Abstract
Background
The Enterovirus genus of the Picornaviridae is represented by several viral pathogens that are associated with human disease, namely Poliovirus 1, Enterovirus 71 and Rhinoviruses. Enterovirus 71 has been associated with encephalitis, while Rhinoviruses are a major cause of asthma exacerbations and chronic obstructive pulmonary disease. Based on the structure of both pleconaril and pirodavir, we previously synthesized some original compounds as potential inhibitors of Rhinovirus replication.
Methods
These compounds were explored for in vitro antiviral potential on other human pathogenic Enteroviruses, namely Enterovirus 71 on rhabdo-myosarcoma cells, Coxsackievirus B3 on Vero cells, Poliovirus 1 and Echovirus 11 on BGM cells.
Results
Activity was confirmed for compound against Rhinovirus 14. Furthermore, few compounds showed a cell-protective effect on Enterovirus 71, presented a marked improvement as compared to the reference drug pleconaril for inhibitory activity on both Enterovirus 71 and Poliovirus 1. The most striking observation was the clear cell protective effect for the set of analogues in a virus-cell-based assay for Echovirus 11 with an effective concentration (EC50) as low as 0.3 µM (Selectivity index or SI = 483), and selectivity indexes greater than 857 (EC50 = 0.6 µM) and 1524 (EC50 = 0.33 µM).
Conclusion
Some of the evaluated compounds showed potent and selective antiviral activity against several enterovirus species, such as Enterovirus 71 (EV-A), Echovirus 11 (EV-B), and Poliovirus 1 (EV-C). This could be used as a starting point for the development of other pleconaril/pirodavir-like enterovirus inhibitors with broad-spectrum activity and improved effects as compared to the reference drugs.
Introduction
The genus Enterovirus of the family Picornaviridae is divided into 12 species (enterovirus EV-A to-J, and rhinovirus HRV-A to -C), each of which contains multiple (sero) types, which in turn group a wide variety of strains. Human pathogens like poliovirus (PV), enterovirus (EV), Coxsackievirus (CV) and rhinovirus (HRV) are well-known representatives.1,2 In general, enterovirus infections are sub-clinical or very mild, such as the common cold caused by rhinoviruses. However, PV, Enterovirus 71 (EV71) and several other members of this virus genus are known to cause inflammation of the central nervous system,3,4 and rhinovirus infections have been associated with asthma exacerbation and chronic obstructive pulmonary disease.5,6
Enteroviruses are un-enveloped, icosahedral virions that contain a single-stranded positive-sense RNA genome which encodes, amongst others, four structural proteins (VP-1 to -4). The hydrophobic pocket beneath the groove in the VP1 capsid protein is a well-known target for the development of antivirals, which are also known as ‘capsid binders’: interference with virus entry and uncoating is brought about through interaction with the VP1 capsid protein.7,8 Pleconaril (Schering Plough) and pirodavir (R77975, Janssen) are two of the best studied capsid binders. Pleconaril has broad-spectrum antiviral activity against several entero- and rhinoviruses, and positive clinical outcomes of naturally acquired rhinovirus infections have been reported after pleconaril treatment. 9 However, the side effects of pleconaril treatment (CYP induction) have not outweighed the burden of disease, and the drug has not been approved by the FDA. 10 Pirodavir is a potent in vitro inhibitor of both HRV-A and HRV-B. 7 Despite this in vitro activity, treatment of naturally occurring rhinovirus infections has not produced any clinical benefits and has been associated with side effects, including nasal dryness and an unpleasant taste.11,12 Recently, a clinical phase II trial with vapendavir (BTA798, Biota Holdings), a pirodavir derivative, was successfully completed in asthma patients: treatment with vapendavir significantly reduced the respiratory symptoms caused by naturally acquired rhinovirus-induced asthma exacerbations. 13
We have previously described a panel of novel compounds
Materials and methods
The purpose of the present study was to explore the antiviral potential of the above-mentioned compounds together with an additional set of similar substances (compounds
The antiviral activity of the compounds was evaluated in an MTS (3 -(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium)-based assay. Briefly, HeLa Rh cells were seeded at a density of 1.8 × 104 cells per well (100 mL) in 96-well cell culture plates (Falcon, BD Biosciences). Rhinovirus assays were performed using MEM Rega3 medium supplemented with 2% FBS, 2 mM-glutamine, 0.075% NaHCO3 and 30 mM MgCl2. For the remaining picornaviruses, assays were performed in the same medium in the absence of MgCl2. Cells were allowed to adhere overnight, after which serial dilutions of the compound and virus inoculum (multiplicity of infection (moi) optimized for each virus strain) were added. The cultures were subsequently incubated for three days at 35 or 37°C, for rhinovirus and the other picornaviruses, until complete virus-induced CPEs were observed in the untreated and infected virus control conditions (VC). After removal of the medium, 100 mL Phenol Red-free MEM (Invitrogen) containing 5% MTS-phenazine (Promega) were added. Following a 1 h incubation period at 35 or 37°C, the optical density of each well at 498 nm (OD 498) was determined using a microplate reader (Safire 2, Tecan). The optical density values were converted to control percentages and logarithmic interpolation was used to calculate the 50% effective concentration (EC50) as the compound concentration resulting in a 50% protective effect against virus-induced CPE. In addition, the cell morphology of each compound condition was evaluated microscopically in a multi-cycle assay on a confluent cell layer for minor signs of CPE or for adverse effects caused by the compound. CC50 = 50% cytotoxic concentration, selectivity index (SI), EC50/CC50.
Results
Antiviral activity a against HRV-A2 and HRV-B14.
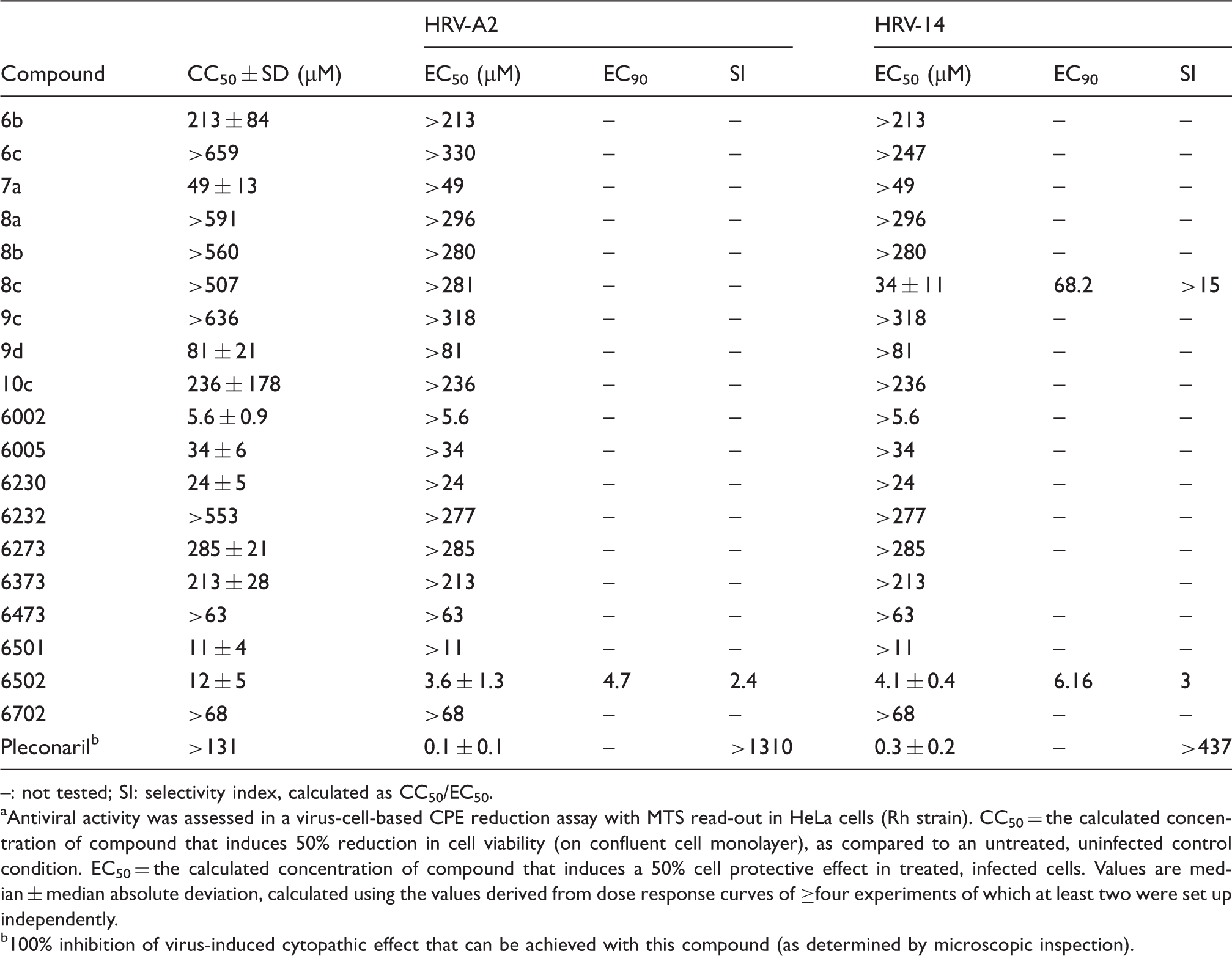
–: not tested; SI: selectivity index, calculated as CC50/EC50.
Antiviral activity was assessed in a virus-cell-based CPE reduction assay with MTS read-out in HeLa cells (Rh strain). CC50 = the calculated concentration of compound that induces 50% reduction in cell viability (on confluent cell monolayer), as compared to an untreated, uninfected control condition. EC50 = the calculated concentration of compound that induces a 50% cell protective effect in treated, infected cells. Values are median ± median absolute deviation, calculated using the values derived from dose response curves of ≥four experiments of which at least two were set up independently.
100% inhibition of virus-induced cytopathic effect that can be achieved with this compound (as determined by microscopic inspection).
Antiviral activity against EV71 (EV-A clade) and CVB3 (EV-B).
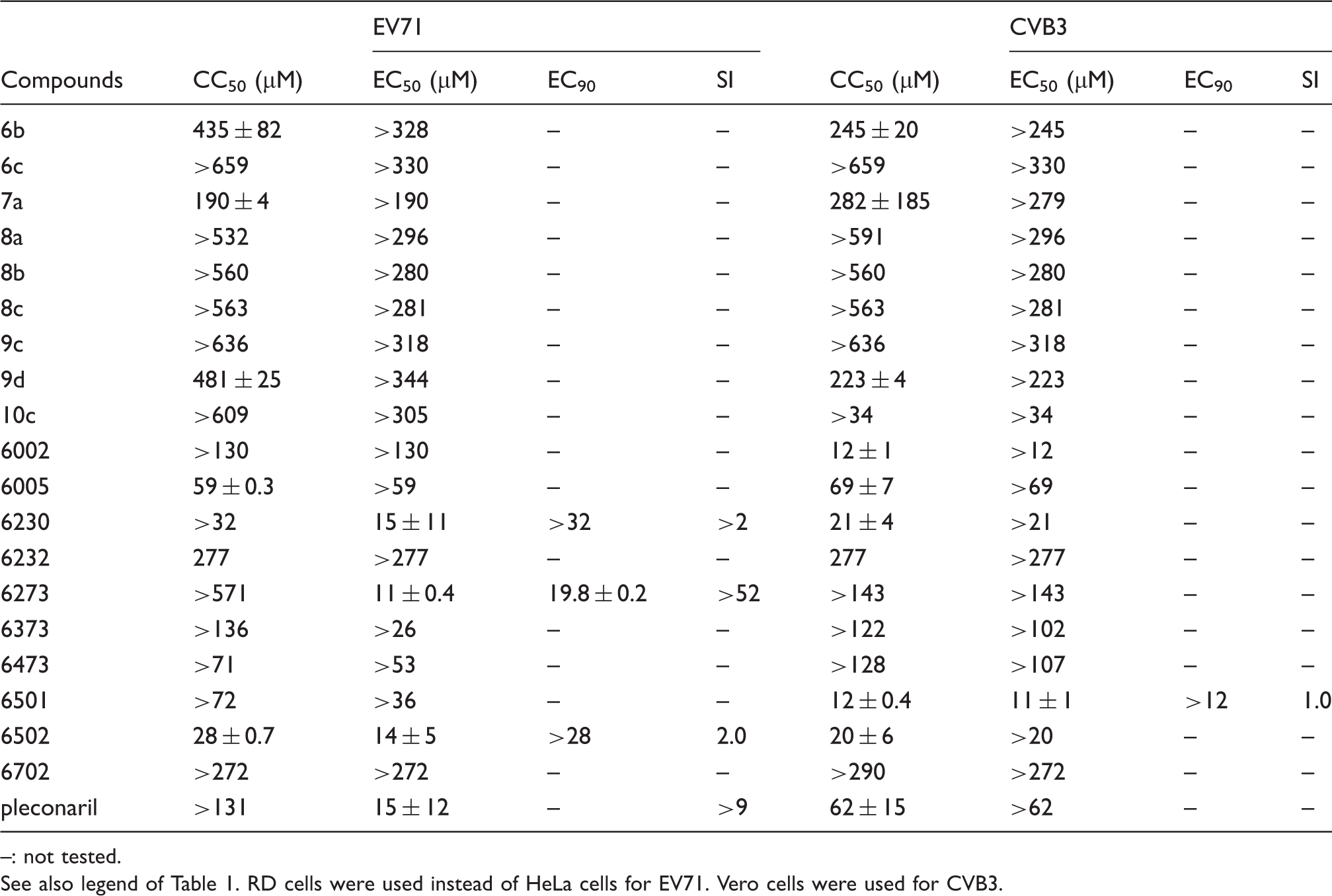
–: not tested.
See also legend of Table 1. RD cells were used instead of HeLa cells for EV71. Vero cells were used for CVB3.
Antiviral activity against echovirus 11 (EV-B) and poliovirus 1 (EV-C).
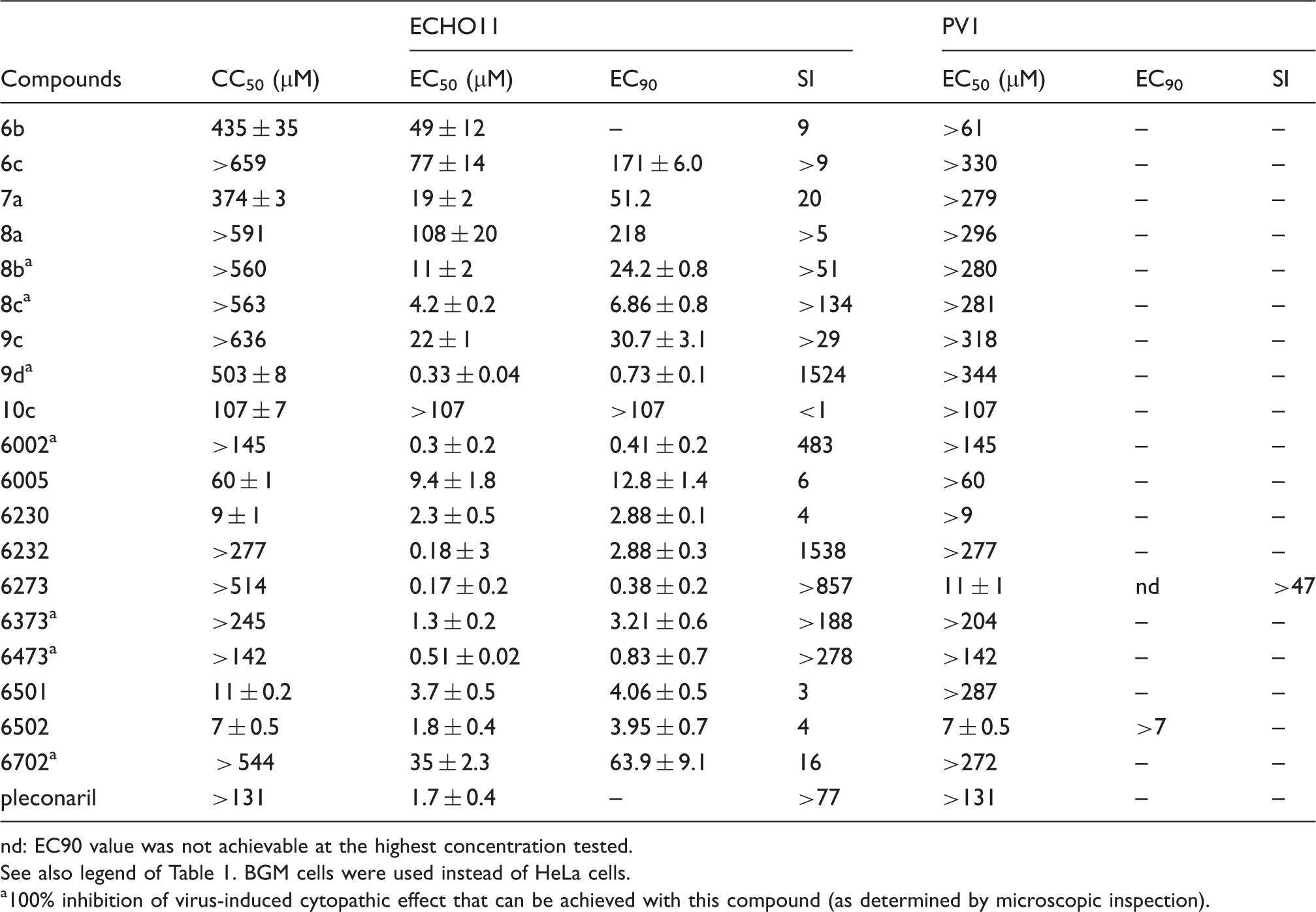
nd: EC90 value was not achievable at the highest concentration tested.
See also legend of Table 1. BGM cells were used instead of HeLa cells.
100% inhibition of virus-induced cytopathic effect that can be achieved with this compound (as determined by microscopic inspection).
In sharp contrast, a cell protective effect was observed for all but one of the compounds in the assay for ECHO11 (EV-B), with EC50s in the range between 0.3 and 108 µM (SI ranging from 3 to >857 and 1524) (Table 3). It is interesting to note that at least one concentration of seven of these compounds was able to completely inhibit the production of virus-induced cytopathic effects, without causing any adverse effects on host cell and monolayer morphology, which was assessed by microscopic inspection. For substances
Discussion
Previously obtained data indicated that compound
In conclusion, some of the evaluated compounds show selective antiviral activity against several enterovirus species, such as EV71 (EV-A), ECHO11 (EV-B) and PV1 (EV-C). This could be used as a starting point for the development of other pleconaril/pirodavir-like enterovirus inhibitors with broad-spectrum activity and a different structure. Of particular interest is the observation of the activity of all the molecules against ECHO 11, with EC50s that reach low µM and even high nM values. It is also worth noting that compound
Footnotes
Acknowledgements
We thank the IFI Company (Rome, Italy) for providing us with compounds
Conflict of interest
None declared.
Funding
Céline Lacroix is supported by a PhD grant from the Agency for Innovation by Science and Technology (IWT, Belgium).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
