Abstract
Extraskeletal osteosarcoma (ESOS) is a very rare entity among renal malignancies. There are few reports of renal ESOS in the database. Renal ESOS was found to have a high rate of local recurrence and distant metastasis. In most reports, the overall survival of patients was less than 1 year. We present a 51-year-old man who presented with gross hematuria and a clinical diagnosis of a staghorn stone in the left kidney. He underwent radical nephrectomy. The pathologic diagnosis of osteosarcoma was evident.
Introduction
Sarcomas account for only 1-2 percent of renal malignancies. 1 Extraskeletal osteosarcoma (ESOS) of the kidney is a rare sarcoma, accounting for less than four percent of all osteosarcomas according to previous studies. 2 As we noted in the literature review, there have been 36 cases of ESOS of the kidney since 1936. The median survival time for renal ESOS is 15 months. 3 The role of adjuvant treatments after surgery, such as chemotherapy and radiotherapy, has not been clarified so far. In this article, we present a new case of renal ESOS whose story is thought-provoking.
Case History
A 51-year-old man presented a year ago with complaints of gross hematuria. He had undergone open surgery on his left kidney 30 years ago for a kidney stone. Noncontrast-enhanced computed tomography (CT) of the abdomen revealed a huge calcified mass like lesion similar to staghorn stone, measuring 81 × 40 mm in the inferior calyx and pelvis of the left kidney. The cortical thickness of the left kidney was reduced. There was also moderate hydronephrosis on the left (Figure 1). Renal function tests revealed a creatinine clearance of 62 mL/min, and the left kidney had negligible function on dimercaptosuccinic acid (DSMA) scan (Figure 2). The right kidney and other genitourinary organs were normal in all examinations. In laboratory tests, alkaline phosphatase was 436 (with an upper normal limit of 306) and C-reactive protein (CRP) was 3+; other tests such as dipstick urinalysis, electrolytes and liver function tests were normal. Non-contrast-enhanced computed tomography (CT) scan of the abdomen. A huge calcified mass like lesion similar to staghorn stone. DSMA scan shows non-functional left kidney.

Because the left kidney was nonfunctional, the patient underwent left nephrectomy. In macroscopic view, the total weight of the specimen was 650 g with dimensions of 145 × 100 × 75 mm. The thickness of the left renal parenchyma was 15 mm in most parts. The renal pelvis contained hemorrhagic fluid. The pilocalyceal space contained a massive irregular mass of 100 × 65 × 60 mm extending to the renal parenchyma and proximal of the ureter, with a hard bony consistency and small areas of soft consistency and hemorrhage.
Microscopic examination revealed that the renal parenchyma was infiltrated by a hypercellular mesenchymal neoplasm consisting of sheets of atypical osteoblasts with thin, lace-like osteoid, broad, irregular trabeculae, and cartilage. No necrosis was noted (Figure 3). The tumor extended to the renal parenchyma, perinephric adipose tissue, and ureter, and invasion of the small vessels was observed. No lymphatic invasion was noted. Numerous fine sections and careful microscopic examination of the specimen revealed no evidence of renal or urothelial cell carcinoma. The specimen was positive for CD10 and negative for PAX8 and cytokeratins (CK) (Figure 4). Morphologic and immunohistochemical (IHC) findings were in favor of primary renal high-grade chondroblastic osteosarcoma. H&E study. (a): Kidney parenchyma infiltrated by a sarcomatous neoplasm. (b): Sarcomatous tumor cells with densely eosinophilic cytoplasm, lace-like osteoid, abd broad, irregular trabeculae. (c): Thin lace-like osteoid. IHC study. (a): positive immunostaining for CD10. (b): Negative immunnostaining for PAX8. (c): Negative immunnostaining for CK.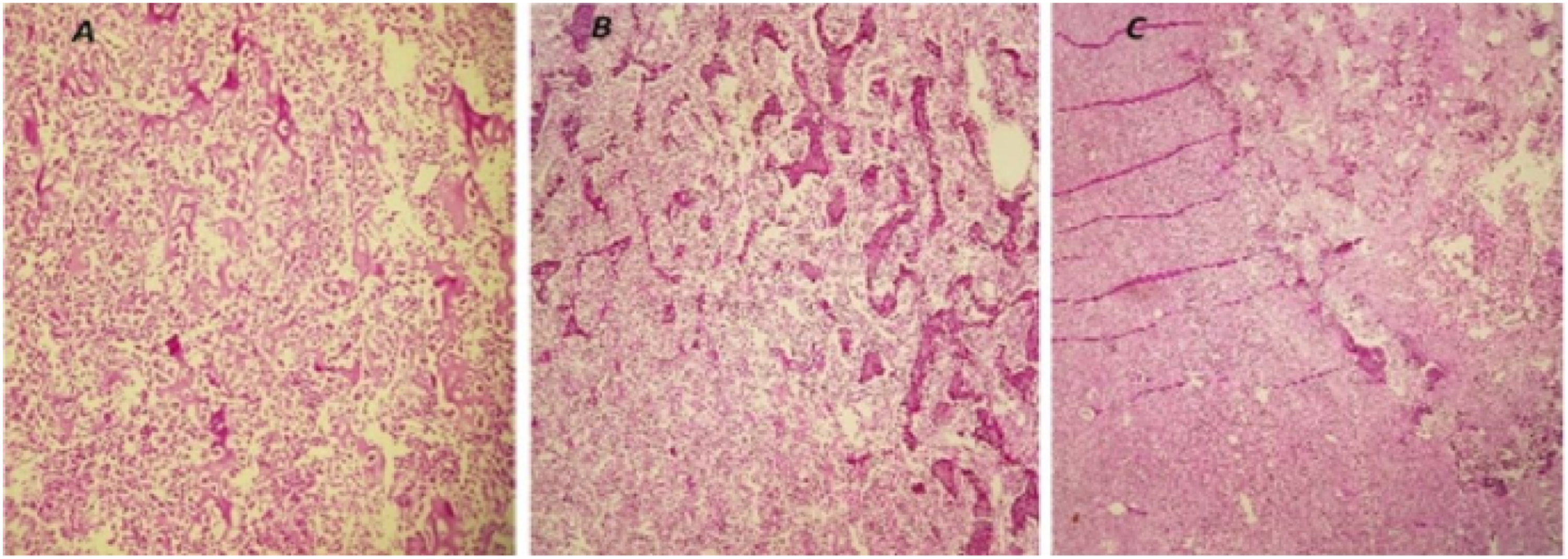

Imaging studies including a thoracic and abdominopelvic CT with contrast and a whole-body bone scan ruled out distant metastasis. The patient had no follow-up and returned after 4 months with a bulging and heaviness in his left flank. Imaging showed a 131 × 96 × 139 mm heterogeneous hypodense mass anterior to the spleen with enhancement on post-contrast CT. The fat plane between the mass and the abdominal wall muscles had disappeared. This finding and abdominal wall muscle stranding indicated tumor invasion. The fat plane between the mass and some intestinal loops had also disappeared. The mucosal wall of the adjacent loops was thickened, suggesting invasion. A similar 132 × 96 × 62 mm mass was noted in the left psoas muscle (Figure 5). In the absence of distant metastases, the patient was proposed for neoadjuvant radiotherapy and then surgery in a multidisciplinary decision-making process. The total dose of 50 Gy in 25 fractions was administered using a three-dimensional (3D) conformal radiotherapy technique. Three weeks after completion of radiotherapy, imaging follow-up was performed. Multiple hypodense enhancing foci of up to 40 × 47 mm were seen in segment seven of the liver on post-contrast CT scan images. There was also a 125 × 98 × 95 mm mass in the subsplenic space and a 47 × 42 mm mass in the psoas muscle. Core needle biopsy (CNB) of the liver mass revealed a poorly differentiated neoplasm. IHC examination was negative for CK, PAX8 and chromogranin, which was in favor of metastatic renal osteosarcoma. The patient was eligible for palliative chemotherapy, and cisplatin and doxorubicin-based chemotherapy was prescribed every 3 weeks. The patient has received four cycles of chemotherapy to date. He tolerated the treatment well, and there has been no progression of the tumor. Post-contrast CT scan. (a) muscle stranding of the abdominal wall indicated tumor invasion (asterisk). (b) Disappearence the fat plane between the mass and some loops of the intestine (arrow)suspicious of invasion. (c) infiltrated mass in the left psoas muscle (arrow).
Discussion
There are at least 36 reports of ESOS of the kidney in the literature review. The first report was from a postmortem study in 1936. 4 Not all data were accessible from some reports, but the available information suggests that the disease occurs predominantly in males, with a male-to-female ratio of approximately three to two. The median age of onset is 51 years, with a range of 29–82 years. Our patient is also a 51-year-old male.
The pathogenesis of ESOS remains unclear. Since 1884, only one theory, known as Virchow’s theory and still referred to, has been proposed, namely, reversion of renal capsular connective tissue to pluripotent embryonic mesenchyme and re-differentiation to osteosarcoma. 5
Commonly reported symptoms include flank pain, gross hematuria, and constitutional symptoms such as weakness, fatigue, and weight loss. Our patient was found to have gross hematuria. ESOS appears to behave aggressively when diagnosed late in advanced stages. Few reports show long-term survival without recurrence or distant metastasis.2,3 All of these cases were diagnosed at an early stage, demonstrating the importance of early detection for the outcome of renal ESOS. Most reported cases were locally invasive at presentation or developed metastases during their clinical course. A study of high-grade extraskeletal osteosarcomas found a 5-year local relapse-free survival of 47% and a disease-specific survival of 53%. 6 However, in renal ESOS, the results appear to be worse, and overall survival was approximately 10 months. 7 This may be due to late presentation and diagnosis of the disease. Fifty-two percent of reported cases developed metastases at presentation or during the course of the disease. Huang et al. 7 also reported 86% local recurrence and 32% metastasis at presentation. Most organs affected by distant metastases were lung, liver, and bone, although rare sites such as the lip were also reported.
Imaging studies of renal osteosarcoma may show a calcified mass on plain radiograph, a sunburst appearance of calcification on CT or an intravenous urogram, and a nonfunctioning kidney on a radioisotope scan.7,8 The differential diagnosis of a calcified mass on radiography may be metaplastic, bone-forming renal cell carcinoma, which can be excluded by IHC. In addition, renal ESOS is usually hypovascular compared with the relatively hypervascular nature of RCC. 9
Differential diagnoses of extraskeletal sarcoma include other sarcomas that produce tumoral osteoid or bone, such as dedifferentiated liposarcoma, leiomyosarcoma, and solitary fibrous tumor, as well as sarcomas that contain reactive metaplastic bone, such as synovial sarcoma and sarcomas with dense collagen that mimics osteoid, such as spindle cell/sclerosing rhabdomyosarcoma, and also benign soft tissue lesions such as phosphaturic mesenchymal tumors. 10 Because the prognoses of ESOS and these mimic lesions differ significantly, accurate diagnosis with proper sampling technique, preparation, and adequate volume of biopsied specimens is essential.
In addition, sarcomatoid carcinoma may contain osteoid, bone and cartilage formation and is one of the differential diagnoses. Sarcomatoid carcinoma is a high-grade neoplasm that contains both epithelial and mesenchymal elements. On IHC examination, this entity reacts with either epithelial-specific markers such as cytokeratin or mesenchymal-specific markers such as vimentin. 11 Therefore, it is very important for the pathologist to look for the epithelial component in a renal mass containing osteoid, bony, and cartilaginous components by evaluating the specimen with multiple thin sections and distinguishing RCC with sarcomatoid differentiation from osteosarcoma of the kidney.
Considering the challenges of diagnosing ESOS in the kidney, immunohistochemical examination is helpful. The specimen should be positive for mesenchymal markers, vimentin (VIM) and S-adenosylmethionine (SAM) and negative for epithelial markers, RCC and CK. 10
Enbloc resection with negative margin is a positive prognostic factor and the only treatment that can be curative. Resection margin, size of primary tumor, and depth of invasion are other prognostic factors for ESOS. It appears that these factors also apply to renal osteosarcoma. 6
Patients who present at early stages and undergo complete resection, may have a better chance of disease control or even cure compared to advanced stages. It is unclear whether adjuvant RT could be omitted after complete resection, and how much it can contribute to local tumor control. It seems that some high-risk patients should be identified and receive adjuvant therapies to prevent treatment failure. An example of this is our patient who had local recurrence after 4 months despite resection with a negative margin and no adjuvant treatment. In this case, the local recurrence presented as a large mass that could be identified by observation of the abdomen. The mass had remarkable shrinkage during radiotherapy. However, despite this good response to RT, the tumor metastasized to the liver. No standard chemotherapy regimen is proposed.
Most of the regimens prescribed in the reports are ifosfamide and cisplatin based.7,8,12 Fan et al. 6 suggested treating ESOS like soft tissue sarcomas with the same chemotherapy regimen because they have similar behavior and prognostic factors. As we know, there are significant data limitations in applying this idea to the treatment of renal ESOS. We started chemotherapy with cisplatin and adriamycin every 3 weeks, and 4 months after the discovery of metastases, his disease course has been stable so far.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Research Ethics Committees of Mashhad University of Medical Science (Approval ID: IR. MUMS.REC.1401.338).
Informed consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
