Abstract
Objectives
This study aimed to compile a list of light-sensitive injectables in a tertiary hospital, determine the reasons and impacts of light protection, and suggest light protective methods.
Methods
Using a descriptive study design, injectables listed in the drug formulary of a tertiary academic hospital were screened for light-sensitive injectables through Package Inserts (PIs), both physical and online, for light protection information. Additional references like Micromedex, British Pharmacopoeia, United States Pharmacopeia and Trissel’s Handbook on Injectable Drugs, were consulted for verification. Where information was insufficient, manufacturers were contacted for photostability data. A literature search was conducted to suggest possible light-protective measures.
Results
Of 322 injectables from the formulary, 193 were identified as light-sensitive. Instructions on protective methods were found in 109 (56.5%) PIs, while 45 (23.3%) PIs mentioned the acceptable light exposure duration or stages requiring protection. Only 13 (6.74%) PIs provided the rationale for light protection. Rationales and details pertaining to light protection from PIs and other sources were presented in a table, with identified discrepancies discussed. Light protective methods were subsequently suggested.
Conclusion
The study highlights a lack of detailed, accessible information on light protection in PIs beyond the instruction to “Protect from light”. More comprehensive guidance on when and how to implement light protection, would improve the handling of light-sensitive injectables. Also, incorporating evidence, such as photostability studies, to validate manufacturers’ claims would empower healthcare professionals to make informed decisions, ultimately reducing the risk of photodegradation.
Introduction
Light-sensitive medications require special storage conditions as light exposure may affect their photostability. Photostability refers to a drug’s behaviour under light which can result in altered drug properties. 1 This may be due to the change in a drug’s susceptibility to photodegradation, where a light-sensitive molecule degrades after absorbing electromagnetic radiation, such as ultraviolet (UV) and visible light. 2
Varying levels of light protection may be required as the effect of different spectrums of light on a drug depends on the drug’s chemical structure, photoreactivity and dosage form. 1 Drug properties can be affected physically (e.g. changes in appearance), chemically (e.g. formation of precipitates) and pharmacologically (e.g. altered safety and efficacy). 3
Light protection is often considered during storage, but it is also crucial in other steps of the Medication Use Process (MUP),
4
mainly dispensing and administering (as well as reconstitution and dilution) in Figure 1. Medication use process (Modified from reference 4).
Light-sensitive injectables are especially vulnerable to light as the drug is usually in a suspension or solution before administration. Compared to solid formulation, photodegradation is accelerated due to increased molecular mobility where light is absorbed by a greater percentage of molecules. 5 Also, injectables are usually administered in transparent primary packaging, such as clear plastic. 6 This allows for easy inspection of the drug’s appearance before administration to avoid serious health risks. However, this means injectables may suffer inadequate light protection upon removal of their secondary packaging. Primary packaging refers to packaging directly in contact with the medication while secondary packaging refers to packaging that is not directly in contact with the medication i.e. a covering over the primary packaging. 7 Consequently, appropriate light protective measures may be required.
Currently, there are no published guidelines or standardized practices on the labelling of light-sensitive medications. 5 Labelling requirements are often set by national/regional regulatory authorities but these requirements regarding light protection tend to result in labelling that are vague and lack details in the package inserts (PIs). 5 The Health Sciences Authority of Singapore (HSA) issued guidance on therapeutic product registration in Singapore. Under “Online Appendix 7: Points to consider for Singapore labelling”, it is mandatory to include special precautions for storage (e.g. protect from light). However, it did not mention the need to include recommendations for light protection, let alone the rationale behind light protection. 8
The most recent publications by Elste et al 9 in 2023 and Sánchez-Quiles et al 3 in 2011 presented storage conditions related to light protection in a list of light-sensitive injectables. However, some of the injectables that are used in Singapore were not included in these lists. These publications also lacked details on the rationale behind light protection and did not recommend ways to protect these injectables. More comprehensive information is needed to understand the degree of light protection required and implement protective measures.
Thus, the aim of this study was to identify light-sensitive injectables in one of Singapore’s tertiary restructured hospitals, determine the reasons for and impact of light protection, and suggest appropriate light protective measures.
Methods
This study focused on light-sensitive injectables in the drug formulary of a tertiary academic hospital in Singapore. It is a major tertiary hospital which offers diverse healthcare services, and it has an extensive formulary covering most drugs available in Singapore. As such, the information collected will also apply to injectables in other restructured hospitals in Singapore.
Institutional Review Board approval was not required as no human subject was involved. Figure 2 summarizes the methods of the study. Summary of study methodology. NUH = National University Hospital, PI = prescribing information, TPN = Total Parenteral Nutrition, USP = US Pharmacopeia.
Compiling the list of injectables
Injectables were compiled by screening the hospital drug list. Injectables stored in the ward (e.g. dialysis fluids, continuous renal replacement therapy fluids) were excluded. Products that are routinely protected from light (e.g. total parenteral nutrition, laboratory preparation, chemotherapy medications) were also excluded.
Identifying light-sensitive injectables
PIs were the primary references used to obtain information on light protection, and they were first physically retrieved from the products. If inaccessible, PIs were downloaded from HSA’s Register of Therapeutic Products or credible sources such as countries’ regulatory websites, manufacturer’s websites, and online drug databases. The injectables’ packaging was also referred to when PIs were unavailable. Additional sources e.g. Micromedex, 10 Wolters Kluwer Clinical Drug Information, 11 British Pharmacopoeia, 12 US Pharmacopeia, 13 Trissel’s Handbook on Injectable Drugs, 14 Gahart’s Intravenous Medications, 15 were also reviewed to ensure the accuracy of information. The keywords ‘protect’, ‘light’ and ‘light-sensitive’ were searched in these sources. 9 If PI mentioned the need for light protection, the injectable would be deemed as light-sensitive. Other storage requirements, such as temperature, were excluded when compiling information on storage conditions as they were outside the scope of this study.
Obtaining rationale for light protection
The rationale behind light protection was collected from PIs and additional sources.10–15
This was supplemented with primary literature searched from PubMed and online assessment reports by regulatory agencies. Relevant manufacturers were also contacted to obtain photostability data when there was insufficient information online.
Analysis of findings
The data collected was analyzed and presented using descriptive statistics. Information on the light-sensitive injectables were compiled in a table and further analyzed for the actual need for light protection. The details under “Stages of Handling” column were based on the storage conditions across PI and additional sources.10–15 Discrepancies between the PI and additional sources10–15 regarding the need for light protection were noted in the “Light Sensitive?” column of the Supplemental Table with source of information specified. Relevant results of photostability studies were documented under the column for “Rationale behind light protection” in the Supplemental Table to support the claim for the need for light protection. The information in the “Need Actual Light Protection?” column is specific to the listed injectables as the collation of rationale behind light protection was done with brand specific information as much as possible. Where discrepancies were noted in the “Light sensitive?” column, conclusion for the “Need Actual Light Protection?” column was made based on results of photostability studies as collated in the “Rationale behind light protection” column. Should it be concluded that light protection is deemed unnecessary based on primary literature, we specified the conditions studied e.g. light intensity and duration of exposure under the “Need Actual Light Protection?” column. The actual need for light protection was deemed to be “Inconclusive” if the contradictory information was unresolvable or there was a lack of credible up-to-date photostability results. Light protection is still recommended for these injectables as a precautionary measure.
Results
A total of 193 out of 322 injectables (59.9%) in the drug formulary were identified as light-sensitive. Instructions on light protective measures, e.g. keeping injectables in secondary packaging, were mentioned in 109 (56.5%) PIs of light-sensitive injectables. Also, 45 (23.3%) PIs provided the acceptable duration of light exposure and/or the stages where light protection is required. However, only 13 (6.74%) PIs stated the rationale behind light protection.
Summary of compiled information of selected light-sensitive injectables in hospital drug list.
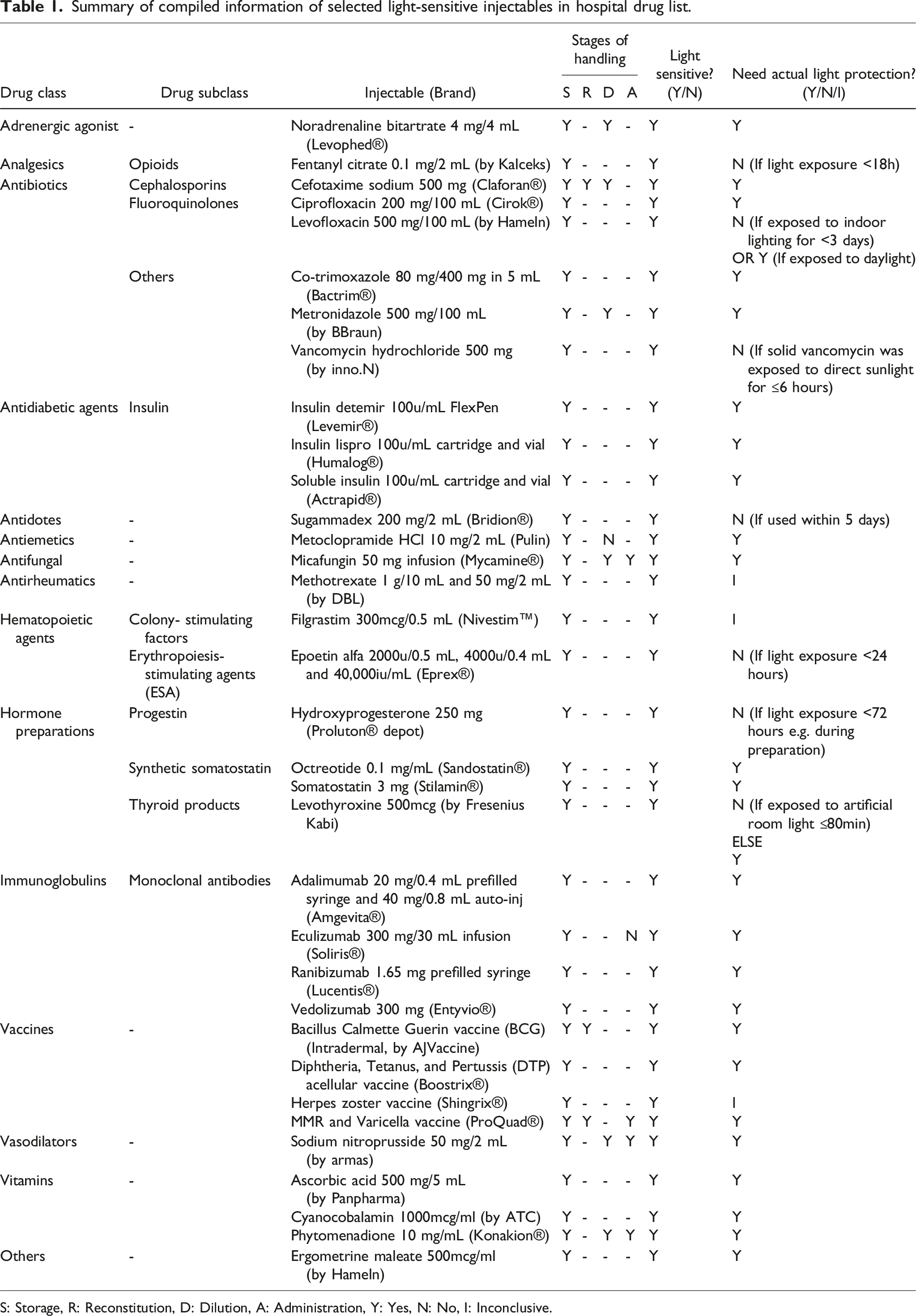
S: Storage, R: Reconstitution, D: Dilution, A: Administration, Y: Yes, N: No, I: Inconclusive.
Discussion
Light exposure may result in photodegradation of drugs, which alters their safety and efficacy. 3 Hence, it is necessary to identify light-sensitive injectables and gather the rationale behind light protection, to implement appropriate light protective measures.
Key drug classes
Additional information on the effect of light was gathered and presented in the Supplemental Table. This highlights the patterns of light sensitivity in drugs, and potentially flags out other light-sensitive injectables within the same drug class.
Insulins
Light exposure is a major concern for protein stability as light is absorbed by amino acids, such as tryptophan and tyrosine. 16 For example, tyrosine residues in Humalog® 17 are susceptible to light. Moreover, both UV and visible light from indoor fluorescent lighting are capable of inducing protein degradation. 18 Photodegradation may alter the structure and function of proteins, 16 affecting the formulation’s potency. 19 Photodegradants formed may also be immunogenic, 16 compromising patient safety.
Immunoglobulins
Similar to insulins, immunoglobulins are susceptible to light due to the reactivity of certain amino acids. 18 Exposing monoclonal antibodies to light may lead to aggregation, fragmentation and decreased bioactivity. 19 One study 20 showed increased concentrations of eculizumab aggregates with prolonged exposure to UV light and potential reduction of eculizumab’s specific activity if degradation occurred in its hypervariable regions.
Hematopoietic agents (HA)
Similar to insulin and immunoglobulins, the protein nature of HAs makes them vulnerable to photodegradation. For example, epoetin alfa may be adversely affected if exposed to light for more than 24 hours. 14
Vaccines
Most vaccines are light-sensitive, highlighting the need for adequate light protection. For instance, BCG vaccine degrades even under short periods of light exposure, 21 resulting in decreased viability. 22 The damage due to light is cumulative and inappropriate storage may result in a complete loss in potency. 23
Antibiotics
Since antibiotics are used to treat bacterial infections, their stability is critical for patient safety. This is especially so for antibiotics given intravenously as they are administered directly into the systemic circulation. Several commonly used antibiotics were found to require light protection. For instance, fluoroquinolones experienced reduced antibacterial activity and increased phototoxicity under light. 24
Significance of findings
Lack of detailed information on light protection and/or exposure
Majority of PIs only stated “Protect from light” or “Protect from light by storing in outer carton”, and did not provide evidence to substantiate these recommendations. This was shown by the small percentage of PIs (7%) that provided the rationale for light protection. Additionally, only 24% of PIs mentioned the acceptable duration of light exposure and/or the stages where light protection is required. This information is useful as it helps healthcare professionals (HCPs) understand the importance of proper storage and handling of light-sensitive injectables, which improves adherence to the instructions stated on the PI.
Acceptable duration of light exposure
While some injectables are widely thought to be light-sensitive, our primary literature search has uncovered studies indicating the duration that the drug remained stable under light. This allows for re-examination of the need for light protection during short periods of light exposure. For example, fentanyl was thought to undergo photodegradation as its chemical structure is prone to numerous degradation pathways. 25 However, fentanyl was found to be stable for 18 hours under sunlight and artificial light. 25 Thus, photostability studies are necessary to determine the duration that injectables can be safely exposed to light.
Effect of different lighting conditions
Varying light wavelengths and intensities may have different effects on drugs. A study 14 on phytonadione intravenous solutions found that sunlight exposure led to 43-63% degradation in 3 hours, while exposure to fluorescent lighting had a smaller impact where 50% degradation was detected in 15 days. More photostability studies conducted under common lighting conditions are thus needed for a more accurate understanding of the actual need for light protection in clinical settings.
Actual need for light protection
There are situations where manufacturers recommended light protection as a conservative approach although the formulation was not proven to be light-sensitive. For instance, Manufacturer 26 provided photostability data demonstrating slight changes in Entyvio® under light, which were acceptable and no significant effect was observed even with bright sunlight exposure. Despite this, the manufacturer recommended keeping the vial in its outer carton for light protection, as the stability data is continuously monitored and subjected to changes. Hence, photostability data may be useful in identifying injectables which are significantly affected by light, facilitating the development of standard operating procedures for light protection within the institution.
Discrepancies in the need for light protection
Discrepancies between the PI and additional sources,10–15 regarding storage conditions on light, were noted in the Supplemental Table. One possible explanation may be formulation differences whereby an excipient may require or negate the need for light protection. Excipients are inactive substances that aid medication delivery, which may initiate or participate in reactions with the active pharmaceutical ingredient (API). 27 Also, excipients may degrade under light, 28 in turn, affecting the formulation’s stability. For example, polysorbates can undergo photodegradation via autoxidation and form peroxides that further intensify oxidation. 28 Nonetheless, excipients are also able to stabilize formulations as USP 13 stated that metoclopramide injection requires light protection unless formulated with antioxidants, which is a type of excipient. Since various brands may contain different excipients, it is necessary to refer to the PIs or brand-specific photostability data to ensure appropriate storage.
Recommendations on light protective measures
Determining the actual need for light protection helps to reduce healthcare institutions’ logistical burden of having to protect all injectables deemed as light-sensitive by their PIs. Light protective measures should be reserved for injectables which truly require light protection.
Keeping light-sensitive injectables in their secondary packaging helps to protect the products from light. 23 This method is commonly suggested by manufacturers as seen in the majority of the PIs (57%) that provided light protection instructions. Indeed, this is the most commonly adopted light protective measure in the local setting. Auxiliary labels stating ‘light-sensitive’ may be placed on the secondary packaging or instructions may be stated on the bins where light-sensitive injectables are stored. These labels may also recommend the retention of these injectables in their secondary packaging. 5 This serves as a visual aid to alert HCPs of the appropriate storage and handling.
Additional measures may be required when injectables are removed from their secondary packaging for various reasons such as ward return or partial dispensing from original packaging, especially when the primary packaging alone does not provide sufficient light protection. The light protection measures for injectables removed from secondary packaging vary across local pharmacies. To address this issue, the following recommendations may be considered. An opaque black or brown polyethene bag may be used to block off light until the time of administration. A study 29 tested the changes in the quality of tablets stored under LED lighting, fluorescent lighting and in the dark for 28 days. The results demonstrated that brown light-shielding plastic bags were more effective than normal plastic bags as the extent of colour change of the tablets was smaller. However, there is a need to open the bag when the pharmacy or nursing staff is verifying the injectable’s identity, which can be time-consuming. Another option is the storage of light-sensitive injectables in semi-opaque low-density polyethylene (LDPE) plastic bottles. A study on liquid preparations found that when compared to a transparent bottle, the semi-opaque LDPE bottle, amber polyvinyl chloride (PVC) plastic bottle and amber glass bottle were effective in providing light protection to the extent of 91.3%, 90.5% and 72.6% respectively. Therefore, semi-opaque LDPE plastic bottles provided better light protection than amber bottles (which are commonly used to protect medications against light) for drugs tested in the study. 30
It should be noted that the light protective measures suggested above may not be generalized to all formulations. For instance, capping of injectables formulated in pen form, such as Levemir® FlexPen® is sufficient for light protection.
There are benefits in tailoring light protective measures to specific injectables but this may be less practical or feasible due to complications and the need for more resources. In contrast, the proposed light protective measures mentioned above were chosen for their applicability to a wide range of light-sensitive injectables which allows for standardization.
Applicability to a larger context
Storage requirements are specific to a drug’s formulation and different brands of the same API may be used across hospitals. Therefore, information on APIs was obtained to estimate the likelihood of a particular drug requiring light protection. However, it is still necessary to refer to the relevant PIs to ensure that the storage conditions are consistent with the findings of this study.
Limitations
While PIs were useful in identifying storage conditions, the majority did not provide evidence to substantiate the need for light protection. Although literature searches were conducted, there was a lack of accessible photostability studies and data for some light-sensitive injectables. Most of the available studies could be outdated or were not specific to the brands in use. To strengthen the applicability of the data, assessment reports by regulatory agencies (e.g. European Medicines Agency, Therapeutic Goods Administration) containing brand-specific information had to be obtained.
Additionally, the rationale behind light protection for several light-sensitive injectables could not be found. While 42 manufacturers were contacted to obtain photostability data, the response rate was only 21% (9/42) and 5 (56%) of the nine responders were unable to provide data as the information was considered proprietary. Regulatory agencies may consider mandating specification of the stages of handling which require light protection and the appropriate light protective measures or sharing of photostability data in PI.
Conclusion
A total of 193 light-sensitive injectables were identified and their rationale behind light protection was compiled. Light protective measures were subsequently suggested. Overall, there is a lack of easily accessible detailed information regarding light protection for most products beyond the phrase, “Protect from light”.
Only 7% of PIs stated the rationale for light protection, while 24% of PIs mentioned the acceptable duration of light exposure and/or the stages where light protection is needed. Regulatory authorities may consider mandating companies to provide clearer information on the stages of handling which require light protection and the appropriate light protective measures for their products. Additionally, the availability of data from photostability studies to substantiate manufacturers’ recommendations for light protection, will enable HCPs to make an informed decision on the actual need for light protection. This facilitates appropriate handling and storage of light-sensitive injectables, to maintain their quality, safety and efficacy.
Supplemental Material
Supplemental Material - Evaluating light-sensitive injectables: Gaps in manufacturers’ package inserts and recommendations for best practices
Supplemental Material for Evaluating light-sensitive injectables: Gaps in manufacturers’ package inserts and recommendations for best practices by Shannon Fang Ting Lim, Sze Yi Ong, How Priscilla, Elaine Lo Ah gi in Proceedings of Singapore Healthcare.
Footnotes
Acknowledgments
We would like to thank Mr Wong Zhi Yen for his editorial assistance.
Author contributions
S.F.T.L. Design, Literature search, Data collection, Data analysis, Preparation of the manuscript, Editing of the manuscript, Review of the manuscript. S.Y.O. Design, Literature search, Data collection, Data analysis, Preparation of the manuscript, Editing of the manuscript, Review of the manuscript. H.P. Concepts, Design, Editing of the manuscript, Review of the manuscript. E.L.A.G. Concepts, Design, Data collection, Editing of the manuscript, Review of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data Availability Statement
Data supporting this study are included within the article and/or supporting materials.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
