Abstract
Introduction and importance: Even though insulinoma is the most frequent neuroendocrine tumor, it represents only 2% of pancreatic 2% of all pancreatic neoplasms. Diagnosis is relatively simple, and surgery after accurate determination of the tumors location within the pancreas is the cornerstone of its treatment. Case presentation: We herein report 4 patients undergoing various surgeries for benign secreting insulinomas, after extensive radiological and endoscopic exploration. Clinical discussion: Diagnosis is relatively simple relying on clinical and biological criteria, it must be followed by an extensive and accurate preoperative determination of the tumors localization. The laparoscopic tumoral enucleation is the treatment of choice for small isolated tumors, but open surgery still has its indications. Conclusion: Pancreatic insulinoma is a rare neuroendocrine tumor that can be life-threatening due to hypoglycemic manifestations. The diagnosis is based on clinical and biological criteria. echo endoscopy and to a lesser extent radiological exploration can precisely determine the tumors location. Laparoscopic surgical enucleation of the tumor remains the preferred curative treatment.
Introduction
Digestive neuroendocrine tumors are a large group of tumors that originate from cells of the diffuse endocrine system, the most common of which are insulinomas. 1 The vast majority of insulinomas originate in the beta cells of Langerhans and are manifested by excessive and rapid insulin production, the half-life of which is no longer modulated by blood glucose levels, 2 thus constituting an unusual but usually curable cause of hypoglycemia. It is a rare tumor that represents 2% of all pancreatic neoplasms, with an annual incidence of 1 to 2 per 100,000 population. 3
The diagnosis of insulinoma is based on clinical and biological criteria, as outlined by Whipple and Frantz. These criteria include a triad consisting of: symptoms of hypoglycemia, plasma glucose levels below 50 mg/dl, and symptom resolution following glucose administration. 4 Topographic assessment using a wide array of imaging and endoscopic technics is mandatory, for it guides the therapeutic approach.5,6
Most insulinomas can be successfully treated with surgery. The choice of surgical procedure depends on the size and location of the tumor
We report 4 cases of insulinoma with neurological manifestations admitted and surgically managed between 2020 and 2023.
Presentation of Cases
This study encompassed all patients who underwent surgery for pancreatic insulinoma at the Digestive Surgical Department -C- of University Hospital Ibn Sina, Rabat, Morocco, over a 4-year period from January 2020 to December 2023.
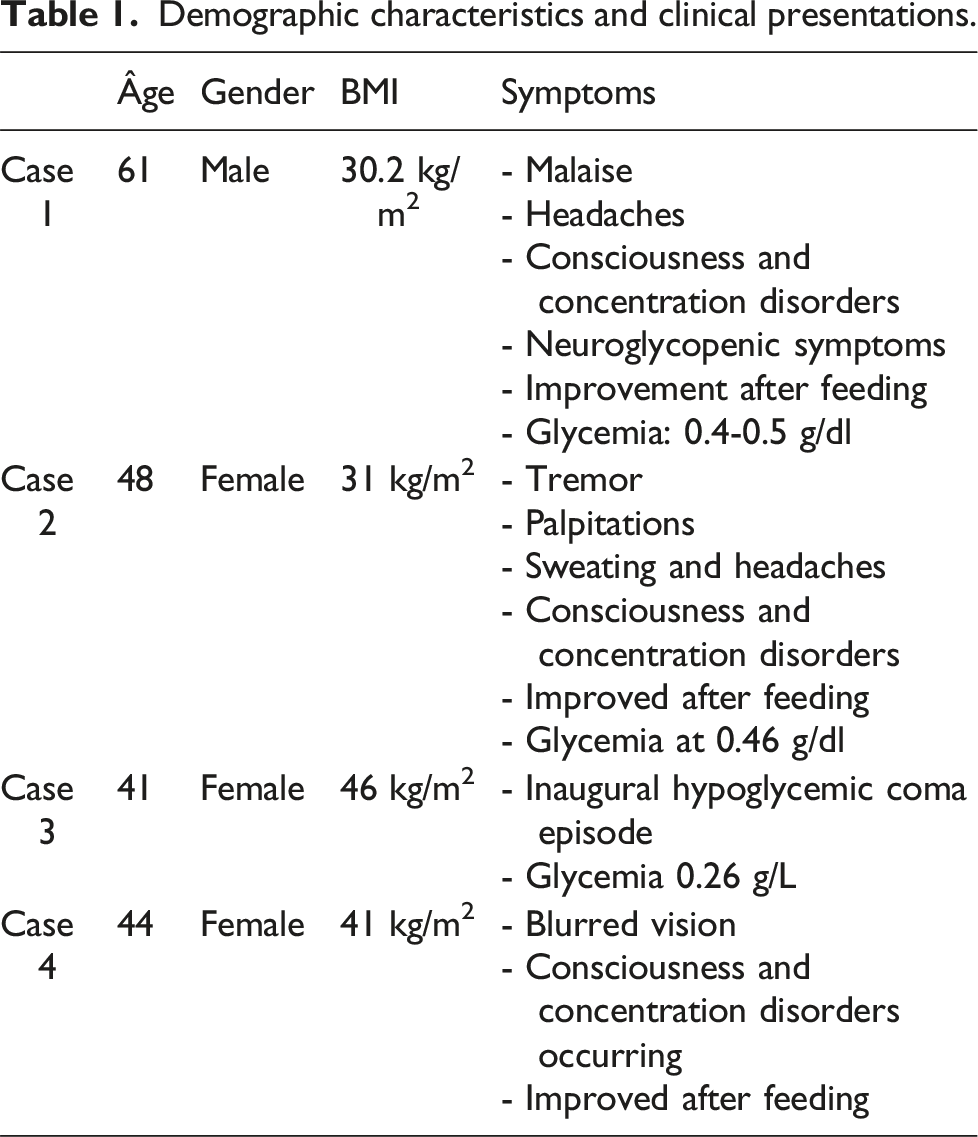
Demographic characteristics and clinical presentations.
Radiological examinations for each case and their findings.

Surgical treatment with postoperative course and histopathological outcomes.

Clinical Presentation
Our study included four patients, with a mean age of 48.5 years (range: 41–61 years). The sex-ratio was 0.3 (3 women, 1 man). The median interval between onset of symptoms and diagnosis of 3.5 years (range 1–7 years).
Three patients presented chronic neuroglycopenic symptoms such as confusion, personality changes, blurred vision, or loss of consciousness and one patient presented an inaugural hypoglycemic coma episode. The most common adrenergic symptoms were asthenia, headaches, sweating, and tachycardia which were all alleviated by oral glucose administration. Interestingly, all patients reported weight gain, with an average body mass index (BMI) of 34.3 kg/m2.
The range of plasma glucose at the time of hypoglycemia was 0.3–0.6 g/dl.
Diagnosis
Abdominal computed tomography (CT) scans were performed on all patients and none found any specific lesion. All cases presented a small (>2 cm) often difficult to characterize nodular formation justifying MRI and endoscopic follow-up, which allowed for better characterization of the lesion and to suggest the diagnosis. Two were corporeal and two were cephalic. For one patient (case 3), radiological and echo endoscopic explorations were ineffective, and the diagnosis was based on clinical criteria and extensive surgical resection was necessary.
It showed a non-specific lesion in the pancreas in three patients (75%). Endoscopic ultrasonography was performed in two patients (50%). One patient underwent a magnetic resonance imaging (MRI).
No Preoperative tissue diagnosis was performed because of equipment unavailability. (Figure 1) Abdominal CT scan showing a nodular formation adjacent to the tail of the pancreas (17 × 15 mm), favoring insulinoma (red arrow indicating the location).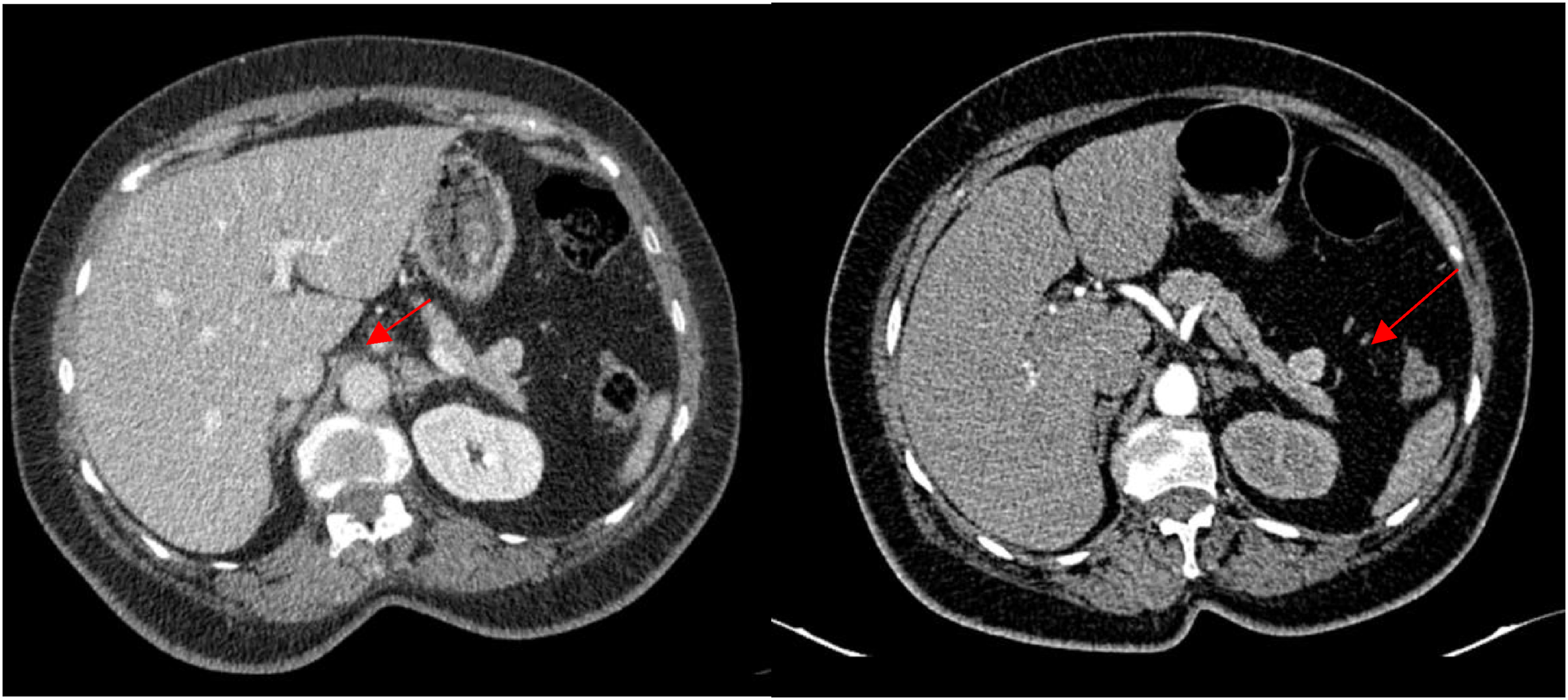
Surgical Management
All patients underwent surgical interventions, with two patients (50%) operated on via median laparotomy, and two patients (50%) operated on laparoscopically.
The choice of the surgical procedure depended on the tumor location, its relationship to the pancreatic vessels and ducts and the degree of diagnosis certainty. Laparoscopic enucleation was performed for two patients with a strong diagnosis certainty while two patients underwent open surgery for additional surgical exploration with digital palpation of the organ resulting in one simple tumor enucleation and one Whipple cephalic duodeno-pancreatectomy with Child’s continuity reconstruction, due to the presence of a cephalic tumor (20 mm) with suspicious centimetric lymphadenopathies at the hepatic pedicle suggesting a malignant lesion. (Figure 2) Surgical specimen of the tumor after excision.
Postoperative Course and Follow-Up
The immediate postoperative courses were uneventful, with no occurrences of postoperative pancreatic fistulas, bleeding, or sepsis. However, one patient developed type 1 diabetes postoperatively with a median hospital stay of 3 days. The 6 month follow-up was favorable, characterized by resolution of the hypoglycemic episodes. Notably, no mortality was observed in this study.
Anatomopathological examination with immunohistochemistry of the surgical specimen confirmed the neuroendocrine nature of the tumor in all our patients.
Discussion
Insulinomas are the most common type of pancreatic endocrine tumor 7 occurring at any age and with a slight female predominance. 8 In 90% of cases, these neoplasms are benign and isolated, developing predominantly on the cephalic and corporeal portions of the pancreas. 9 However, in 5%–10% of cases, they may present as multiple neoplasms and can even be associated with genetic syndromes, mainly Multiple Endocrine Neoplasia type 1 (MEN-1) rarely in von Hippel–Lindau disease, neurofibromatosis 1 and tuberous sclerosis. 10
The non-specificity of the clinical signs of insulinomas explains the often-delayed diagnosis, up to 5 years in 20% of patients.10,11 These signs are manifestations of the hypoglycemia resulting from endogenous hyperinsulinism. 3 These symptoms most commonly occur on an empty stomach and away from meals, and may also occur during exercise, 12 however, in some cases, they can develop independently of food intake or even postprandially 10 and can be intermittent first becoming more frequent and persistent as the tumor grows. 13 It is important to note that severity of the symptoms does not correlate to the tumor’s size. 13
Clinical diagnosis of insulinoma is based on the pathognomonic “Whipple’s triad”: low blood glucose (<50 mg/dl) at the time of symptoms, occurrence of neuroglycopenic signs, and resolution of symptoms after administration of glucose and correction of hypoglycemia. 3 After the triad is documented, biological explorations focus on detecting hypoglycemia, either spontaneously or during a 72-h fasting test in uncertain cases, in conjunction with elevated levels of insulin and C-peptide. 8
Sensitivity and specificity of the diagnostic techniques. 35 Contrast enhanced ultrasound is a low-cost, well-spread and non-invasive technique that exploits insulinomas’ high vascularization to detect the tumor (sensitivity of 89.2% and a specificity of 86.5%).10,15 It can also be used intraoperatively combined with digital palpation of the pancreas to reach 95% sensitivity 10 at the cost of being more invasive.
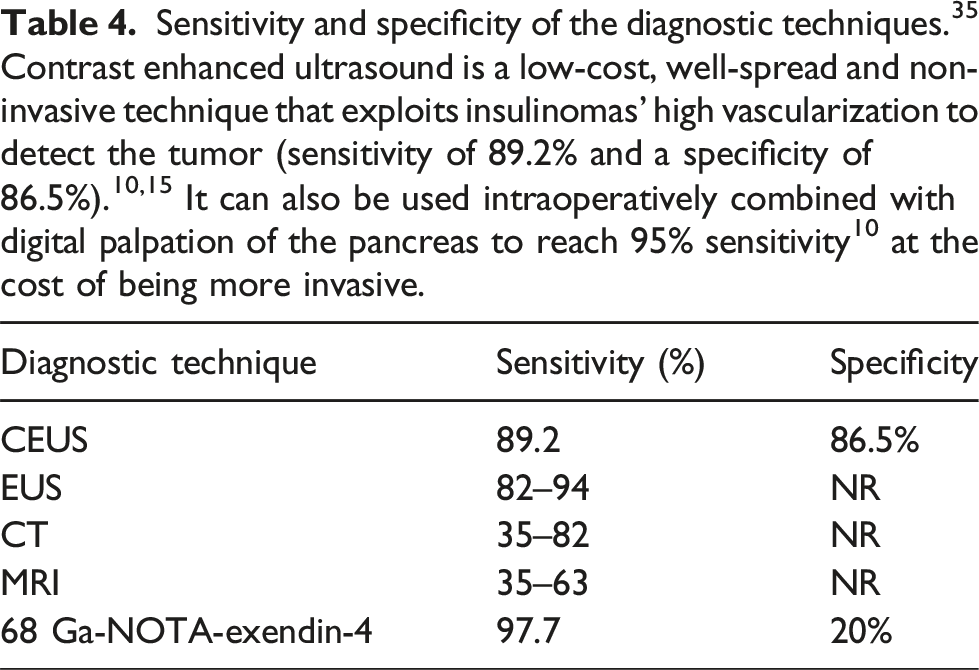
Failure to obtain diagnosis through CT or MRI should lead to further assessment using endoscopic ultrasound (EUS). 19 EUS is the most accurate diagnostic tool for insulinoma, detecting even tumors as small as 5 mm. 20 Its detection rate for insulinomas alone is 94%, increasing to nearly 100% when combined with computed tomography, especially for cephalic and corporeal tumors, 21 and can also define the tumor’s relationship with ductal and vascular structures, 22 analyze adjacent lymph nodes 23 allowing for biopsy and cytology through fine needle aspiration,10,24 and marking of the lesion with a marking agent.10,25 Reducing the need to perform blind distal pancreatectomy from 25% to about 0%. 26 However, it is an invasive and highly operator-dependent tool, with poor sensitivity for caudal pancreatic tumors. 10 PET scans have not been useful for diagnosis due to the low proliferation rate of insulinomas. 10 However, a recent study documented the 68-Ga-NOTA-exedin-4 scan as a promising technique, localizing insulinomas in nine out of ten patients. 27
In practice, the positive fasting test and endoscopic-ultrasonography are enough for the diagnosis and treatment decision. 24
Once an insulinoma has been diagnosed, the therapeutic goal is twofold: to control hormonal secretions and to remove the tumor. 28 The medical treatment aims to control the symptoms and control the tumoral growth and therefore is reserved to patients awaiting surgery or not eligible for it, as well as malignant metastatic presentations. 29 Initial managements involve dietary measures that can be sufficient to achieve symptom control, if these measures prove insufficient, glucose intravenous infusion may be necessary. 30 Diazoxide (50–600 mg/d) is a nondiuretic benzothiazide analog that directly acts on Langerhans beta cells, inhibiting insulin secretion and stimulating glycogenolysis. 28 It is considered to be the primary treatment that controls the symptoms in 50%–60% of cases. 15 Other pharmacological agents, including Verapamil, beta-blockers, phenytoin, and glucocorticoids, have been proposed for their effects on insulin production. However, their efficacy has not been demonstrated. 10 Somatostatin analogues (octreotide, Ianreotide) can be used in case of diazoxide failure to control symptoms (30%–50% of cases) or as primary treatment for their antiproliferative effects to control the disease when surgery isn’t possible. 31
Surgery remains the cornerstone of a curative treatment of insulinomas. 19 Given the typically small, benign, and solitary nature of insulinomas they are particularly well-suited for a laparoscopic approach, especially in obese patients. 19 The Laparoscopic management of insulinomas was first reported by Gagner et al. in 1996 and has since been adopted by numerous centers. 19 This technique currently demonstrates a resection success rate between 70 and 100%, with a cure rate ranging from 77 to 100%, 4 and a postoperative overall survival rate of 95% at 5 years and 88% at 10 years. 19
However, a mini-invasive approach is not recommended before accurate preoperative determination of the tumor localization because of, firstly, the lack of intraoperative tactile feedback, secondly, the trocar and patient positioning relies on this tumor localization within the pancreas, 19 therefore open surgery with intraoperative ultrasound is required when insulinomas are not localised preoperatively. 32 The laparoscopic approach must also not be considered in presence of MEN-1 or malignant presentations which requires a more extensive resection. 19
Tumor enucleation with optimal preservation of the pancreatic parenchyma (reducing the risk of pancreatic endocrine or exocrine insufficiency) is the procedure of choice. 33 It is indicated for benign, single, superficial tumors that are small (<2 cm) and not adherent to the pancreatic ducts. 3 Pancreatic excision is required for large, locally advanced tumors that are suspected of malignancy or that come into intimate contact with the pancreatic and bile ducts and vessels. 16 Most often, this involves caudal or corporeo-caudal pancreatectomy, cephalic duodeno-pancreatectomy, middle pancreatectomy, 34 or even total duodeno-pancreatectomy. 8
For patients unfit for surgery or refusing it, Endoscopic ultrasound-guided radiofrequency ablation of insulinomas has emerged as a safe and effective technic, that enables selective and precise ablation of insulinomas while minimizing damage to surrounding tissues and avoiding the postoperative complications associated with pancreatic surgeries. 35 This technic is indicated for small (<2 cm), solitary and benign (grade 1 or 2) insulinomas with no association with genetic syndromes and a minimal 1 mm distance from main pancreatic duct. 35
After a curative resection of a pancreatic insulinoma, it is recommended to have a follow-up within 3–6 months and then every 6–12 months for at least 7 years. 36
Conclusion
Pancreatic insulinoma is a rare neuroendocrine tumor that can be life-threatening due to hypoglycemic manifestations. The non-specificity of clinical signs often leads to delayed diagnosis. The diagnosis is relatively easy based on clinical and biological criteria.
Radiological screening and echoendoscopy is used to diagnose the tumor’s location using a variety of invasive and non-invasive techniques. Laparoscopic surgical resection of the tumor remains the preferred curative treatment.37–51
Footnotes
Author contributions
All authors contributed to the conduct of this work. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
