Abstract
Background
The reverse transcriptase (RT) of human immunodeficiency virus type 1 (HIV-1) is an attractive target for the development of drugs used in the treatment of HIV-1 infection and acquired immune deficiency syndrome (AIDS). We have continued the search for novel anti-HIV-1 agents using the structure–activity relationships of the successful 1,3-disubstituted and 1,3,6-trisubstituted uracil-type HIV-1 RT inhibitors.
Methods
A series of new triazine analogs were synthesized using an established method. The anti-HIV-1 activities of these compounds were determined based on the inhibition of virus-induced cytopathogenicity in MT-4 cells. The cytotoxicity of the compounds was evaluated by assessing the viability of mock-infected cells.
Results
Some of the compounds showed good-to-moderate activities against HIV-1, with half-maximal effective concentrations (EC50) in the submicromolar range. In particular, a dihydro-1-(4-aminobenzyl)triazine analog showed satisfactory anti-HIV-1 activity with an EC50 of 0.110 µM and a selectivity index (SI) of 909. Furthermore, molecular modeling analyses were performed to explore the major interactions between HIV-1 RT and potent inhibitors. These results may be important for further development of this class of compounds as anti-HIV-1 agents.
Conclusion
The satisfactory anti-HIV-1 activity of triazine analogs may serve as the basis for further investigations of the behavior of this class of compounds against drug-resistant mutants.
Introduction
The reverse transcriptase (RT) of human immunodeficiency virus type 1 (HIV-1) is an attractive target for the development of drugs used in HIV-1 infection and AIDS treatment. It is a multifunctional enzyme that is crucial to viral replication. Currently, two functionally distinct classes of HIV-1 RT inhibitors (nucleoside and non-nucleoside) have been discovered and are used clinically. In particular, non-nucleoside RT inhibitors (NNRTIs), with a wide range of chemically diverse structures, have gained an important place in clinical use given their unique antiviral potency, generally low toxicity, and favorable pharmacokinetic properties.1–3
Among the more than 50 different series of NNRTIs reported hitherto, 1-[(2-hydroxyethoxy)methyl]-6-(phenylthio)thymine (HEPT, Figure 1), which possesses a uracil skeleton,4,5 was the first compound shown to target HIV-1 RT specifically. Since HEPT discovery, related uracil derivatives have been synthesized. In particular, emivirine (Figure 1),6–9 formerly known as MKC-442, was chosen as a candidate for clinical trials in AIDS patients, and is the best-known HEPT derivative. It also possesses an uracil skeleton. However, the phase III study was halted when emivirine was found to activate the liver enzyme cytochrome P450, which metabolizes protease inhibitors.
10
Structures of 1-[(2-hydroxyethoxy)methyl]-6-(phenylthio)thymine (HEPT) and emivirine.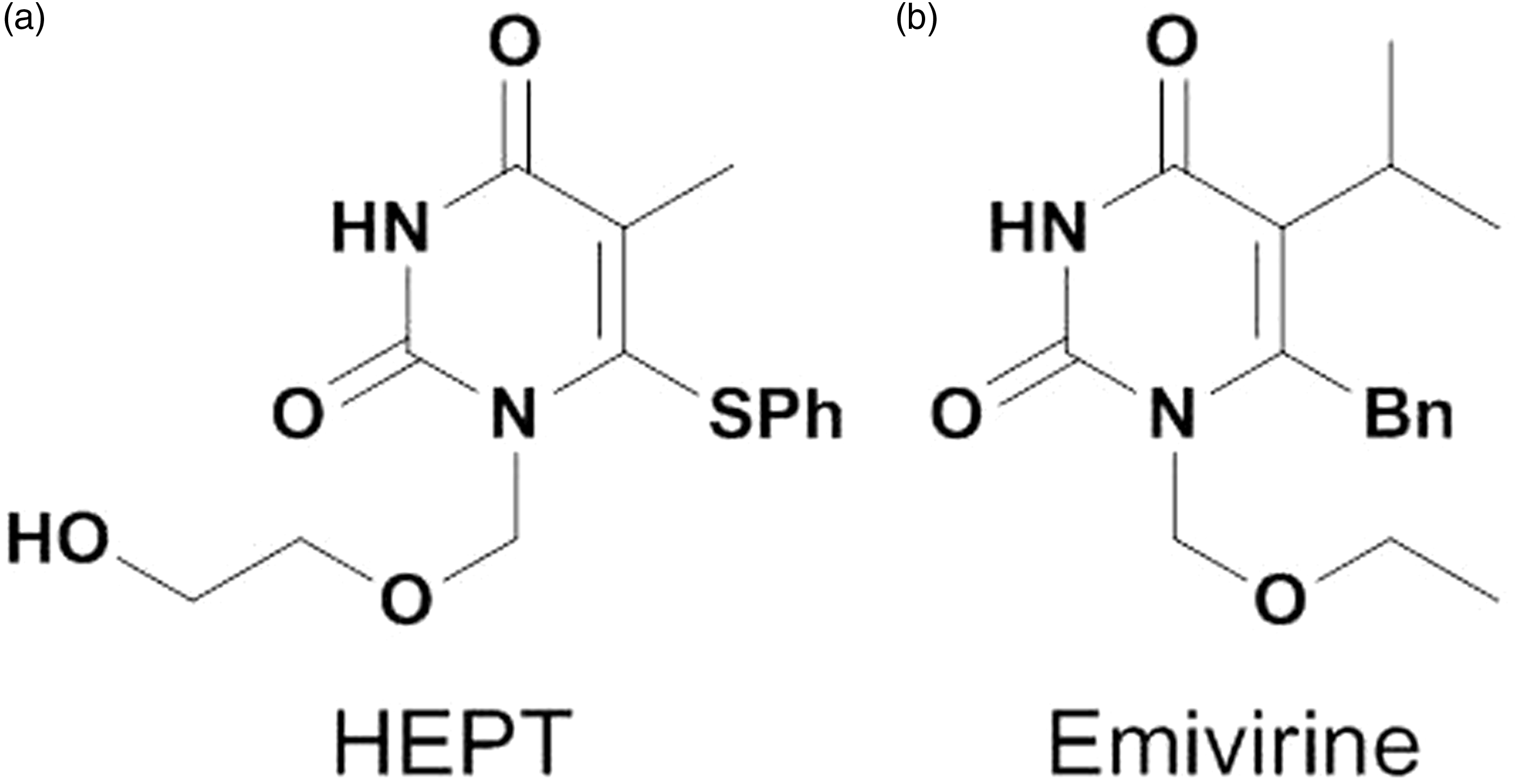
Structures and anti-HIV-1 activity of 1-substituted-3-(3,5-dimethylbenzyl)uracil derivatives
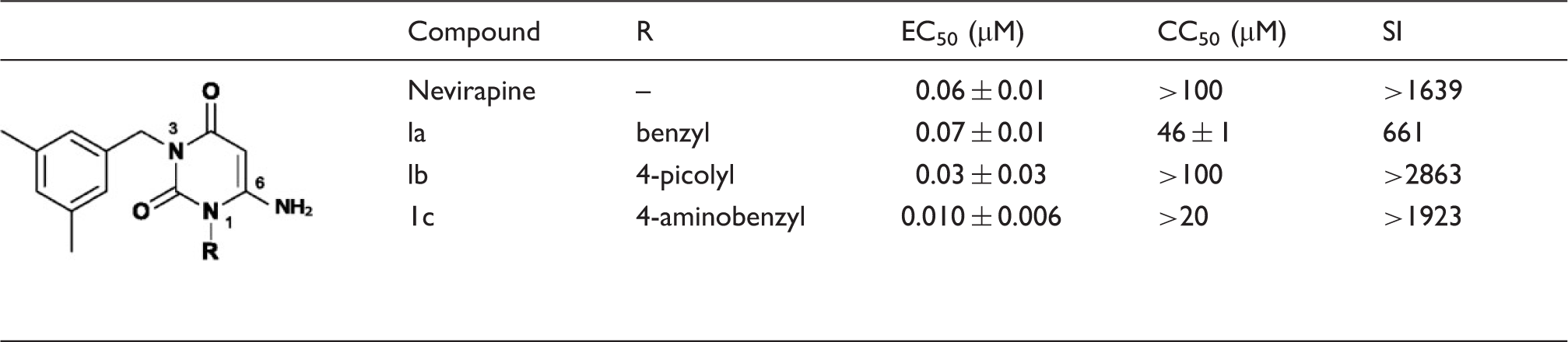
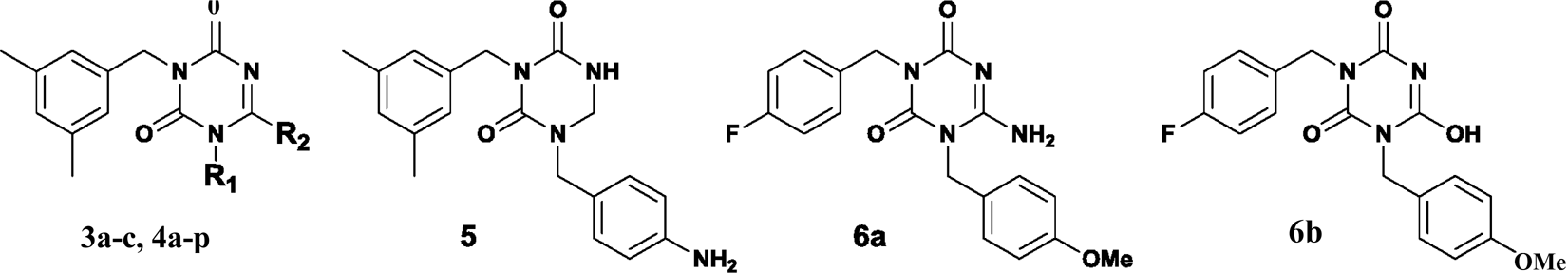
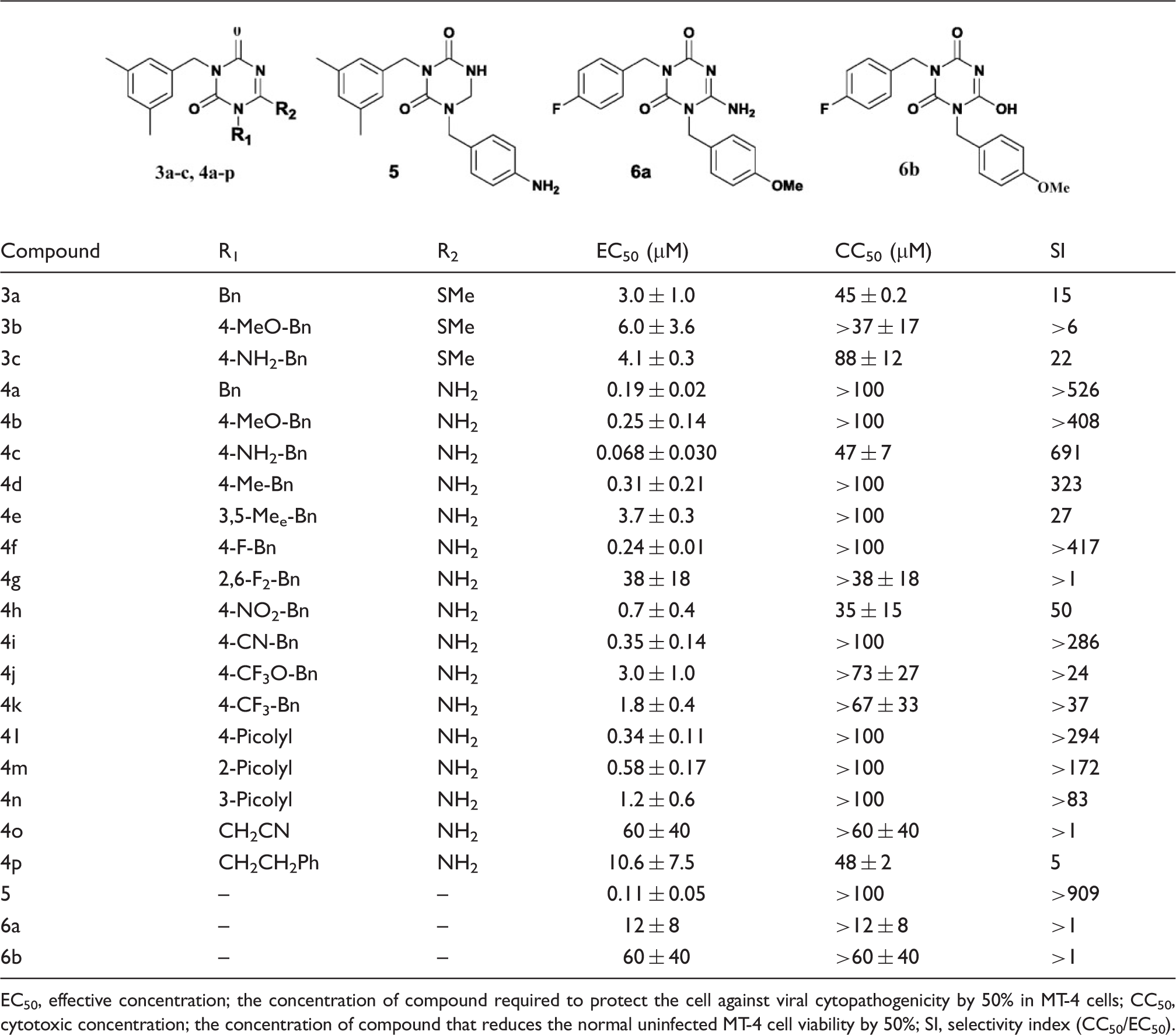
In an ongoing effort to discover potential NNRTIs with high anti-HIV-1 activity and low cytotoxicity, we designed 22 structurally relevant, novel classes of 1-substituted 3-(3,5-dimethylbenzyl)triazine analogs ( Design of 1,3,6-trisubstituted triazine derivatives. Antiviral activity of 3-(3,5-dimethylbenzyl)triazine analogs against HIV-1. EC50, effective concentration; the concentration of compound required to protect the cell against viral cytopathogenicity by 50% in MT-4 cells; CC50, cytotoxic concentration; the concentration of compound that reduces the normal uninfected MT-4 cell viability by 50%; SI, selectivity index (CC50/EC50).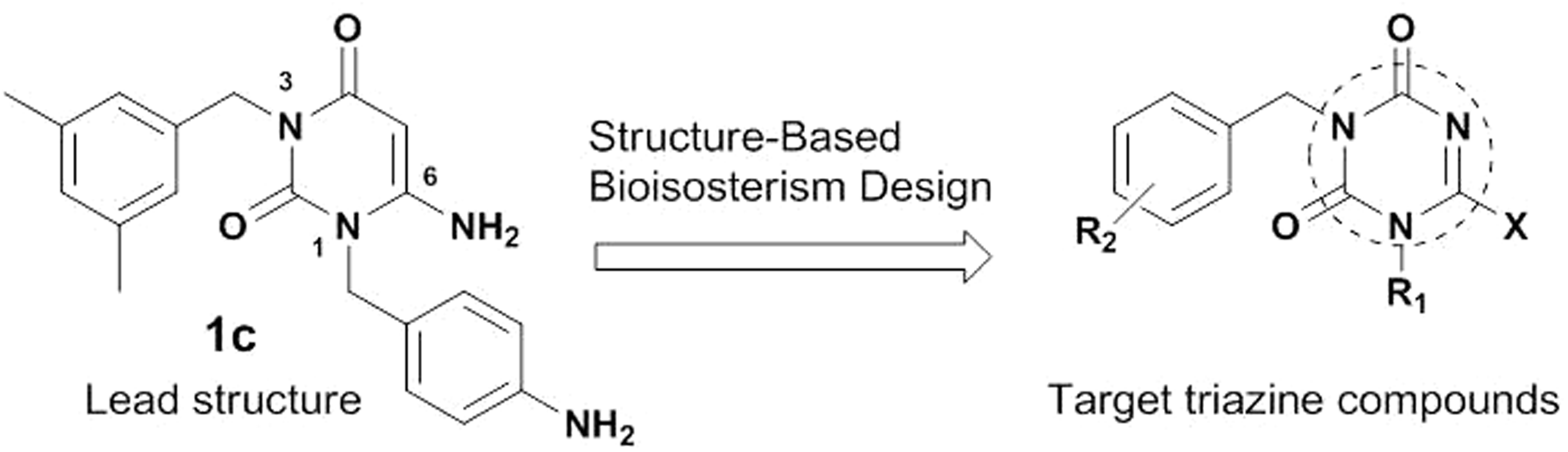
Methods
Chemistry
3-(3,5-Dimethylbenzyl)-6-methylthio-1,3,5-triazine-2,4(1 Synthesis of 3-(3,5-dimethylbenzyl)triazine derivatives 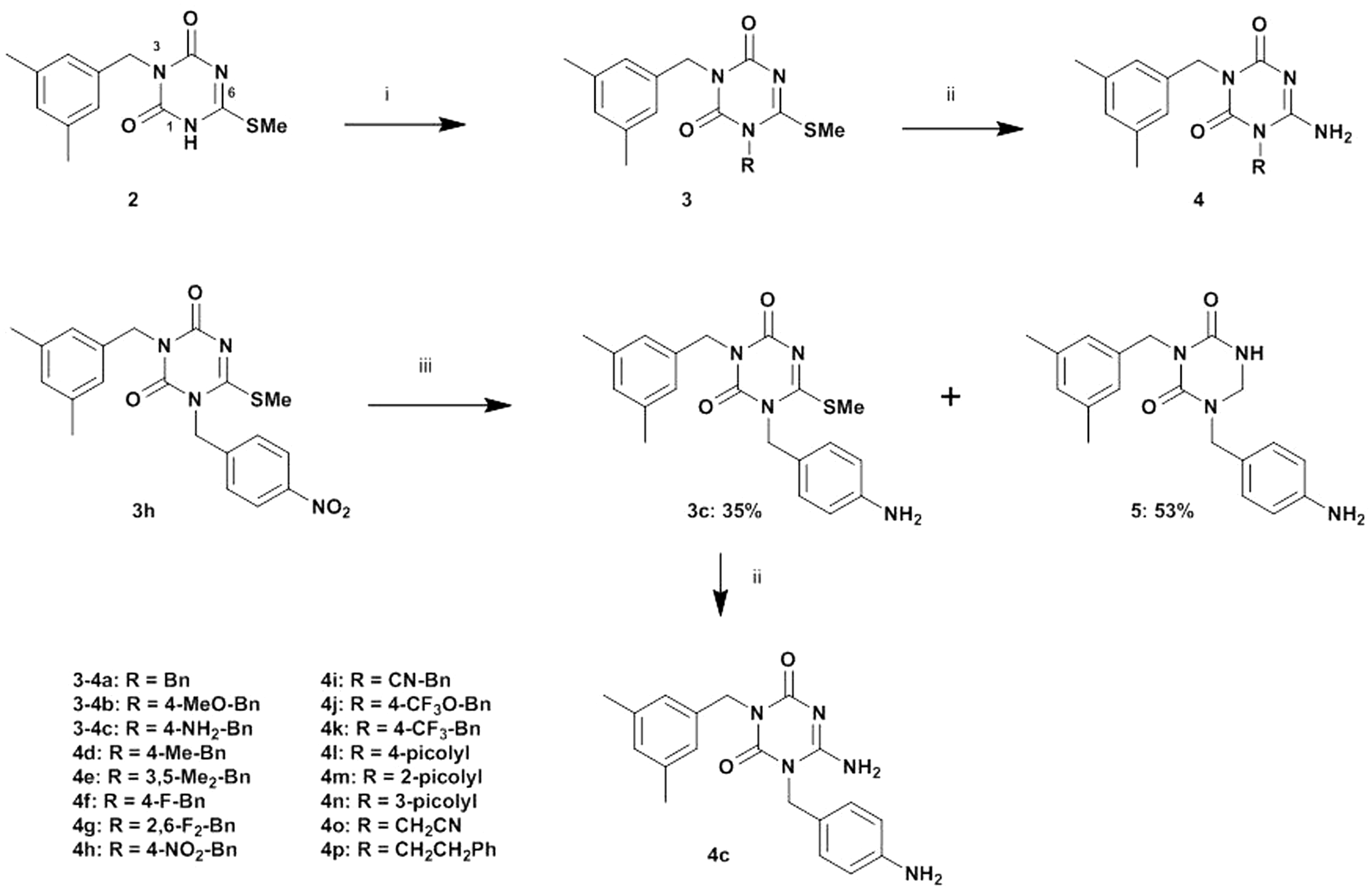
First, compound (
Anti-HIV-1 assay
MT-4 cells were maintained in RPMI 1640 medium supplemented with 10% heat-inactivated fetal bovine serum (FBS), 100 U/mL of penicillin G, and 100 mg/mL of streptomycin. The IIIB strain of HIV-1 was used throughout the experiment. The virus was propagated and titrated in MT-4 cells. Virus stocks were stored at −80°C until use. The anti-HIV-1 activity of the test compounds was determined by the inhibition of virus-induced cytopathogenicity in MT-4 cells. 24 Briefly, MT-4 cells (1 × 105 cells/mL) were infected with HIV-1 at a multiplicity of infection (MOI) of 0.1 and were cultured in the presence of various concentrations of the test compounds. After four-day incubation at 37°C in 5% CO2, the number of viable cells was monitored by the water-soluble tetrazolium dye MTT. The cytotoxicity of the compounds was evaluated in parallel with their antiviral activity, based on the viability of mock-infected cells, as determined by the MTT method.
Materials
Instrumentation
1H NMR and 13C NMR spectra were taken with an Ultrashield™ 400 Plus FT NMR System (BRUKER, Germany). Chemical shifts and coupling constants (
Compounds
General procedure for the synthesis of 3
A solution of compound
1-Benzyl-3-(3,5-dimethylbenzyl)-6-methylthio-1,3,5-triazine-2,4(1H,3H)-dione (3a)
Yield 95%; white solid; 1H NMR (400 MHz, CDCl3): δ 7.31 (5H, m, Bn), 7.08 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 5.12 (2H, s, Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.54 (3H, s, SMe), 2.28 (6H, s, 3,5-Me2-Bn); 13 C NMR (100 MHz, CDCl3): δ 170.1, 152.3, 150.5, 138.0, 136.0, 134.3, 129.6, 128.8, 128.3, 127.6, 126.7, 48.4, 45.6, 21.3, 15.3; HRMS (ESI) Calcd for C20H21N3NaO2S+ [M+Na]+: 390.12467. Found 390.12416; melting point (mp): 136.3–137.9°C.
3-(3,5-Dimethylbenzyl)-1-(4-methoxybenzyl)-6-methylthio-1,3,5-triazine-2,4(1H,3H)-dione (3b)
Yield 82%; white solid; 1H NMR (400 MHz, CDCl3): δ 7.29 (2H, d,
1-(4-Aminobenzyl)-3-(3,5-dimethylbenzyl)-6-methylthio-1,3,5-triazine-2,4(1H,3H)-dione (3c)
Yield 35%; brown solid; 1H NMR (400 MHz, CDCl3): δ 7.17 (2H, d,
General procedure for the synthesis of 4
Compound
6-Amino-1-benzyl-3-(3,5-dimethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4a)
Yield 53%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-methoxybenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4b)
Yield 58%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-1-(4-aminobenzyl)-3-(3,5-dimethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4c)
Yield 86%; pale yellow solid; 1H NMR (400 MHz, CD3OD): δ 6.93 (2H, d,
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-methylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4d)
Yield 80%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-1-(3,5-dimethylbenzyl)-3-(3,5-dimethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4e)
Yield 81%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-fluorobenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4f)
Yield 62%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-1-(2,6-difluorobenzyl)-3-(3,5-dimethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4g)
Yield 30%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-nitrobenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4 h)
Yield 61%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-1-(4-cyanobenzyl)-3-(3,5-dimethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4i)
Yield 69%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-trifluoromethoxybenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4j)
Yield 92%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-trifluoromethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4k)
Yield 56%; white needle; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-picolyl)-1,3,5-triazine-2,4(1H,3H)-dione (4l)
Yield 69%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(2-picolyl)-1,3,5-triazine-2,4(1H,3H)-dione (4m)
Yield 58%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(3-picolyl)-1,3,5-triazine-2,4(1H,3H)-dione (4n)
Yield 79%; white solid; 1H NMR (400 MHz, DMSO-
6-Amino-1-cyanomethyl-3-(3,5-dimethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (4o)
Yield 59%; brown solid; 1H NMR (400 MHz, DMSO-
6-Amino-3-(3,5-dimethylbenzyl)-1-(4-phenethyl)-1,3,5-triazine-2,4(1H,3H)-dione (4p)
Yield 55%; white solid; 1H NMR (400 MHz, DMSO-
Dihydro-1-(4-aminobenzyl)-3-(3,5-dimethylbenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (5)
Compound
6-Amino-3-(4-fluorobenzyl)-1-(4-methoxybenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (6a)
White solid; 1H NMR (400 MHz, DMSO-
3-(4-Fluorobenzyl)-6-hydroxy-1-(4-methoxybenzyl)-1,3,5-triazine-2,4(1H,3H)-dione (6b)
White solid; 1H NMR (400 MHz, DMSO-
Results
Biological activity
The antiviral activities of the 6-methylthio (
In contrast, three types of 6-methylthiolated derivatives (
Additionally, the dihydro-
Unfortunately, the 6-(cyanomethyl) and 6-(ethylphenyl) analogs (
Discussion
Molecular modeling analysis
The X-ray co-crystal structure (PDB: 1VRT) of HIV-1 RT with bound nevirapine was taken from PDB
2
for use in docking studies. A docking model comprising ligand (a) Molecular docking of 6-amino-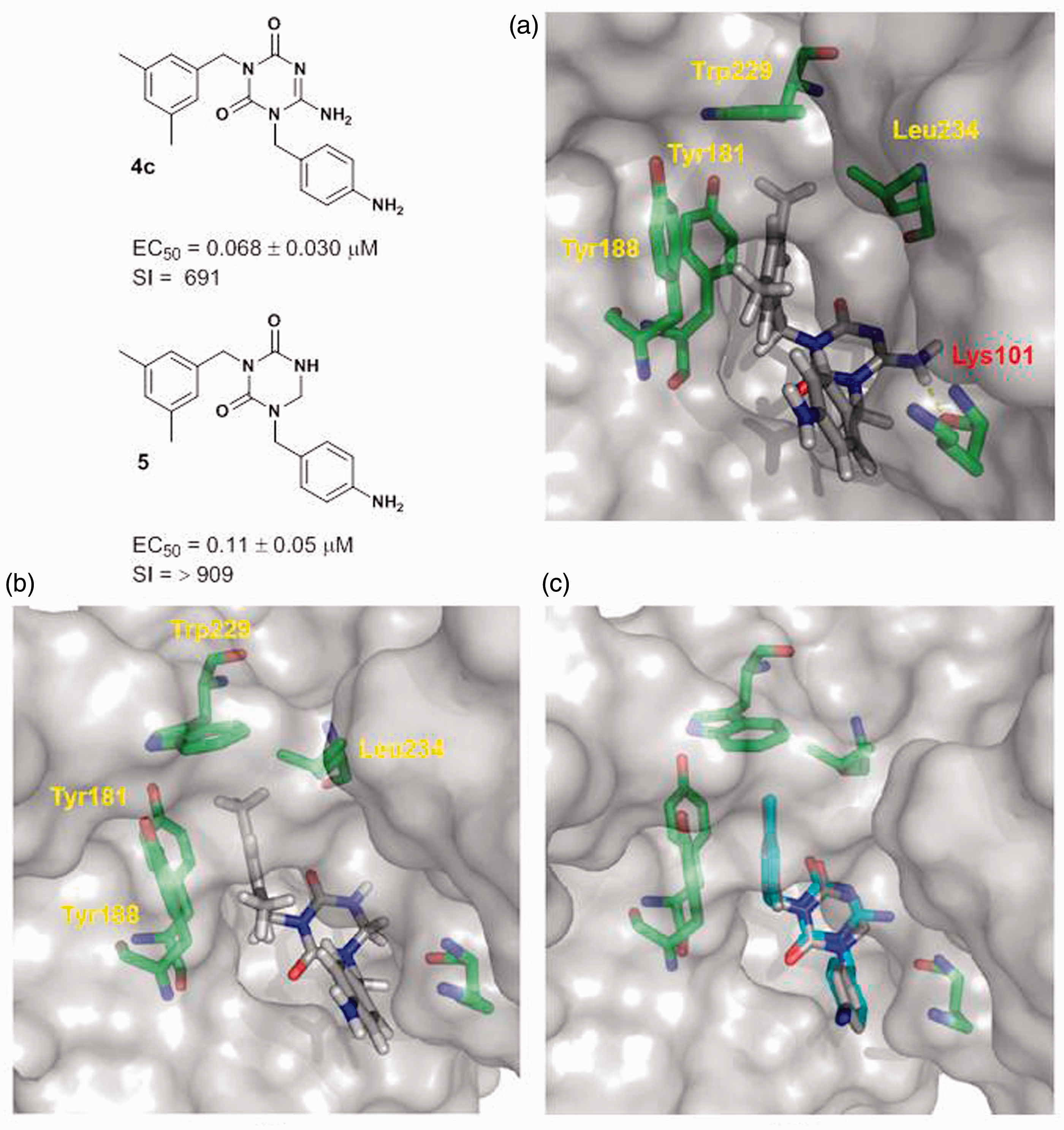
Figure 3(c) shows the overlay of the docked conformations of the 6-amino analog
In conclusion, we designed 22 structurally relevant, novel classes of 1-substituted 3-(3,5-dimethylbenzyl)triazine analogs, and 3-(4-fluorobenzyl)-1-(4-methoxybenzyl)triazine derivatives. The satisfactory anti-HIV-1 activity of triazine analogs may serve as the basis for further investigations of the behavior of this class of compounds against drug-resistant mutants.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: partially supported by a Grant-in-Aid for Young Scientists (B), No. 24790123, from the Japan Society for the Promotion of Science (JSPS).