Abstract
Background
Natural product-inspired synthesis is a key incorporation in modern diversity-oriented synthesis to yield biologically novel scaffold. Inspired by β-carboline fused system, we have designed molecules with multi ring fused scaffold by modifying the tricyclic pyrido[3,4-b]indole ring with imidazo[1,2-a]isoquinoline.
Methods
A highly convergent approach with new C–N and C–C bond formation to synthesize multiring fused complex scaffold imidazo[1,2-a]isoquinolinies as fluorophores. N-nucleophile-induced ring transformation of 2H-pyran-2-one followed by in situ cis-stilbene-type oxidative photocyclization yielded new C–C bond formation without additional oxidant. The cytotoxicity, effective concentrations, and the mode of action of the synthesized analogs were determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT),, plaque reduction, time of addition, and reverse transcriptase Polymerase Chain Reaction (PCR).
Results
Novel imidazo[1,2-a]isoquinoline analogs were prepared, and the results revealed that trans isomer of cyclopropyl analog (EC50 35 and 37.5 µg/ml) and trans isomer of citric acid salt of phenyl analog (EC50 38.2 and 39.8 µg/ml) possess significant anti-Herpes Simplex Virus (HSV) activity with selectivity index of >10. The kinetic study demonstrated that both the analogs inhibited HSV-1F and HSV-2G at 2–4 h postinfection. Finally, western blot and reverse transcriptase PCR assays revealed that both the analogs suppressed viral immediate early transcription.
Conclusion
Novel imidazo[1,2-a]isoquinoline analogs were synthesized from pyranone with appropriate amines. Two compounds showed better antiviral profile on HSV-infected Vero cells, compared to the standard drug acyclovir (ACV). Overall, we discovered a promising scaffold to develop a nonnucleoside lead targeting the viral immediate early transcription for the management of HSV infections.
Introduction
Diversity-oriented natural product based synthesis has drawn a special attention due to its potential to discover significant biologically active molecules. 1 More than 70% of new chemical moieties discovered in the recent past were natural products or natural product derived or mimetics. 2 The traditional total synthesis of natural products has been the key to medicinal chemistry since ages, although this type of synthesis is cumbersome and time consuming. Modern approach diversifies the synthesis and explores a wider chemical space.3,4
β-Carboline alkaloids, saturated or unsaturated, both of them have been reported to be active against either HSV-1 or HSV-2 or both (Figure in supporting information).5–8 Previously, we had described that βC alkaloids exhibit anti-HSV activity, although with high toxicity (safe up to 50 mg/kg body weight).6,7 Moreover, in continuation to our pyranone scaffold exploration toward antiviral molecule synthesis,9,10 chiral amines were inserted into pyranone moiety to yield a natural product like “N-containing multi ring heterocycles” with imidazo[1,2-a]isoquinoline core. This has several biological implications,11,12 including anti-HSV property. 13 Furthermore, the organic molecules with fluorescent properties have added advantages to its biological applications. For example, it can act as DNA staining dyes, work as intracellular probe or they can also be covalently linked to specific protein to study its mode of action. 14
Diverse synthetic approaches are utilized to synthesize a fused isoquinoline system. The traditional routes such as Bischler−Napieralski, Pictet−Gams, and Pomeranz−Fritsch reactions require harsh conditions and highly active substrates. The transition metal-catalyzed reactions provide an efficient route 15 and are widely used 16 for coupling of C(aryl)–C, C(aryl)–N, C(aryl)–O, and C(aryl)–S bonds but is generally expensive. Metal-catalyzed 17 or non-metal-catalyzed 18 cycloaddition reactions are yet another efficient way to synthesize N-containing multiple fused ring system. Herein we present a one-pot synthesis of fluorophoric [1,2-a]isoquinoline analogs using key intermediate pyranone and N-nucleophiles to demonstrate anti-HSV potential of novel scaffold.
Materials and methods
General chemistry
The reactions were carried out in oven-dried glassware. The chemicals and solvents were purchased from Spectrochem, Across, Rankem, or Sigma Aldrich. Melting points were recorded on Veego melting point apparatus. Analytical thin layer chromatography (TLC) was performed on precoated plates (silica gel 60 F-254). Purification by gravity column chromatography was carried out on silica gel (100–200 mesh). Elico UV/Vis spectrophotometer was used for recording the UV spectra; while 1H,
13
C NMRs were determined in a Varian/Bruker (300/400 MHz) spectrometer using CDCl3 or DMSO-d6 as solvent. Peaks are recorded with: s (singlet), bs (broad singlet), d (doublet), t (triplet), q (quartet), m (multiplet), and J (coupling constant, Hertz). Fluorescence emission spectrum was measured in Shimadzu RF-5301 spectrofluorometer. All the intermediates and final compounds were adequately characterized by spectroscopic analysis, and one of the compounds ORTEP diagram of 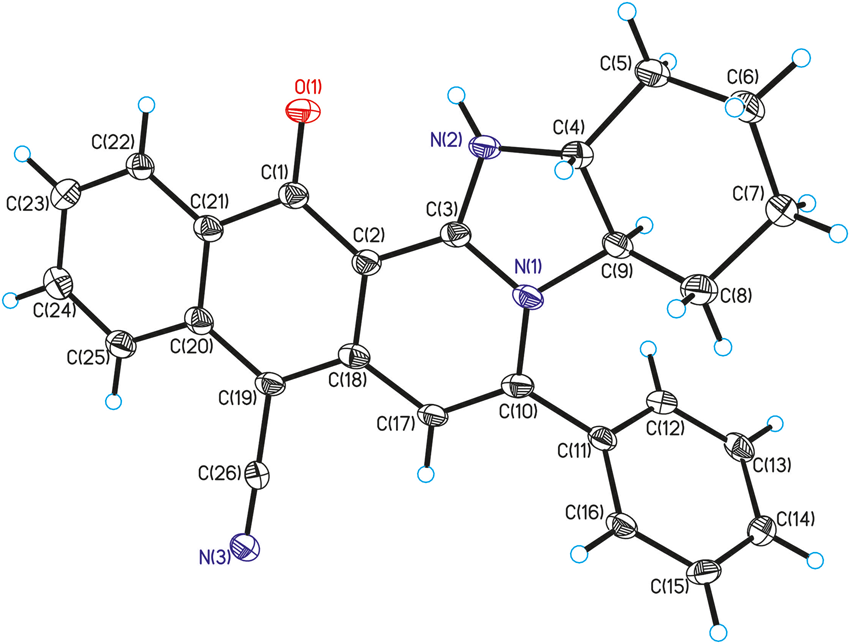
Procedure for the synthesis of [3a–f]
The suspension of NaH (3.3 mmol) and
Procedure for synthesis [3g–h]
Similar procedure was followed using
Procedure for synthesis [4a–b]
Characterization of all the new compounds are given in supporting information.
Measurement of fluorescence spectra
Fluorescence emission spectrum was measured on Shimadzu RF-5301 spectrofluorometer. The molecules were excited at λ max (380 nm) using methanol as the solvent. Fluorescence spectrum was measured at different pH using HCl and NaOH solutions in the range of 0.5–12. Spectrum was further obtained using aprotic solvent DMF instead of MeOH dimethylformamide.
Antiviral activity study
Cells and viruses
Vero cells (ATCC, USA) were cultured in Dulbecco’s Modified Eagle’s medium (DMEM; nvitrogen, USA) with 5% fetal bovine serum (FBS; Invitrogen, USA), 100 U/ml penicillin, and 100 U/ml streptomycin at 37°C in 5% CO2. The viral strains used were HSV-1F (ATCC 733) and HSV-2G (ATCC 734), purchased from the ATCC, USA.
In vitro cytotoxicity and anti-HSV activity study
The in vitro cytotoxicity and antiviral activity were determined by MTT and plaque reduction assays (PRAs).7,19 In brief, the Vero cells were exposed to various concentrations of the analogs and incubated at 37°C in 5% CO2, using ACV and DMSO (0.1%) as controls. After 72 h, MTT assay was carried out following manufacturer’s protocol (MTT; Sigma) and the OD was read at 570 nm. The 50% cytotoxic concentration (CC50) was calculated by linear regression of the dose–response curves. For antiviral activity, the PRA was used. Briefly, Vero cells infected with HSV-1F and HSV-2G (100 PFU) were exposed to different concentrations of test drugs for 1 h and then overlaid with 1% methylcellulose containing test drug. The plaques developed after 72 h were counted and virus titers were calculated by scoring the plaque-forming units. The effective concentration of test drugs that reduced plaques’ number by 50% (EC50) was interpolated from the dose–response curves. 7
Mode of action
Time of addition assay
Following three different approaches, for understanding the preinfection, cells were treated with test drugs either for 3 or 1 h and then infected with HSV-1F in DMEM containing 2% FBS at 37°C. For studying the coinfection, cells were subsequently infected and treated with test drugs. These were removed after 1 h and added with fresh media. Whereas for postinfection (pi) the cells were first infected with HSV and then treated with the test drugs at 0–24 h time intervals, with an incubation period of 1 h in each case. The mixture was removed and added with fresh media. The PRA was carried out as described,7,19 after incubation at 37°C in 5% CO2 for 48 h. DMSO (0.1 %) and ACV (5.0 microg/ml) were used as control for all these experiment.
Attachment and penetration assay
To investigate the role of test drugs on viral attachment the Vero cells were prechilled at 4°C for 1 h and challenged with HSV-1 (100 PFU/well) in the presence of
Quantitative reverse transcriptase PCR (RT-PCR)
The effects of analog addition on HSV-1 RNA expression within the cell was performed by RT-PCR analysis. Briefly, the HSV-1 (5 Multiplicity of infection (MOI)) infected Vero cells were treated with
Results and discussions
A convergent one-pot route was followed to synthesize multi ring-fused imidazo[1,2-a]isoquinolines Synthesis of key intermediates 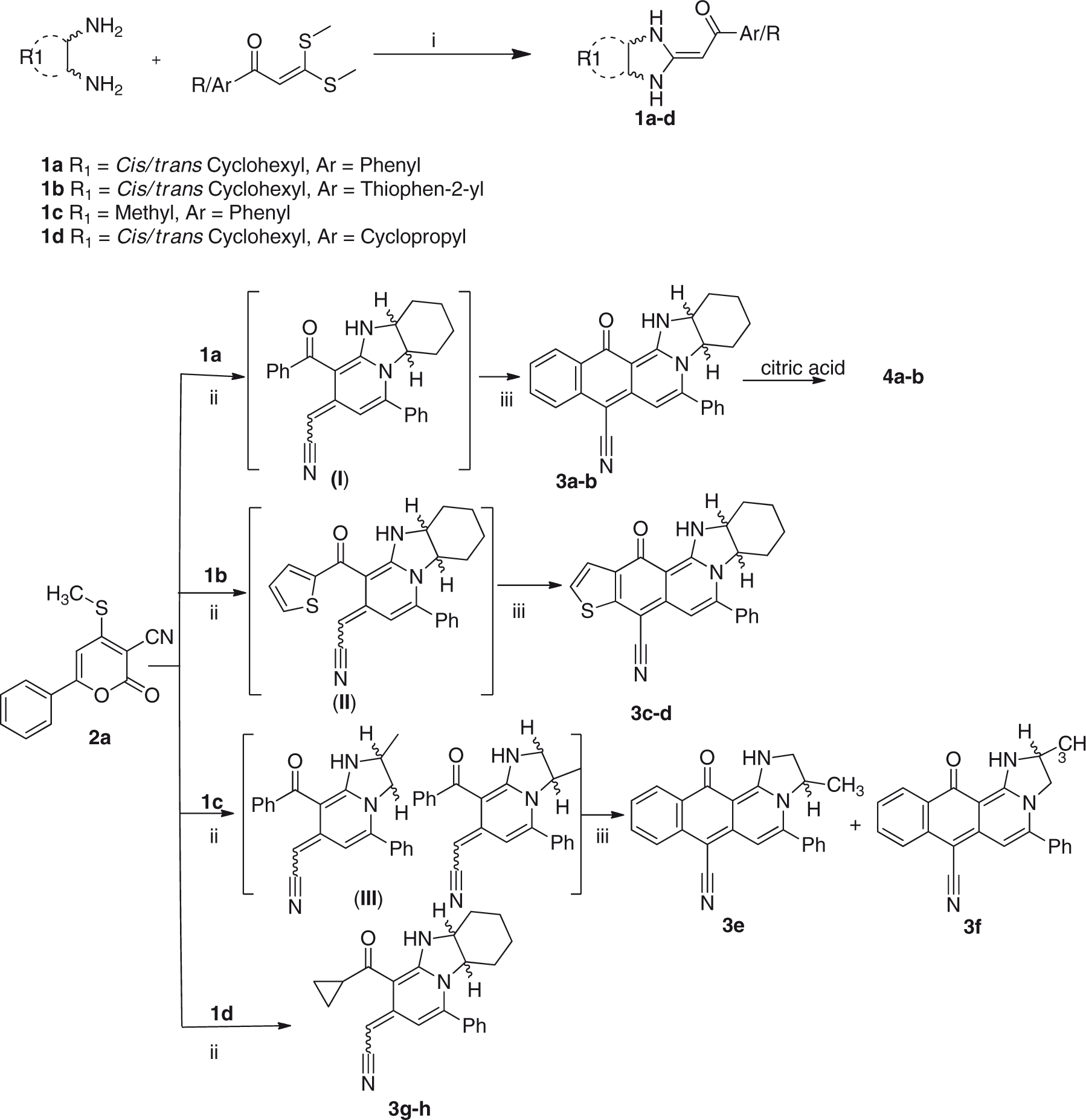
The reaction of
The products
Fluorescent properties
Significant fluorescent properties were shown by (a) Absorbance and emission spectra of 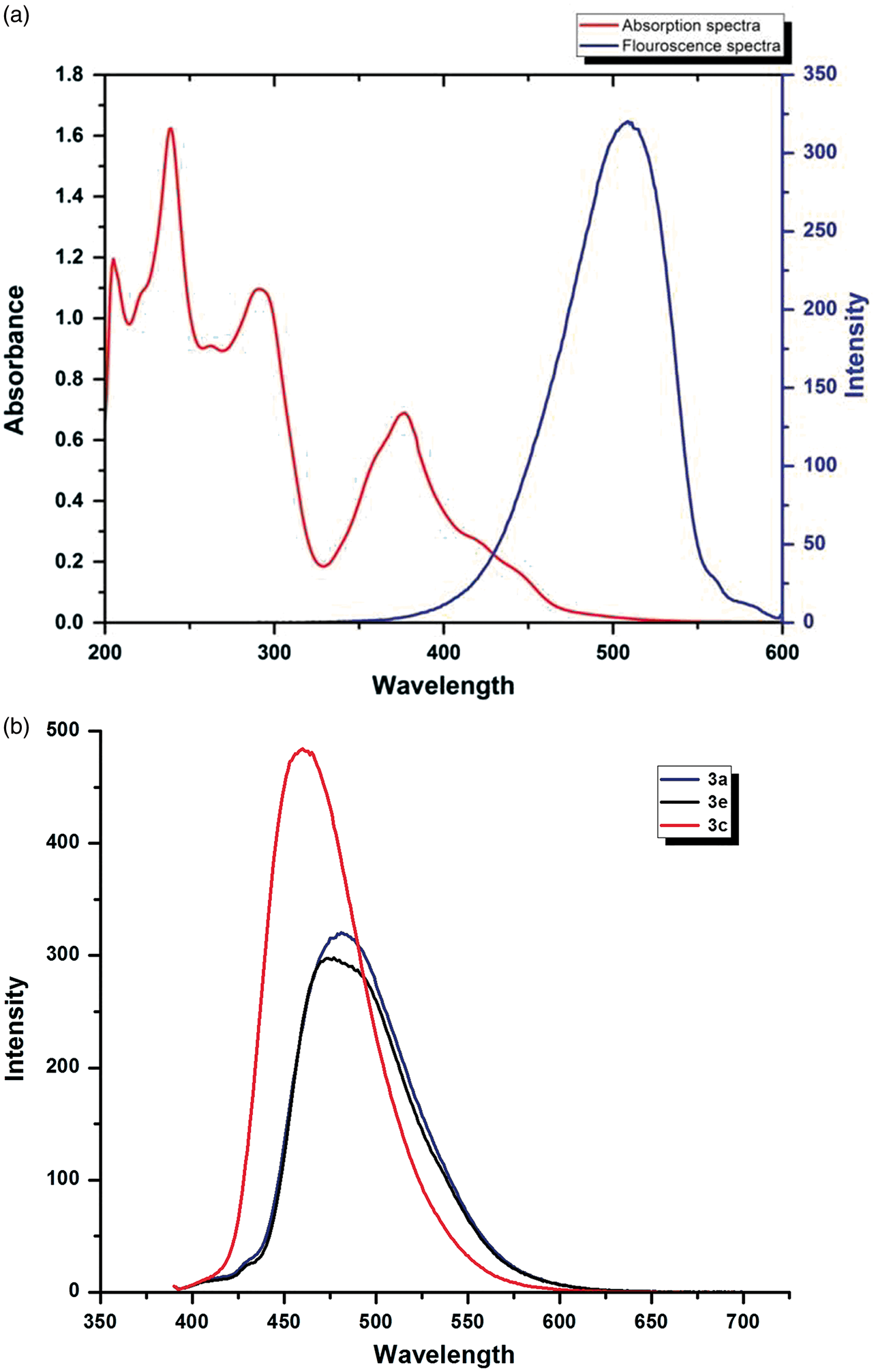
Cytotoxicity and antiviral activity study
Anti-HSV activity spectrum on HSV-infected Vero cell.
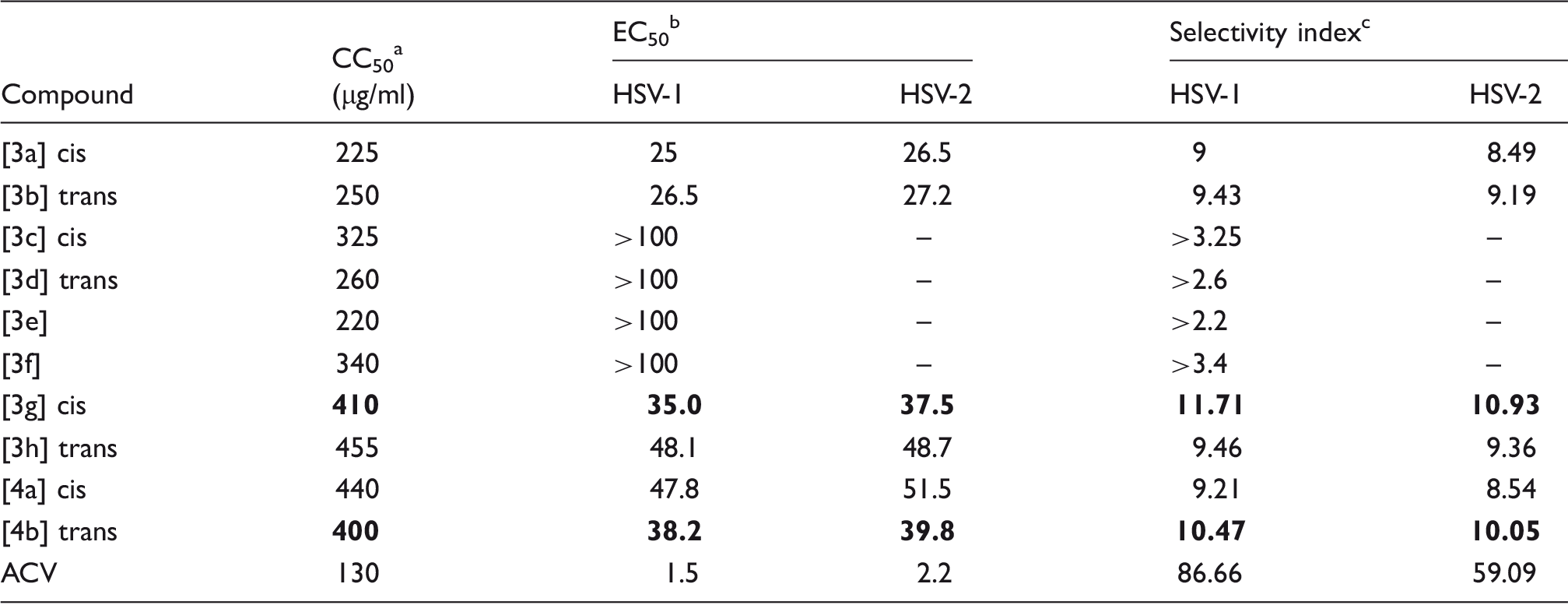
ACV: Acyclovir; CC: cytotoxic concentration; EC: Effective Concentration; HSV: Herpes Simplex Virus.
The 50% cytotoxic concentration for Vero cells in µg/ml.
Concentration of compound in µg/ml producing 50% inhibition of virus-induced cytopathic effect. All experiments were repeated for three times.
Selectivity index = CC50/EC50.
To investigate the possible stage of viral life cycle interfered by the test analogs, we performed the time-of-addition assay. The results demonstrated that Time of addition assay of 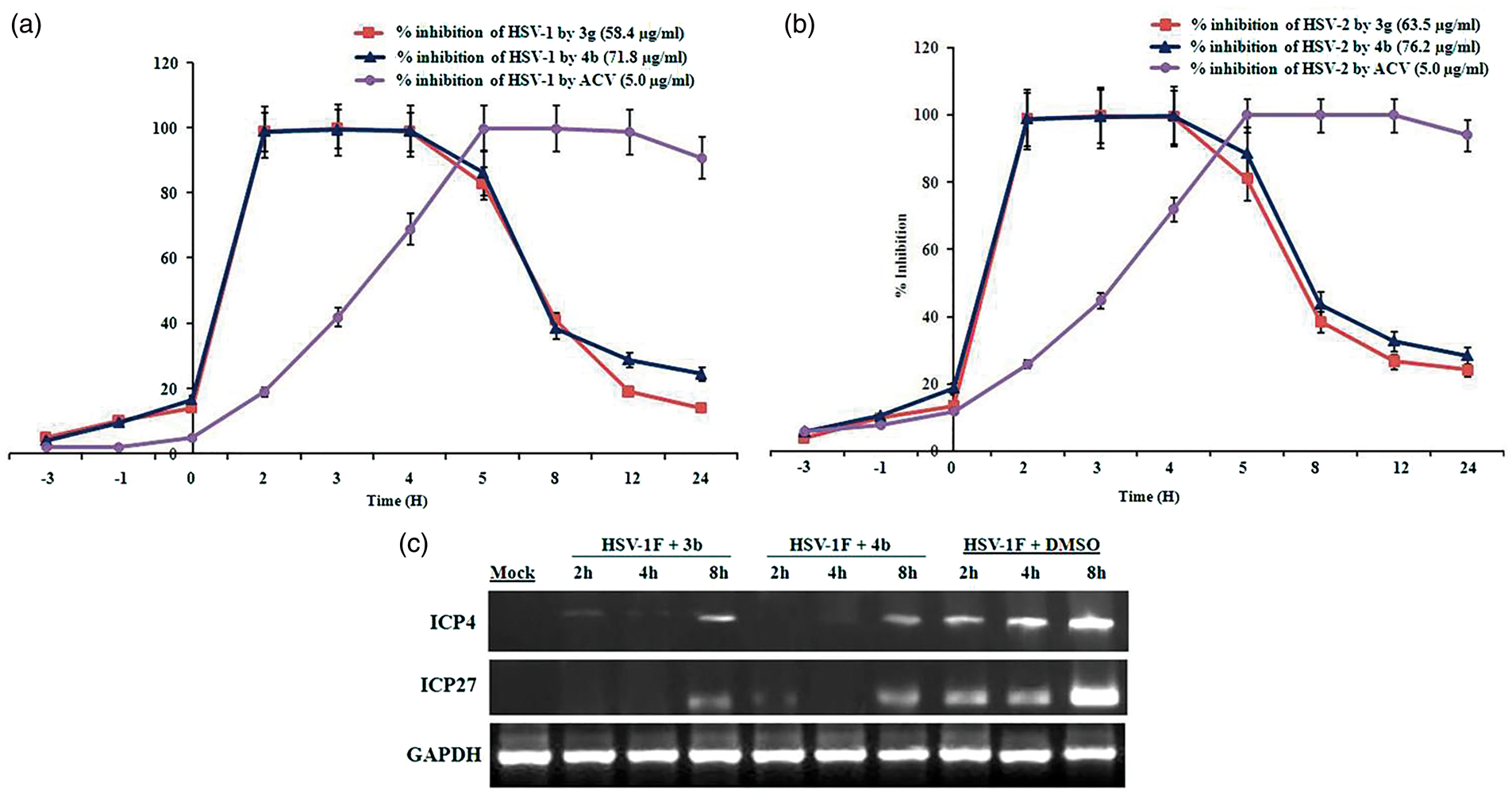
The expressions of two IE gene products ICP4 and ICP27 were determined to analyze the effect of
During lytic infection, the gene expression of HSV occurs in a regulated fashion commencing with the synthesis of IE gene products, followed by the synthesis of early and late gene products.28,29 The HSV synthesizes five IE proteins as infected cell polypeptides (ICP) 0, 4, 22, 27, and 47. Except for ICP47, all these proteins are reported to affect the expression of viral genes, and the genes encoding ICP22 and ICP47 can be deleted from the viral genome with no effect on the growth and viability of the virus in most cell types.30,31Mutant HSV-1 deleted for the ICP0 gene grows poorly in cell culture, particularly at low MOIs but remains viable. 32 On the other hand, viral mutants carrying deletions or temperature-sensitive lesions in ICP4 and ICP27 demonstrated that these proteins provide essential functions during productive infection.26,27 We previously reported that Harmaline, an isolated βC alkaloid, recruits lysine-specific demethylase-1 and it binds on IE complex of ICP0 promoter which leads to the inhibition of viral IE gene synthesis and suppressed the expression of ICP4 and ICP27. 6 It could hence be suggested that both the test analogs interfere with the viral IE-transcriptional events, which are a critical component of herpes virus reactivation. Our study demonstrated that the test analogs may help to prevent the multiplication of HSV and provide an interesting molecular target for the development of lead for better therapeutic and efficient management of HSV infection with minimal animal toxicity (safe up to 200 mg/kg, shown in supporting information).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from Department of Biotechnology (DBT), Delhi, India through grant no BT/PR13759/PBD/17/686/2010. HC thanks BIT, Mesra for Institute Research Fellowship. Author thanks to ILS, Hyderabad and CIF, BIT Mesra for analytical support. AS & CB acknowledge DST FIST support SR/FST/CSI-242/2012.
