Abstract
Cystic echinococcosis is a widely endemic helminthic disease worldwide but occurs only rarely in Central Europe. Humans are infected as ‘aberrant’ hosts by Echinococcus granulosus and develop cysts in numerous different organs. 20%–30% of the affected individuals develop hydatid disease in the lungs with associated complications including pleuritis, lung abscess and pneumothorax. Radiologically, the pulmonary lesions of cystic echinococcosis occasionally pose difficulties in the differential diagnosis of primary lung carcinoma or metastatic disease and vice versa. Herein we report on a case of pulmonary hydatid disease in a 25-year-old Iraqi male presenting with a cystic lesion of the lung associated with thoracic pain and involuntary weight loss. Despite of its rare occurrence in Central Europe, clinicians, radiologists and pathologists should be aware of this entity and its pulmonary manifestations. During frozen section examination, imprint cytology specimens may facilitate the detection of the pathogens.
Introduction
Human echinococcosis is a zoonotic infection transmitted by dogs in livestock raising areas. The causative agent is Echinococcus granulosus belonging to the Taeniidae family of the cestoda class.1,2 Based on their genetic structure and biological properties, the genus Echinococcus includes six parasite species of cyclophyllid tapeworms, of which four are of public health concern and have significant economic impact: E. granulosus (which causes cystic echinococcosis, also termed hydatid disease), E. multilocularis (which causes alveolar echinococcosis) as well as E. Vogeli and E. oligarthrus (which cause polycystic echinococcosis). Molecular studies using mitochondrial DNA sequences have identified 10 distinct genetic types (G1-10) within E. granulosus. These encompass two sheep strains (G1 and G2), two bovid strains (G3 and G5), a horse strain (G4), a camelid strain (G6), a pig strain (G7) and a cervid strain (G8).3,4 The sheep strain (G1) is the most prevalent form and is most commonly associated with human infections. 3
The life cycles of these parasites involve two mammalian hosts. The adult cestode inhabits the small intestine of a carnivore (definitive host) and produces eggs containing infective oncospheres. Either cestode segments (proglottids) containing eggs or free eggs are released from the intestinal tract of the carnivore into the environment. After oral uptake of eggs by an intermediate host animal, a larval stage (the metacestode), develops in internal organs. The mature metacestode produces numerous protoscoleces, with each having the potential to develop into an adult cestode after being ingested by a suitable definitive host. Accidentally, eggs are also ingested by humans and other ‘aberrant’ hosts that do not play a role in the natural cycle. Whereas the infection of carnivores with immature or mature intestinal stages of E. granulosus does not cause morbidity, the invasion of various organs (mainly liver and lungs) of intermediate or aberrant hosts by metacestodes can cause severe and even fatal disease. 2
A hydatid cyst consists of three layers with intraluminal hydatid fluid. The outermost layer is called the pericyst (or ectocyst, respectively) and is formed by the host tissues’ reaction to the parasite including granulation tissue and fibrosis. The next layer is the laminated membrane (exocyst), and the innermost layer is termed the germinative layer or endocyst. The latter actively produces laminated membrane outward and brood capsules and protoscoleces inward. A brood capsule is formed by inward budding of the germinal layer of the hydatid cyst and is a vesicle-like structure containing protoscoleces. The laminated layer functions to maintain the physical integrity of the cyst and protects the germinal layer from host immune responses. The size of cysts in the human body is highly variable and usually ranges between 1 and 15 cm, but much larger cysts (>20 cm in diameter) may also occur.5,6,9
In humans, cysts may develop in numerous anatomic sites. This form of echinococcosis is termed primary cystic echinococcosis (CE). Secondary CE, predominantly in the abdominal and/or pleural cavity, results from spontaneous or trauma-induced cyst rupture and the release of protoscoleces and/or small cysts, which can grow to larger cysts.1,2 Approximately 40%–80% of patients with primary CE have single-organ involvement and harbour a solitary cyst. Hydatid fluid is antigenic, thus immune-mediated reactions such as urticaria, asthma, membranous nephropathy and anaphylaxis can occur.5,9 Because oncospheres enter the circulation via the gastrointestinal tract, the most common site of this disease in humans is the liver (50%–70%), followed by the lungs (20%–30%), and less frequently other organs including kidneys, spleen, muscles, skin, abdominal and pelvic cavities.2,7
The diagnosis of CE in individual patients is based on identification of cyst structures by imaging techniques, predominantly ultrasonography, computed tomography, X-ray examinations and confirmation by detection of specific serum antibodies by immunodiagnostic tests.1,2,8 Approximately 10%–20% of patients with hepatic cysts and about 40% with pulmonary cysts do not produce detectable specific serum antibodies (IgG) and therefore give false-negative results.2,3 Due to the slow growth of the cyst development, CE can go undetected for years. Depending on their size and anatomic location, cysts can eventually exert pressure on adjacent structures and can be associated with unspecific symptoms, and when in the lungs might provoke chronic cough, dyspnoea, pleuritic chest pain and haemoptysis. Cysts may rupture either spontaneously or due to blunt trauma and cause pleural hydatidosis with simple or tension pneumothorax and empyema or bronchial fistula. Treatment options depend on the extent of organ involvement and the number of cysts include both medical and surgical approaches.2,9
Case report
A 25-year-old male consulted the outpatient clinic of our hospital complaining about back and thoracic pain with increasing intensity and an involuntary weight loss of about 5 kg during the preceding 2 months. Three years before, he had emigrated from Iraq to Germany and had travelled to his native country 1 year before admission. Occasionally, the patient had noted blood-expectorating cough. Upon admission, elevated serological inflammatory parameters were observed. A chest radiograph (Figure 1(a)) and a subsequent contrast-enhanced computed tomography scan (Figure 1(b) and (c)) revealed a smoothly delineated cystic tumour with a maximal diameter of 9 cm in the lower lobe of the left lung with attenuation of contrast media in the periphery of the lesion, whereas the remainder lung parenchyma appeared inconspicuous. Radiologically, either a tuberculous cavern or echinococcosis was suspected. Bronchoscopy revealed no pathological changes. A mycobacterial infection could not be verified by PCR neither in the serum nor in a bronchoalveolar washing. Of note, serological testing for Echinococcus species was negative. Consequently, lobectomy of the affected lower left lobe was performed and submitted to the Institute of Pathology for diagnostic work-up.

A smoothly delineated cystic tumour with a maximal diameter of 9 cm is visualised in the lower lobe of the left lung in a chest radiograph (a) and in computed tomography scans ((b) and (c), asterisks). Note the attenuation of contrast media in the periphery of the lesion ((c), arrow). The adjacent lung parenchyma appears inconspicious.
Macroscopically, the resected lobe showed a cystic lesion with a whitish fibrous rim containing turbid fluid (Figure 2(a)). The adjacent lung parenchyma appeared normal. Histomorphologically, the periphery of the cystic lesion showed a thick fibrous rim, consisting of an outer acellular laminated membrane and a germinal membrane with nucleated lining with numerous protoscolices with occasional hooklets (Figure 2(b) and (c)). During workup for frozen section, cytological smear preparations had been performed, which also clearly showed protoscolices and hooklets, facilitating the frozen section diagnosis. Some of the smears were later stained according to Papanicolau (Figure 2(d)). In the periphery of the lesion, the lung parenchyma was dystelectatic with numerous intraalveolar macrophages and occasional multinucleated giant cells. The adjacent lung appeared normal with regular bronchi and blood vessels. The pleura covering the lesion was oedematous and thickened with granulation tissue admixed with polymorph nuclear granulocytes and covered by fibrin. Written informed consent was obtained from the patient before submission of this case report. The preparation of the clinical data was performed according to the rules outlined in the Declaration of Helsinki.
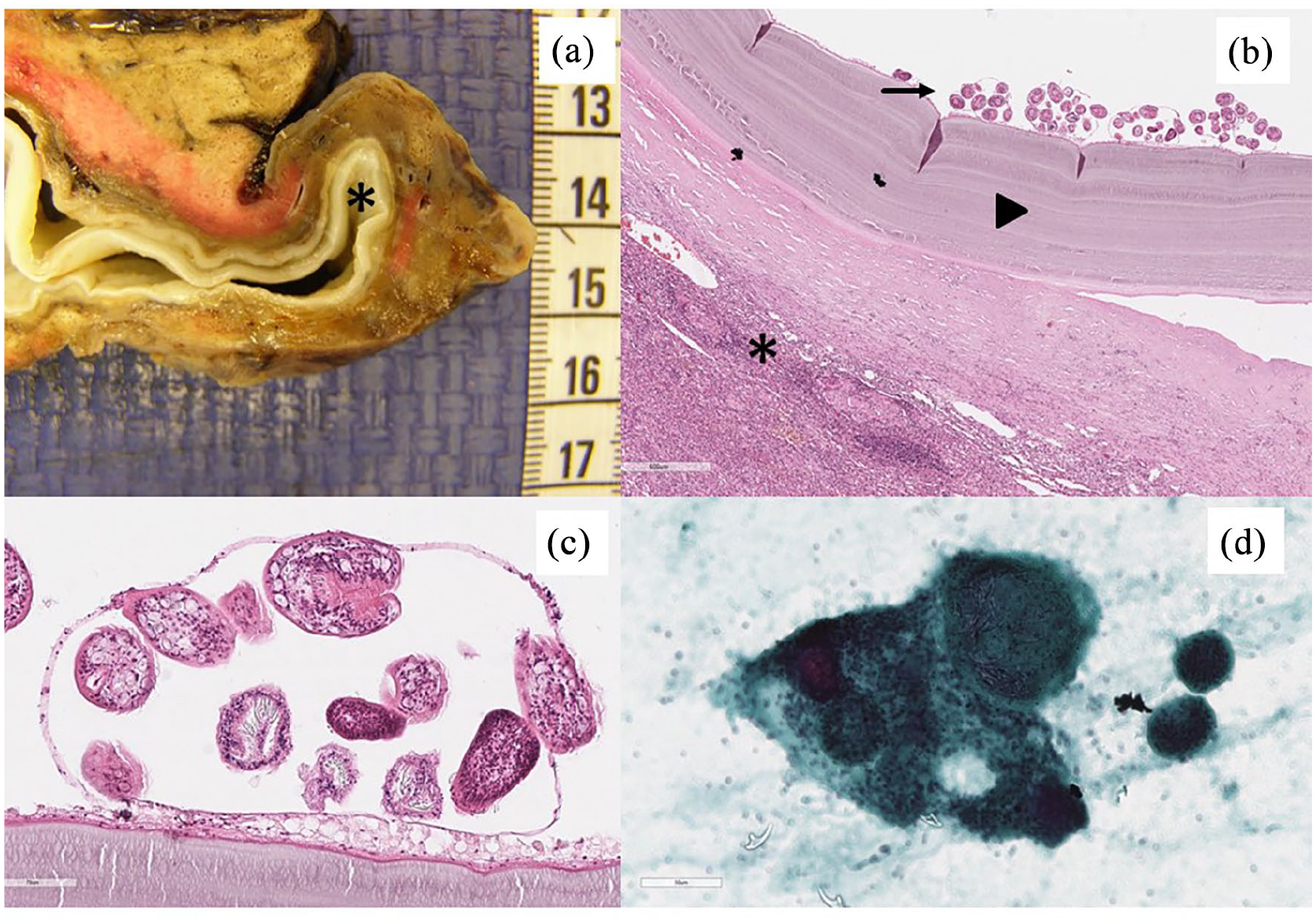
Surgical resection specimen of the lower lobe of the left lung with a cystic lesion ((a), asterisk). Histomorphologically, a hydatid cyst shows an outermost layer (pericyst or ectocyst, respectively) consisting of granulation tissue and fibrosis and multinucleated giant cells (asterisk). Adjacent to the pericyst is a laminated membrane or exocyst (arrowhead). The innermost layer is termed the germinative layer or endocyst, which is actively producing both laminated membrane outward and brood capsules and protoscoleces inward (arrow) (b). Close-up of the germinative layer with brood capsules containing protoscoleces (c). During work-up for frozen section examination, a smear cytology was performed, which facilitates the diagnosis by identification of brood capsules containing protoscoleces. This smear preparation was later stained according to Papanicolau (d).
Discussion
According to the World Health Organisation (WHO), E. granulosus is endemic in South America, Eastern Europe, Russia, the Middle East and China, where human incidence rates are as high as 50 per 100.000 person-years. 10 In contrast, the Robert-Koch-Institute (German federal government agency and research institute responsible for disease control and prevention) reports that in Germany almost no autochthonous E. granulosus infections occur. The majority of the infections observed in Central Europe including Germany are diagnosed in migrants from endemic regions. Infections by imported dogs have been noted. Cystic echinococcosis in tourists visiting endemic countries is rare. 11 Clinical diagnosis can be challenging because symptoms are unspecific. 9 Uncomplicated small cysts located in the periphery of the lung often remain asymptomatic and are detected incidentally on chest radiography. 12 Clinical symptoms occur, when the cysts are large enough to exert mechanical effects on adjacent structures and cysts larger than 5 cm in diameter can cause bronchial obstruction. 13 Complications include rupture, secondary infection with lung abscess and pneumothorax. Radiologically, lesions of ED can be mistaken for centrally lung carcinoma 14 and misinterpretations of hydatid cysts for pulmonary metastases of breast cancer, respectively, have been reported. 15 Vice versa, cystic pulmonary hamartomas, so-called benign metastasising leiomyoma of the uterus and bronchiectasis have been mistaken for hydatid disease.16–18
Conclusion
Cystic echinococcosis is rare in Central Europa including Germany but is frequently observed in other parts of the world, especially in the Middle East. Diagnosis is made in a synopsis of clinical, microbiological and radiological data and ultimately by histomorphological confirmation. Radiologically, pulmonary manifestations of hydatid disease can be mistaken for primary lung cancer or pulmonary metastases. In contrast, some benign entities including cystic pulmonary hamartomas and bronchiectasis can mimic hydatid disease. Especially in a younger patient group from endemic regions, pulmonary echinococcosis should be considered in the diagnostic work-up of pulmonary lesions. During workup frozen section, preparation of a cytology specimen can facilitate the detection of the pathogens.
Footnotes
Acknowledgements
We would like to thank the lab technicians at the Institutes of Pathology at the Diakonissenkrankenhaus Flensburg for their technical assistance.
Contributorship
Kristina Greimelmaier and Jeremias Wohlschlaeger wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript. In particular, the individual contributions of the authors were as follows:
Greimelmaier Kristina: Preparation of the draft
Hager Thomas: Scanning of the figures, critical revision of the draft
Moskalenko Vasily: Surgical procedure, acquisition of clinical data
Mueller-Huelsbeck Stefan: Radiological work-up, intellectual input, critical revision of the draft
Feist Henning: Frozen section examination of the specimen, critical revision of the draft
Schmid Kurt Werner: Intellectual input, critical revision of the draft
Seidel Alice: Critical revision of the draft
Jonigk Danny: Critical revision of the draft
Wohlschlaeger Jeremias: Preparation of the draft and figures
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Diakonissenkrankenhaus Flensburg does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymised information to be published in this article (original is separately uploaded as supplementary material).
