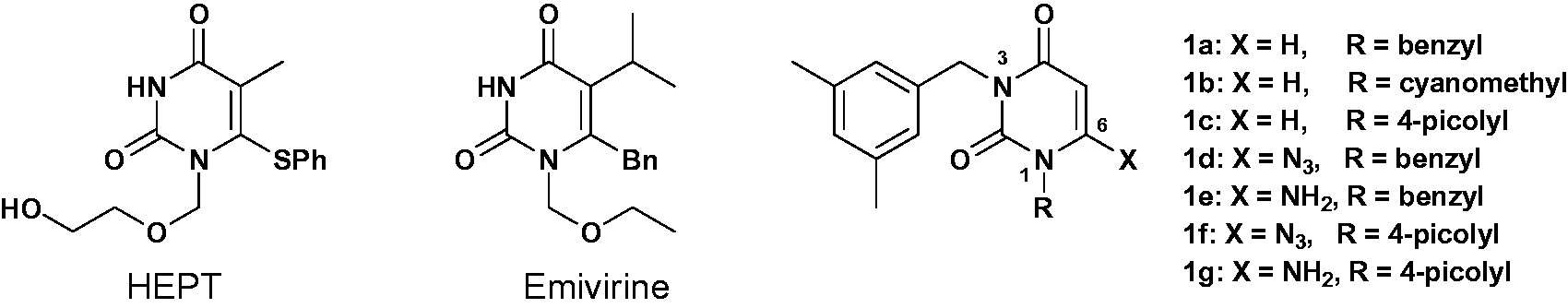
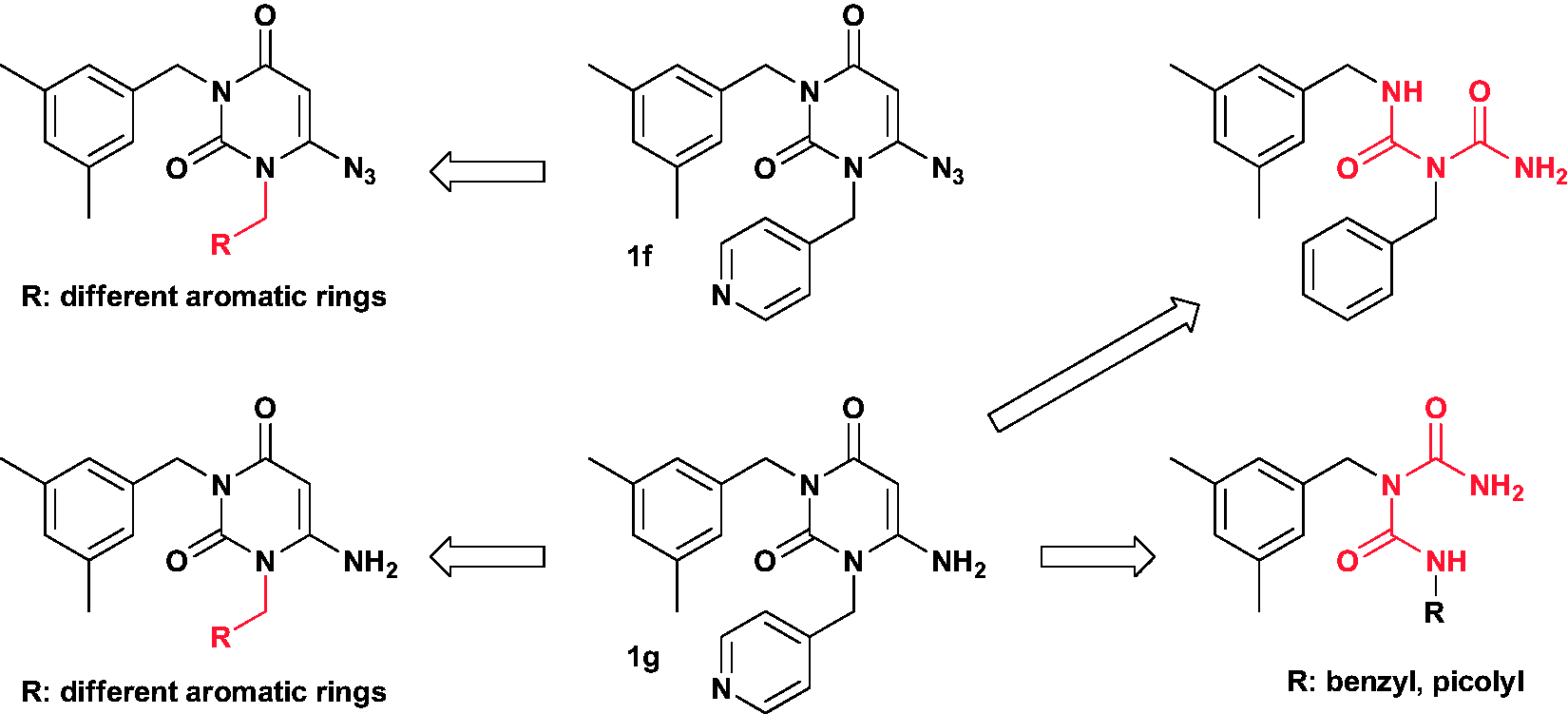
Compounds
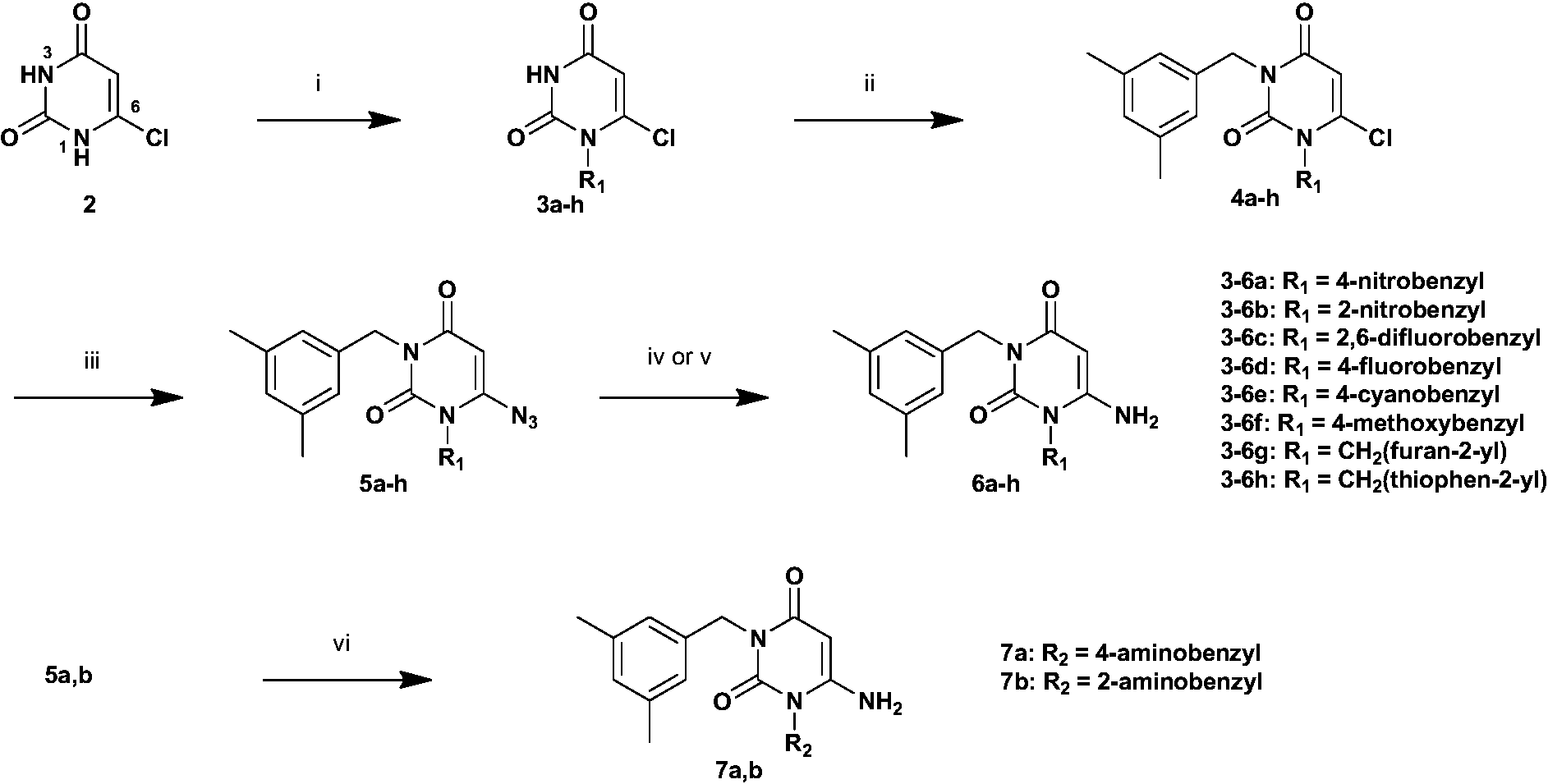
General procedure for the synthesis of 3a–h
A solution of compound 2 (1.03 g, 7.0 mmol), appropriate alkyl halide (8.4 mmol) and K2CO3 (0.51 g, 3.68 mmol), in dry DMF (25.0 mL) was heated at RT –70°C. After 2–18 h stirring, the mixture was extracted with silica gel column chromatography (AcOEt). The organic extracts were washed with water and saturated sodium chloride solution, dried with sodium sulfate, and then evaporated. The residue was purified by silica gel column chromatography to afford 3a–h.
6-Chloro-1-(4-nitrobenzyl)uracil [3a]
Yield 75%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 11.81 (1H, brs, 3-NH), 8.22 (2H, d, J 8.8, 4-NO2-Bn), 7.57 (2H, d, J 8.8, 4-NO2-Bn), 6.05 (1H, s, H-5), 5.29 (2H, s, 4-NO2-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 161.0, 150.4, 146.8, 146.4, 144.1, 127.6, 123.8, 102.7, 47.8; HRMS (ESI) Calcd for C11H8ClN3NaO4+ [M+Na]+: 304.00955. Found 304.02556; mp: 111.9–114.6°C.
6-Chloro-1-(2-nitrobenzyl)uracil [3b]
Yield 44%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 11.81 (1H, brs, 3-NH), 8.08 (1H, d, J 8.0, 2-NO2-Bn), 7.78 (1H, dd, J 8.0 and 8.0, 2-NO2-Bn), 7.61 (1H, dd, J 8.0 and 8.0, 2-NO2-Bn), 7.39 (1H, d, J 8.0, 2-NO2-Bn), 6.09 (1H, s, H-5), 5.47 (2H, s, 2-NO2-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 161.1, 150.4, 147.0, 146.4, 134.7, 131.5, 128.8, 127.1, 125.3, 102.9, 46.1; HRMS (ESI) Calcd for C11H8ClN3NaO4+ [M+Na]+: 304.00955. Found 304.00970; mp: 145.9–147.5°C.
6-Chloro-1-(2,6-difluorobenzyl)uracil [3c]
Yield 21%; white crystal; 1H NMR (400 MHz, CDCl3): δ 8.66 (1H, brs, 3-NH), 7.30 (1H, m, 2,6-F2-Bn), 6.93 (2H, m, 2,6-F2-Bn), 5.89 (1H, s, H-5), 5.37 (2H, s, 2,6-F2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.4 (d, J 28), 160.4 (d, J 212), 159.9, 149.6, 147.6, 130.2, 111.9, 103.1, 39.0; HRMS (ESI) Calcd for C11H7ClF2N2NaO2+ [M+Na]+: 295.00563. Found 295.00600; mp: 76.2–77.4°C.
6-Chloro-1-(4-fluorobenzyl)uracil [3d]
Yield 60%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 11.75 (1H, brs, 3-NH), 7.33 (2H, m, 4-F-Bn), 7.19 (2H, m, 4-F-Bn), 6.00 (1H, s, H-5), 5.14 (2H, s, 4-F-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 162.6, 161.0, 150.5, 146.6, 132.4 (d, J 12), 128.9 (d, J 36), 115.5 (d, J 84), 102.5, 47.5; HRMS (ESI) Calcd for C11H9ClFN2O2+ [M+H]+: 255.03311. Found 255.03365; mp: 93.4–95.0°C.
6-Chloro-1-(4-cyanobenzyl)uracil [3e]
Yield 59%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 11.78 (1H, brs, 3-NH), 7.84 (2H, d, J 8.4, 4-CN-Bn), 7.49 (2H, d, J 8.4, 4-CN-Bn), 6.04 (1H, s, H-5), 5.24 (2H, s, 4-CN-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 161.0, 150.5, 146.4, 142.0, 132.6, 127.3, 118.6, 110.2, 102.7, 47.9; HRMS (ESI) Calcd for C12H9ClN3O2+ [M+Na]+: 262.03778. Found 262.03830; mp: 131.0–132.0°C.
6-Chloro-1-(4-methoxybenzyl)uracil [3f]
Yield 47%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 11.78 (1H, brs, 3-NH), 7.22 (2H, d, J 8.8, 4-CH3O-Bn), 6.92 (2H, d, J 8.8, 4-CH3O-Bn), 5.97 (1H, s, H-5), 5.08 (2H, s, 4-CH3O-Bn), 3.74 (3H, s, 4-CH3O-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 160.9, 158.6, 150.5, 146.8, 128.3, 128.1, 114.0, 102.3, 55.1, 47.6; HRMS (ESI) Calcd for C12H12ClN2O3+ [M+H]+: 267.05310. Found 267.05375; mp: 123.5–124.8°C.
6-Chloro-1-[(2-furanyl)methyl]uracil [3g]
Yield 29%; brown crystal; 1H NMR (400 MHz, CDCl3): δ 9.25 (1H, brs, 3-NH), 7.38 (1H, dd, J 2.0 and 0.4, CH2(furan-2-yl)), 6.41 (1H, d, J 3.2, CH2(furan-2-yl)), 6.35 (1H, dd, J 3.2 and 2.0, CH2(furan-2-yl)), 5.90 (1H, s, H-5), 5.23 (2H, s, CH2(furan-2-yl));
13
C NMR (100 MHz, CDCl3): δ 160.8, 149.8, 148.1, 147.5, 143.0, 110.7, 110.1, 103.0, 42.1; HRMS (ESI) Calcd for C9H7ClN2NaO3+ [M+Na]+: 249.00374. Found 249.00441; mp: 130.6–132.4°C.
6-Chloro-1-[(2-thiophenyl)methyl]uracil [3h]
Yield 70%; white crystal; 1H NMR (400 MHz, CDCl3): δ 9.25 (1H, brs, 3-NH), 7.38 (1H, dd, J 2.0 and 0.4, CH2(thiophen-2-yl)), 6.41 (1H, d, J 3.2, CH2(thiophen-2-yl)), 6.35 (1H, dd, J 3.2 and 2.0, CH2(thiophen-2-yl)), 5.90 (1H, s, H-5), 5.23 (2H, s, CH2(thiophen-2-yl));
13
C NMR (100 MHz, CDCl3): δ 160.4, 149.7, 147.2, 136.4, 128.7, 126.9, 126.7, 103.0, 44.0; HRMS (ESI) Calcd for C9H7ClN2NaO2S+ [M+Na]+: 264.98090. Found 264.98138; mp: 138.2–139.4°C.
General procedure for the synthesis of 4a–h
A solution of compound 3a–h (1.93 mmol), triphenylphosphine (0.66 g, 2.51 mmol), 3,5-dimethylbenzylalcohol (0.28 g, 2.02 mmol), and TMAD (0.43 g, 2.51 mmol) in THF (16.3 mL) was stirred at 50°C. After 18 h stirring, the solution was filtered and concentrated to a small volume. The residual solution was purified by silica gel column chromatography to form 4a–h.
6-Chloro-3-(3,5-dimethylbenzyl)-1-(4-nitrobenzyl)uracil [4a]
Yield 69%; clear oil; 1H NMR (400 MHz, CDCl3): δ 8.20 (2H, d, J 8.8, 4-NO2-Bn), 7.45 (2H, d, J 8.8, 4-NO2-Bn), 7.03 (2H, s, 3,5-Me2-Bn), 6.91 (1H, s, 3,5-Me2-Bn), 6.01 (1H, s, H-5), 5.34 (2H, s, 4-NO2-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 160.2, 151.1, 147.7, 145.0, 142.6, 138.1, 135.9, 129.6, 128.2, 126.6, 124.1, 103.1, 49.2, 45.0, 21.3; HRMS (ESI) Calcd for C20H18ClN3NaO4+ [M+Na]+: 422.08780. Found 422.08734.
6-Chloro-3-(3,5-dimethylbenzyl)- 1-(2-nitrobenzyl)uracil [4b]
Yield 84%; brown oil; 1H NMR (400 MHz, CDCl3): δ 8.18 (1H, d, J 8.0, 2-NO2-Bn), 7.62 (1H, dd, J 8.0 and 8.0, 2-NO2-Bn), 7.50 (1H, dd, J 8.0 and 8.0, 2-NO2-Bn), 7.08 (1H, d, J 8.0, 2-NO2-Bn), 7.03 (2H, s, 3,5-Me2-Bn), 6.91 (1H, s, 3,5-Me2-Bn), 6.05 (1H, s, H-5), 5.60 (2H, s, 2-NO2-Bn), 5.05 (2H, s, 3,5-Me2-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 160.3, 151.0, 147.4, 145.5, 138.1, 135.9, 134.2, 131.7, 129.6, 128.5, 126.6, 126.4, 125.8, 103.1, 47.8, 45.0, 21.3; HRMS (ESI) Calcd for C20H18ClN3NaO4+ [M+Na]+: 422.08780. Found 422.08740.
6-Chloro-1-(2,6-difluorobenzyl)-3-(3,5-dimethylbenzyl)uracil [4c]
Yield 60%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.27 (1H, m, 2,6-F2-Bn), 7.00 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 6.89 (2H, m, 2,6-F2-Bn), 5.94 (1H, s, H-5), 5.39 (1H, s, 2,6-F2-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.4 (d, J 28), 160.5, 159.9 (d, J 28), 150.9, 145.5, 137.9, 136.1, 130.0 (t, J 40), 129.4, 126.3, 111.7 (q, J 24), 102.7, 44.9, 39.8 (t, J 16), 21.3; HRMS (ESI) Calcd for C20H17ClF2N2NaO2+ [M+Na]+: 413.08388. Found 413.08344; mp: 120.5–122.2°C.
6-Chloro-3-(3,5-dimethylbenzyl)- 1-(4-fluorobenzyl)uracil [4d]
Yield 84%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.27 (2H, m, 4-F-Bn), 7.00 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 6.89 (2H, m, 4-F-Bn), 5.94 (1H, s, H-5), 5.39 (2H, s, 4-F-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 163.7, 161.3, 160.4, 151.2, 145.5, 138.2 (d, J 52), 136.1, 129.6 (q, J 32), 126.5.5, 124.9, 115.9 (d, J 84), 102.7, 49.2, 44.9, 21.3; HRMS (ESI) Calcd for C20H19ClFN2O2+ [M+Na]+: 373.11136. Found 373.11074; mp: 99.0–99.9°C.
6-Chloro-1-(4-cyanobenzyl)- 3-(3,5-dimethylbenzyl)uracil [4e]
Yield 71%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.64 (2H, d, J 8.4, 4-CN-Bn), 7.40 (2H, d, J 8.4, 4-CN-Bn), 7.03 (2H, s, 3,5-Me2-Bn), 6.91 (1H, s, 3,5-Me2-Bn), 6.00 (1H, s, H-5), 5.29 (2H, s, 4-CN-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 160.2, 151.1, 145.1, 140.6, 138.1, 135.9, 132.7, 129.6, 128.0, 126.5, 118.3, 112.2, 103.0, 49.4, 45.0, 21.3; HRMS (ESI) Calcd for C21H18ClN3NaO2+ [M+Na]+: 402.09798. Found 402.09728; mp: 97.6–98.5°C.
6-Chloro-3-(3,5-dimethylbenzyl)- 1-(4-methoxybenzyl)uracil [4f]
Yield 83%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.28 (2H, d, J 8.8, 4-CH3O-Bn), 7.03 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 6.83 (2H, d, J 8.8, 4-CH3O-Bn), 5.93 (1H, s, H-5), 5.19 (2H, s, 4-CH3O-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 3.79 (3H, s, 4-CH3O-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 160.5, 159.5, 151.3, 145.7, 138.0, 136.2, 129.3, 127.5, 126.5, 124.8, 114.1, 102.5, 55.3, 49.4, 44.8, 21.3; HRMS (ESI) Calcd for C21H21ClN2NaO3+ [M+Na]+: 407.11329. Found 407.11293; mp: 96.4–97.2°C.
6-Chloro-3-(3,5-dimethylbenzyl)-1-[(2-furanyl) methyl]uracil [4g]
Yield 69%; brown crystal; 1H NMR (400 MHz, CDCl3): δ 7.37 (1H, m, CH2(furan-2-yl)), 7.04 (2H, s, 3,5-Me2-Bn), 6.89 (1H, s, 3,5-Me2-Bn), 6.38 (1H, d, J 3.2, CH2(furan-2-yl)), 6.34 (1H, dd, J 3.2 and 2.0, CH2(furan-2-yl)), 5.94 (1H, s, H-5), 5.23 (2H, s, CH2(furan-2-yl)), 5.02 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 160.5, 150.8, 148.5, 145.3, 142.8, 138.0, 136.2, 129.5, 126.6, 110.6, 109.9, 102.7, 44.9, 42.9, 21.2; HRMS (ESI) Calcd for C18H17ClN2NaO3+ [M+Na]+: 367.08199. Found 367.08146; mp: 51.8–52.3°C.
6-Chloro-3-(3,5-dimethylbenzyl)-1-[(2-thiophenyl) methyl]uracil [4h]
Yield 68%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.28 (1H, dd, J 5.2 and 0.8, CH2(thiophen-2-yl)), 7.16 (1H, d, J 3.2, CH2(thiophen-2-yl)), 7.05 (2H, s, 3,5-Me2-Bn), 6.97 (1H, dd, J 5.2 and 3.2, CH2(thiophen-2-yl)), 6.90 (1H, s, 3,5-Me2-Bn), 5.93 (1H, s, H-5), 5.39 (2H, s, CH2(thiophen-2-yl)), 5.04 (2H, s, 3,5-Me2-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 160.4, 150.9, 144.9, 138.0, 136.7, 136.1, 129.5, 128.6, 126.8, 126.6, 126.5, 102.7, 44.8, 44.7, 21.3; HRMS (ESI) Calcd for C18H17ClN2NaO2S+ [M+Na]+: 383.05915. Found 383.05870; mp: 94.9–95.5°C.
General procedure for the synthesis of 5a–h, 10, and 14
Compound 4a–h, 9, or 13 (0.58 mmol) was dissolved in dry DMF (4.0 mL), and NaN3 (0.04 g, 0.67 mmol) was added to the solution, which was stirred for 30 min at room temperature. The mixture was extracted with AcOEt, washed with saturated aqueous sodium chloride solution, dried with sodium sulfate, and then evaporated. The residue was purified by silica gel column chromatography (70% AcOEt in hexane) to form C6-azido derivatives.
6-Azido-3-(3,5-dimethylbenzyl)-1-(4-nitrobenzyl) uracil [5a]
Yield 90%; brownish crystal; 1H NMR (400 MHz, CDCl3): δ 8.19 (2H, d, J 8.4, 4-NO2-Bn), 7.47 (2H, d, J 8.4, 4-NO2-Bn), 7.03 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 5.60 (1H, s, H-5), 5.11 (2H, s, 4-NO2-Bn), 5.05 (2H, s, 3,5-Me2-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 160.0, 151.1, 150.3, 147.7, 142.9, 138.0, 136.2, 128.6, 126.4, 124.8, 124.0, 88.3, 46.2, 44.7, 21.3; HRMS (ESI) Calcd for C20H18N6NaO4+ [M+Na]+: 429.12817. Found 429.12714; mp: 143.6–146.7°C.
6-Azido-3-(3,5-dimethylbenzyl)-1-(2-nitrobenzyl) uracil [5b]
Yield 80%; colorless needle crystal; 1H NMR (400 MHz, CDCl3): δ 8.13 (1H, dd, J 8.4 and 1.6, 2-NO2-Bn), 7.59 (1H, m, 2-NO2-Bn), 7.48 (1H, m, 2-NO2-Bn), 7.09 (1H, d, J 8.0, 2-NO2-Bn), 7.03 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 5.63 (1H, s, H-5), 5.47 (2H, s, 2-NO2-Bn), 5.06 (2H, s, 3,5-Me2-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 161.0, 151.1, 150.6, 147.8, 138.0, 136.0, 134.0, 131.9, 129.4, 128.6, 126.9, 126.5, 125.5, 88.3, 44.7, 44.5, 21.3; HRMS (ESI) Calcd for C20H18N6NaO4+ [M+Na]+: 429.12817. Found 429.12757; mp: 127.6–128.3°C.
6-Azido-1-(2,6-difluorobenzyl)- 3-(3,5-dimethylbenzyl)uracil [5c]
Yield 98%; brown crystal; 1H NMR (400 MHz, CDCl3): δ 7.25 (1H, m, 2,6-F2-Bn), 6.99 (2H, s, 3,5-Me2-Bn), 6.88 (2H, m, 2,6-F2-Bn), 6.87 (1H, s, 3,5-Me2-Bn), 5.56 (1H, s, H-5), 5.17 (1H, s, 2,6-F2-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.26 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.5 (d, J 28), 161.3, 160.0 (d, J 32), 150.8 (d, J 28), 137.9, 136.4, 129.9 (t, J 40), 129.2, 126.2, 111.8 (d, J 24), 111.5 (t, J 12), 88.2, 44.6, 36.8 (t, J 12), 21.3; HRMS (ESI) Calcd for C20H17F2N5NaO2+ [M+Na]+: 420.12425. Found 420.12268; mp: 96.8–99.8°C.
6-Azido-3-(3,5-dimethylbenzyl)- 1-(4-fluorobenzyl)uracil [5d]
Yield 86%; brown crystal; 1H NMR (400 MHz, CDCl3): δ 7.34 (2H, m, 4-F-Bn), 7.02 (2H, s, 3,5-Me2-Bn), 7.01 (2H, m, 4-F-Bn), 6.89 (1H, s, 3,5-Me2-Bn), 5.56 (1H, s, H-5), 5.05 (2H, s, 3,5-Me2-Bn), 4.99 (2H, s, 4-F-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 163.7, 161.3 (d, J 40), 151.2, 150.6, 137.9, 136.4, 131.6 (d, J 12), 130.0 (d, J 24), 129.3, 126.3, 115.8 (d, J 84), 88.1, 46.3, 44.6, 21.3; HRMS (ESI) Calcd for C20H18FN5NaO2+ [M+Na]+: 402.13367. Found 402.13242; mp: 78.8–82.5°C.
6-Azido-1-(4-cyanobenzyl)-3-(3,5-dimethylbenzyl) uracil [5e]
Yield 96%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.62 (2H, d, J 8.0, 4-CN-Bn), 7.41 (2H, d, J 8.0, 4-CN-Bn), 7.02 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 5.59 (1H, s, H-5), 5.07 (2H, s, 4-CN-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 161.0, 151.1, 150.3, 141.0, 138.0, 136.3, 132.6, 129.4, 128.4, 126.4, 118.4, 112.1, 88.2, 46.5, 44.7, 21.3; HRMS (ESI) Calcd for C21H18N6NaO2+ [M+Na]+: 409.13834. Found 409.13718; mp: 131.3–133.4°C.
6-Azido-3-(3,5-dimethylbenzyl)- 1-(4-methoxybenzyl)uracil [5f]
Yield 87%; brown crystal; 1H NMR (400 MHz, CDCl3): δ 7.30 (2H, d, J 8.4, 4-CH3O-Bn), 7.03 (2H, s, 3,5-Me2-Bn), 6.88 (1H, s, 3,5-Me2-Bn), 6.85 (2H, d, J 8.4, 4-CH3O-Bn), 5.54 (1H, s, H-5), 5.05 (2H, s, 3,5-Me2-Bn), 4.97 (2H, s, 4-CH3O-Bn), 3.79 (3H, s, 4-CH3O-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 161.3, 159.4, 151.2, 150.8, 137.9, 136.5, 129.6, 129.3, 127.9, 126.3, 114.1, 88.0, 55.3, 46.5, 44.6, 21.3; HRMS (ESI) Calcd for C21H21N5NaO3+ [M+Na]+: 414.15366. Found 414.15233; mp: 93.4–98.7°C.
6-Azido-3-(3,5-dimethylbenzyl)-1-[(2-furanyl) methyl]uracil [5g]
Yield 65%; brown oil; 1H NMR (400 MHz, CDCl3): δ 7.35 (1H, dd, J 1.6 and 0.8, CH2(furan-2-yl)), 7.03 (2H, s, 3,5-Me2-Bn), 6.88 (1H, s, 3,5-Me2-Bn), 6.35 (1H, d, J 3.2, CH2(furan-2-yl)), 6.32 (1H, dd, J 3.2 and 1.6, CH2(furan-2-yl)), 5.56 (1H, s, H-5), 5.03 (2H, s, 3,5-Me2-Bn), 5.01 (2H, s, CH2(furan-2-yl)), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 161.2, 150.8, 150.5, 148.8, 142.7, 137.9, 136.5, 129.3, 126.5, 110.6, 109.6, 88.1, 44.5, 39.9, 21.3; HRMS (ESI) Calcd for C18H17N5NaO3+ [M+Na]+: 374.12236. Found 374.12183.
6-Azido-3-(3,5-dimethylbenzyl)- 1-[(2-thiophenyl)methyl]uracil [5h]
Yield 86%; brown oil; 1H NMR (400 MHz, CDCl3): δ 7.26 (1H, dd, J 5.2 and 0.8, CH2(thiophen-2-yl)), 7.14 (1H, d, J 3.6, CH2(thiophen-2-yl)), 7.04 (2H, s, 3,5-Me2-Bn), 6.95 (1H, dd, J 5.2 and 3.6, CH2(thiophen-2-yl)), 6.88 (1H, s, 3,5-Me2-Bn), 5.54 (1H, s, H-5), 5.17 (2H, s, CH2(thiophen-2-yl)), 5.04 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 161.1, 150.8, 150.2, 137.9, 137.1, 136.4, 129.3, 128.4, 126.7, 126.4, 126.4, 88.1, 44.5, 41.6, 21.3; HRMS (ESI) Calcd for C18H17N5NaO2S+ [M+Na]+: 390.09952. Found 390.09913.
1-[4-(Acetoxy)benzyl]-6-azido-3- (3,5-dimethylbenzyl)uracil [10]
Yield 96%; yellow crystal; 1H NMR (400 MHz, CDCl3): δ 7.35 (2H, d, J 8.8, 4-AcO-Bn), 7.06 (2H, d, J 8.8, 4-AcO-Bn), 7.04 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 5.96 (1H, s, H-5), 5.24 (2H, s, 4-AcO-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.29 (3H, s, 4-AcO-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 169.4, 161.2, 151.2, 150.7, 150.4, 138.0, 136.4, 133.3, 129.3, 129.3, 126.3, 121.9, 88.0, 46.3, 44.6, 21.3, 21.1; HRMS (ESI) Calcd for C22H21N5NaO4+ [M+Na]+: 442.14858. Found 442.14776; mp: 39.6–41.5°C.
6-Azido-3-(3,5-dimethylbenzyl)- 1-(4-pyrimidinylmethyl)uracil [14]
Yield 89%; brown oil; 1H NMR (400 MHz, CDCl3): δ 9.13 (1H, s, 4-pyrimidinylmethyl), 8.69 (1H, d, J 5.2, 4-pyrimidinylmethyl), 7.20 (1H, d, J 5.2, 4-pyrimidinylmethyl), 7.01 (2H, s, 3,5-Me2-Bn), 6.89 (1H, s, 3,5-Me2-Bn), 5.64 (1H, s, H-5), 5.13 (2H, s, 4-pyrimidinylmethyl), 5.05 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 163.5, 161.2, 158.9, 157.2, 151.1, 150.7, 137.9, 136.3, 129.3, 126.2, 118.4, 88.1, 47.1, 44.6, 21.3; HRMS (ESI) Calcd for C18H17N7NaO2+ [M+Na]+: 386.13359. Found 386.13924.
General procedure for the synthesis of 6a–d, 6f–h, and 12
Compound 5a-h or 10 (0.64 mmol) was dissolved in dry THF (10.0 mL) under nitrogen atmosphere. To this stirred solution, we carefully added LiAlH4 (0.02 g, 0.77 mmol) at 0°C. Stirring was continued at 0°C for 5 min, and the reaction quenched by the addition of AcOEt (5.0 mL) until no effervescence was observed. Aqueous 1N HCl (2.2 mL) was then added, and the product was extracted with AcOEt. The combined organic extracts were washed with water and saturated sodium chloride solution, dried with sodium sulfate, and then evaporated. The residue was purified by silica gel column chromatography (10% MeOH in AcOEt) to form C6-amino analogs.
6-Amino-3-(3,5-dimethylbenzyl)- 1-(4-nitrobenzyl)uracil [6a]
Yield 71%; yellowish crystal; 1H NMR (400 MHz, CDCl3): δ 8.12 (2H, d, J 8.4, 4-NO2-Bn), 7.30 (2H, d, J 8.4, 4-NO2-Bn), 6.93 (2H, s, 3,5-Me2-Bn), 6.84 (1H, s, 3,5-Me2-Bn), 5.13 (2H, s, 4-NO2-Bn), 5.07 (2H, s, 3,5-Me2-Bn), 4.99 (1H, brs, 6-NH2), 4.93 (1H, s, H-5), 2.23 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.9, 153.7, 151.8, 147.6, 142.5, 138.0, 137.1, 129.1, 127.2, 125.8, 124.2, 78.6, 45.5, 44.4, 21.3; HRMS (ESI) Calcd for C20H20N4NaO4+ [M+Na]+: 403.13768. Found 403.13612; mp: 109.3–112.2°C.
6-Amino-3-(3,5-dimethylbenzyl)-1-(2-nitrobenzyl) uracil [6b]
Yield 67%; yellowish crystal; 1H NMR (400 MHz, CDCl3): δ 8.11 (1H, dd, J 8.4 and 1.2, 2-NO2-Bn), 7.63 (1H, m, 2-NO2-Bn), 7.50 (1H, m, 2-NO2-Bn), 7.24 (1H, d, J 8.0, 2-NO2-Bn), 7.00 (2H, s, 3,5-Me2-Bn), 6.86 (1H, s, 3,5-Me2-Bn), 5.50 (2H, s, 2-NO2-Bn), 5.04 (1H, s, H-5), 5.04 (2H, s, 3,5-Me2-Bn), 4.71 (2H, brs, 6-NH2), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.5, 152.9, 152.0, 147.6, 137.9, 137.2, 134.5, 130.7, 129.1, 127.4, 126.1, 126.1, 125.7, 79.1, 44.4, 43.2, 21.3; HRMS (ESI) Calcd for C20H20N4NaO4+ [M+Na]+: 403.13768. Found 403.13663; mp: 177.1–181.6°C.
6-Amino-3-(3,5-dimethylbenzyl)- 1-(2,6-difluorobenzyl)uracil [6c]
Yield 89%; yellowish crystal; 1H NMR (400 MHz, CDCl3): δ 7.23 (1H, m, 2,6-F2-Bn), 6.97 (2H, s, 3,5-Me2-Bn), 6.91 (2H, m, 2,6-F2-Bn), 6.83 (1H, s, 3,5-Me2-Bn), 5.20 (1H, s, 2,6-F2-Bn), 5.02 (2H, s, 3,5-Me2-Bn), 4.97 (1H, s, H-5), 4.78 (2H, brs, 6-NH2), 2.25 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.7, 162.4 (d, J 28), 159.9 (d, J 28), 153.3, 151.6, 137.8, 137.3, 130.3 (t, J 40), 128.8, 125.7, 112.0 (q, J 28), 111.0, 79.1, 44.3, 21.2; HRMS (ESI) Calcd for C20H19F2N3NaO2+ [M+Na]+: 394.13375. Found 394.13294; mp: 53.1–57.2°C.
6-Amino-3-(3,5-dimethylbenzyl)- 1-(4-fluorobenzyl)uracil [6d]
Yield 84%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.21 (2H, m, 4-F-Bn), 7.04 (2H, m, 4-F-Bn), 7.01 (2H, s, 3,5-Me2-Bn), 6.86 (1H, s, 3,5-Me2-Bn), 5.07 (2H, s, 4-F-Bn), 5.05 (2H, s, 3,5-Me2-Bn), 5.00 (1H, s, H-5), 4.55 (1H, brs, 6-NH2), 2.26 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 163.8, 162.7, 153.6, 152.1, 137.9, 137.3, 130.8 (d, J 12), 129.0, 128.2 (d, J 32), 126.1, 116.4 (d, J 84), 79.4, 45.6, 44.3, 21.3; HRMS (ESI) Calcd for C20H20FN3NaO2+ [M+Na]+: 376.14318. Found 376.14182; mp: 166.8–168.4°C.
6-Amino-3-(3,5-dimethylbenzyl)- 1-(4-methoxybenzyl)uracil [6f]
Yield 88%; yellowish oil; 1H NMR (400 MHz, CDCl3): δ 7.14 (2H, d, J 8.8, 4-CH3O-Bn), 6.99 (2H, s, 3,5-Me2-Bn), 6.87 (1H, s, 3,5-Me2-Bn), 6.86 (2H, d, J 8.8, 4-CH3O-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 5.02 (2H, s, 4-CH3O-Bn), 4.97 (1H, s, H-5), 4.71 (1H, brs, 6-NH2), 3.78 (3H, s, 4-CH3O-Bn), 2.25 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.8, 159.6, 154.1, 152.2, 137.8, 137.4, 128.9, 127.7, 127.0, 126.0, 114.7, 79.0, 55.3, 45.8, 44.3, 21.3; HRMS (ESI) Calcd for C21H23N3NaO3+ [M+Na]+: 388.16316. Found 388.16225.
6-Amino-3-(3,5-dimethylbenzyl)-1-[(2-furanyl) methyl]uracil [6g]
Yield 53%; red-brown crystal; 1H NMR (400 MHz, CDCl3): δ 7.37 (1H, m, CH2(furan-2-yl)), 7.04 (2H, s, 3,5-Me2-Bn), 6.86 (1H, s, 3,5-Me2-Bn), 6.45 (1H, d, J 3.2, CH2(furan-2-yl)), 6.37 (1H, dd, J 3.2 and 2.0, CH2(furan-2-yl)), 5.05 (2H, s, 3,5-Me2-Bn), 5.04 (1H, s, H-5), 5.01 (2H, s, CH2(furan-2-yl)), 4.89 (1H, brs, 6-NH2), 2.26 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.6, 153.6, 151.6, 148.9, 142.7, 137.8, 137.3, 129.0, 126.3, 111.2, 110.1, 78.0, 44.3, 39.2, 21.3; HRMS (ESI) Calcd for C18H19N3NaO3+ [M+Na]+: 348.13186. Found 348.13110; mp: 69.8–74.8°C.
6-Amino-3-(3,5-dimethylbenzyl)- 1-[(2-thiophenyl)methyl]uracil [6h]
Yield 79%; yellow crystal; 1H NMR (400 MHz, CDCl3): δ 7.26 (1H, m, CH2(thiophen-2-yl)), 7.01 (1H, d, J 3.6, CH2(thiophen-2-yl)), 7.00 (2H, s, 3,5-Me2-Bn), 6.94 (1H, dd, J 5.2 and 3.6, CH2(thiophen-2-yl)), 6.85 (1H, s, 3,5-Me2-Bn), 5.21 (2H, s, CH2(thiophen-2-yl)), 5.04 (2H, s, 3,5-Me2-Bn), 4.97 (1H, s, H-5), 4.82 (1H, brs, 6-NH2), 2.25 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.7, 153.6, 151.7, 137.9, 137.7, 137.2, 129.0, 127.0, 126.4, 126.4, 125.9, 79.4, 44.3, 41.6, 21.3; HRMS (ESI) Calcd for C18H19N3NaO2S+ [M+Na]+: 364.10902. Found 364.10866; mp: 72.3–78.5°C.
6-Amino-3-(3,5-dimethylbenzyl)- 1-(4-hydroxybenzyl)uracil [12]
Yield 95%; colorless oil; 1H NMR (400 MHz, CD3OD): δ 6.95 (2H, d, J 8.4, 4-HO-Bn), 6.76 (2H, s, 3,5-Me2-Bn), 6.72 (1H, s, 3,5-Me2-Bn), 6.62 (2H, d, J 8.4, 4-HO-Bn), 4.89 (2H, s, 3,5-Me2-Bn), 4.89 (2H, s, 4-HO-Bn), 4.86 (1H, s, H-5), 2.11 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CD3OD): δ 165.2, 158.2, 157.1, 153.5, 138.9, 138.8, 129.6, 129.0, 127.6, 126.3, 116.5, 77.1, 46.2, 45.0, 21.4; HRMS (ESI) Calcd for C20H21N3NaO3+ [M+Na]+: 374.14751. Found 374.14671.
6-Amino-1-(4-cyanobenzyl)- 3-(3,5-dimethylbenzyl)uracil [6e]
To a refluxing mixture of compound 5e (0.08 g, 0.20 mmol) and NaBH4 (0.008 g, 0.20 mmol) in THF (3.0 mL), MeOH (0.3 mL) was added over a period of 15 min. The mixture was the allowed to cool to room temperature, sat. NaHCO3 aq (5.0 mL) was added, and the solution was extracted with AcOEt, washed with saturated aqueous sodium chloride solution, dried with sodium sulfate, and then evaporated. The residual solution was purified by silica gel column chromatography (20% MeOH in AcOEt) to form a white crystal 6e (0.07 g, 0.19 mmol, 95%). 1H NMR (400 MHz, CDCl3): δ 7.67 (2H, d, J 8.0, 4-CN-Bn), 7.35 (2H, d, J 8.0, 4-CN-Bn), 7.04 (2H, s, 3,5-Me2-Bn), 6.89 (1H, s, 3,5-Me2-Bn), 5.18 (2H, s, 4-CN-Bn), 5.06 (1H, s, H-5), 5.05 (2H, s, 3,5-Me2-Bn), 4.27 (1H, brs, 6-NH2), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.4, 152.7, 151.9, 140.4, 137.9, 137.0, 133.1, 129.2, 127.0, 126.3, 118.1, 112.5, 80.1, 45.9, 44.4, 21.3; HRMS (ESI) Calcd for C21H20N4NaO2+ [M+Na]+: 383.14785. Found 383.14664; mp: 128.5–131.6°C.
General procedure for the synthesis of 7a,b
Compound 5a,b (0.20 mmol) was dissolved in the 3:1 mixture of dry MeOH (3.0 mL) and dry THF (1.0 mL), and NaBH4 (0.03 g, 0.92 mmol) and NiCl2· 6H2O (0.08 g, 0.32 mmol) were added to the solution, which was stirred for 30 min at 0°C. The mixture was extracted with AcOEt, washed with saturated aqueous sodium chloride solution, dried with sodium sulfate, and then evaporated. The residue was purified by silica gel column chromatography (20% MeOH in AcOEt) to form diamino derivative 7a,b.
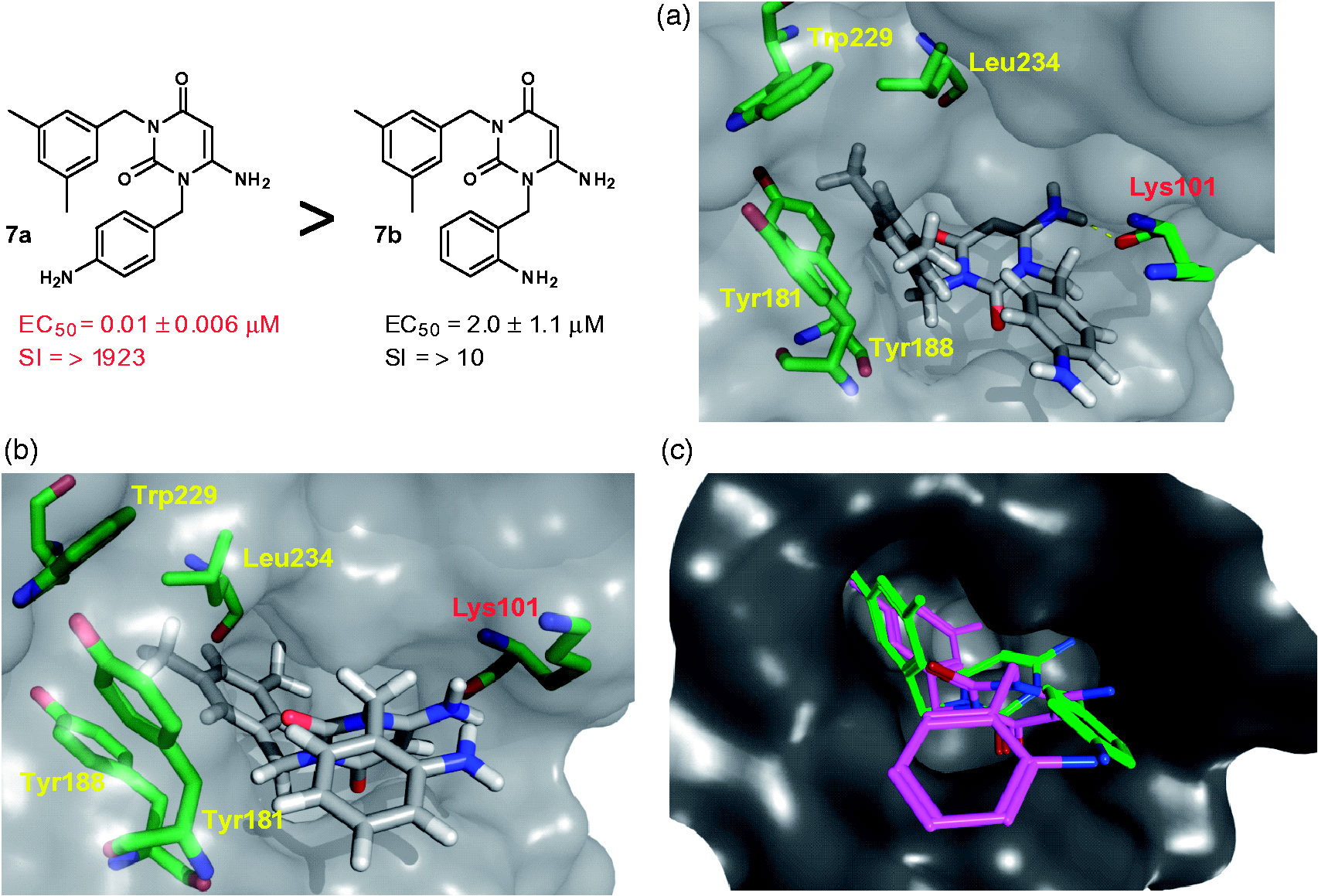
6-Amino-3-(3,5-dimethylbenzyl)- 1-(4-aminobenzyl)uracil [7a]
Yield 80%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.01 (2H, d, J 8.4, 4-NH2-Bn), 7.00 (2H, d, J 8.4, 4-NH2-Bn), 6.85 (2H, s, 3,5-Me2-Bn), 6.61 (1H, s, 3,5-Me2-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 4.97 (2H, s, 4-NH2-Bn), 4.91 (1H, s, H-5), 4.65 (2H, brs, 6-NH2), 3.76 (2H, brs, 4-NH2-Bn), 2.26 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 162.8, 154.2, 152.2, 146.5, 137.8, 137.5, 128.9, b127.7, 126.0, 124.5, 115.7, 79.0, 46.0, 44.3, 21.3; HRMS (ESI) Calcd for C20H22N4NaO2+ [M+Na]+: 373.16350. Found 373.16279; mp: 167.5–168.9°C.
6-Amino-3-(3,5-dimethylbenzyl)- 1-(2-aminobenzyl)uracil [7b]
Yield 80%; yellowish needle crystal; 1H NMR (400 MHz, CD3OD): δ 6.96 (1H, m, 2-NH2-Bn), 6.88 (1H, d, J 7.2, 2-NH2-Bn), 6.80 (2H, s, 3,5-Me2-Bn), 6.74 (1H, s, 3,5-Me2-Bn), 6.70 (1H, d, J 7.6, 2-NH2-Bn), 6.61 (1H, m, 2-NH2-Bn), 4.91 (2H, s, 2-NH2-Bn), 4.91 (2H, s, 3,5-Me2-Bn), 4.86 (1H, s, H-5), 2.14 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CD3OD): δ 165.1, 157.1, 135.7, 154.8, 138.9, 138.8, 129.8, 129.6, 128.0, 126.3, 122.7, 120.6, 118.7, 76.9, 45.1, 42.6, 21.4; HRMS (ESI) Calcd for C20H22N4NaO2+ [M+Na]+: 373.16350. Found 373.16246; mp: 157.3–162.2°C.
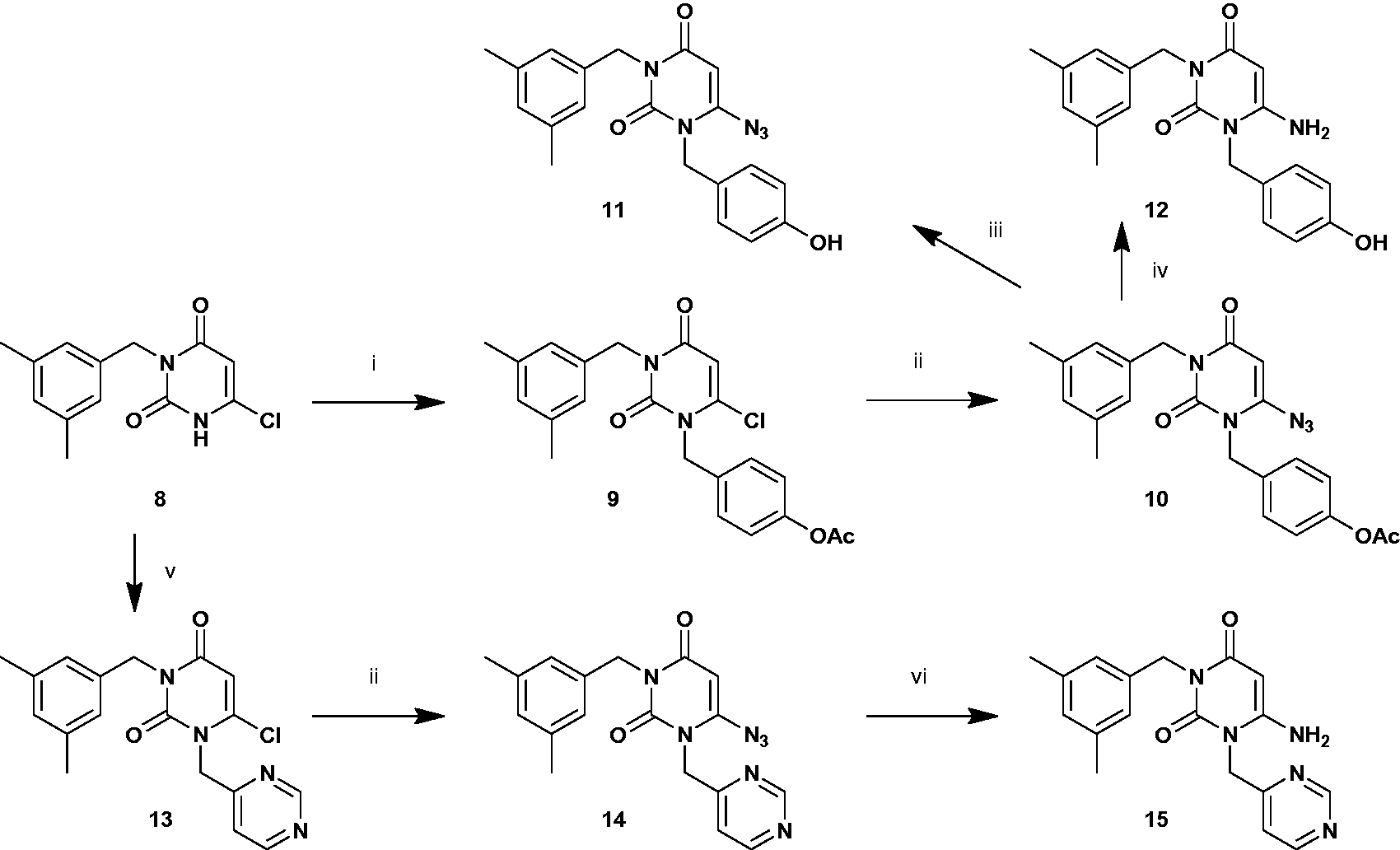
1-[4-(Acetoxy)benzyl]-6-chloro-3- (3,5-dimethylbenzyl)uracil [9]
A solution of compound 8 (0.26 g, 1.00 mmol), triphenylphosphine (0.34 g, 1.30 mmol), 4-(acetoxy)benzyl alcohol (0.28 g, 2.02 mmol), and TMAD (0.22 g, 1.30 mmol) in THF (8.5 mL) was stirred at 50°C. After 24 h stirring, the solution was filtered and concentrated to a small volume. The residual solution was purified by silica gel column chromatography (50% AcOEt in hexane) to form syrup 9 (0.26 g, 0.64 mmol, 64%). 1H NMR (400 MHz, CDCl3): δ 7.35 (2H, d, J 8.8, 4-AcO-Bn), 7.06 (2H, d, J 8.8, 4-AcO-Bn), 7.04 (2H, s, 3,5-Me2-Bn), 6.90 (1H, s, 3,5-Me2-Bn), 5.96 (1H, s, H-5), 5.24 (2H, s, 4-AcO-Bn), 5.04 (2H, s, 3,5-Me2-Bn), 2.29 (3H, s, 4-AcO-Bn), 2.28 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 169.3, 160.5, 151.2, 150.5, 145.6, 138.0, 136.1, 132.9, 129.5, 128.9, 126.5, 122.0, 102.7, 49.3, 44.9, 21.3, 21.1; HRMS (ESI) Calcd for C22H21ClN2NaO4+ [M+Na]+: 435.10821. Found 435.10700.
6-Azido-3-(3,5-dimethylbenzyl)- 1-(4-hydroxybenzyl)uracil [11]
Compound 10 (0.06 g, 0.15 mmol) was dissolved in dry MeOH (5.0 mL), and K2CO3 (0.42 g, 3.00 mmol) was added to the solution, which was stirred for 30 min at room temperature. The mixture was extracted with AcOEt, washed with saturated aqueous sodium chloride solution, dried with sodium sulfate, and then evaporated. The residue was purified by AcOEt to form a red-brown crystal 11 (0.04 g, 0.12 mmol, 78%). 1H NMR (400 MHz, CDCl3): δ 7.17 (2H, d, J 8.8, 4-HO-Bn), 7.02 (2H, s, 3,5-Me2-Bn), 6.86 (1H, s, 3,5-Me2-Bn), 6.74 (2H, d, J 8.8, 4-HO-Bn), 6.16 (1H, brs, 4-HO-Bn), 5.14 (1H, s, H-5), 5.05 (2H, s, 3,5-Me2-Bn), 4.97 (2H, s, 4-HO-Bn), 2.25 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 163.3, 161.3, 155.8, 151.2, 137.9, 136.8, 129.7, 129.2, 126.2, 115.5, 78.2, 57.0, 45.0, 44.4, 21.3; HRMS (ESI) Calcd for C20H19N5NaO3+ [M+Na]+: 400.13801. Found 400.11876; mp: 79.6–83.8°C.
6-Chloro-3-(3,5-dimethylbenzyl)- 1-(4-pyrimidinylmethyl)uracil [13]
A solution of compound 8 (0.27 g, 1.00 mmol), 4-(chloromethyl)pyrimidine (0.22 g, 1.72 mmol), K2CO3 (0.24 g, 1.72 mmol), and NaI (0.03 g, 0.20 mmol) in DMF (4.0 mL) was stirred at room temperature. After 48 h stirring, the solution was extracted with AcOEt, washed with saturated aqueous sodium chloride solution, dried with sodium sulfate, and then evaporated. The residual solution was purified by silica gel column chromatography (50% AcOEt in hexane) to form a brown crystal 13 (0.16 g, 0.44 mmol, 44%). 1H NMR (400 MHz, CDCl3): δ 9.15 (1H, s, 4-pyrimidinylmethyl), 8.70 (1H, d, J 5.2, 4-pyrimidinylmethyl), 7.22 (1H, d, J 5.2, 4-pyrimidinylmethyl), 7.01 (2H, s, 3,5-Me2-Bn), 6.89 (1H, s, 3,5-Me2-Bn), 6.03 (1H, s, H-5), 5.34 (2H, s, 4-pyrimidinylmethyl), 5.04 (2H, s, 3,5-Me2-Bn), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 163.2, 160.4, 159.0, 157.3, 151.1, 154.6, 138.0, 135.9, 129.4, 126.3, 118.3, 102.7, 49.9, 44.9, 21.3; HRMS (ESI) Calcd for C18H17ClN4NaO2+ [M+Na]+: 379.09322. Found 379.09275; mp: 97.1–98.2°C.
6-Amino-3-(3,5-dimethylbenzyl)- 1-(4-pyrimidinylmethyl)uracil [15]
To a refluxing mixture of compound 14 (0.07 g, 0.21 mmol) and NaBH4 (0.008 g, 0.21 mmol) in THF (3.0 mL), MeOH (0.3 mL) was added over a period of 15 min. The mixture was then allowed to cool to room temperature, sat. NaHCO3 aq (5.0 mL) was added, and the solution was extracted with AcOEt, washed with saturated aqueous sodium chloride solution, dried with sodium sulfate, and then evaporated. The residual solution was purified by silica gel column chromatography (20% MeOH in AcOEt) to form a yellowish crystal 15 (0.04 g, 0.12 mmol, 58%). 1H NMR (400 MHz, CDCl3): δ 9.14 (1H, d, J 0.8, 4-pyrimidinylmethyl), 8.78 (1H, d, J 5.2, 4-pyrimidinylmethyl), 7.59 (1H, dd, J 5.2 and 0.8, 4-pyrimidinylmethyl), 7.00 (2H, s, 3,5-Me2-Bn), 6.85 (1H, s, 3,5-Me2-Bn), 5.86 (1H, brs, 6-NH2), 5.08 (1H, s, H-5), 5.07 (2H, s, 3,5-Me2-Bn), 4.99 (2H, s, 4-pyrimidinylmethyl), 2.25 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 163.2, 162.7, 158.7, 158.4, 154.6, 151.9, 137.9, 137.2, 129.1, 126.1, 121.6, 79.9, 47.8, 44.3, 21.3; HRMS (ESI) Calcd for C18H19N5NaO2+ [M+Na]+: 360.14310. Found 360.14230; mp: 85.8–89.0°C.
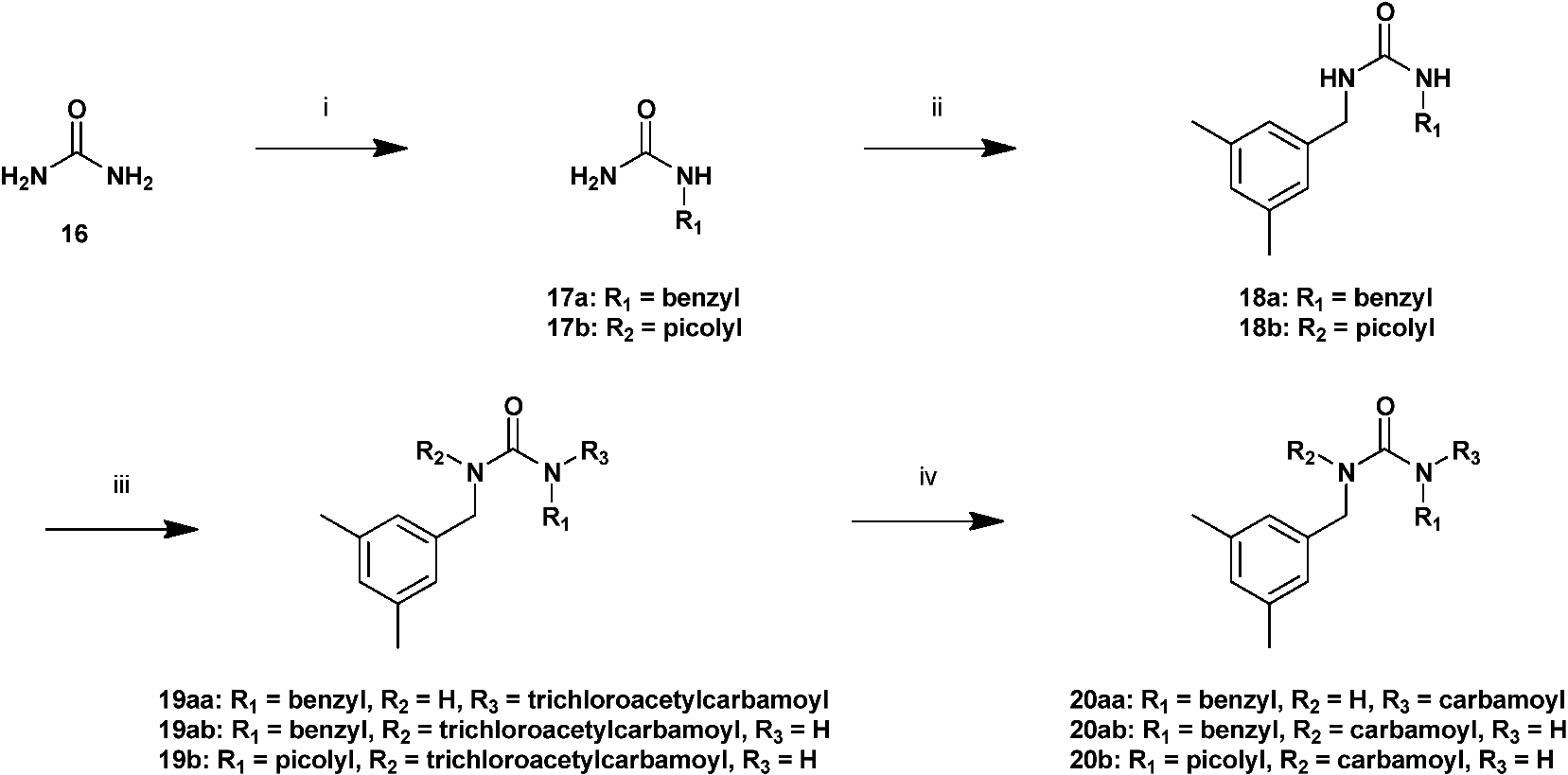
General procedure for the synthesis of 17a and 17b
A mixture of urea 16 (4.32 g, 72.0 mmol) and benzylamine or 4-aminomethylpyridine (60.0 mmol) in H2O (30.0 mL) was stirred at 130 °C. After 12 h stirring, the solution was filtered and concentrated to a small volume. The residual solution was purified by silica gel column chromatography to form a benzylated or picolylated product.
N-Benzylurea [17a]
Yield 61%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 7.19–7.32 (5H, m, Ph), 6.39 (1H, t, J 6.0, NH-CH2-Ph), 5.50 (2H, s, NH2), 4.16 (2H, d, J 6.0, NH-CH2-Ph);
13
C NMR (100 MHz, DMSO-d6): δ 158.7, 140.9, 128.2, 127.0, 126.5, 42.8; HRMS (ESI) Calcd for C8H10N2NaO [M+Na]+: 173.06853. Found 173.06964; mp 146.2–149.2°C.
N-(4-picolyl)urea [17b]
Yield 74%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 8.47–8.48 (2H, m, picolyl), 7.22–7.23 (2H, m, picolyl), 6.53 (1H, t, J 6.0, NH-CH2-), 5.63 (2H, s, NH2), 4.19 (2H, d, J 6.0, NH-CH2-);
13
C NMR (100 MHz, DMSO-d6): δ 158.7, 150.1, 149.4, 121.9, 41.8; HRMS (ESI) Calcd for C7H9N3NaO [M+Na]+: 174.06378. Found 174.06470; mp 187.0–187.2°C.
General procedure for the synthesis of 18a and 18b
A mixture of 3,5-dimethylbenzaldehyde (0.40 mL, 3.0 mmol), benzylurea 17a or 17b (1.5 mmol), and titanium (IV) isopropoxide (0.50 mL, 1.7 mmol) were slurred in THF (2.0 mL). This slurry was stirred at 50°C under nitrogen. After 2 h stirring, the solution was then cooled to 0°C, and NaBH4 (0.06 g, 1.5 mmol) was added. The ice bath was removed, and the resulting slurry was allowed to stir for 30 min. The slurry was again cooled to 0°C and quenched by dropwise addition of 1 N HCl aq (6.0 mL), the solution was extracted with AcOEt, washed with saturated aqueous sodium chloride solution, dried with sodium sulfate, and then evaporated. The residual solution was purified by silica gel column chromatography to form a crystal 18a or 18b (0.32 g, 1.18 mmol, 79%).
N-benzyl-N′-(3,5-dimethylbenzyl)urea [18a]
Yield 79%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 7.20–7.32 (5H, m, Ph), 6.84 (3H, s, 3,5-Me2-Bn), 6.40 (1H, t, J 6.0, NH-CH2-Ph), 6.37 (1H, t, J 6.0, NH-CH2-Ph), 4.22 (2H, d, J 6.0, NH-CH2-Ph), 4.16 (2H, d, J 6.0, NH-CH2-Ph), 2.23 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 158.1, 141.0, 140.7, 137.1, 128.2, 127.9, 126.9, 126.5, 124.7, 42.9, 42.9, 20.9; HRMS (ESI) Calcd for C17H20N2NaO [M+Na]+: 291.14678. Found 291.14575; mp 146.0–147.6°C.
N′-3,5-dimethyl-N-(4-picolyl)urea [18b]
Yield 20%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 8.47–8.48 (2 H, m, picolyl), 7.22–7.24 (2 H, m, picolyl), 6.84 (3 H, s, 3,5-Me2-Bn), 6.50–6.56 (2 H, m, NH),4.25 (2 H, d, J 6.4, NH-CH2), 4.15 (2 H, d, J 6.0, NH-CH2), 2.24 (6 H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 158.04, 150.16, 149.35, 140.57, 137.08, 127.92, 124.73, 121.86, 42.91, 41.99, 20.90; HRMS (ESI) Calcd for C16H19N3NaO2 [M+Na]+: 292.14203. Found 292.14097; mp 138.1–138.8°C.(
N-benzyl-N′-3,5-dimethylbenzyl-N-trichloroacetylcarbamoylurea [19aa] and N-benzyl-N′-3,5-dimethylbenzyl-N′-trichloroacetylcarbamoylurea [19ab]
To a solution of compound 18a (0.13 g, 0.5 mmol) in THF (5.0 mL), trichloroacetyl isocyanate (0.24 mL, 2.0 mmol) was added dropwise. After 1 h stirring, the solution was filtered and concentrated to a small volume. The residual solution was purified by silica gel column chromatography (50% t-butyl methyl ether in CH2Cl) to form milky white oils 19aa (0.07 g, 0.16 mmol, 32%) and 19ab (0.09 g, 0.19 mmol, 38%), respectively.
Compound 19aa
1H NMR (400 MHz, CDCl3): δ 7.18–7.39 (5H, m, Ph), 6.85 (1H, s, 3,5-Me2-Bn), 6.82 (2H, s, 3,5-Me2-Bn), 5.11 (2H, s, N-CH2-Ph), 4.28 (2H, d, J 4.8, NH-CH2-Ph), 2.16 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 161.0, 157.1, 152.1, 138.3, 138.1, 137.1, 128.5, 128.2, 127.3, 126.2, 124.6, 92.2, 44.9, 43.5, 20.9.
Compound 19ab
1H NMR (400 MHz, CDCl3): δ 6.95–7.28 (5H, m, Ph), 6.94 (1H, s, 3,5-Me2-Bn), 6.79 (2H, s, 3,5-Me2-Bn), 4.91 (2H, s, N-CH2-Ph), 4.34 (2H, d, J 5.2, NH-CH2-Ph), 2.23 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 156.5, 151.2, 139.5, 136.2, 134.9, 130.3, 128.9, 128.0, 124.4, 92.2, 47.0, 45.1, 21.2.
N′-3,5-dimethylbenzyl-N′-trichloroacetylcarbamoyl-N-(4-picolyl)urea [19b]
To a solution of compound 18b (0.29 g, 1.06 mmol) in THF (15.0 mL), trichloroacetyl isocyanate (0.15 mL, 1.27 mmol) was added dropwise. After 10 min stirring, the solution was filtered and concentrated to a small volume. The residual solution was purified by silica gel column chromatography (20% MeOH in AcOEt) to form yellowish crystal 19b (0.34 g, 0.74 mmol, 70%). 1H NMR (400 MHz, DMSO-d6): δ 9.03 (1H, t, J 6.0, NH-CH2), 8.47–8.48 (2H, m, picolyl), 7.25–7.26 (2H, m, picolyl), 6.79–6.88 (3H, m, 3,5-Me2-Bn), 4.89 (2H, s, N-CH2), 4.39 (2H, d, J 6.0, NH-CH2), 2.20 (6H, s, 3,5-Me2-Bn); mp 186.1–186.9°C.
General procedure for the synthesis of 20aa, 20ab, and 20b
A mixture of urea 19aa, 19ab, or 19b (0.046 mmol) and silica gel (0.5 g) in MeOH 5.0 mL) was stirred at 50°C. After 18 h stirring, the solution was filtered and concentrated to a small volume. The residual solution was purified by silica gel column chromatography to form 20aa, 20ab, or 20b (6.54 g, 43.6 mmol, 61%).
N-benzyl-N-carbamoyl-N′-(3,5-dimethylbenzyl) urea [20aa]
Yield 74%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.26–7.39 (5H, m, Ph), 6.88 (1H, s, 3,5-Me2-Bn), 6.75 (2H, s, 3,5-Me2-Bn), 5.02 (2H, s, N-CH2-Ph), 4.36 (2H, d, J 5.2, NH-CH2-Ph), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 157.5, 156.1, 138.2, 137.9, 136.7, 129.3, 129.0, 128.0, 126.3, 125.0, 47.0, 44.7, 21.2; HRMS (ESI) Calcd for C18H21N3NaO2 [M+Na]+: 334.15260. Found 334.15086; mp 123.5–124.6°C.
N-benzyl-N′-carbamoyl-N′-3,5-dimethylbenzylurea [20ab]
Yield 83%; white crystal; 1H NMR (400 MHz, CDCl3): δ 7.17–7.32 (5H, m, Ph), 6.91 (1H, s, 3,5-Me2-Bn), 6.84 (2H, s, 3,5-Me2-Bn), 4.93 (2H, s, N-CH2-Ph), 4.44 (2H, d, J 5.2, NH-CH2-Ph), 2.27 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, CDCl3): δ 157.8, 156.6, 139.0, 138.2, 136.6, 129.7, 128.6, 127.3, 127.3, 124.0, 46.9, 44.7, 21.3; HRMS (ESI) Calcd for C18H21N3NaO2 [M+Na]+: 334.15260. Found 334.15074; mp 118.1–119.5°C.
N′-carbamoyl-N′-3,5-dimethylbenzyl-N- (4-picolyl)urea [20b]
Yield 81%; white crystal; 1H NMR (400 MHz, DMSO-d6): δ 8.96 (1H, t, J 5.6, NH-CH2), 8.37–8.38 (2H, m, picolyl), 7.22 (2H, s, NH2), 7.06–7.07 (2H, m, picolyl), 6.81 (1H, s, 3,5-Me2-Bn), 6.71 (2H, s, 3,5-Me2-Bn), 4.82 (2H, s, N-CH2), 4.29 (2H, d, J 5.6, NH-CH2), 2.17 (6H, s, 3,5-Me2-Bn);
13
C NMR (100 MHz, DMSO-d6): δ 157.0, 156.1, 149.4, 148.8, 138.1, 137.2, 128.1, 123.9, 121.8, 44.9, 42.5, 20.9; HRMS (ESI) Calcd for C17H20N4NaO2 [M+Na]+: 335.14785. Found 335.14757; mp 151.2–153.3°C.