Abstract
Epithelioid hemangioendothelioma (EHE) is a low-grade, malignant vascular neoplasm that frequently involves the liver, lungs, bone, and soft tissue. Although not commonly associated with a paraneoplastic syndrome, paraneoplastic syndromes in the setting of EHE have been reported. Acute disseminated encephalomyelitis (ADEM) is an acute, autoimmune, demyelinating disorder of the central nervous system that most commonly occurs after an infection or vaccination. We present the case of a 23 year old female who developed the acute onset of fevers, tremors, right sided hemiplegia, global aphasia, and incontinence of urine and stool. MRI demonstrated findings consistent with a demyelinating disorder and brain biopsy confirmed the diagnosis of ADEM. The patient’s work up revealed multiple liver lesions which were biopsy proven EHE. This case report discusses the diagnosis and treatment of two concurrent rare disease processes and the possible association of the processes via a paraneoplastic syndrome.
Introduction
Epithelioid hemangioendothelioma (EHE) is a low-grade, malignant vascular neoplasm that most frequently involves the liver, lungs, bone, and soft tissue. 1 The incidence is estimated to be one case per one million people. 2 Despite commonly being a low-grade malignant tumor, one study found a metastasis rate of 75%. 3 Treatment of choice in EHE with liver involvement is radical liver resection, or in cases with more diffuse liver involvement, liver transplantation. 2 Although not commonly associated with a paraneoplastic syndrome, paraneoplastic syndromes in the setting of EHE have been reported.4–7
Acute disseminated encephalomyelitis (ADEM) is an acute, autoimmune, demyelinating disease of the central nervous system known to predominantly affect the white matter tracts of the brain and spinal cord. 8 The reported incidence is between 4 and 8 cases per one million people.9–11 The disease is more common in children than adults.8,12 The pathogenesis is thought to occur secondary to molecular mimicry 9 as ADEM most commonly occurs following an infection or vaccination. 13
Classically, there is a delay that can last up to 2 months after the febrile illness before the onset of the symptoms of ADEM. Common presenting symptoms include headache, nausea, emesis, fever, acute hemiparesis, mental status changes/encephalopathy, and gait abnormality.13,14 Symptoms are often multifocal and rapidly progressive requiring hospitalization, with 15%–25% of children requiring intensive care unit admission.12,15 Neuroimaging is critical in establishing a diagnosis of ADEM. MRI is the imaging study of choice and abnormalities are identified on T2 weighted and fluid-attenuated inversion recovery (FLAIR) sequences as patchy areas of signal intensity, often with poor margins. There are typically multiple lesions that are large and asymmetric. 8 Although ADEM most commonly presents after an infection or vaccination, multiple case reports have shown an association between neoplasms and ADEM presenting as a possible paraneoplastic syndrome.16–22
We present a case of EHE causing ADEM by a paraneoplastic process in a 23 year old female who presented with acute onset of fevers, tremors, right sided hemiplegia, global aphasia, and incontinence of urine and stool.
Case presentation
A 23 year old female with multifocal EHE without distant metastasis diagnosed by liver biopsy 10 months prior at an outside hospital. She presented to an outside hospital emergency department complaining of severe headaches, nausea, and emesis. She denied any recent sinusitis, pharyngitis, or upper respiratory tract infections. She denied any recent vaccinations. Twelve hours later, she returned to the same emergency department with tremors, right sided hemiplegia, global aphasia, and incontinence of urine and stool with a reported temperature 100.9°F. Head CT demonstrated left frontal lobe hypodensity suggestive of edema with underlying mass. MRI of the brain (Figure 1(a)) was performed and showed a left frontal lobe lesion with vasogenic edema and midline mass effect. Initial concern was for intracranial mass and she was started on intravenous (IV) dexamethasone 4 mg every 6 hours and was transferred to our tertiary care center for neurosurgical evaluation.

(a) Initial Brain MRI Axial T2 FLAIR images demonstrating a large infiltrative FLAIR hyperintensity centered within the left frontal lobe with heterogeneous, incomplete peripheral enhancement and extension along the corpus callosum into the right frontal lobe. There were no diffusion restricting correlations, (b) Repeat Brain MRI Axial T2 FLAIR 2 days after initial brain MRI demonstrating interval progression of the large infiltrative FLAIR hyperintensity within the left frontal lobe with heterogeneous, incomplete peripheral enhancement and extension along the corpus callosum into the right frontal lobe, and (c) Repeat Axial T2 FLAIR after left hemicraniectomy and brain biopsy. There is persistent FLAIR hyperintensity within the left frontal lobe with heterogeneous, incomplete peripheral enhancement and extension along the corpus callosum into the right frontal lobe. There are residual blood products within the biopsy cavity.
Upon initial examination, she was alert and intermittently followed one-step commands. She had dense aphasia, left gaze preference, bilateral papilledema, and right hemiplegia. A repeat MRI T2-weighted and FLAIR (Figure 1(b)) sequence showed progression of the infiltrative lesion centered in the left frontal lobe with ring enhancement and patchy uptake within the lesion when compared to the outside hospital MRI. Funduscopic exam showed papilledema. Infectious etiology was considered given rapid progression of symptoms and funduscopic evidence of increased intracranial pressure. She underwent a lumbar puncture showing 0 red blood cells, 99 white blood cells of which 93% were neutrophils, glucose of 80 mg/dL (plasma glucose 135 mg/dL), and protein of 55 mg/dL. She was empirically started on vancomycin 1 g q12hrs, ceftriaxone 2 g q12hrs, amphotericin B 10 mg/kg, micafungin 150 mg, and metronidazole 500 mg q6hrs. MRI chest, abdomen, and pelvis with contrast was performed and demonstrated multiple peripherally enhancing central hypo-attenuating liver lesions. She was scheduled to undergo a stereotactic left frontal lobe lesion biopsy. Before biopsy could be done, she had five to six generalized tonic-clonic seizures and developed a fixed left gaze deviation with an intermittently fixed and dilated 4 mm left pupil. She was intubated and underwent an emergent left decompressive hemicraniectomy with biopsy due to concern for a herniation syndrome. Operative findings showed no evidence of purulent or fibrous material on the brain suggestive of infection. A large wedge resection of the left frontal area was made and specimens were sent for pathology evaluation. Repeat brain MRI post-operatively showed persistent FLAIR hyperintensity in the left frontal lobe with heterogeneous, incomplete peripheral enhancement and extension of the hyperintensity across the corpus callosum in to the right frontal lobe (Figure 1(c)).
Pathology of the left frontal subcortical white matter showed multiple foci of perivascular chronic inflammation with pallor and aggregates of macrophages which were consistent with a demyelinating disorder (Figure 2). Given these results, concern for an infectious process was less likely and all antimicrobials and antifungals were discontinued. CSF gram stain, bacterial, and fungal cultures were negative. Blood cultures were negative and a transthoracic echocardiogram was negative for any evidence of vegetation. Viral titers including herpes simplex virus, Epstein-Barr virus, varicella zoster virus, and cytomegalovirus were negative. Serology for CSF VDRL, Lyme disease, Histoplasma, Toxoplasma, and Bartonella were negative. HIV-1/2 antibodies and RPR were negative. Urine Histoplasma antigen was negative. CSF cytology was notable for a marked acute inflammation without evidence of malignancy. Additionally, CSF flow cytometry did not identify any malignant cells. No oligoclonal bands were present in the patient’s CSF. Given the patient’s negative workup, the source of her demyelination was thought to be ADEM.
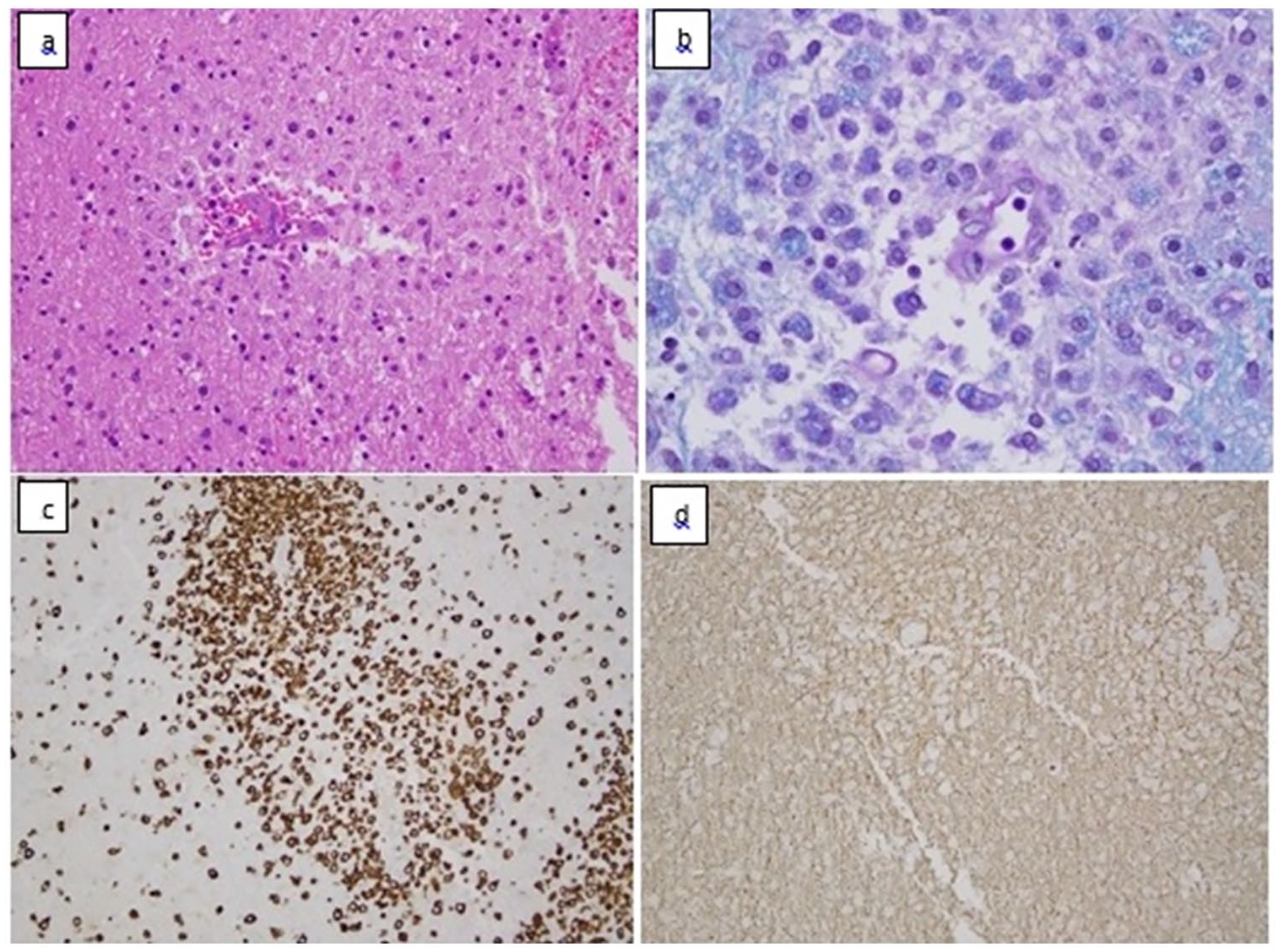
Lesion frontal lobe histology: (a) H&E: perivenous foci of palor and demyelination, collections of inflammatory cells and macrophages, (b) LFB-PAS: patchy perivascular demyelination with numerous fragments of myelin debris in macrophages, (c) CD68 and CD163: Myelin pallor and inflammatory cells showing demyelination with relative axon preservation accompanied by macrophages and a microglial and mononuclear cell infiltrate, and (d) Neurofilament: relatively well preserved axons in areas of demyelination.
The patient was given IV methylprednisolone 1 g daily for 5 days for immunosuppression and was then started on daily prednisone and sirolimus for chronic immunosuppression. Records from outside hospital were obtained in order to determine etiology of multiple liver lesions seen on CT imaging. Outside records revealed that 10 months prior the patient was admitted to an outside hospital for intractable emesis and diarrhea. Abdominal CT at that time demonstrated pancolitis with multiple hepatic lesions. At that time, she underwent a liver biopsy demonstrating EHE (Figure 3) but the patient was lost to follow-up. Given the outside records, imaging of hepatic lesions, and newly diagnosed ADEM on brain biopsy this admission, there was concern for a hepatic EHE with paraneoplastic features. PET scan was negative for metastatic disease but did show hypermetabolic and hypoattenuating lesions in the liver consistent with known EHE. Blood tests revealed normal levels of carcinoembryonic antigen, and cancer antigen 125. Autoimmune encephalopathy panel and a paraneoplastic autoantibody panel were negative. LGI1-IgG, CASPR2-IgG, NMO/AQP4, and MOG-IgG1 were negative.
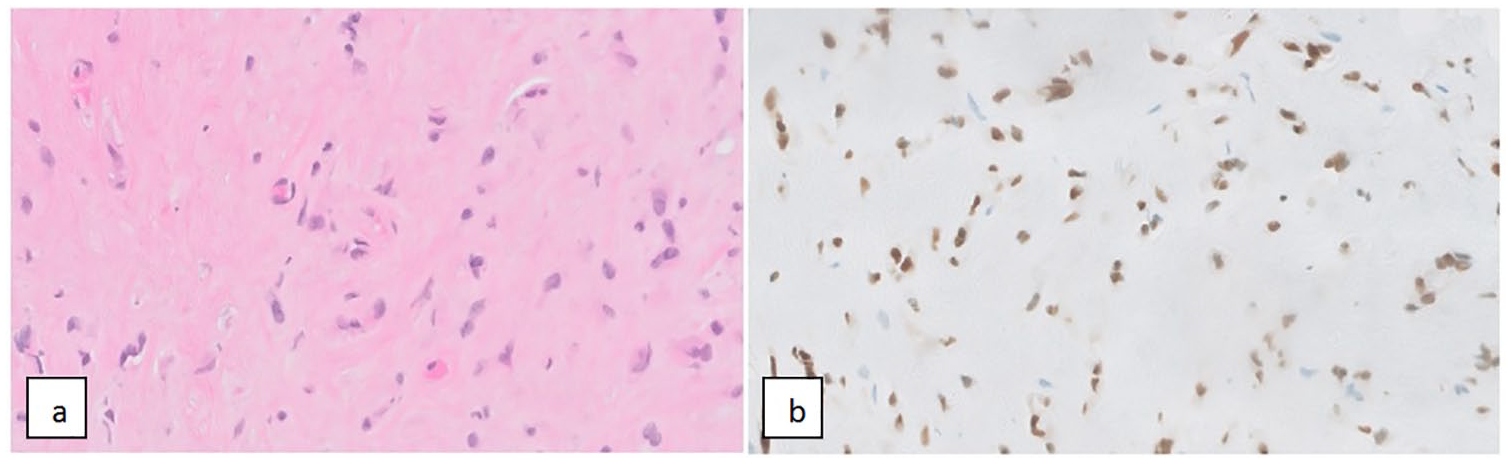
Liver histology: (a) H&E ×60: the biopsy of the liver shows complete replacement of the parenchyma with loosely cohesive cords and singly dispersed cells with rounded to irregular nuclei, and moderate amount of eosinophilic cytoplasm are dispersed in a hyalinized stroma and (b) ERG ×60: marker for vascular differentiation including benign and malignant vascular tumors neoplasms such as hemangioendothelioma.
MRI of the liver and abdomen were performed to determine anatomic disease burden and to guide further treatment with possible ablation, resection, or liver transplantation. MRI of the liver showed 25 hyperintense diffusion restricting and peripheral rim-enhancing lesions involving all lobes of the liver. Given the multilobar involvement, she was not a candidate for ablation or resection.
She was continued on prednisone for immunosuppression for 5 weeks with therapeutic dosing of sirolimus (12–16 ng/mL). Over the course of 3 weeks, her aphasia significantly improved and she regained full functional strength of her right upper and lower extremity with physical, occupational, and speech therapy while admitted to an acute inpatient rehabilitation service. She is scheduled to undergo a liver transplantation in hopes for a definitive treatment for her EHE.
Discussion
EHE is a rare tumor derived from endothelial cells that can involve different organs and soft tissues but the most frequent topography is the liver and lungs. Various causative agents have been theorized to be risk factors such as vinyl chloride, oral contraceptives, and polyurethane based products.2,23 However, no definitive correlation between development of EHE and these risk factors has been found. 2 EHE is a diagnosis that requires imaging to locate and even then, a diagnosis cannot truly be made without a tissue sample.
ADEM is typically a monophasic, inflammatory, autoimmune, CNS demyelinating disease. The presentation of neurological symptoms is variable, but the most common symptoms include headache, nausea, emesis, fever, acute hemiparesis, mental status changes/encephalopathy, and gait abnormality.13,14 Typically, the diagnosis of ADEM can be made without biopsy since the radiographic picture is characteristic with MRI T2-FLAIR sequences demonstrating patchy, poorly marginated areas of increased signal intensity. Due to concern for possible underlying brain lesion, our patient underwent a brain biopsy. Thus, the diagnosis of ADEM was aided by histopathology showing lesions with perivenular infiltrates of macrophages and foci of demyelination with axonal preservation on H&E, LPB-PAS, CD68/CD163, and neurofilament immunostain.
Although EHE is not typically associated with paraneoplastic syndrome, multiple case reports have described a paraneoplastic syndromes as a result of EHE. The most commonly reported paraneoplastic syndrome associated with EHE is secondary hypertrophic osteoarthropathy. Secondary hypertrophic osteoarthropathy is a paraneoplastic syndrome characterized by digital clubbing, periosteal reactions, polyarthralgia, arthritis, and synovitis. 24 Hypertrophic osteoarthropathy is commonly associated with pulmonary carcinomas and has been observed in primary EHE of the lungs 6 as well as hepatic EHE with metastasis to the lungs. 7 Additional paraneoplastic syndromes have also been attributed to EHE. Boukhris et al. 4 describe a case of a man with hypercalcemia related to tumor production of PTH-rp who was ultimately diagnosed with EHE. Additionally, El Mahou et al. 5 describe a case of a patient with a 13-year history of Cushing’s syndrome of unknown etiology despite extensive workup and hypophysectomy. However, after the discovery, diagnosis, and surgical excision of EHE, the patient’s hypercortisolism resolved and ACTH levels promptly normalized. Although paraneoplastic syndrome associated with EHE is uncommon, there are multiple reported examples in the literature. Therefore, it is possible that EHE was associated with a paraneoplastic process in this patient.
Our patient had no antecedent febrile illness, respiratory infection, or vaccination to serve as a more likely cause of her ADEM. Although the patient’s paraneoplastic antibody panel was negative, no specific antibody associated with paraneoplastic ADEM has been successfully isolated. 16 Thus, a negative paraneoplastic panel would not rule out paraneoplastic syndrome, but rather fail to identify any paraneoplastic syndrome associated with the tested antibodies. Negative paraneoplastic panels have also been observed in other cases of suspected paraneoplastic syndrome. 22 Further, as above, EHE has been described to cause paraneoplastic disease and there are numerous reports suggesting demyelination as a result of paraneoplastic syndrome.16–22 Although a causal relationship cannot be established at this time, we believe that the two processes in this patient are related, likely via a paraneoplastic syndrome, rather than by chance association. As an outpatient, the patient continues to follow up with regarding management of her EHE as well as a potential liver transplant.
In this report, we have described a patient with no other confounding medical co-morbidities who developed ADEM 10 months after being diagnosed EHE. To our knowledge and after review of the literature, we believe that this represents a rare presentation of ADEM by a paraneoplastic processes from untreated EHE.
Footnotes
Author contributions
Adem Aktas wrote the initial draft of the manuscript. Daniel Probst assisted in the revisions and edits. All authors reviewed and edited the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written signed consent was obtained from the patient and is attached as a separate document.
