture of 3′,5′-bis-phosphorylated by-products, which is not suitable in case of large-scale synthesis.
Coupling mediated by NMI, which forms a labile imidazolium intermediate with phosphorylating agent, favors the selective phosphorylation of the primary hydroxyl group at the 5′-position of the nucleoside. However, the final outcome in terms of regioselectivity and yield with NMI or
To avoid the formation of undesired 3′,5′- and 3′-phoshoramidates, usually the selective protection of the free 3′-hydroxyl group in the sugar part of a NA prior to the coupling reaction with a phosphorylating reagent
Regioselective synthesis of 5′-O-phosphoramidates.
When the key phosphorylating agents are used as a pair of diastereoisomers at the phosphorus centre (1:1 ratio
Over the past 10 years, the main focus of researchers was concentrated on the development of diastereoselective strategies toward phosphoramidates obtained as a single isomer. Demand for efficient diastereoselective methods appeared to increase particularly after the discovery of a significant difference in the antiviral activity between
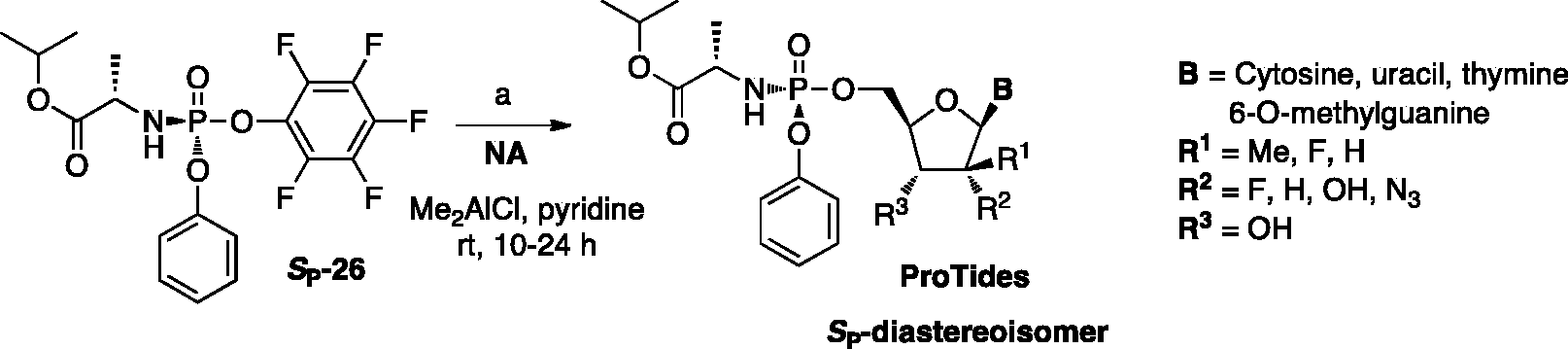
A diastereoselective method to obtain phosphoramidates using a chiral auxiliary-bearing phosphoramidating reagent
Asymmetric synthesis of phosphoramidates via chiral auxiliary.
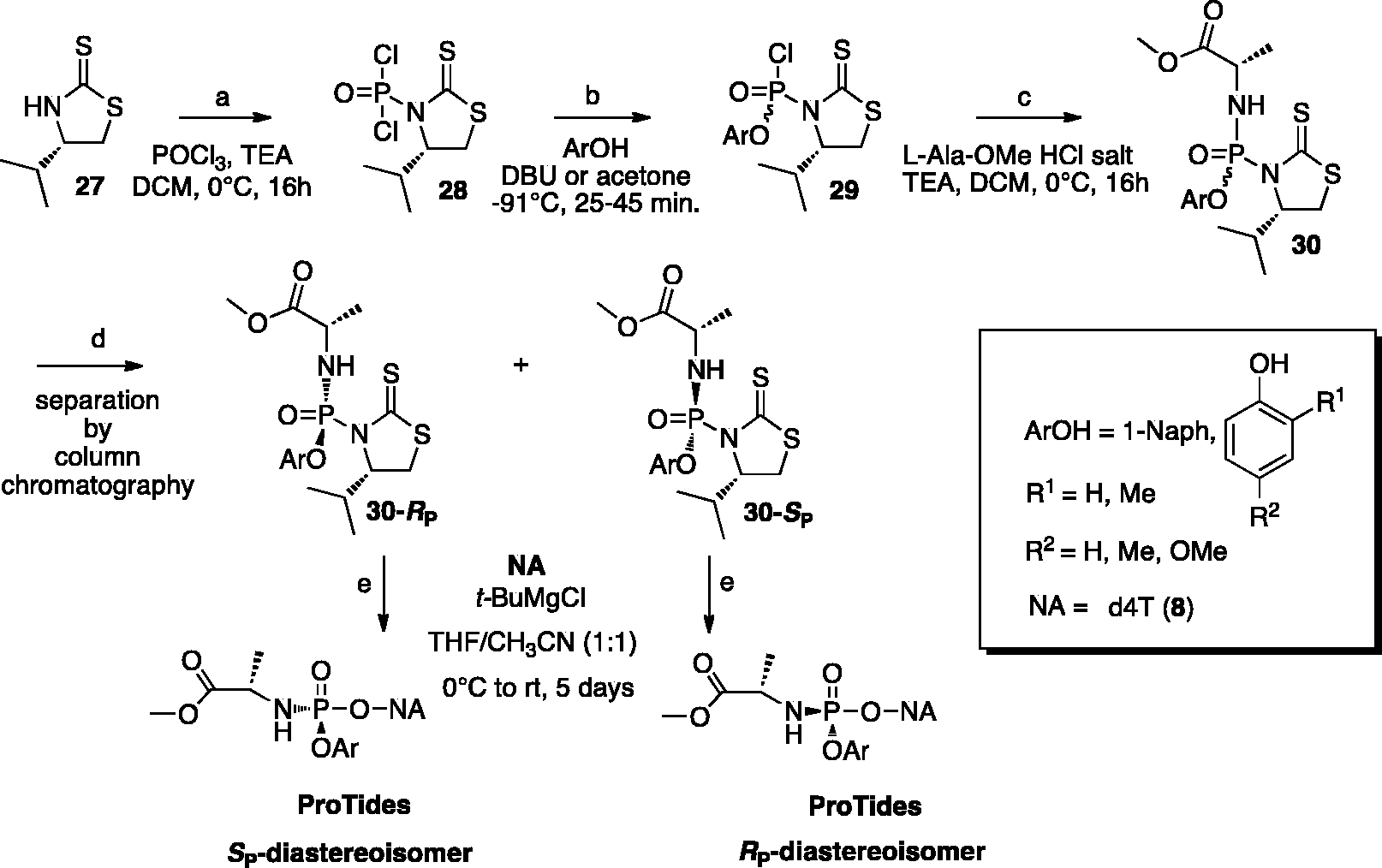
A novel approach to develop a diastereoselective synthesis of aryloxyphosphoramidates was reported more recently by Ross and colleagues.
100
In this approach, a diastereomerically pure phosphoramidating agent with substituted phenolic leaving groups such as
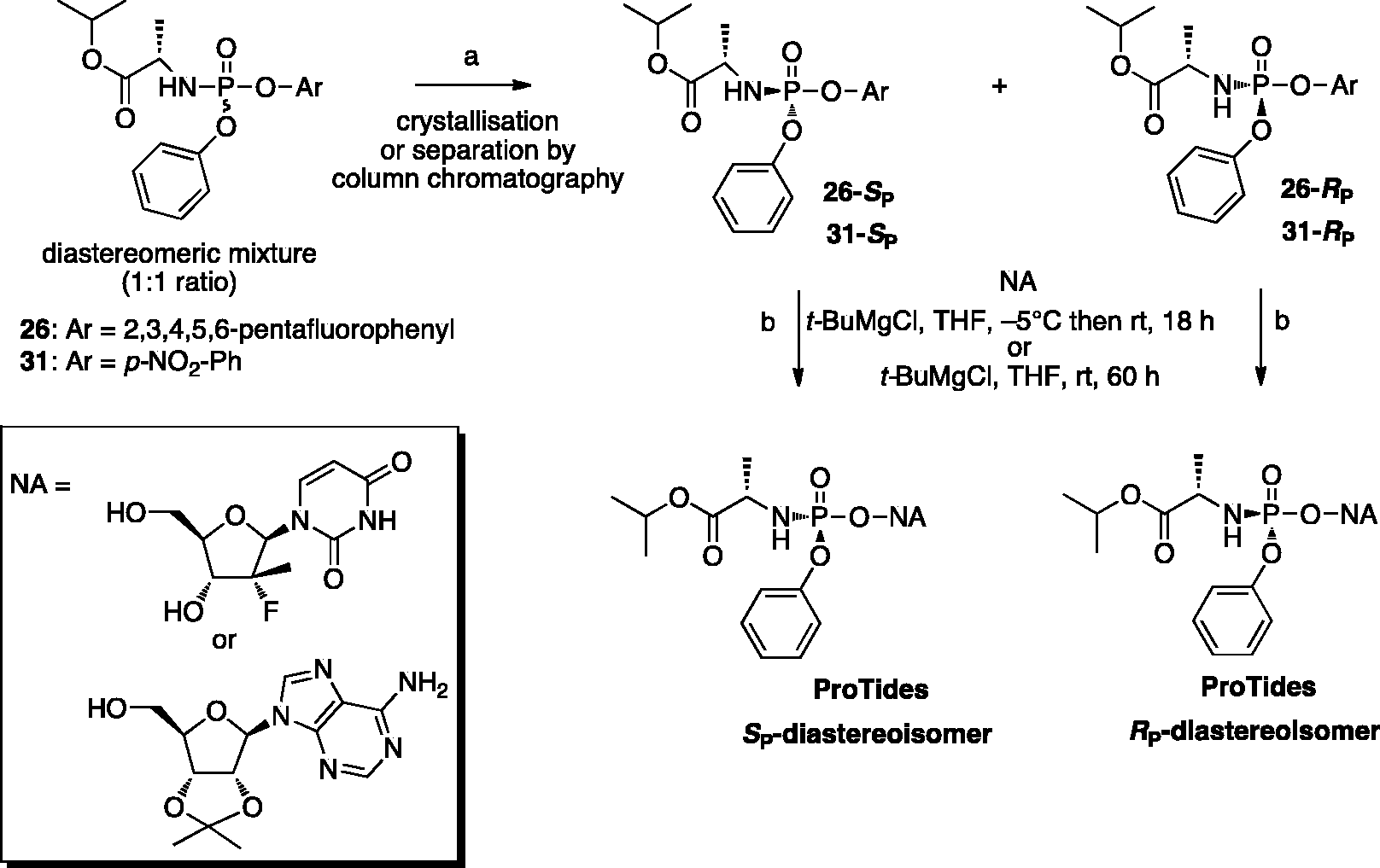
Diastereoselective synthesis of aryloxyphosphoramidates using a single isomer of 2,3,4,5,6-pentafluorophenyloxy or para-nitrophenyloxy phosphorylating agents.
A diastereoselective method for the synthesis of
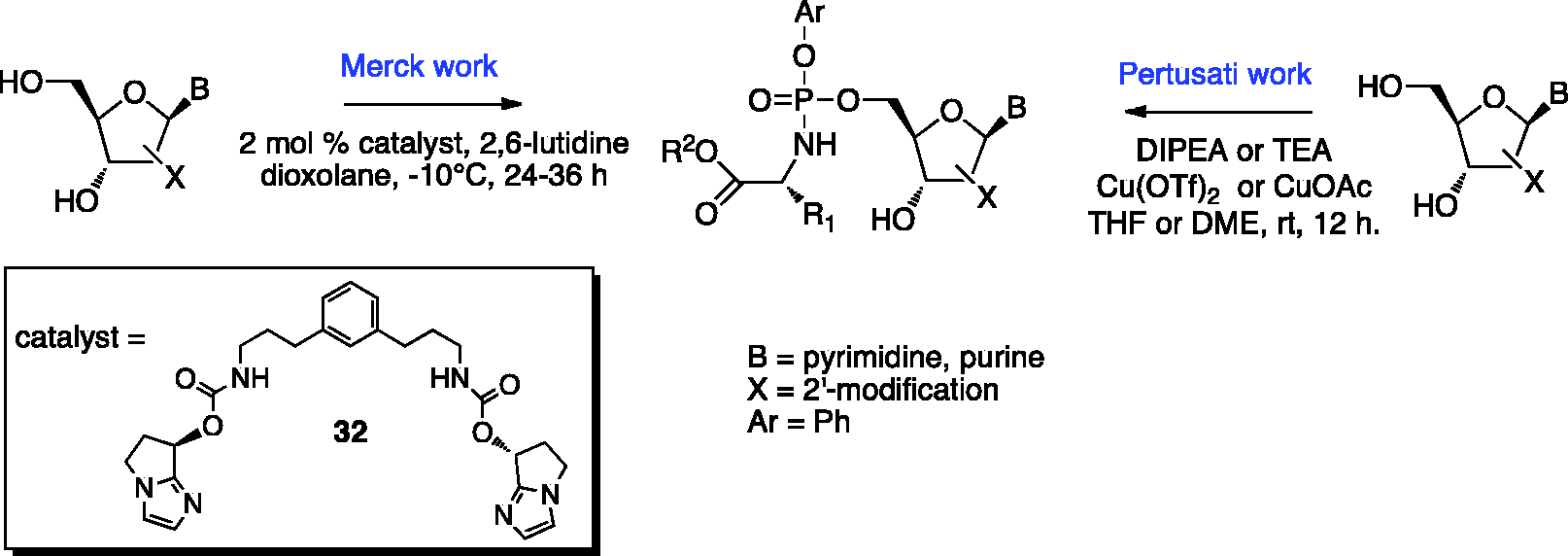
Catalyst-mediated diastereoselective synthetic approaches to phosphoramidates.
Another catalytic stereoselective method that attains high selectivity for nucleoside phosphoramidation was reported in 2017 by researchers at Merck. In this methodology a metal free small-molecule catalyst
All aryloxyphosphoramidates synthetic strategies described above are based on the phosphoramidation process performed at the level of either protected or unprotected NAs. A different synthetic approach, involving the preparation of an aryloxyphosphoramidate ribose derivative as the key building block was recently testified by Gao et al.
118
This aryloxyphosphoramidate ribose bearing
Aryloxyphosphonamidates
The preparation of phosphonamidate prodrugs of ANPs is generally accomplished from the corresponding phosphonic acid via two general procedures A and B (Scheme 6).
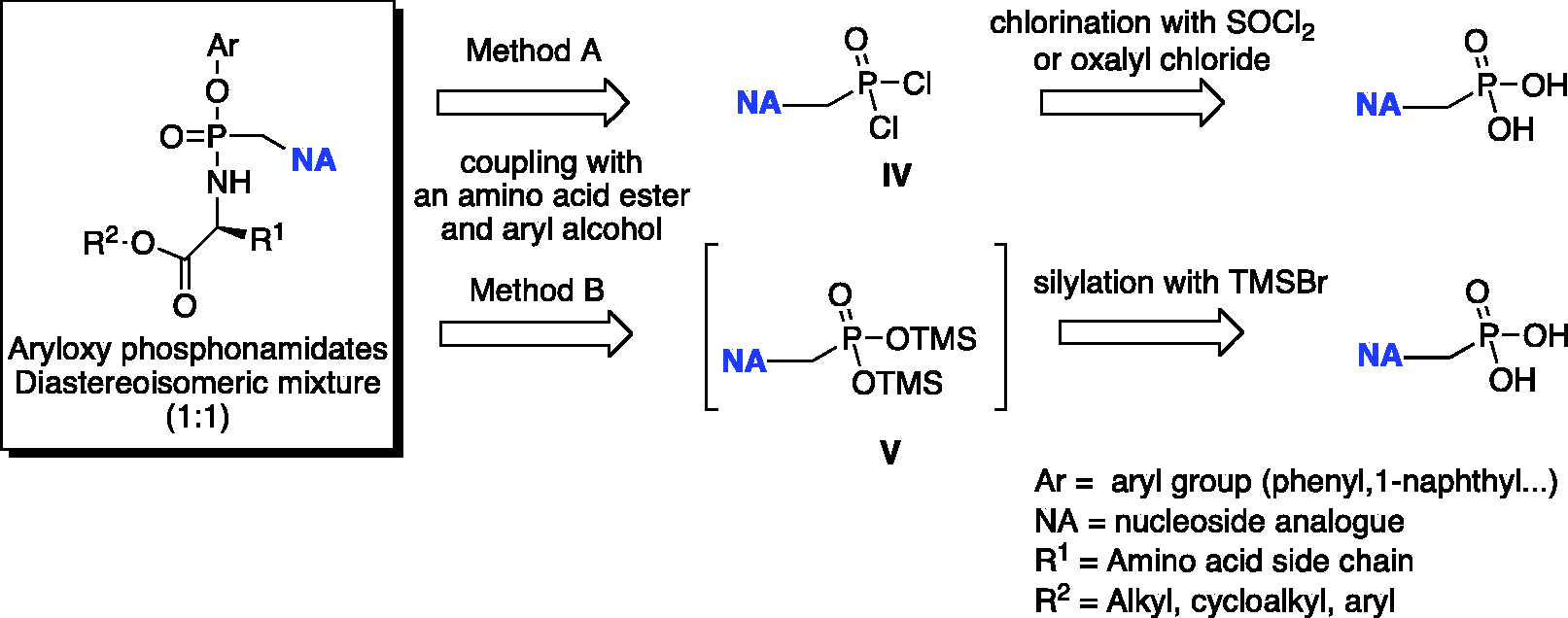
Retrosynthetic analysis for the conventional synthesis of phosphonamidates.
Method A, developed in McGuigan’s laboratory
119
consists of the formation of the nucleoside phosphorodichloridate
Method B is based on a modified methodology for the synthesis of symmetrical bis-amidate prodrugs of ANPs, which was reported first by Janeba and colleagues.
123
This latter procedure consists of the synthesis of the silyl ester of the phosphonic acid via the reaction of a selected ANP with an excess of trimethylsilylbromide (TMSBr) in acetonitrile. The silyl ester was not isolated but immediately reacted with the desired amino acid ester in pyridine and triethylamine used as a coupling reagent with a mixture of triphenyl phosphine and 2,2′-dipyridyldisulfide (Aldrithiol-2). Phosphonodiamidates of several ANPs were obtained in high yields. This method is operatively simpler when compared to the procedures described above and offers the advantage that either the free phosphonic acids or the corresponding alkyl esters can be used as a starting material. This is of a great advantage, considering the difficulties generally encountered in the purification of free phoshonic acids. McGuigan’s group adapted this methodology to the synthesis of phosphonamidate ANPs prodrugs.
66
To accomplish that, the silyl ester
Metabolic activation pathway
The biological activity of aryloxyphosphor(n)amidate prodrugs is expressed after their metabolic activation to the intracellularly released corresponding monophosph(on)ate nucleoside, further phosphorylated to the corresponding active di– and –triphosphate forms by nucleotide kinases. 124
The early reports on the phosphoramidate activation pathway date back to the late 1990s.64,125 In these preliminary investigations, the metabolism of d4T phosphoramidates with pig liver carboxylesterase (CES) was studied 125 using in situ 31 P NMR analysis, a technique following that is now routinely employed in McGuigan’s laboratories as a predictive tool for the likely in vitro biological activity as well as for SAR establishment. The original protocol of this enzymatic experiment was later adapted to study the ProTide first activation step with carboxypeptidase Y enzyme, to prove the nucleoside monophosphate release in biological matrix such as cell lysate 126 or to test the prodrug stability in human serum. 76
Over the same period, these investigations were then extended to AZT phosphoramidates, where stability was tested with pig liver CES and different biological media such as human lymphocyte CEM cell extract, human serum, and mouse serum.64,125
The results of these studies suggested that the carboxylester group linked to the amino acid moiety has pronounced influence on the pharmacokinetics of the prodrugs and their associated stability. Introducing a
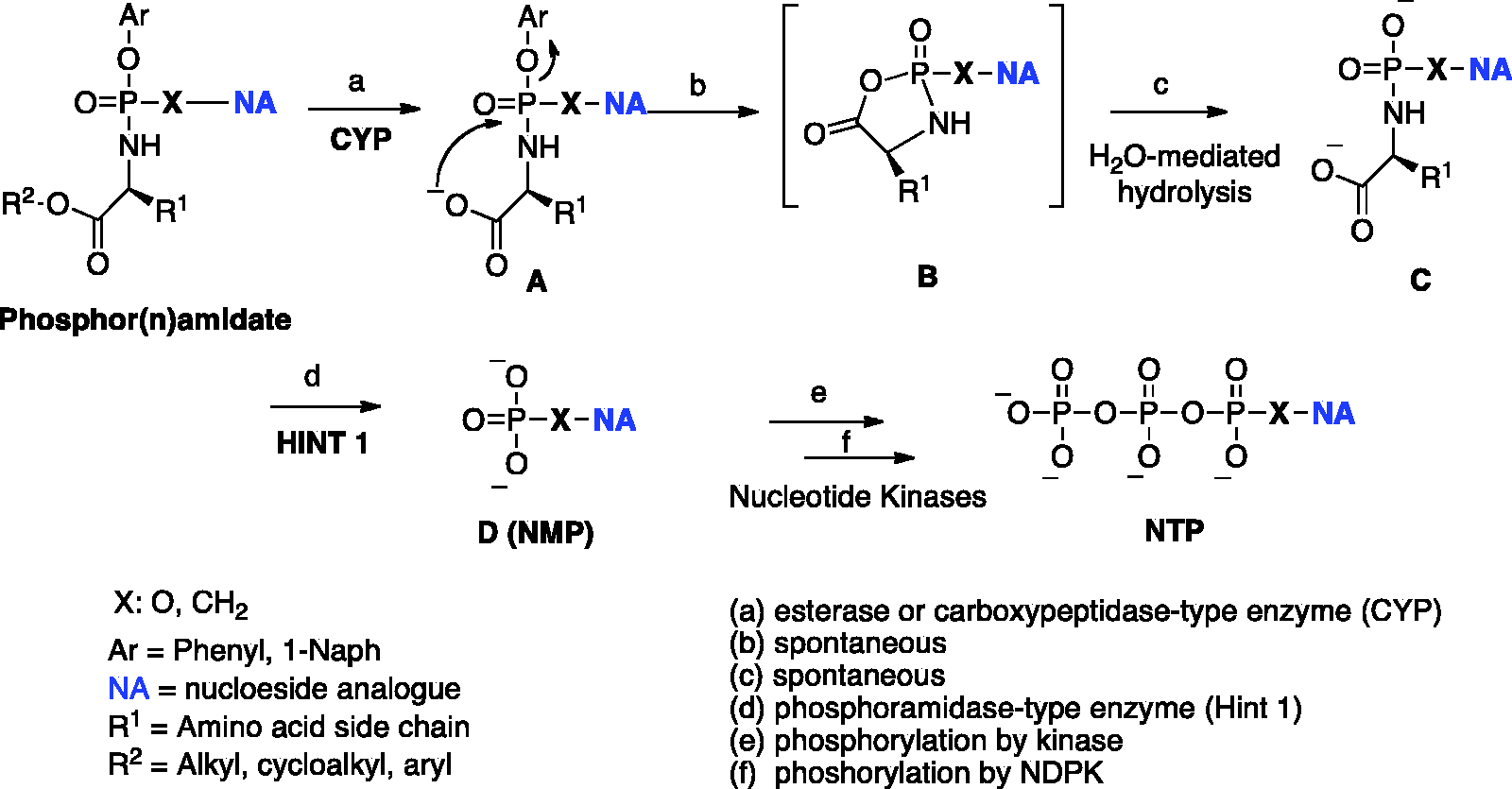
General metabolic pathway for phosphor(n)amidate ProTides.
A detailed mechanism of the hHint1-catalyzed hydrolysis of nucleoside phosphoramidates (in particular sofosbuvir) was recently proposed on the basis of crystallographic studies using a combination of more slowly hydrolyzed substrates and a catalytically inactive mutant enzyme.
134
Molecular modeling of phosphoramidates and their corresponding amino acyl intermediate
ProTide approach application to antiviral nucleosides
Chris McGuigan’s work: From early reports to his latest investigations
The first prototype of phosphoramidates of a nucleoside, reported by McGuigan et al., in 1990, displayed two alkyl amines masking the monophosphate group on AZT. 136 AZT suffers from an absolute dependence on host cell kinase-mediated activation, which can lead to poor activity, emergence of drug resistance, and clinical toxicity. In order to address AZT limitations, McGuigan designed the above-mentioned compounds as membrane-soluble prodrugs of the bioactive nucleotide, capable to bypass the first phosphorylation step. Among different compounds, terminal substituted alkyl amines showed pronounced anti-HIV effect in vitro, which was observed to decline when increasing the length of the methylene spacer. These results were considered consistent with a mechanism of action involving intracellular cleavage of the phosphoramidate P–N bond, and the release of the nucleotide, or a derivative thereof. Thereafter, phosphate triester derivatives of AZT were designed and evaluated against HIV-1 in vitro. 137 During these studies, it was found that simple dialkyl phosphate derivatives of AZT as well as other NAs such as d4T, were inactive as anti-HIV agents, whereas substituted dialkyl phosphates were active. In particular, compounds bearing at the phosphorus centre a trichloro- or trifluoroethyl group and a carboxyl-protected, amino-linked amino acid displayed potent anti-HIV activity and low host toxicity.
Continuing to explore different structures, the phosphorus center was then masked with an ester-containing group in combination with either a simple alkyl moiety or a trichloroethyl group or another ester-containing group. 138 The results of these investigations revealed the presence of two ester-substituted groups enhances activity relative to having only one substituted group. Furthermore, suggesting that a trihaloethyl group may substitute for an ester-containing group but with reduced potency. In several cases, these phosphate derivatives were found to be more selective in their antiviral activity than AZT due to their low toxicity in comparison to the parent nucleoside. Overall, the data supported the conclusion that these phosphate derivatives exert their biological effects via intracellular release of the nucleotide form.
In this report McGuigan stated: “If these in vitro findings could be translated into a demonstrable in vivo advantage, such phosphate pro-drugs could have merit as candidates for clinical development.” 138 This was clearly an anticipation of what would have happened 25 years later. His investigations underlaid the importance of the masked phosphate approach, and had significant implications for what became the future design of chemotherapeutic NAs.
The ProTide series of AZT is the earliest example of aryloxyphosphoramidate technology reported by McGuigan’s group in the early 1990s.139,140
In vitro evaluation revealed these compounds had a pronounced, selective anti-HIV activity in CEM cells; the magnitude of the biological effect varied considerably depending on the nature of the phosphate-blocking groups. Moreover, several of the compounds retained marked antiviral activity in TK- (thymidine kinase-deficient) mutant CEM cells in which AZT was virtually inactive. Diaryl phosphate derivatives of the anti-HIV NA AZT were also investigated as potential prodrugs of the bioactive free nucleotide. The compounds were shown to be inhibitors of HIV replication in several cell lines, and show reduced cytotoxicity in vitro, by comparison to the parent nucleoside. However, in contrast to the previously reported aryloxyphosphoramidate derivatives, the diaryl phosphates of AZT showed to be poorly active in HIV-infected TK-deficient CEM cells. The results clearly pointed to the aryloxyphosphoramidate as the most promising structure for the delivery of the nucleotide and paved the way for the development of this class of prodrugs.
Thereafter, the ProTide technology was extensively and successfully applied to a high number of nucleoside phosphates with antiviral activity. In particular, following AZT studies, extensive SAR studies investigating the aryl, amino acid, and ester moieties were carried out on d4T phosphoramidates.141–145 In these reports, the preliminary results on the activation mechanism of such prodrugs were also described.125,146 The nature of the amino acid appeared to be extremely important for the eventual antiviral action. Among the amino acids studied,
As part of research project (sponsored by GlaxoSmithKline in Research Triangle Park North Carolina) devoted to discover anti-HIV and anti-HBV agents, McGuigan’s team applied the ProTide approach to 2′,3′-didehydro-2′,3′-dideoxyadenosine and other 2′,3′-dideoxy nucleosides including 2′,3′-dideoxyuridine, -adenosine, -3′-fluoroadenosine, -uridine147–153 and also to the carbocyclic nucleoside ABC
The phosphoramidate technology was also applied to uridine-based NAs. Although their triphosphate forms were found to posses inhibitory activity on uridine triphosphate (UTP) incorporation into RNA of influenza virus, in general they are characterized by poor antiviral activity which may be related to their inefficient phosphorylation. 99 However, in this case the ProTide approach was not very successful leading to compounds with weak antiviral activity. The slow release of the active monophosphate species of these compounds observed in cell lysate, as well as inefficient di- or triphosphorylation of 5′-monophosphate forms were considered as possible explanations for their weak antiviral activity.
A different outcome was instead obtained with ProTides of 6-modified 2′-fluoro-2′-deoxyguanosines, which showed marked antiviral activity in vitro assays proving that this class of prodrugs can be pursued for influenza virus therapy. 157 Rapid metabolic activation in enzymatic assays with yeast carboxypeptidase Y or crude cell lysate supported the antiviral results. Evidence for efficient removal of the 6-substituent on the guanine part was provided by enzymatic studies with adenosine deaminase, and by molecular modeling of the nucleoside 5′-monophosphates in the catalytic site of a model of this enzyme (ADAL1), thus indicating the utility of the double prodrug concept.
No improvement or broadening of the antiviral activity of the parent nucleoside RBV was obtained with a family of ProTides. 158 Again, a likely explanation for this lack of activity was attributed to their poor activation to the free 5′-monophosphate, as evidenced by cell lysate incubation studies. While enzymatic studies with carboxypeptidase Y indicated that the first step in the activation of RBV ProTides was efficient, molecular modeling data with the Hint enzyme suggested that subsequent amino acid cleavage to liberate the necessary free 5′-monophosphate was most probably impeded in this case. 158 Other examples in which the ProTide technology showed a lack of significant improvement of the antiviral activity versus the parent compounds are the phosphoramidates of 2-fluoro derivatives of the bicyclic NA Cf1743, the most potent anti-VZV agent reported to date, 159 and of 2′-deoxy-2′,2′-difluoro-5-halouridine. 160
A collaboration between McGuigan’s group (Cardiff University, UK) and Van Calenbergh’s laboratories (Ghent University, Belgium) led to the investigation of α-
More recently, the phosphoramidate approach was applied by the same authors to the family of apionucleosides
162
such as 2′,3′-dideoxy-β-
While all target nucleosides failed to show significant antiviral activity, the authors demonstrated that the triphosphate of 2′,3′-deoxy-
Application of the ProTide approach to ACV was also extensively investigated in the McGuigan laboratories. Although ACV makes an important contribution to the therapy of herpes infections, it has some limitations such as low oral bioavailability and drug resistance caused by mutation in either the TK or DNA polymerase.17,18 Though the oral bioavailability can be increased by the amino acid prodrugs of ACV, these compounds can be cleaved in the gut and the liver by hydrolase enzymes. Interestingly, ACV was reported to inhibit HIV in human herpes virus (HHV) co-infection in tissue cultures.
165
This activity was found to be correlated with the phosphorylation of the parent drug to the monophosphate form mediated by HHV-encoded kinase(s), whereas further phosphorylation steps provided the active triphosphate form of ACV able to inhibit HIV-RT. Because, HIV does not encode an enzyme that recognizes ACV as a substrate for its activation (phosphorylation) step, the HHV coinfection is needed for ACV to exhibit activity. ACV was therefore a perfect substrate for the application of ProTide technology. Based on these observations, McGuigan and colleagues presented the synthesis and initial biological evaluation (against HSV-1 and HSV-2 and against HIV-1 and -2) of a series of ACV ProTides.135,166,167 The application of this strategy was efficient to overcome two main issues associated with ACV: to bypass its poor efficiency of diffusion through intact cell membranes and the first limiting phosphorylation step. SAR studies showed that in general ester and aryl variations were well tolerated, whereas the variation of the amino acid moiety seemed to be tolerated only in the case of HSV. Regarding in vitro HIV screening, good results were obtained only for the
In the absence of HHV infection, the prodrug compounds showed antiviral activity, demonstrating their nucleoside kinase independence. These findings were also supported by a different study, where ACV phosphate prodrugs showed a full retention of antiviral activity against HSV-1 and VZV TK-deficient strains. Enzymatic and molecular modeling studies were performed to better understand the antiviral behavior of these compounds. These indicated that ProTides with diminished biolability toward carboxypeptidase translate to poor anti-HIV agents and vice versa. Given that, this enzymatic assay became a predictive tool regularly used to assess potential activity of phosphoramidate prodrugs of other NAs. To overcome the cytotoxicity observed with these prodrugs, very recently a virtual screening on a library of ACV derivatives was reported. 168 Docking experiments with a database of 3600 compounds against three different enzymes encompassing HIV reverse transcriptase, adenylate or guanylate kinase, and a model of DNA polymerase γ resulted in the selection of five NAs as potentially strong RT inhibitors and weak cellular DNA polymerase inhibitors including GCV, PCV, 6-Cl-PCV, 6-OMe-PCV and 2′-SH-GCV. Several phosphoramidate prodrugs of the selected NAs were synthesized and assessed for their potency against HIV, HSV, VZV, and HCMV. Most of the compounds exhibited inhibitory activity against HIV with activity in the low micromolar range, but again some toxicity was observed.
As reported before, over the last 20 years, ANPs have emerged as a novel class of clinically effective antiviral agents.
168
Explorations of various types of nucleoside phosphonate prodrugs have also led to the design and development of their aryloxyamidate prodrugs. ProTides originally designed to deliver nucleoside monophosphates, have also been successfully applied to nucleoside phosphonates. McGuigan’s group was the first to report the synthesis and biological evaluation of ProTides based on adefovir and tenofovir.
119
Results of these studies indicate similar SARs for such prodrugs as earlier noted for NAs like d4T (
This early work was followed by investigation into the synthesis and antiviral evaluation of a broad family of phosphonamidates of adefovir and tenofovir. 66 ProTides, synthesized with a more efficient methodology, showed improved in vitro anti-HIV activity, compared to previously reported data for the parent ANPs. Phosphonamidates bearing 5,6,7,8-tetrahydro-1-naphthol unit which was for the first time introduced as hydrolysable aryl unit in the ProTide motif displayed an improved antiviral activity compared to the “common”-naphthyl and phenyl ProTide units. Enzymatic studies showed that this novel aryloxy group was processed through the “standard” metabolic pathway. In this view 5,6,7,8-tetrahydro-1-naphthol was considered as a good aryl group for future improvement of the ProTide motif and thus was subsequently used by the same authors to design phosphonamidates of C5-substituted pyrimidine acyclic nucleosides functionalized with but-2-enyl-chain. 67
In the search of anti-HCV agents, McGuigan’s group started a project (funded by Roche Palo Alto), involving application of the ProTide technology to the ribonucleoside analogues 4′-azidouridine,
169
4′-azidoadenosine,
170
and 4′-azidocytidine.
171
Although 4′-azidouridine and 4′-azidoadenosine did not inhibit HCV, their triphosphate forms showed potent inhibitory activity against HCV RNA polymerase. Several phosphoramidates of these NAs were prepared, including variations in the aryl, ester, and amino acid regions. Among a number of 4′-azidouridine and -adenosine prodrugs with sub-micromolar inhibition of HCV replication in cell culture, the 1-naphthyl
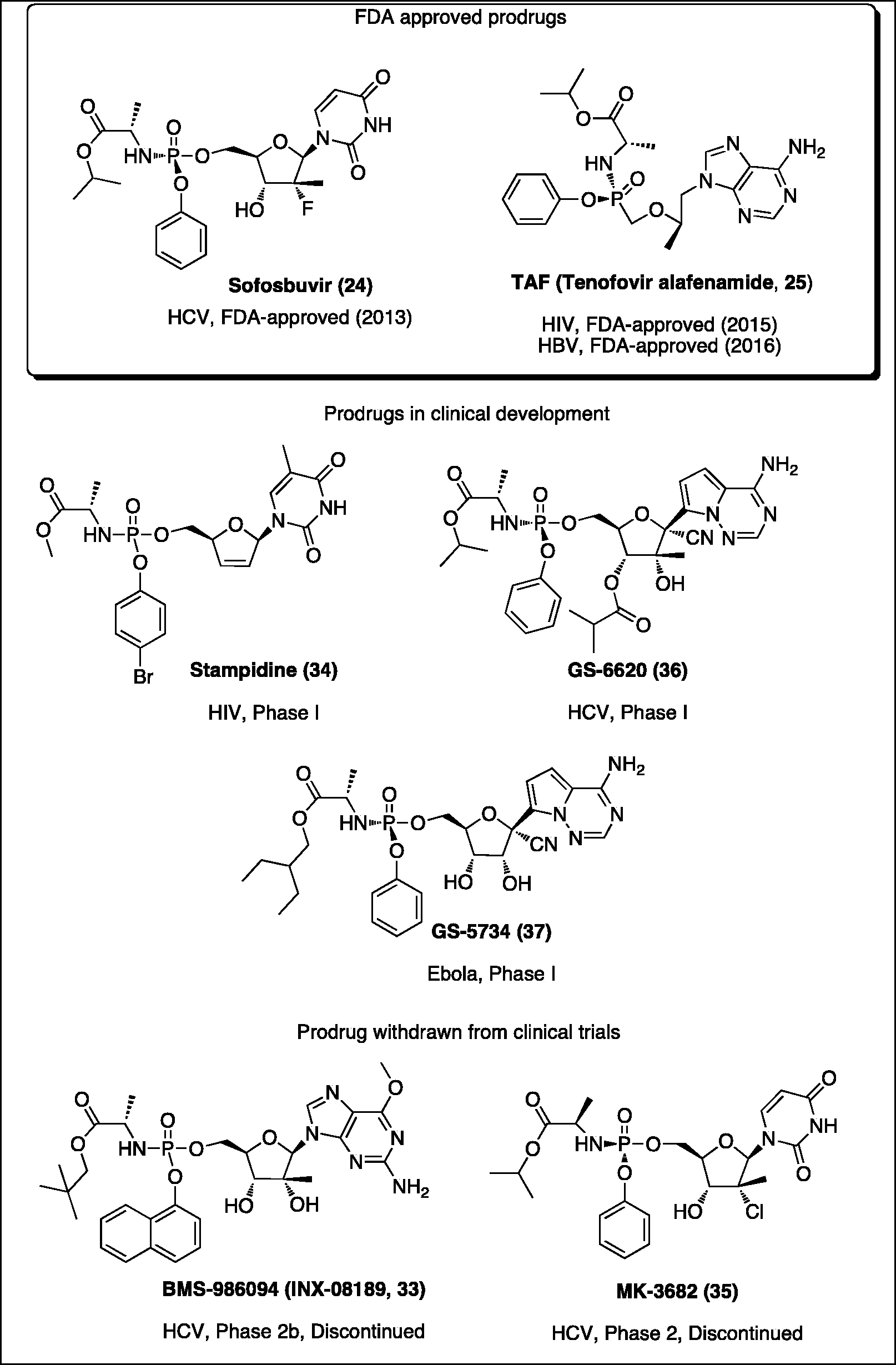
Antiviral phosphor(n)amidates in clinical use or in clinical development.
INX-08189 exhibited nanomolar activity in vitro in HCV replicon assay, with EC50s of 10 nM against genotype 1b, 12 nM against genotype 1a, and 0.9 nM against genotype 2a after 72 h of exposure.
174
It was also tested in the S282T mutant replicon and showed reduced activity with a 10-fold change in the EC50 yet still being capable to complete inhibition of HCV replication with EC90 value of 344 nM. In the replicon inhibition studies in which INX-08189 was used in combination with RBV, a high degree of synergy against the wild-type (WT) and S282T mutant replicons was observed. Intracellular metabolism of INX-08189 and its conversion to the active 5′-triphosphate form was investigated in the HCV genotype 1b replicon assay, showing an intracellular concentration of the triphosphate form (2′-
ProTides in clinical use or in clinical development as antiviral agents
The research undertaken in McGuigan’s laboratories during the last 25 years was of great inspiration for many scientists all over the world. Nowadays ProTide technology is recognized as a prodrug strategy with proven capacity to generate new drug candidates for nucleoside-based antiviral indications. In fact, a potential of this prodrug approach was confirmed with the discovery of agents that are currently in clinical use (sofosbuvir and TAF), or in clinical development as antiviral drugs including stampidine
Sofosbuvir
During the time of McGuigan’s research on HCV, phosphoramidate prodrugs of the 5′-phosphate β-
Tenofovir alafenamide (TAF)
As mentioned above, one of the most successful example of ANP prodrug is represented by TDF (
To circumvent these major problems, Gilead Sciences developed TAF (TAF,
In preclinical animal studies, TAF exhibits enhanced distribution of tenofovir into PBMCs and the lymphatic organs after oral administration, in comparison to tenofovir disoproxil fumarate. Twenty-four hours after a single dose of TAF in dogs, the concentration of tenofovir in lymphatic organs is between 5- and 15-fold greater than an equivalent dose of TDF. Intracellular tenofovir concentrations, measured by AUC0–24, in PBMCs after a single oral dose of TAF in dogs are ∼38-fold greater after an equivalent oral dose of TDF and ∼100-fold greater than those observed after subcutaneous administration of tenofovir. These in vivo pharmacokinetics studies showed also that both (GS-7340 and GS-7339) were rapidly eliminated in plasma relative to tenofovir with the
Moreover, intracellular activation and antiviral activity of TAF are adversely non-affected by other medications (often administered in combination) such as HIV and HCV protease inhibitors, 205 except for telaprevir and boceprevir that non-specifically inhibit cathepsin A, the key enzyme responsible for the activation of the prodrug. 127
Furthermore, the rapid intracellular cleavage step catalyzed by cathepsin A in HIV- and HBV target cells, coupled with the formation of poorly permeable metabolites (the charged phosphates) effectively trapped in cells, accounts for the substantial accumulation of the pharmacologically active metabolite tenofovir diphosphate and increased therapeutic efficacy of TAF with respect to other prodrugs. Because of this accumulation inside the infected cells, TAF can be administered in lower therapeutic doses in comparison to TDF. Phase I/II clinical study to asses the pharmacokinetics, safety and anti-HIV activity of TAF showed that administration of 40 mg of TAF for 14 days in HIV-infected patient resulted in lower tenofovir
TAF was also investigated for the treatment of HBV infections. In particular, Phase III clinical trials studies evaluating investigational use of once-daily 25 mg dose of TAF in treatment-naive and treatment-experienced adults with HBeAg-negative and HBeAg-positive chronic HBV infections, demonstrated again that TAF was non-inferior to TDF based on the percentage of patients having low HBV DNA levels after 48 weeks of therapy. In addition to high efficacy, the results of these studies reflect improved renal and bone safety parameters similar to those seen in clinical studies evaluating TAF-based regimens for HIV. In November 2016, TAF received FDA approval for the treatment of chronic HBV. 209
Stampidine
Stampidine
Uprifosbuvir
Uprifosbuvir (MK-3682, IDX-21437,
GS-6620
Prodrugs in clinic and clinical development.
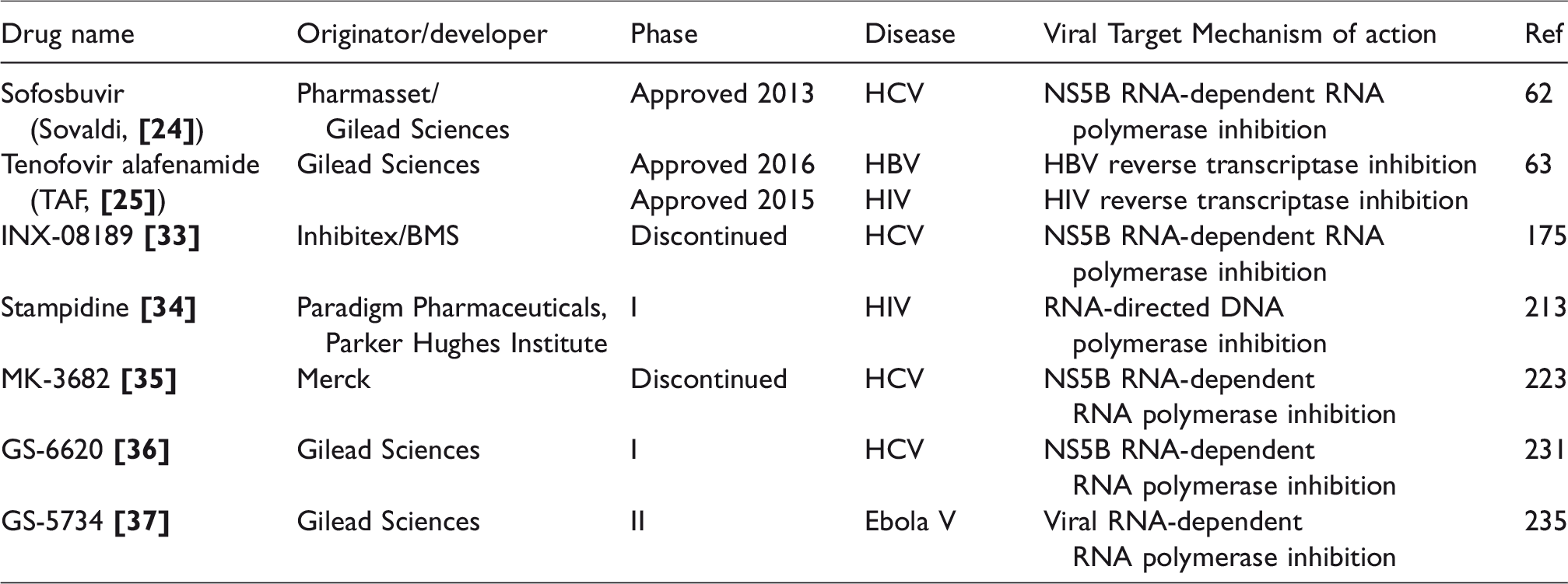
GS-5734
GS-5734
Conclusion
The idea of phosph(on)ates prodrugs with aryloxyphosphoramidates (ProTides) in particular, was extensively investigated over the last three decades. Designed and pioneered by McGuigan and his group, the ProTide approach was successfully applied to a great variety of modified NAs with antiviral and anticancer activity. In this review, we presented a summary of the “ProTide technology” development commencing from its discovery to the most recent applications in the antiviral field. We outlined a discovery and clinical development of recently FDA-approved sofosbuvir (Sovaldi®), one of the best-selling drugs for treatment of hepatitis C infection. We highlighted all synthetic methodologies towards aryloxphosphor(n)amidates including the latest, more sophisticated regio- and stereoselective procedures. Given its increasing interest in the scientific community, as well as a significant commercial impact, the ProTide technology will continue to be used as a strategy to improve the efficacy profile of nucleoside and also non-nucleoside existing and/or novel therapeutics. Certainly, McGuigan’s discovery and extensive research on the phosphoramidate approach has provided an inspiration and the solid foundation for bringing the ProTide concept into the clinical success.
Footnotes
Acknowledgement
We would like to acknowledge all the scientists from both pharmaceutical companies and academic institutions, who for many years believed in and supported Chris’s research, driving ProTide technology to success. We particularly wish to note the valuable contribution of Jan Balzarini, who was both a long-term collaborator and a dear friend of Chris. The joint efforts of Chris and Jan, coupled with their passion and dedication to investigate new medicines were undoubtedly crucial to the development of ProTide technology.
We also wish to express our sincere thanks to the work of the over 100 postdocs and PhD students who together formed part of Chris’s group for over 25 years. Their contribution was invaluable to the successful results achieved.
We also wish to extend a special thanks to Helen Murphy and Julie Hayward, for their assistance to Chris’s group, as his PA during these 25 years.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: McGuigan's laboratory actively collaborated with Inhibitex inc., and BMS (whose agent INX-08189 is discussed herein) received financial support for this work.
