Abstract
Nucleoside analogs represent the largest class of small molecule-based antivirals, which currently form the backbone of chemotherapy of chronic infections caused by HIV, hepatitis B or C viruses, and herpes viruses. High antiviral potency and favorable pharmacokinetics parameters make some nucleoside analogs suitable also for the treatment of acute infections caused by other medically important RNA and DNA viruses. This review summarizes available information on antiviral research of nucleoside analogs against arthropod-borne members of the genus
Introduction
The genus
Flaviviral infections are accompanied by a wide spectrum of distinct clinical manifestations, ranging from relatively mild fevers and arthralgia to severe viscerotropic symptoms (YFV and DENV), hemorrhagic fevers (KFDV and OHFV), encephalitis/myelitis (JEV, WNV, and TBEV), and neuropathic or teratogenic manifestations (ZIKV). More than 200 million clinical cases of flaviviral infections, including numerous deaths, are reported annually worldwide. 12 Currently no specific antiviral therapies are available to treat patients with flaviviral infections, thus the search for safe and effective small-molecule inhibitors that would be active against these viruses represents a high research priority. 13
Nucleoside analog inhibitors have figured prominently in the search for effective antiviral agents. 14 Nucleoside analogs are synthetic, chemically modified nucleosides that mimic their physiological counterparts (endogenous nucleosides) and block cellular division or viral replication by impairment DNA/RNA synthesis or by inhibition of cellular or viral enzymes involved in nucleoside/tide metabolism (Figure 1). 15 The first antiviral analogs were developed in the late 1960s and currently there are over 25 approved therapeutic nucleosides used for the therapy of viral infections of high medical importance, such as HIV/AIDS (tenofovir),16,17 hepatitis B (lamivudine/entecavir),18,19 hepatitis C (sofosbuvir), 20 or herpes infections (acyclovir). 21 So far, numerous nucleoside analogs have been described to inhibit arthropod-transmitted flaviviruses. Since these viruses are closely related to the hepatitis C virus (HCV), for which many potent inhibitors are being currently developed, anti-HCV nucleoside analogs represent promising tools to be repurposed against other viruses within the Flaviviridae family. 12
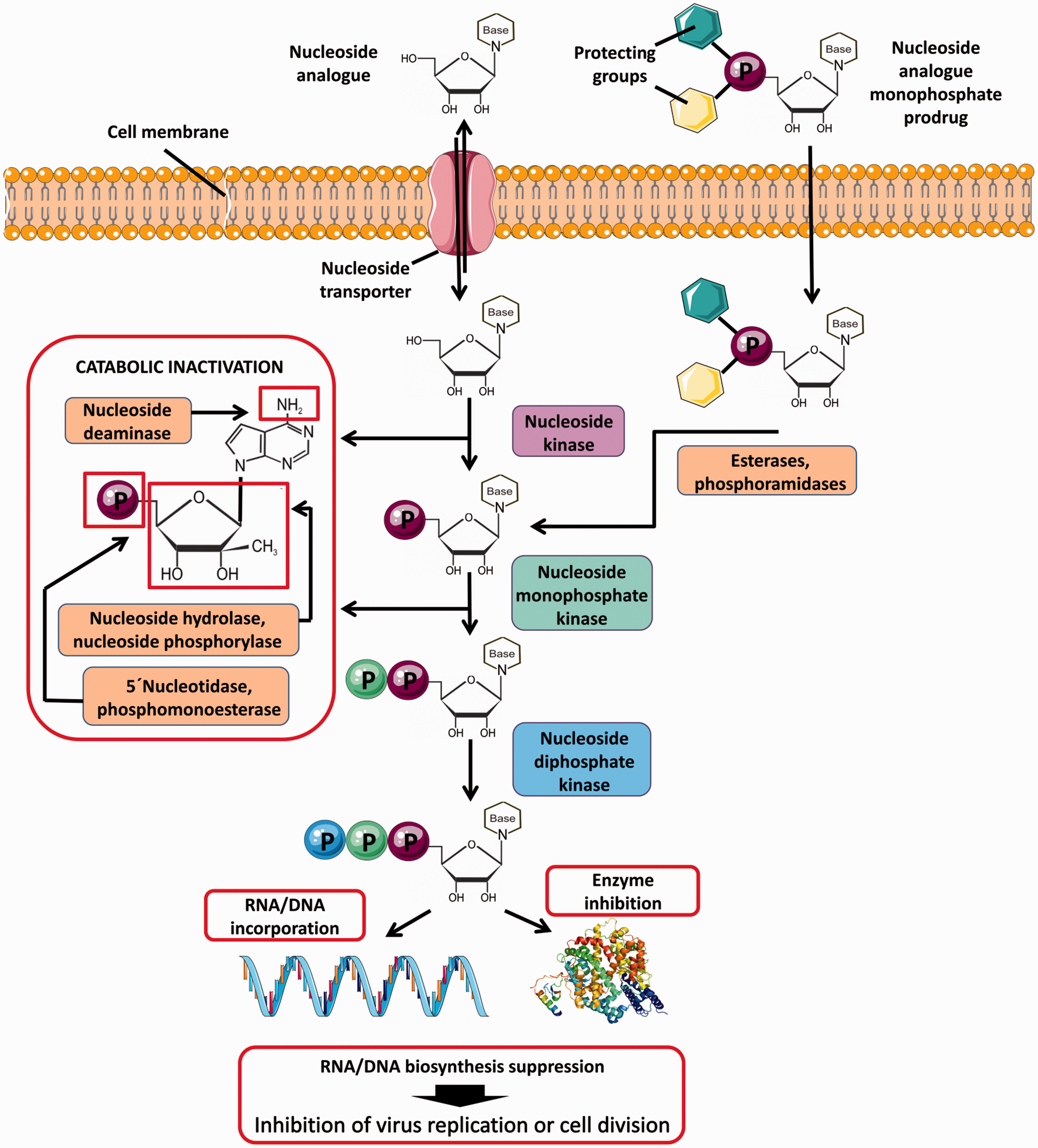
Intracellular uptake and metabolism of nucleoside analogs and nucleoside analog prodrugs. Nucleoside analogs enter cells through specific plasma membrane nucleoside transporters. Inside the cell, the compounds are phosphorylated by cellular nucleoside kinases resulting in formation of nucleoside mono-, di-, and triphosphates. The first kinase phosphorylation is the rate-limiting step of the triphosphate conversion, which can be overcome by the monophosphate prodrug approach based on the introduction of a phosphorylated group into the 5′ nucleoside position. The phosphorylated group includes protecting moieties to increase hydrophobicity and facilitate the cellular uptake of the prodrug. Monophosphate prodrugs enter cells independently of membrane transporters and the protecting groups are removed by intracellular esterases or phosphoramidases after cell penetration. The triphosphates of nucleoside species represent the active forms of nucleoside analogs that act by inhibiting cellular or viral enzymes, such as DNA/RNA polymerases. During DNA/RNA replication, nucleoside analogs are incorporated into nascent DNA or RNA chains resulting in termination of nucleic acid synthesis or in accumulation of mutations in viral genomes to suppress viral replication due to error catastrophe. At normal physiological conditions, intracellular nucleoside concentrations are maintained at low levels due to nucleoside/nucleotide catabolic pathways, such as deamination (oxidation) of heterocyclic base, hydrolysis or phosphorolysis of heterocyclic base, and hydrolysis of phosphomonoester bonds. These catabolic reactions also concern most nucleoside analogs containing the natural
The aim of this review is to provide an overview of known antiviral agents targeting selected arthropod-borne flaviviruses and to discuss their characteristic properties, modes of action, and advantages or limitations of their therapeutic use. Moreover, the important challenges and complications in antiflavivirus nucleoside analog development are highlighted and possible strategies to overcome their shortcomings are suggested.
Major classes of antiflavivirus nucleosides
Nucleoside analogs active against arthropod-borne flaviviruses are usually classified according to their targets in the viral life cycle. Such antiviral molecules act as inhibitors of flaviviral RdRp,22–26 MTase,27,28 and helicase/NTPase.29,30 Other nucleoside scaffolds suppress host cell enzymes involved particularly in nucleoside biosynthetic pathways31–34 or in some cases, exhibit multiple modes of action.32,35 In vitro antiviral effects and cytotoxicity profiles of the most important antiflavivirus nucleoside analogs are summarized in Table 1. An overview of in vivo antiflaviviral activities of selected nucleosides as evaluated in different animal models is presented in Table 2.
In vitro antiviral activities and cytotoxicity profiles of selected nucleoside inhibitors of flaviviral replication.

AHFV: Alkhurma hemorrhagic fever virus; CFI: cellular flavivirus immunodetection; CPE: cytopathic effect reduction assay; DC: dendritic cells; DENV: dengue virus; JEV: Japanese encephalitis virus; KFDV: Kyasanur Forest disease virus; LGTV: Langat virus; LIV: louping ill virus; ND: not determined; OHFV: Omsk hemorrhagic fever virus; PA: plaque reduction assay; PBMC: peripheral blood mononuclear cells; PHM: primary human macrophages; POWV: Powassan virus; TBEV: tick-borne encephalitis virus; USUV: Usutu virus; VTR: viral titer reduction assay; VYR: viral yield reduction assay; WESSV: Wesselsbron virus; WNV: West Nile virus; YFV: yellow fever virus; ZIKV: Zika virus.
aEC50 and CC50 values are expressed as µg/ml.
bEC90 values, expressed as µg/ml.
cCC50 (24 h): the cell culture was treated for 24 h with the appropriate compound to obtain the cytotoxicity data.
dIC50 values obtained from enzyme-based inhibition assays.
eCC50 values for confluent compound-treated cells.
fEC50 (sim): the cell culture was TBEV infected and simultaneously treated with the appropriate compound.
Examples of in vivo antiflaviviral activities of selected nucleoside analogs.
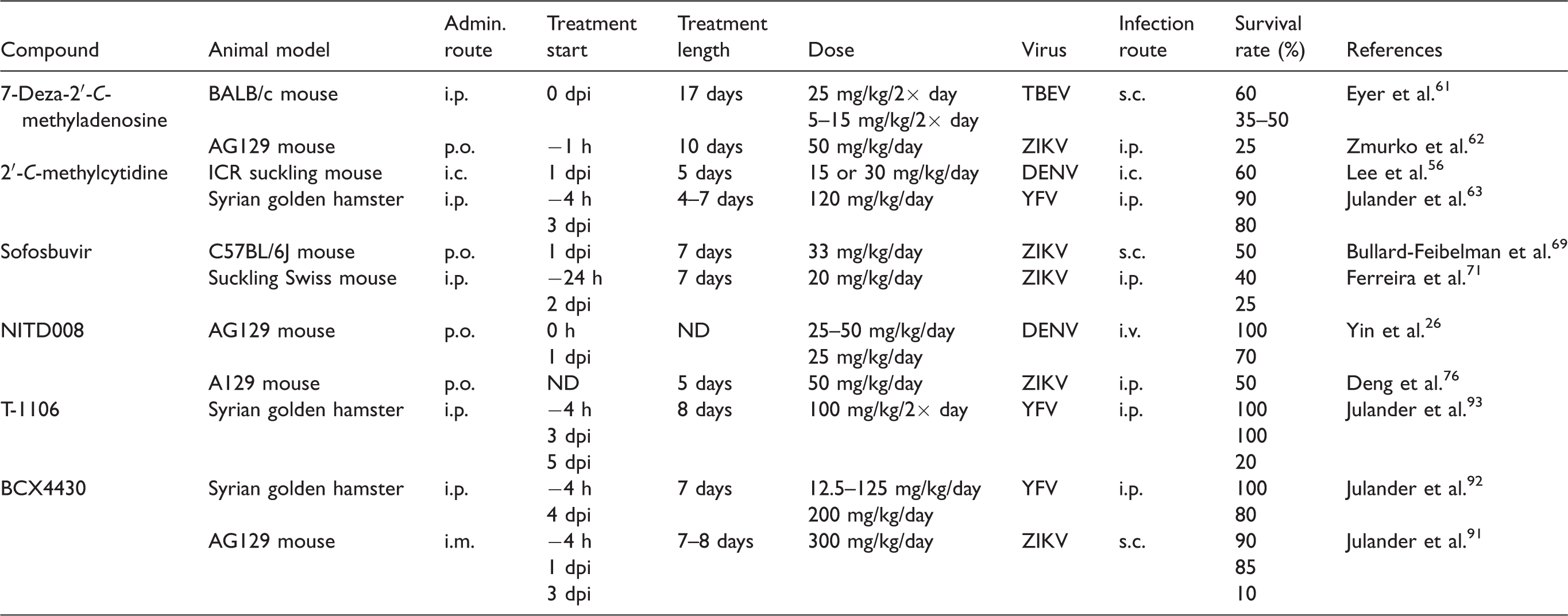
DENV: dengue virus; i.c.: intracerebral administration; i.m.: intramuscular administration; i.p.: intraperitoneal administration; i.v.: intravenous administration; ND: not determined; p.o.:
Inhibitors of flaviviral NS5 RdRp
The flavivirus NS5 protein is approximately 900 amino acids in length and consists of the NH2-terminal MTase domain required for the 5′-RNA capping process and the COOH-terminal RdRp domain responsible for the replication of the viral RNA genome.10,36 Flaviviral RdRp is a right hand-shaped structure with fingers, palm, and thumb domains; the palm domain is the catalytic domain carrying the polymerase active site that coordinates two Mg2+ ions essential for catalyzing the polymerization reaction. 37 Nucleoside inhibitors of flaviviral RdRp are the most attractive targets for antiviral drug design; as human replication/transcription enzymes lack RdRp activity, such compounds are expected to show fewer deleterious side effects and favorable safety profiles.12,15,38,39
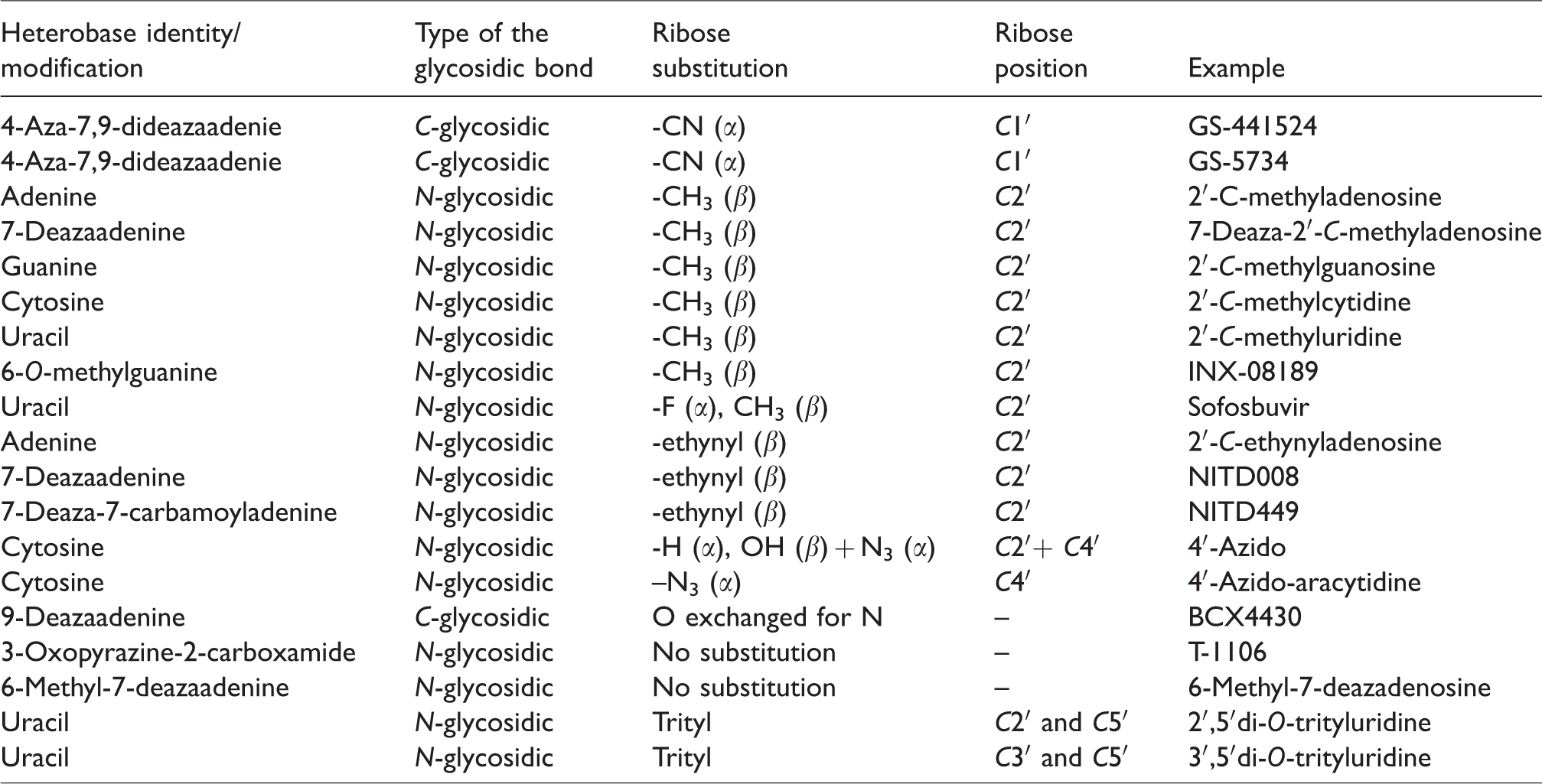
The mode of action for nucleoside RdRp inhibitors is based on the premature termination of viral nucleic acid synthesis. 40 Following the intracellular phosphorylation, the 5′-triphosphate metabolites are competitively incorporated into the flaviviral RNA nascent chains (Figure 1). This prevents further extension of the incorporated analog by addition of the next nucleoside triphosphate resulting in formation of incomplete (nonfunctional) viral RNA chains. 41 Nucleoside inhibitors of flaviviral RdRp act as “nonobligate chain terminators,” as their 3′-hydroxyl group is conformationally constrained or sterically/electronically hindered, thus decreasing the potency to form a phosphodiester linkage with the incoming nucleoside triphosphate. 40 The nonobligate terminators differ from “obligate chain terminators,” in which the 3′-hydroxy group is completely missing, as exemplified by numerous nucleoside reverse transcriptase inhibitors for the treatment of HIV infections. 42 Chemical modifications of the heterobase moiety, different types of glycosidic bonds, and substitutions at different positions of the sugar ring, which are typical modifications for nucleoside inhibitors of flaviviral RdRps, are shown in Table 3.
Heterobase substitutions and ribose modifications of selected flaviviral RdRp nucleoside inhibitors.
1′-Cyano substituted nucleosides
GS-441524, a 1′-cyano substituted
A phosphoramidate prodrug of GS-441524, referred to as GS-5734, recently entered Phase II clinical trials for treatment of Ebola infections, displayed 10- to 40-fold higher antiviral effect against members of the TBEV serocomplex when compared with its parental analog GS-441524. The increased antiviral potency could be related to an improved conversion of the prodrug to the biologically active form; however, the reported SI values (2.4–8.3) indicate a low therapeutic potential for this nucleoside to treat flaviviral infections. 45 Further substitutions of GS-441524 molecule at the C1′ position with methyl, vinyl, or methyl–ethynyl moieties yielded compounds with considerably reduced potency and a narrower spectrum of antiviral activity. 43
2′-C -methyl substituted nucleosides
2′-
The 2′-C-methyl substituent introduced at the nucleoside β-face appears to be an important structural element for highly selective micromolar inhibition of tick-borne flaviviruses, particularly for TBEV, AHFV, KDFV, OHFV, and POWV, when assayed in porcine stable kidney cells (PS), human neuroblastoma cells UKF-NB4, or adenocarcinomic human alveolar basal epithelial cells A549.24,54,55 From mosquito-transmitted flaviviruses, 2′-
Strong antiflaviviral activity for several 2′-
2′-Fluoro-2′-C -methyl substituted nucleosides
Similar to the 2′-
Sofosbuvir was demonstrated to inhibit the ZIKV RdRp in a recombinant polymerase assay 68 and to suppress ZIKV replication in different cell-based systems using U87 glioblastoma cells, baby hamster kidney fibroblasts (BHK-21), SH-sy5y neuroblasts, hepatocarcinoma Huh-7 cells, Jar human placental choriocarcinoma cells, neural stem cells, and brain organoids with nanomolar or low micromolar inhibitory activity.25,69 Interestingly, no inhibition of ZIKV replication with sofosbuvir even at the 50 µM level was observed in Vero cells, 23 indicating that the sofosbuvir-mediated anti-ZIKV effect is strongly cell-type dependent. 70 Sofosbuvir protected inbred C57BL/6J mice, which were previously immunosuppressed with a single dose of anti-Ifnar1 blocking monoclonal antibody, against ZIKV-induced mortality. 69 Moreover, this compound reduced viral titer in blood plasma, spleen, kidney, and brain in suckling Swiss albino mice and prevented virus-induced neuromotor impairment in ZIKV-infected animals. 71
Surprisingly however, sofosbuvir was inactive against TBEV, when screened on both PS and UKF-NB4 cells.
54
Another 2′-α-fluoro-2′-β-
2′-C -ethynyl substituted nucleosides
2′-
Introduction of the C7 carbamoyl moiety to NITD008 molecule provided another 2′-ethynyl modified derivative, referred to as NITD449. Despite its low micromolar anti-DENV efficacy, this nucleoside disappointingly exhibited only low levels in plasma when dosed orally. 72 To increase the oral bioavailability, the isobutyryl ester prodrug of NITD449, designed as NITD203, was synthesized. NITD203 successfully exhibited both nanomolar anti-DENV activity as well as improved pharmacokinetic parameters. 72 Although NITD008 and NITD203 showed anti-DENV potency in rodent models, even when the treatment was delayed up to 48 h after infection, both nucleosides failed in preclinical toxicity studies in rats and dogs due to their insufficient safety profiles.26,72 NITD008 failed to achieve no-observed-adverse-effect levels (NOAEL) when rats (10 mg/kg/day) and dogs (1 mg/kg/day) were dosed daily for two weeks. 26 Similarly, NOAEL was not achieved for NITD203 in the two-week toxicity test when rats were dosed at 30 and 75 mg/kg/day. 72
Further substitutions of the C7 position of NITD008 with fluoro or cyano moieties provided compounds with nano- or low micromolar anti-DENV activities and acceptable cytotoxicity profiles. In contrast, 2′-
2′-O -substituted nucleosides
2′-
3′-C - and 3′-O -substituted nucleosides
Methylation of the C3′ or O3′ position to generate the corresponding 3′-
4′-Azido substituted nucleosides
Using high-throughput screening of large nucleoside libraries in combination with a rational drug design approach by investigators at Roche, several cytidine analogs with an azido group at the C4′ position were identified as potent inhibitors of HCV replication in subgenomic replicon assays.79–81 It was later shown that these compounds were also highly active against henipaviruses and other paramyxoviruses. 82 Two 4′-azido modified nucleoside analogs, 4′-azidocytidine (R-1479) and 4′-azido-aracytidine (RO-9187), showed nano- or micromolar in vitro antiviral activity against TBEV. 54 Moreover, both compounds were found to be active also against WNV (L. Eyer, manuscript in preparation).
The anti-TBEV activity of RO-9187 (the arabino-counterpart of 4′-azidocytidine) was unexpected, as this compound lacks the 2′-α-hydroxy moiety, a determinant which was generally considered to be crucial for specific hydrogen-bonding interactions with RdRps during the RNA replication process.54,80 Some additional interactions of the polymerase active site with both the 2′-β-hydroxy substituent and the 4′-azido substituent are thought to compensate for the loss of the 2′-α-hydroxy interaction, resulting in the strong and selective anti-TBEV activity of RO-9187. Interestingly, the anti-TBEV efficacy of both 4′-azido modified nucleosides was cell-type dependent; the compounds were active only in PS cells, but not in UKF-NB4 cells. 54
An ester prodrug of 4′-azidocytidine, denoted as balapiravir, was completely inactive against TBEV in vitro, probably because of its poor intracellular uptake or insufficient kinase phosphorylation in the tested host cell lines. 54 In contrast, balapiravir was reported to show strong in vitro antiviral activity against DENV of various serotypes; it was the first direct antiviral agent tested clinically for DENV infection. Unfortunately, this compound failed to achieve antiviral efficacy in DENV patients, which was reflected in a negligible reduction of DENV viremia and persistence of clinical symptoms, even though the plasma concentration of the compound was higher than the 50% effective concentration. 83 One of the possible explanations is that DENV infection stimulates PBMCs to produce cytokines, which are responsible for the decreased efficiency of the conversion of balapiravir to its triphosphate form. 84
Interestingly, nucleoside analogs combining the 4′-azido moiety with the 2′-
Imino-C -nucleoside analog BCX4430
BCX4430, developed by BioCryst Pharmaceuticals Inc., is an adenosine analog with the furanose oxygen on the ribose ring replaced by nitrogen and the heterobase nitrogen 9 replaced by carbon.
87
This interesting nucleoside is classified as imino-
BCX4430 is active against numerous mosquito-transmitted flaviviruses, such as WNV (EC50 of 2.33 µM) 90 and representatives of both the African and Asian lineages of ZIKV (3.8–11.7 µg/ml). 91 For YFV, JEV, and DENV-2, micromolar EC50 values were reported. 89 In vivo efficacy of BCX4430 was also demonstrated in a lethal hamster model of YFV infection and in a mouse model of ZIKV infection.91,92 Low micromolar antiviral activity of BCX4430 was also described for some of the medically important tick-borne flaviviruses, such as TBEV, LIV, and KFDV. 90
Heterocyclic base-modified nucleosides
Heterocyclic base-modified nucleosides with demonstrated antiflaviviral activities include T-1106,93–95 6-methyl-7-deazaadenosine,
96
and numerous
6-Methyl-7-deazaadenosine is a hydrophobic mimic of adenosine showing nanomolar antiviral activity against DENV-2 in a Vero cell-based screening system, as well as in luciferase-driven DENV-2 replicon assay, with no cytotoxic effects noted after 7 h of treatment. 96 Related compounds such as 6-methyl-1-deazaadenosine and 6-methyl-4-deazaadenosine were completely inactive when tested against DENV-2. Mechanistic studies of the 5′-triphosphate of 6-methyl-7-deazaadenosine revealed that this nucleotide is an efficient substrate for viral RdRp (screened against polio RdRp) and is incorporated into nascent viral RNA strains mimicking both ATP and GTP. 96
Tritylated nucleosides
In a large-scale cell-based screening campaign of alkylated, silylated, or acylated pyrimidine nucleosides, 2′,5′di-
The mechanisms of action of tritylated nucleosides are not completely understood. Based on the observed inhibition of DENV replication in subgenomic replicon assays, it is assumed that these compounds may be acting as inhibitors of intracellular viral replication events rather than suppressing either early or late processes of viral infection, such as entry or assembly. 103 Although the presence of large, hydrophobic trityl moieties does not make these structures ideal candidates for further drug development, their chemical structures may provide valuable information for advanced mechanistic studies and for further development of related nucleoside scaffolds with improved biological parameters. 104
Nucleoside inhibitors of flaviviral MTase
The NH2 domain of flaviviral NS5 protein is associated with the virus’s MTase activity, which is involved in methylation of the 5′-cap structure of genomic RNA.8,105 The flaviviral cap structure is formed by the conserved dinucleotide sequence AG (m7GpppAm) and is crucial for mRNA stability and efficient translation.
106
Two topologically distinct methylation reactions are mediated by the flaviviral NS5 MTase: the
SAH and the nucleoside antibiotic sinefungin are natural nonselective inhibitors of many eukaryotic and viral MTases, including those of DENV108,109 or ZIKV.
61
Chemical derivatization of SAH at the
Recently, a novel series of flexible nucleoside analogs known as “fleximers” have exhibited activity against several hard to treat viruses, including filoviruses such as Ebola and Sudan,
112
coronaviruses including Severe Acute Respiratory Virus and Middle East Respiratory Virus,
113
as well as most recently, flaviviruses including ZIKV and DENV. The fleximers feature a “split” purine nucleobase that has been shown to impart significant activity to the nucleoside scaffold as well as to allow it to overcome resistance related to point mutations.114–116 The most recent series combined the fleximer approach with the acyclic nucleoside acyclovir, an FDA approved drug for herpes virus. While these analogs inhibited the aforementioned viruses, acyclovir shows no activity against any of those viruses, thereby underscoring the importance of the fleximer approach. Since those initial findings, these compounds have also demonstrated potent activity against DENV and ZIKV (K. Seley-Radtke, manuscript in preparation). Preliminary results indicate that these compounds target the DENV and ZIKV NS5 in at least its cap-MTase activity, with negligible effects on the cognate human
Nucleoside inhibitors of flaviviral NTPase/helicase
Flaviviral NTPase and helicase activities are associated with the COOH-proximal domain of the NS3 protein. 117 Flaviviral helicases are capable of unwinding duplex RNA structures during viral replication by disrupting the hydrogen bonds keeping the two strands together.118,119 The helicase activity is strictly associated with NTP hydrolysis (NTPase activity); the released chemical energy is used for the translocation of the enzyme along the double-helix structures, capturing the exposed single strand regions or for a direct disruption of the hydrogen bonds between the two RNA strains. 120
Specific nucleoside inhibitors of flaviviral NTPase/helicase can interact with dsRNA or DNA resulting in the weakened stability of double-helix structures or by steric hindrance of translocation of the enzyme along the polynucleotide chain. Such a mechanism could modulate the efficacy of the unwinding reaction or NTPase activity of the enzyme and therefore, affect the viral replication process.117–121 Weak inhibitory effects on flaviviral NTPase/helicases were described for ribavirin triphosphate,124,125 5′-
Ribavirin and other nucleoside synthesis inhibitors
Ribavirin, a nucleoside analog featuring a [1,2,4]triazole ring for a nucleobase, is a licensed drug against various RNA viruses. The predominant inhibitory mechanism for ribavirin against flaviviral replication is the suppression of de novo biosynthesis of guanine nucleotides through direct inhibition of inosine monophosphate dehydrogenase, an enzyme converting inosine monophosphate to xanthosine monophosphate, a precursor in GTP biosynthesis. 32 Speculation over other modes of action for ribavirin includes specific inhibition of the viral RdRp, 128 accumulation of mutations in viral genomes resulting in error catastrophe,129,130 interference with mRNA capping guanylation, 131 and immuno-modulation promoting the Th1 antiviral response.32,132,133
Ribavirin was shown to exert a moderate inhibitory effect for multiple mosquito-borne flaviviruses in various cell cultures,31,134,135 often being used as a positive control in many in vitro25,59,92 and in vivo antiviral studies.91–94 Ribavirin administered to YVF-infected hamsters challenged intraperitoneally, resulted in significant improvement in survival rates, even if the therapy was started two days post-YFV infection.93,94 In contrast however, in primates, only a weak prophylactic effect on viremia in rhesus monkeys challenged with DENV was observed.
136
Several ribavirin derivatives were recently synthesized and showed interesting bioactivity profiles: ETAR (1-β-
Two nucleosides whose antiviral activity is based on the depletion of the intracellular nucleoside pool are 6-azauridine and 5-aza-7-deazaguanosine. 6-Azauridine and its derivatives are inhibitors of orotidine monophosphate decarboxylase blocking cellular de novo pyrimidine biosynthesis.31,33 6-Azauridine proved to be active against numerous arthropod-borne flaviviruses, 55 however exhibited slight cytotoxicity with an inhibitory effect on the growth of host cells. 24 A triacetate prodrug of 6-azauridine showed low micromolar activity against AHFV and WNV in vitro 55 and very low toxicity in both animal and human studies. 138 Another derivative, 2-thio-6-azauridine, exerted a moderate inhibitory effect on WNV. 33 Similar results were achieved using 5-aza-7-deazaguanosine (ZX-2401) 139 ; this compound exhibited synergistic in vitro anti-YFV activity in combination with interferon. The mechanism of action for 5-aza-7-deazaguanosine is currently unknown; however, it is conceivable that it likely resembles that of ribavirin. 34
Rigid amphipathic nucleosides
Nucleoside derivatives containing bulky aromatic substituents (perylene or pyrene moieties) attached to the heterocyclic base were originally synthesized as fluorescent nucleoside probes140–142; later these structures were identified as inhibitors of herpes simplex virus, type 1 and 2 (HSV-1 and HSV-2), vesicular stomatitis virus, and Sindbis virus replication. 143 Further studies demonstrated their broad-spectrum antiviral activity against other enveloped viruses, such as influenza virus, murine cytomegalovirus, and HCV. 144 The mechanism of action of these rigid amphipathic nucleosides is based on their incorporation into the viral or cellular membranes, preventing fusion.143,144 Alternatively, these nucleosides may also function by photosensitization of lipid membranes, resulting in irreversible damage of enveloped virion particles. 145
5-(Perylen-3-yl)ethynyl-arabinouridine and 5-(perylen-3-yl)ethynyl-2′-deoxyuridine were shown to act as strong inhibitors of TBEV in PEK cell culture. 101 The perylene moiety as well as the rigid ethynyl linker appeared as crucial structural elements for nanomolar anti-TBEV activity and low cytotoxicity (>50 µM). Interestingly, uracil nucleosides bearing the pyrene moiety, such as 5-[(pyren-3-yl)methoxypropyn-1-yl]-2′-deoxyuridine and 5-(pyren-1-yl)ethynyl-2′-deoxyuridine, showed almost a 10-fold lower anti-TBEV potency compared to their perylene-substituted counterparts. Such compounds, if not used as therapeutic agents, could still contribute to a better understanding of different modes of action of various nucleoside scaffolds. 101
Challenges and complications of antiflavivirus nucleoside analog development
Introduction of various chemical substituents into different positions of the nucleoside scaffold can dramatically affect the physicochemical properties of the compound. Such modifications can also significantly influence the compound’s biological/pharmacokinetic parameters, such as cellular uptake,15,146 the ability of the compound to be activated (phosphorylated) by cellular kinases, 147 degradation by nucleoside catabolic enzymes, 148 or cellular toxicity.149,150 Use of nucleoside analogs can also result in the undesirable emergence of drug-resistant virus mutants.151–153 This section highlights the most important challenges and complications toward the development of nucleoside inhibitors of arthropod-transmitted flaviviruses and suggests possible strategies to surmount these difficulties.
For most nucleoside analogs, the first kinase phosphorylation is the rate-limiting step for the conversion to the nucleoside triphosphates. This limitation has a major influence on nucleoside analog antiviral activity147,154 but can be bypassed by the use of a monophosphate prodrug approach based on the introduction of the phosphorylated group into the 5′ nucleoside position. The phosphorylated group includes masking moieties on the charged phosphate leading to a neutral and eventually hydrophobic entity able to deliver the nucleoside 5′-monophosphate into the cells (Figure 1).
15
The monophosphate prodrug approach has been shown to convert some inactive nucleosides into strong inhibitors, or, has improved the kinetics parameters of intracellular nucleoside triphosphate formation.
85
This strategy, together with enantioselective purification, led to the development of the phosphoramidate prodrug sofosbuvir, which exhibited considerably increased phosphorylation efficacy compared to the parent nucleoside, 2′-
Rapid degradation of nucleoside analogs by nucleoside catabolic pathways (Figure 1) is another undesirable phenomenon, which can adversely affect the antiviral potency of some nucleoside analogs.
148
To address this problem, appropriate structural changes can be introduced into the nucleoside scaffolds to protect the nucleosides from metabolic deactivation.
157
Such a strategy was successful for 2′-
Individual host cellular types can display differences in expression levels of nucleoside kinases and other enzymes/proteins involved in nucleoside metabolism and transport. This can then result in cell-type dependent antiviral activity as manifested by different EC50 values for the same inhibitor when assayed on different cell lines.158,159 Strong anti-TBEV activity for 2′-
Another possible complication in nucleoside drug development is an undesirable toxicity profile for the nucleoside inhibitor, which can be related to poor selectivity between viral and human enzymes.39,149,150 A typical example of a nonselective nucleoside analog is 7-deazaadenosine (tubercidin), which exhibits high in vitro cytotoxicity. This has been attributed to the incorporation of tubercidin monophosphate into cellular DNA/RNA by human polymerases.
50
In contrast, two derivatives of tubercidin, 7-deaza-2′-
In that regard, the availability of suitable animal models is crucial for the successful evaluation of a nucleoside’s therapeutic in vivo antiviral efficacy. Some flaviviruses, particularly DEVN and ZIKV, do not readily replicate or cause pathology in immunocompetent mice and, therefore, the use of these rodents as animal infection models is substantially limited.164–166 To overcome this problem, suckling or young mice, 167 AG129 mice lacking INF-α/β and INF-γ receptors, 168 IFNAR−/− mice lacking only the IFN-α/β receptor, 169 or immunosuppressed mice 170 can be used as appropriate models to evaluate nucleosides in vivo. However, as these animals are defective in an immune response, this model may also underestimate the real efficacy of the test compounds. Rodent-adapted flavivirus strains, such as the hamster-adapted YFV strain Jimenez, 171 mice-adapted DENV strain D2S10, 26 or ZIKV African strain Dakar 41519, 172 represent other possible options for in vivo antiviral studies; the biological properties of such viruses can be, however, considerably different compared with those of the parent human-adapted strains. 169
Another issue is related to the length of therapeutic treatment. Most tick- and mosquito-borne flaviviruses cause acute infections, in which short-time treatment duration is expected, ranging between several days to weeks. 173 This is in contrast to chronic diseases, such as HCV, HBV, and HIV infections, which require long-lasting, and sometimes lifelong, treatment regimens.72,173 The differences between the acute and chronic diseases should be considered during preclinical development of antiflaviviral inhibitors. Thus, some compounds that show insufficient safety profiles when tested for treatment of chronic infections can be still suitable and safe for short-term therapy of acute flaviviral diseases. 72
Antiviral therapies based on chemical inhibitors of viral replication can be accompanied with a rapid emergence of drug-resistant mutants which substantially complicates the course of infection treatment, as seen in HIV, HBV, or HCV infections.152,153 In flaviviruses, a rapid evolution of resistance to 2′-
Conclusions
More than 200 million clinical cases caused by arthropod-borne flaviviruses, including numerous deaths, are reported worldwide annually. So many cases of infection indicate the importance for the pursuit of new small molecule-based therapeutics to combat emerging viral pathogens. Among these, inhibitors of flaviviruses represent a critical unmet medical need. In that regard, nucleoside inhibitors of flaviviral RdRps are the most attractive targets for antiviral drug design. Nucleosides with the methyl- or ethynyl- modification at the C2′ position and their 2′-fluoro derivatives are the best understood antiflavivirus nucleoside analogs, many of which were initially developed for treatment of HCV infections and later reemployed to suppress replication of other non-HCV flaviviruses. Other important antiflavivirus nucleosides are represented by inhibitors of nucleoside biosynthesis, whose mode of action is predominantly based on depletion of the intracellular nucleoside pool. Nucleoside inhibitors of flaviviral MTase and NTPase/helicase, as well as some newly discovered flavivirus inhibitors, such as tritylated nucleosides, rigid amphipathic nucleosides, or
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Ministry of Health of the Czech Republic (grant no. 16–34238A), Czech Science Foundation (grant no. 16–20054S) (to DR and RN), and by Project “FIT” (Pharmacology, Immunotherapy, nanotoxicology; CZ.02.1.01/0.0/0.0/15_003/0000495), which was funded by the European Regional Development Fund (to DR).
