Abstract
Objective
To synthesize 3,7-dideazaneplanocin and evaluate its antiviral potential.
Methods
The target 3,7-dideazaneplanocin has been prepared in five steps from a readily available cyclopentenol. A thorough in vitro antiviral analysis was conducted versus both DNA and RNA viruses.
Results
A rational synthesis of 3,7-dideazaneplanocin was conceived and successfully pursued in such a way that it can be adapted to various analogs of 3,7-dideazaneplanocin. Using standard antiviral assays, no activity for 3,7-dideazaneplanocn was found.
Conclusion
Two structural features are necessary for adenine-based carbocyclic nucleosides (like neplanocin) for potential antiviral properties: (i) inhibition of S-adenosylhomocysteine hydrolase and/or (ii) C-5′ activation via the mono-nucleotide. These two requisite adenine structural features to fit these criteria are not present in in the target 3,7-dideazaneplanocin: (i) an N-7 is necessary for inhibition of the hydrolase and the N-3 is claimed to be essential for phosphorylation at C-5′. Thus, it is not surprising that 3,7-dideazaneplaoncin lacked antiviral properties.
Introduction
Since the synthesis of aristeromycin Aristeromycin, neplanocin A and related synthetic analogs.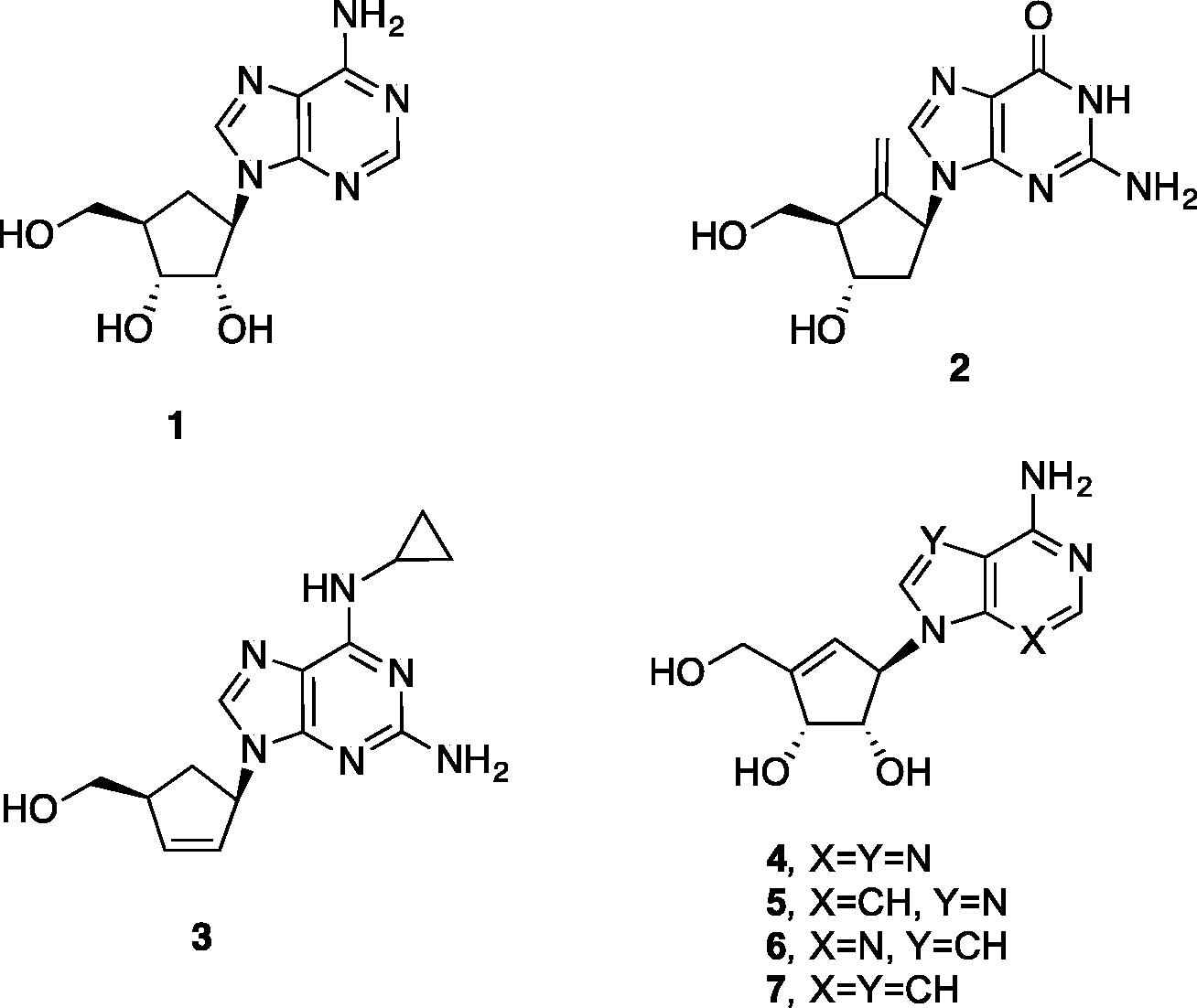
Neplanocin A
Results and discussion
Synthesis
The synthesis of target Synthesis of 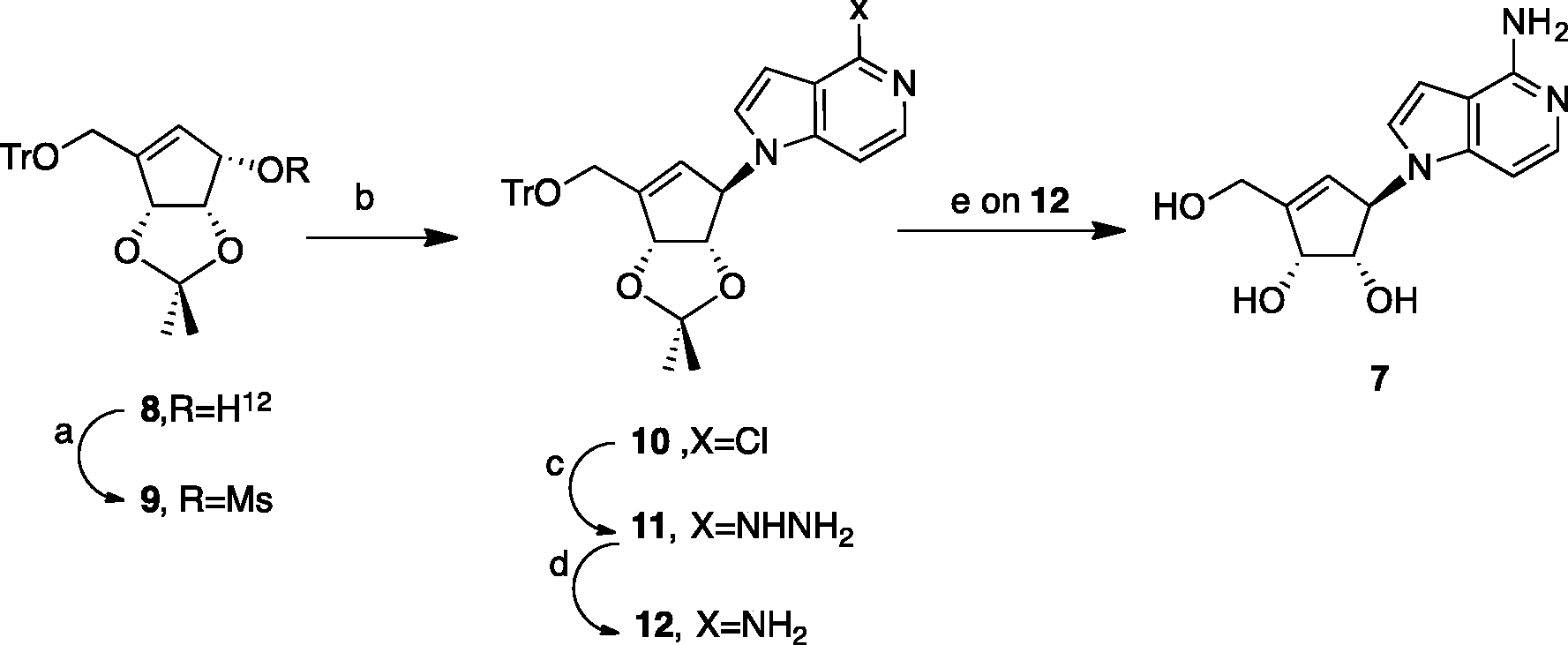
Antiviral results
Compound
Conclusion
Two structural features are necessary for adenine-based carbocyclic nucleosides to demonstrate potential antiviral properties: (i) inhibition of S-adenosylhomocysteine hydrolase
16
and/or (ii) C-5′ activation via the mono-nucleotide.
16
These two requisite adenine structural features that fit these criteria are not present in
Experimental section
Chemistry
The combustion analyses were performed at Atlantic Microlab, Norcross, GA. 1H and 13C NMR spectra were recorded on either a Bruker AV 600 spectrometer (600 MHz for proton and 150 MHz for carbon) or a Bruker AV 400 spectrometer (400 MHz for proton and 100 MHz for carbon), referenced to internal tetramethylsilane at 0.0 ppm. The reactions were monitored by thin-layer chromatography using 0.25 mm Whatman Diamond silica gel 60-F254 precoated plates with visualization by irradiation with a Mineralight UVGL-25 lamp. Column chromatography was performed on Whatman silica, 230–400 mesh, and 60 Å using elution with the indicated solvent system.
To a solution of
To a solution of
Antiviral assays
These assays are presented in Chen et al. 15
Footnotes
Acknowledgments
We are also indebted to the National Institute of Allergy and Infectious Diseases in vitro assay team for the viral data presented herein. The assistance of Dr Erik De Clercq of the Rega Institute is also recognized.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by funds from the NIH (AI 56540) for which we are grateful.
