Abstract
Background
Rilpivirine (TMC278LA) is a promising drug for pre-exposure prophylaxis of HIV-1 because of its sub-nanomolar potency and long-acting formulation; however, increasing transmission of non-nucleoside reverse transcriptase inhibitor-resistant HIV-1 with potential cross-resistance to rilpivirine could reduce its preventive efficacy. This study investigated rilpivirine cross-resistance among recombinant subtype C HIV-1 derived from 100 individuals failing on first-line non-nucleoside reverse transcriptase inhibitor-containing antiretroviral therapy in South Africa whose samples were sent for routine HIV-1 drug resistance testing to Lancet Laboratories (Johannesburg, South Africa).
Methods
Plasma samples were selected from individuals with HIV-1 RNA > 10,000 copies/ml and ≥1 non-nucleoside reverse transcriptase inhibitor-resistance mutation in reverse transcriptase. Recombinant HIV-1LAI-containing bulk-cloned full-length reverse transcriptase sequences from plasma were assayed for susceptibility to nevirapine (NVP), efavirenz (EFV) and rilpivirine in TZM-bl cells. Fold-change (FC) decreases in drug susceptibility were calculated against a mean IC50 from 12 subtype C HIV-1 samples from treatment-naïve individuals in South Africa. Cross-resistance was evaluated based on biological cutoffs established for rilpivirine (2.5-FC) and the effect of mutation combinations on rilpivirine phenotype.
Results
Of the 100 samples from individuals on failing antiretroviral therapy, 69 had 2.5- to 75-fold decreased susceptibility to rilpivirine and 11 had >75-fold resistance. Rilpivirine resistance was strongly associated with K103N especially in combination with other rilpivirine-associated mutations.
Conclusion
The frequently observed cross-resistance of HIV-1 suggests that the preventive efficacy of TMC278LA pre-exposure prophylaxis could be compromised by transmission of HIV-1 from individuals with failure of first-line non-nucleoside reverse transcriptase inhibitor-containing antiretroviral therapy.
Keywords
Introduction
Long-acting injectable antiretroviral agents are attractive as pre-exposure prophylaxis (PrEP) for HIV-1 prevention due to their requirement for infrequent dosing. TMC278LA, the long-acting formulation of the non-nucleoside reverse transcriptase inhibitor (NNRTI) rilpivirine (RPV), has a half-life of 91 days and a sub-nanomolar EC50 to wild-type HIV-1.1,2 TMC278LA administered at a dose of up to 1200 mg via intramuscular injection every four to eight weeks has been found to be safe and well tolerated in several Phase I and II clinical trials.3–5 Mathematical modeling predicts that TMC278LA has the potential to reduce new HIV-1 infections by up to 17% and be cost-effective if implemented in targeted high-risk populations.6,7
A major concern about TMC278LA is the potential risk of selection of NNRTI-resistant HIV-1 in newly infected persons because of the prolonged sub-therapeutic levels of TMC278LA after dosing is delayed or stopped. 8 For example, in an RT-SHIV-infected macaque intravenously administered TMC278LA, the RPV-associated mutation E138Q was selected 17 weeks after the second TMC278 injection and conferred low level (4-fold) RPV resistance. 9 In the Phase I SSAT040 trial, one participant was exposed to HIV-1 approximately 41 days after receiving a 300 mg dose of TMC278LA. Plasma RPV concentration was under 25 ng/µL when infection occurred. This was low enough to allow breakthrough infection of wild-type virus but high enough to subsequently select NNRTI resistance with K101E. RPV remained detectable in plasma below 50 ng/µL more than 200 days post-injection. 10 Sub-therapeutic RPV levels in follow-up samples from the MWRI-01 study also persisted more than 500 days after a single-dose injection of 600 or 1200 mg TMC278LA. 11
A second concern is whether RPV concentrations in the genital tract will be sufficient to protect against infection with either wild-type or NNRTI-resistant variants, the latter which are increasingly prevalent. Nearly six times more RPV was needed to attain EC90 in ectocervical tissues cultured with HIV-1BaL compared to colonic tissue. 12 In explant tissues from participants in MWRI-01 given a single 600 or 1200 mg dose of TMC278LA, HIV-1 was suppressed up to four months post-dosing in rectal tissue cultures, but was not suppressed in cervical and vaginal tissue cultures in the same time period. 5 Sub-optimal drug levels in the genital tract could further increase the risk of infection by HIV-1, in particular by NNRTI-resistant variants.
Several studies have assessed the frequency of RPV cross-resistance against other NNRTI in the context of second- or third-line ART. In a genotypic evaluation of individuals experiencing virologic failure on NNRTI-based therapies that did not include RPV, 20% of sequences were found to contain mutations known to reduce susceptibility to RPV. 13 Fourteen of 38 (37%) clinically derived HIV-1 subtype C clones having one or more NNRTI resistance mutation also demonstrated reduced susceptibility to RPV. This resistance was driven by the number of NNRTI resistance mutations per clone. 14 In vitro analyses of NNRTI resistance mutations in both subtype B and C HIV-1 laboratory variants showed that >20-fold resistance to RPV was achieved by the NNRTI mutations Y181I/V and/or K101P.14,15 HIV-1LAI with different, single NNRTI mutants maintained susceptibility to RPV, except for HIV-1 with Y181I and the double mutant K101P/V179I. 16 No studies to date have examined RPV resistance arising from first-line ART.
The current study evaluated the activity of RPV against drug-resistant clinically derived subtype C HIV-1 arising from failure of first-line NNRTI-containing regimens from South Africa to evaluate the possible risks and benefits of using TMC278LA as a PrEP agent in regions where NNRTI-resistant subtype C HIV-1 may be circulating as a potentially transmissible variant.
Methods
Clinical samples
The sample set used in this analysis has been described previously. 17 Plasma from 100 HIV-1 subtype C-infected individuals whose samples were sent for routine HIV-1 drug resistance testing to Lancet Laboratories (Johannesburg, South Africa) were evaluated. The specimens were from individuals who experienced virologic failure defined as having >10,000 HIV-1 RNA copies/ml after six months of nevirapine (NVP)-containing ART (N = 12 individuals) or efavirenz (EFV)-containing ART (N = 88 individuals; one individual had also taken single-dose NVP to prevent perinatal transmission). The viral population genotype of each sample had at least one major NNRTI resistance mutation in HIV-1 reverse transcriptase (RT) as defined by Stanford HIVdb v7.0.18,19 Plasma samples contained a median of 3 [Q1–Q3: 2–4] NNRTI-associated drug resistance mutations which included A98G, L100I, K101E/H, K103N/S, V106M, V108I, E138A/K, V179D/E Y181C, Y188L/C, G190A, H221Y, P225H, F227L, and M230L. The most frequent EFV and NVP resistance mutations were K103N in 55 of 100 samples (55%), V106M (44%) and G190A (26%). In total, 94 of 100 samples from patients failing ARV treatment also carried HIV-1 NRTI resistane mutations including M184V (82%), K65R (35%), L74I (19%), M41L (17%) or D67N (17%). Resistance mutations in the connection and RNAse H domains of RT spanning amino acids 320–560 included Y318F (3%) and N348I (14%). Additional subtype C resistance-associated C-terminal mutations/polymorphisms observed were G335D (86%), A371V (19%) and A376S (12%). From the treatment-naïve group, eight individuals had G335D and one individual had A376S.
Subtype C samples from 12 HIV-1 infected treatment-naïve individuals from the same geographical location were collected as controls. No NRTI or NNRTI resistance mutations were observed in any of the control sequences. All donor samples were anonymized, and testing was approved by the South African Medical Association Research Ethics Committee (SAMAREC) and the Institutional Review Board of the University of Pittsburgh.
Generation of full-length patient-derived HIV-1xxLAI
Full-length patient-derived virus was generated as previously described. 17 Patient-derived full-length RT was bulk cloned into xxLAIxhoI to preserve sequence diversity using the In-Fusion® HD Cloning System (Clonetech), and plasmid DNA was purified using the PureYield™ Plasmid Midiprep System (Promega). Lipofectamine2000 (Life Technologies) transfection into 293T cells was performed to generate infectious viral clones.
HIV-1 phenotyping
A normalized input of 100 relative light units (RLU) was used to infect untreated or RPV-treated TZM-bl cells in a luciferase-based single-cycle drug susceptibility assay (Britelite Plus; Perkin–Elmer) as previously described. 9 RPV is commercially available ARV for HIV treatment, and the long-acting version TMC278LA is an investigational product for HIV prevention, not yet approved by the FDA. RPV was obtained through the AIDS Research and Reference Reagent Program, Division of AIDS, NIAID, NIH: Rilpivirine (Cat# 12147) from Tibotec Pharmaceuticals, Inc. Four-parameter, non-linear regression for curve fitting was used to generate IC50 values using GraphPad Prism 6 software (GraphPad Software, Inc). All IC50s were evaluated as fold-change (FC) values determined by dividing the IC50 generated for each recombinant plasma-derived virus by the mean IC50 from duplicate determinations of the 12 treatment naïve plasma-derived subtype C viruses collected from the same geographical region against RPV. IC90 was estimated as double the IC50. The conventional biological cutoff (BCO) of 2.5-fold was used to define RPV resistance.20,21 This corresponds to 4 standard deviations above the mean treatment-naïve IC50 of 0.12 ng/mL as determined in this study. To address the effects of protein binding that occurs with human serum, a factor of 18.51 was applied to all IC50 values obtained from cell culture.
HIV-1 population genotyping
A laboratory-developed population genotyping assay was used to sequence HIV-1 from donor plasma at Lancet Laboratories, South Africa, as previously described. 22 Full-length HIV-1 RT from plasma-derived xxLAI viral stocks were also sequenced using six bidirectional primers spanning the entire length of RT. Drug resistance mutations and virus subtype were identified using HIVdb v7.0 (Stanford).18,19 Phylogenetic analysis using the Stanford Calibrated Population Resistance Tool v6.0 23 was done to ensure that plasma-derived cloned virus sequence clustered with virus in the original plasma sample. Overall, the sequences were 99% identical with a median hamming distance of 5/691 (1%). An ambiguity index was calculated as the percentage of ambiguous base calls (R, Y, K, M, S, W, B, D, H, V or N) in each sequence over the number of bases in the sequence. 24 The median ambiguity index for plasma-derived virus (0.0072) was not significantly different from the median ambiguity index for recombinant virus (0.0087) (p > 0.05).
Statistical analysis
Statistical analysis was conducted as previously described. 17 Fisher’s exact test (FET) was used to assess differences in the prevalence of amino acids at all RT codons between samples with >2.5-FC RPV resistance and samples with lower resistance, including those from 12 treatment-naïve individuals (FC ≤ 2.5). A sensitivity analysis was performed by varying the FC cutoff at which RPV resistance was defined. Cutoffs of FC >5, >10 and >76 (the upper limit of the phenotypic resistance assay) were used. The additive effect of individual NNRTI resistance mutations (as defined by Stanford HIVdb v7.0)18,19 was investigated using FET. For each pairwise combination of NNRTI resistance-associated positions, the frequency of samples containing mutations at two codons and at a single codon was compared across resistance groups. A mutation was included in the analysis if it was present at an estimated >25% frequency within a sample as determined by population sequencing and if there were at least three observations of the mutation in the entire study data set. Correction for multiple comparisons was performed by controlling for the false discovery rate (FDR) using the method of Benjamini and Hochberg. 25 An FDR-adjusted p value (or “q value”) of <0.15 was considered significant. Statistical analyses were performed in R (v3.1.2) with the glmnet library.
Results
Cross-resistance to rilpivirine of plasma-derived virus from individuals on failing first-line antiretroviral therapy
Plasma-derived recombinant viruses from 12 treatment-naive individuals yielded a mean RPV 50% inhibitory concentration (IC50) of 0.12 ± 0.04 ng/ml that was used as the wild-type, RPV-susceptible control value. Sixty-nine of 100 (69%) recombinant plasma-derived viruses containing full-length RT from individuals on failing first-line ART exceeded the BCO of 2.5 FC for RPV (Figure 1). Of those 69, 11 exceeded the upper limit of the assay (>IC50 of 9.2 ng/mL or >76 FC compared to the wild type). The median IC50 for the remaining 58 resistant viruses was 0.83 [Q1–Q3: 0.4–3.2] ng/mL or FC 7.0 [Q1–Q3: 3.6–26.4]. Thirty-one percent of specimens with NNRTI-associated drug resistance mutations were susceptible to RPV (FC < 2.5) (Table 1) (Supplementary Table S1). Protein-adjusted IC90 values are also presented in Figure 1 and Table 1.
Cross-Resistance to Rilpivirine of Plasma-Derived Viruses from Individuals on Failing First-Line NNRTI-Based ART.
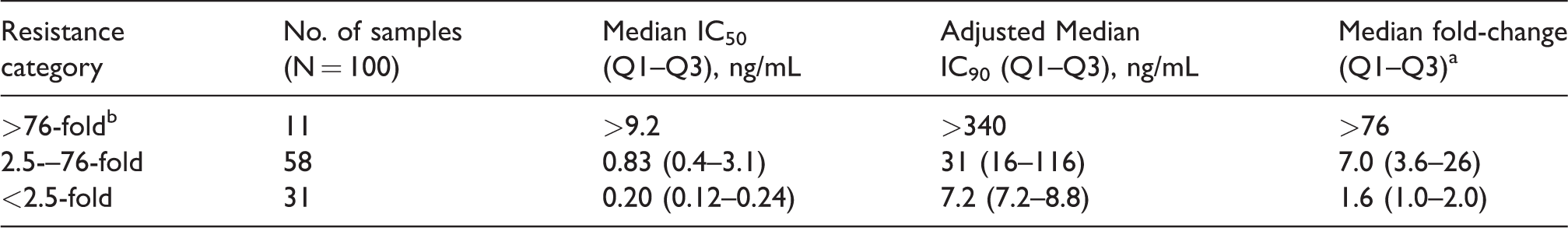
Calculated against a mean IC50 from 12 patient-derived HIV-1subtypeC viruses from South Africa.
Rilpivirine concentrations > 9.2 ng/mL could not be tested due to toxicity.
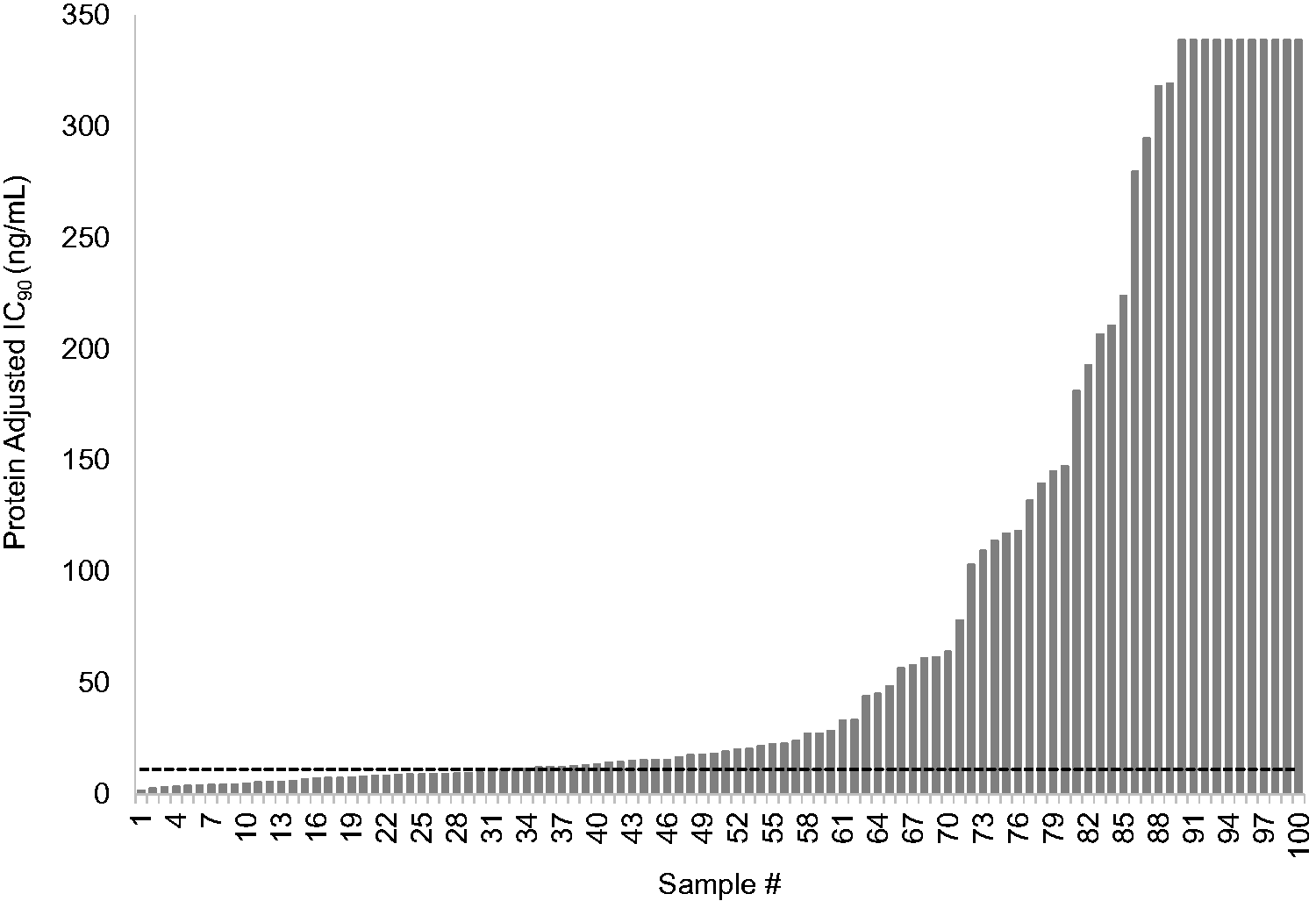
Rilpivirine Cross-Resistance of HIV-1 from 100 Individuals Failing First-Line Antiretroviral Therapy. The adjusted IC90 was calculated by multiplying the IC50 by 2 to approximate IC90 values and applying a rilpivirine (RPV) human serum binding factor (18.5) as determined in rilpivirine susceptibility experiments comparing human serum and fetal bovine serum (data not shown). RPV FC values were determined by dividing the IC90 generated for each plasma-derived virus by a composite IC90 from 12 treatment-naive plasma-derived viruses collected form the same geographical region. The dashed line indicates the RPV phenotypic biological cutoff of 2.5 FC. The IC90 values of samples 90 through 100 exceeded the highest concentration of RPV that could be tested in TZMbl cells without cytotoxicity and are reported as >340 ng/mL.
Prevalence of NNRTI resistance mutations associated with RPV cross-resistance
Sixteen NNRTI drug resistance mutations were analyzed individually to determine if specific mutations were associated with decreased susceptibility to RPV. After adjustment for multiple comparison, K103N was the only mutation associated with significant cross-resistance to RPV from individuals on failing NNRTI therapy at the q < 0.15 cutoff (p < 0.001) (Table 2). However, four out of four samples with K103N as the only NNRTI mutation were susceptible to RPV (FC range 0.9 to 2.0), indicating that additional resistance mutations were necessary for the reduction in RPV susceptibility conferred by K103N (Supplementary Table S1).
Association of NNRTI resistance mutations with rilpivirine cross-resistance.
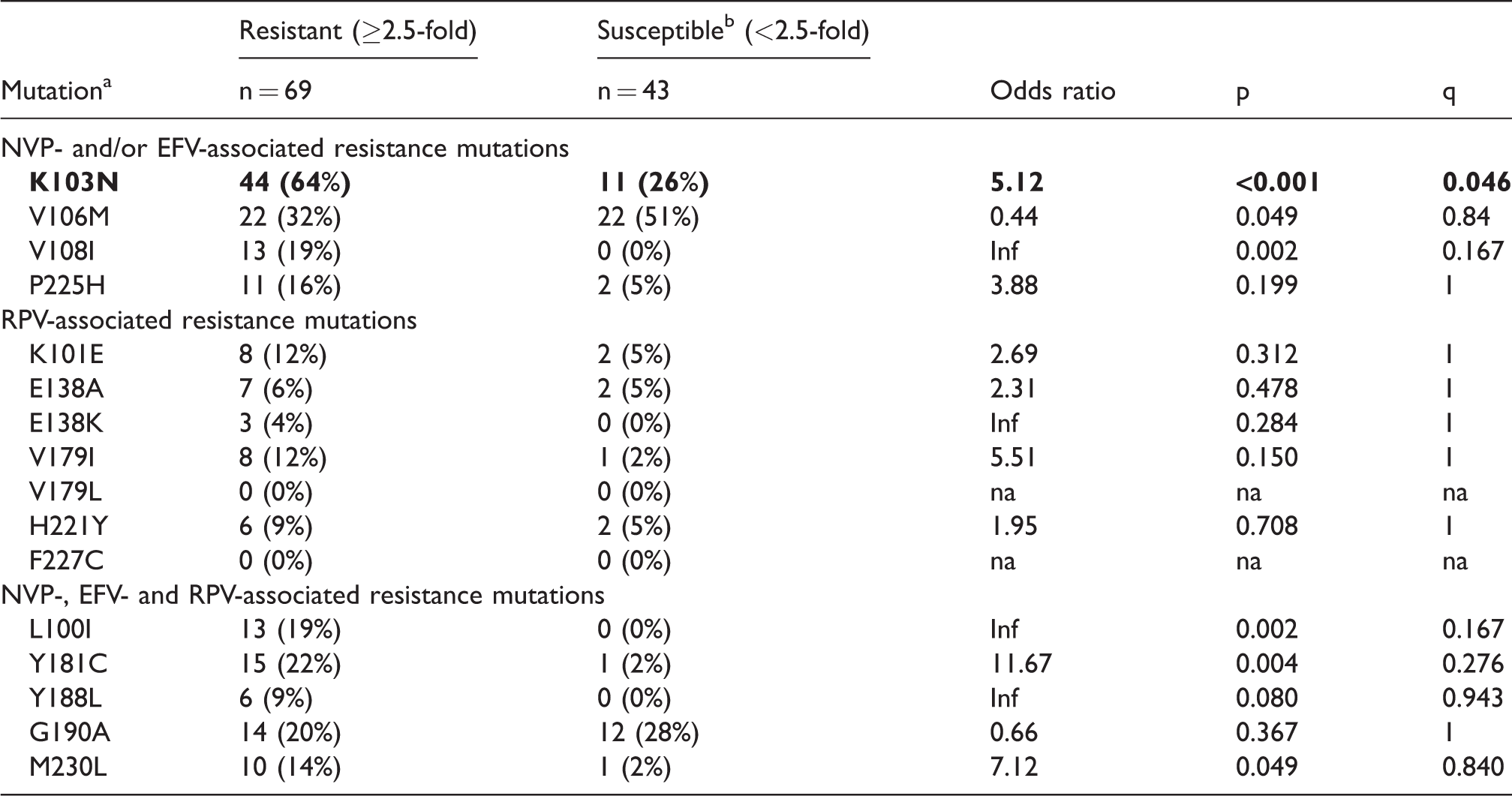
Mutations listed are considered major NVP-, EFV- and/or RPV-associated as reported by Stanford HIV Drug Resistance Database and IAS 2017.19,26
Sample size includes N = 12 “control” samples derived from ARV treatment naïve individuals from South Africa.Statistical significance is indicated with bold-faced text.
L100I, V108I and Y181C also showed a trend toward increased cross-resistance, but when adjusted for multiple comparisons, these were no longer significant. No other RPV-associated mutations in this sample set including K101E, E138A/K, V179I/L, Y188L, G190A, M230L, H221Y or F227C were associated with RPV cross-resistance (Table 2). No single mutation was associated with resistance in the >76 FC category.
Combined effects of NNRTI resistance mutations on RPV cross-resistance
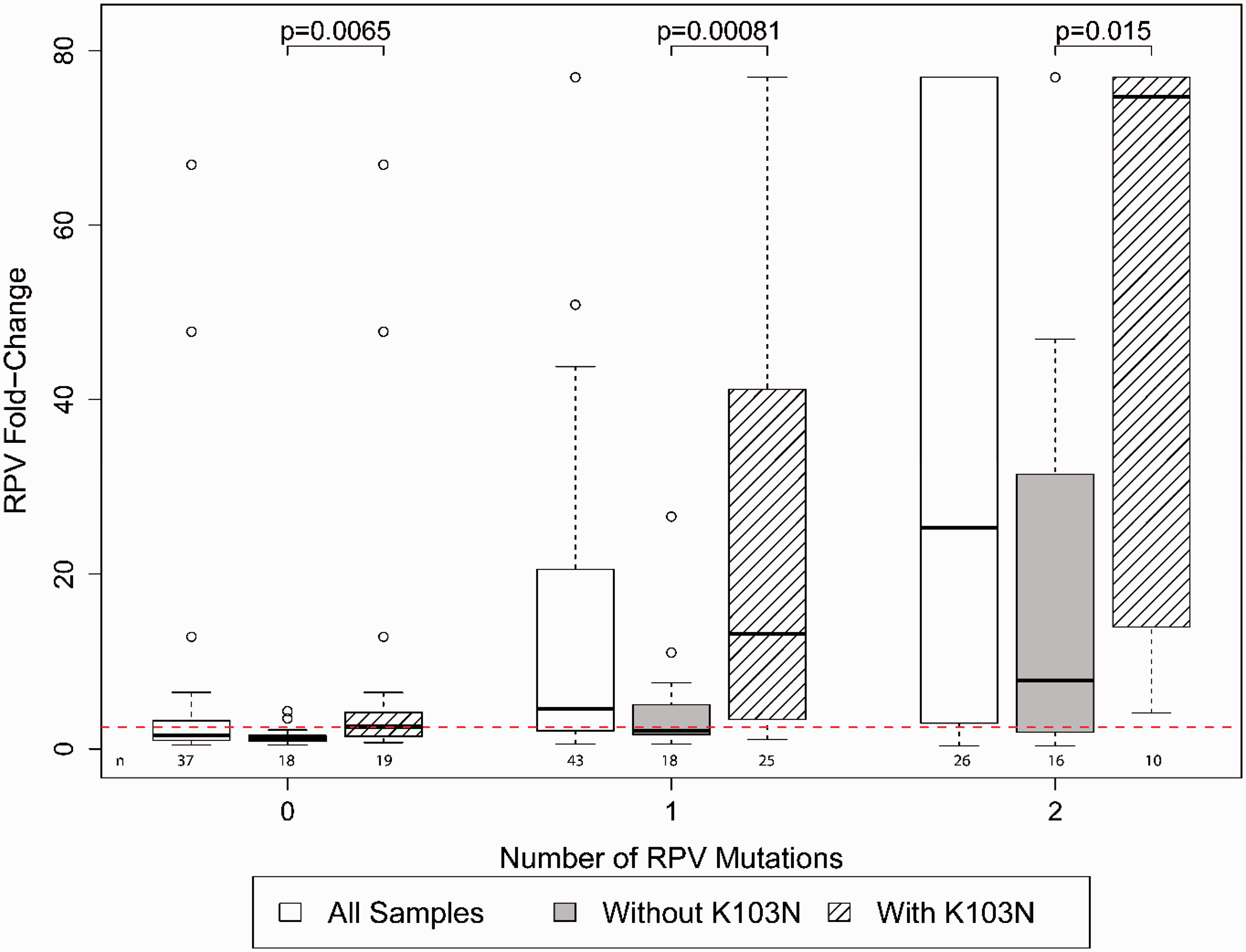
The combination of K103N with other NNRTI resistance mutations was explored for its role in conferring cross-resistance to RPV. Fifty-five of 100 samples had HIV-1 genotypes that included K103N. As expected, the 19 of 55 (34%) samples with K103N and no RPV-associated mutations had relatively low FC values (median 2.6, IQR 1.4–4.2). By contrast, K103N in combination with one or two other RPV-associated mutations (including mutations at codons 100, 101, 138, 179, 181, 188, 221, 227 and/or 230) 26 had a significantly higher median FC than samples with RPV-associated mutations without K103N (p ≤ 0.01). The number of RPV mutations per sample was associated with increased RPV resistance (p < 0.05) and the addition of K103N significantly enhanced the FC (p < 0.001) (Figure 2). In the absence of RPV mutations, a similar trend of higher levels of FC is observed with the addition of K103N with two or more NNRTI mutations, but the numbers were too small to achieve statistical significance.
Discussion
Injectable monthly PrEP formulations could address the challenge of maintaining consistent drug levels over long periods of time to provide continuous protection against HIV-1 infection. This protection, however, could be compromised by exposure to drug-resistant HIV-1 from a partner failing ART, particularly when drug levels in the uninfected partner fall to suboptimal levels. In a recent survey in South Africa of newly diagnosed HIV-1 infections, transmitted NNRTI resistance occurred in 25 of 277 patients (9%) with ≥1 drug resistance mutations. 27 Transmitted drug resistance is on the rise in South Africa and largely from NNRTI resistance.28,29 To evaluate the risk of breakthrough infection with TMC278LA by transmitted drug-resistant HIV-1, we evaluated the cross-resistance of plasma-derived viruses from individuals on failing NNRTI-based regimens to RPV, the active NNRTI in the long-acting formulation TMC278LA.
Overall, our study found that 69% of samples collected from individuals on a failing NNRTI-based ART displayed phenotypic RPV resistance above the BCO (Figure 1), causing concern for the use of TMC278LA as PrEP. The
The most frequently transmitted NNRTI mutation is K103N, 30 but relatively little is known clinically about cross-resistance of K103N to RPV. In a small study of three patients who had HIV-1 with K103N from prior NNRTI-containing therapy with no other NNRTI mutations, all three responded well to a switch from boosted protease inhibitor-based second-line therapies to RPV/tenofovir/emtricitabine. 31 In the ECHO and THRIVE trials, pre-existing K103N alone did not reduce RPV susceptibility in participants who added RPV to an existing NRTI-based ART regimen. 32 In our study, K103N was found in 55 of 100 samples collected from an ART-experienced study population. Additionally, of 69 samples with ≥ 2.5-fold decrease in susceptibility to RPV, 44 (64%) had K103N. The decrease in susceptibility was due to the additive effect of K103N with other RPV-associated NNRTI resistance mutations (Figure 2). Other studies have reported an association of RPV resistance with K103N combined with other NNRTI resistance mutations, especially L100I, in clinical samples.33–35 The clinical implications of the interactions between K103N and NNRTI mutations in conferring RPV resistance is unknown, and should be monitored as the use of RPV expands for both treatment and prevention.
In summary, this study showed that cross-resistance to RPV is common for NNRTI-resistant viruses from individuals experiencing virologic failure while on a first-line regimen and could reduce the efficacy of TMC278LA for HIV-1 prevention. The highest risk of breakthrough infection is likely to occur during the long RPV tail following discontinuation of TMC278LA dosing. The long tail may also be when selection of RPV-resistant HIV-1 is most likely to occur in newly infected individuals. Assessing the preventive efficacy of TMC278LA and monitoring resistance in TMC278LA users will be critical to better understand its role for PrEP.
Supplemental Material
Supplementary table - Supplemental material for Frequent cross-resistance to rilpivirine among subtype C HIV-1 from first-line antiretroviral therapy failures in South Africa
Supplementary Material
Footnotes
Acknowledgements
This work was presented in part as a poster (P22.04) at HIV Research for Prevention (HIVR4P), Cape Town, South Africa, 28–31 October 2014.
Declaration of conflicting interests
KP, CJB, MS-H, KH, KG, RVV, CLW and UMP declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. PRH has received grants from, served as an ad hoc advisor to, or spoke at various events sponsored by Pfizer, Glaxo-SmithKline, Abbott, Merck, Tobira Therapeutics, Virco and Quest Diagnostics and served as a consultant for ViiV Health Care, Tobira Therapeutics, Selah Genomics Inc and Quest Diagnostics. Dr Harrigan is supported by CIHR/GSK Research Chair in Clinical Virology. JWM reports other from Cocrystal Pharma, Inc., grants from Gilead Sciences, personal fees from Gilead Sciences, outside the submitted work. In addition, Dr Mellors has a patent (Patent No.: 8,815,829) issued.
Funding
Funding for this study is from the Bill and Melinda Gates Foundation, Grant# OPP1019228. PRH is supported by a Glen Hillson Professorship in Clinical Virology and the CIHR/GSK Research Chair in HIV/AIDS at the University of British Columbia. CJB is supported by a Postgraduate Fellowship from the Canadian Institutes of Health Research (MFE-146750).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
