Abstract
Background
Coca-Cola and Cola-Cola Zero Sugar are commonly consumed beverages by healthcare professionals. Medical superstitions abound about whether Cola-Cola Zero sugar consumption affects night call outcomes.
Objectives
The current study aimed to determine whether Coca-Cola Zero Sugar, compared to regular Coca-Cola, is associated with any differences in inpatient admissions and mortality of patients on night calls.
Methods
A double-blinded randomized controlled trial was conducted in the Division of Medicine, Singapore General Hospital, Singapore involving junior doctors from January 2024 to February 2024.The primary outcome was the number of ward admissions under the night call doctor’s care. Secondary outcomes involved the number of inpatient deaths, high-dependency or intensive care admissions, the number of hours slept by the doctor, the number of steps walked by the doctor, and the level of sleepiness of the doctor on call.
Results
203 participants consented and were randomized (44.9%). The data of 198 participants were included for analysis (97.5%). No significant differences were detected in the number of ward admissions between the Coca-Cola Zero Sugar group and the regular Coca-Cola group. For secondary outcomes, a significant difference was present between the two groups in the Karolinska Sleepiness Scale score (p-value = .0413). Other secondary outcomes did not demonstrate significant differences between the two groups.
Conclusions
No convincing evidence was present to support the consumption of Coca-Cola Zero Sugar versus the regular Coca-Cola had significant differences in the number of inpatient admissions and mortality in night call duties.
Introduction
During night calls, medical doctors are responsible for attending to newly admitted patients in the hospital as well as hospitalized patients with new medical complaints, also termed “called to see patient” scenarios. Notably, junior doctors often amass critical clinical experiences during their night calls, to grow into mature clinicians capable of assessing complex clinical scenarios. 1 While these night calls contribute significantly to training the clinical acumen of physicians, they also entail reduced sleep and disturbances of circadian rhythms which contribute to a reduced level of alertness and fatigue in doctors from factors such as patient load, mortality and lack of rest.2,3
Hence, junior doctors often desire a less hectic call due to its daunting nature. Although clinical medicine was established based on evidence-based strategies, superstitions for night calls are frequently observed among healthcare professionals to conduct or avoid certain behaviors. These individuals may hold beliefs related to the implications of external or supernatural forces on events occurring in hospitals, leading to the adoption of behavioral changes. Such behaviors have been often attributed as a coping mechanism in dealing with uncertainty, fear, and anxiety in clinical settings. 4 An example of a common superstition includes avoiding the usage of words such as “peaceful” or “wishing you a quiet shift” which is believed to lead to a dramatic increase in workload and sicker patients to be dealt with overnight. 5 Although actual studies have dismissed the existence of such an association, the ritual was still routinely practiced in Asia, especially in the local setting in Singapore and Hong Kong. Another common superstition relates to the consumption of steam buns or “bao” which has been associated with the imagery of a call filled with a lion’s share of corpses. This phenomenon has led to doctors on call avoiding the consumption of steam buns. Although a study failed to demonstrate any significant correlation, consumption of steamed buns was regularly avoided locally. 6
In Singapore, the consumption of Coca-Cola Zero Sugar (Coke Zero) is popular in the medical community as it is believed that consuming it is associated with an auspicious decrease in complicated patient events during night shifts compared to other beverages such as Pepsi or 100PLUS, which enjoyed substantially less popularity. Among junior doctors, it is widely believed that this practice leads to fewer, if not “Zero”, new admissions, less inpatient mortality, and more rest for the on-call doctors, regardless of other factors such as the amount of caffeine intake. Hence, this study aimed to evaluate whether consuming Coke Zero was associated with a different night-call experience in terms of inpatient course and mortality, compared to the consumption of the normal Coke. We primarily hypothesized that the consumption of Coke Zero would lead to significant differences in night call outcomes, including inpatient admissions and mortality compared to Coke consumption.
Materials and methods
Study design and setting
The study was a single-center, double-blinded, randomized controlled trial. The recruitment and data collection took place in the Department of General Medicine at Singapore General Hospital, on weekdays from 8 January 2024 to 2 February 2024. Singapore General Hospital is Singapore’s largest hospital with a total of 1735 beds and delivers medical care to over 1 million local and overseas patients annually. 7 Approval was obtained from SingHealth Centralized Institutional Review Board (CIRB Ref: 2023/2630) on 11/15/2023. The study protocol was registered with ClinicalTrials.gov (Protocol identification number: NCT-06155981) on 12/05/2023. The study was reported following the CONSORT statement for randomized controlled trials. 8
Randomization and intervention
The study was conducted in accordance with the Declaration of Helsinki, and the experimental protocol was approved by SingHealth Centralized Institutional Review Board. 9 All house officers, medical officers, and registrars working in the Department of General Medicine at Singapore General Hospital were eligible to be included in the study. Recruitment of study participants was conducted via text message. Participants were excluded if they declined participation in the study or if they failed to complete the post-study questionnaires. To ensure randomization, a member of the research team created a computer-generated randomization sheet using simple randomization to assign the participants on the daily call roster to either receive regular Coca-Cola (Regular Coke) or Coke Zero randomly, repeated on a daily basis, for 20 days. This member of the research team had no additional involvement with the study and no interaction with any of the participants.
All participants were informed of the purpose of the study, which was to explore any differences in night-call inpatient course between the consumption of Coke Zero versus Coke, without any mention of which type of Coke they would receive. All participants provided written consent before joining the study. Before the participant started their call at 5 p.m. on weekdays, they were contacted via text through phone numbers listed on the official department roster and given the same 7-ounce cup of their designated drink by one of the researchers responsible for the dissemination of the drink in the study, containing either Coke (control group) or Coke Zero (intervention group) without any identification of which drink they received to ensure blinding. The type of drink distributed by the researchers would only be revealed to them on the day of distribution. After the consumption of the Coke drink and the conclusion of the night call the next day at 8 a.m., the on-call doctors received a questionnaire via email that contained questions concerning the course of the previous night’s call. Each day a new randomization sheet was created by the statistician to determine the type of drink each doctor would receive. The study team responsible for distribution was blinded to the type of intervention participants received as the drink was just labeled with a designated number and had no other features or marks suggestive that the drink was Coke or Coke Zero.
Participant information and study outcomes
Basic demographic information was collected from the participants for baseline description of the study participants, including their age, gender, graduating medical institution, number of years post-graduation, and their current rank.
The outcomes of the study involved the use of an online questionnaire which was emailed to the night-call doctors after their call at 8 a.m. on the next day, covering a variety of aspects of the course and inpatient mortality of the night calls. The primary outcomes collected in the questionnaire for the study were the number of general ward admissions under the doctor’s care during the doctor’s night call, the number of high dependency and intensive care unit ward admissions, and the number of inpatient mortality under the care of the doctor.
Secondary outcomes collected included the number of hours that the doctor had slept during the night call, the number of steps the doctor walked if they had used a step tracker, and their level of alertness and sleepiness as measured by the Epworth Sleepiness Scale and the Karolinska Sleepiness Scale.10,11
The Epworth Sleepiness Scale is a self-administered instrument comprising 8 questions, where respondents are requested to rate their risk of falling asleep for eight different activities on a 4-point Likert scale. The total score ranges from 0 to 24, with high scores indicating a higher level of sleepiness. 12 A score of 0-7 indicates that one is unlikely to be sleepy while a score of 16-24 indicates excessive sleepiness. This instrument has been validated for use in Singapore. 13 On the other hand. the Karolinska Sleepiness Scale is a self-reported 9-point scale that assesses the level of situation sleepiness at a particular time of the day. Subjects are required to indicate their psycho-physical state within the last 10 minutes, with higher scores indicating more situational sleepiness. 10 Likewise, this tool has been validated in multiple Asian populations.10,14–16
Statistical analysis
Assuming a power of 80% and a p-value of 0.05, a sample size of 160 was the target for the recruitment of the current study. This was based on a mean difference of 5 between the two groups and a standard deviation of 10 for each of the two groups. A sample size of 64 was needed from each group and it was expected that there would be a 20% loss to follow-up, reaching a final estimated sample size of 160. Analyses were based on intention to treat, and every participant ultimately received the type of intervention they were initially randomly assigned to. We calculated the mean values of the primary and secondary outcomes and conducted a statistical analysis on the differences between the mean values of various primary and secondary outcomes respectively utilizing 95% confidence intervals via Stata ME V15.0 for Mac (StataCorp, Texas Station, USA). Shapiro-Wilk test was employed to determine whether the data were normally distributed. Two-sample Wilcoxon rank-sum (Mann-Whitney) test and two-sample t-test with equal variance were utilized to detect any differences among the study variables depending on whether the study sample values for the outcomes were normally distributed. A p-value <.05 was considered statistically significant.
Results
Ultimately, 203 participants consented and were randomized every day according to the randomization sheet to either the Coke group or the Coke Zero group (100 in the Coke group and 103 in the Coke Zero group). 4 participants were lost to follow-up after the questionnaires were disseminated in the Coke group and 1 participant was lost to follow-up in the Coke Zero group due to persistent nonresponse for questionnaires. Ultimately, the data of 198 participants who completed questionnaires were included in the study (97.5%). Figure 1 demonstrates the flow of the study. Study participants’ recruitment, inclusion, and exclusion.
Demographic characteristics
Characteristics of physicians by randomization group.

SD: standard deviation
Duke-NUS: Duke-NUS Medical School, Singapore (Post graduate medical school)
NUS-YLL: National University of Singapore-Yong Loo Lin School of Medicine, Singapore
NTU-LKC: Nanyang Technological University-Lee Kong Chian School of Medicine, Singapore
Primary and secondary outcomes
Primary and secondary outcomes of the intervention by randomization group.
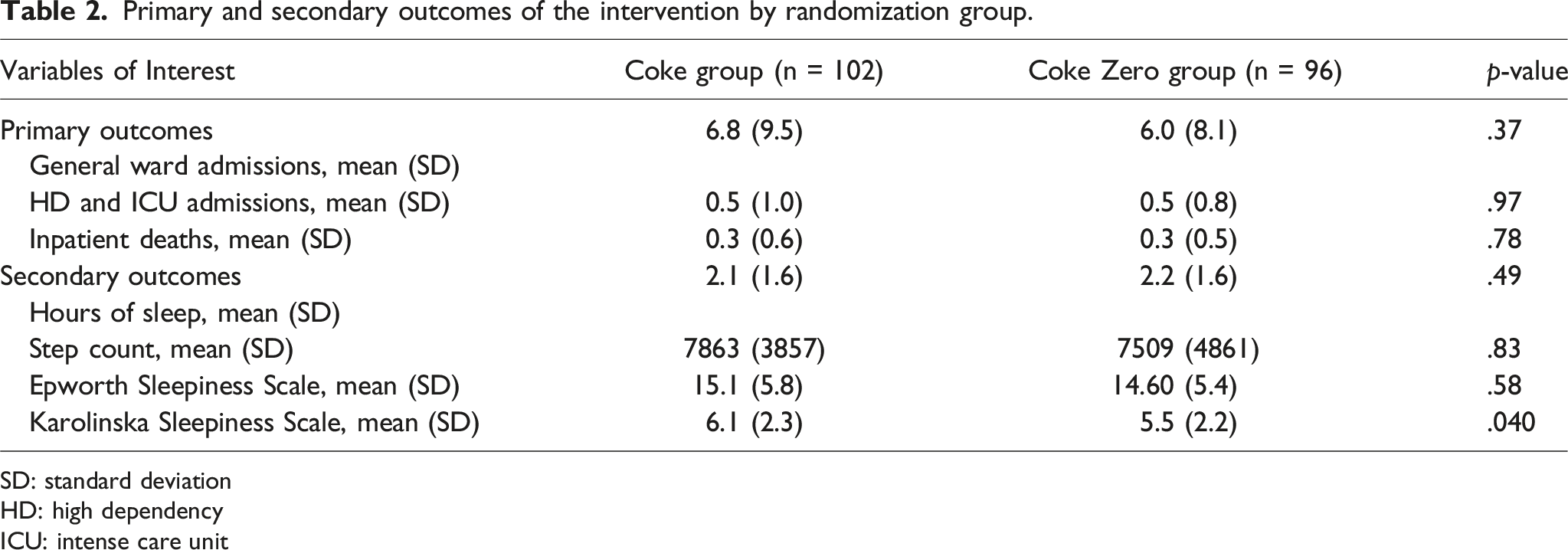
SD: standard deviation
HD: high dependency
ICU: intense care unit
For the secondary outcomes, data from the number of steps walked by the doctor on call was limited (n = 32). Otherwise, the number of steps walked by the doctor on call, the number of hours the doctor on call managed to sleep, and the Epworth Sleepiness Scale were not different between the two groups. However, based on the Karolinska Sleepiness Scale, doctors in the Coke group had more situational sleepiness compared to doctors in the Coke Zero group [6.1 (2.3) versus 5.5 (2.2), p = .04].
Discussion
Principal findings
The high retention rate (97.5%) of the trial reflected a strong interest among the junior doctors to understand the effect of Coke Zero on their night calls. The current study found that the consumption of Coke Zero versus Coke had no significant differences in impacting the inpatient course and mortality as primary outcomes, contrary to the prevailing belief in the local medical community. For the secondary outcomes, the consumption had no significant difference on a variety of variables other than the Karolinska Sleepiness Scale, signifying a potential impact on the level of alertness after the night call.
This study demonstrated that the consumption of Coke Zero made no significant differences in inpatient course and mortality for junior doctors on call. This finding was in keeping with results from other studies that evaluated the effects of medical superstition among healthcare workers during their night duties.6,19 Interestingly, higher scores on the Karolinska Sleepiness scale were noted among participants who took Coke as opposed to Coke Zero. While it remained unclear why the results were not replicated on the Epworth Sleepiness scale, potential reasons may lie in differences in situations where sleepiness was assessed between the two instruments. The Epworth Sleepiness scale focused on sleepiness in performing 8 different activities while the Karolinska Sleepiness scale assessed subjective sleepiness at a time point. Other potential reasons for the difference may lie in the sweeteners used. The use of aspartame has been linked with neuropsychiatric symptoms and problems such as insomnia as it has been postulated to act as a chemical stressor that increases cortisol levels. Consequently, this may have possibly led to users of Coke Zero having lower scores on the Karolinska Sleepiness Scale as opposed to the Epworth Sleepiness Scale.20,21
Importantly, our study showed a significant level of heightened daytime sleepiness and a low number of hours of sleep for doctors on-call, in agreement with prior studies. The mean number of hours junior doctors in the study slept was around 2 hours, which was congruent with previous studies on junior doctors. Of note, no significant changes were noted in comparison with the previous local study in 2008, more than 15 years ago.6,22 In addition, the average Epworth Sleepiness Scale score was from 14 to 15 in the study across the two groups, corresponding to the category of moderate excess daytime sleepiness. Given the negative ramifications of sleep deprivation on patient safety and well-being of doctors, there is a need for policy changes for the implementation of safe working hours and concrete welfare benefits for doctors.23,24
Strengths and limitations
There were several strengths in this study. Firstly, its randomized double-blinded study design helped increase the study findings’ validity. The double-blinded nature increased the study’s validity and minimized any experimenter bias. We recruited doctors from various ranks with adequate sample size and a comparable number of participants between the two groups, at the same time representing the variety of junior doctors in the hospital and minimizing any sampling bias toward the results. The retention rate of the study was 97.5% after the dissemination of questionnaires, which was encouraging given the busy schedule of the doctors.
The study findings should be interpreted in the context of limitations. Firstly, even though the study was conducted in the largest hospital in Singapore, it was conducted in a single center, which may limit the generalizability of the study findings to other healthcare institutions, especially given the limited amount of data obtained for certain outcomes. The difference in demographic characteristics could potentially contribute to differences in outcomes, although they are largely comparable through randomization. Future multi-center studies may aid in confirming the results. Secondly, participants were recruited in January and February 2024. Previous studies have demonstrated seasonal variation in the number of hospital admissions for varying populations such as heat stroke and heart failure patients, and certainly some of the outcomes measured in the study could vary as a result of this seasonal phenomenon. 25 It may be worthwhile to perform longitudinal studies to evaluate if there are seasonal variations related to the consumption of Coke or Coke Zero on hospital admission and mortality. In addition, the study results and reporting could be potentially affected by the study participants’ perception of the drink they consumed as there may be subtle differences in taste of the two drinks, arising from different sweeteners used that might be detectable by some participants. This potential confounder was attempted to be minimized through the blinding process. Additionally, it was difficult for the team to control whether participants were consuming other types of beverages in addition to Coke or Coke Zero, and it would be possible that additional consumption of different beverages could affect the study results, especially given the differing caffeine metabolism and baseline sleep quality among individuals which could potentially affect study outcomes such as scores from the two scales of sleepiness. However, due to the nature of the randomized study, it was likely that these factors were balanced between the two groups. In addition, our sample size in the study was powered towards the primary outcomes of the study, and no adjustment for multiple comparisons was conducted as hypotheses were stated a priori. Hence, there was a possibility that differences among secondary outcomes were not well demonstrated.
Future studies looking into junior doctors’ beliefs on the effect of consuming Coke Zero versus Coke and how their behaviors and mentation were influenced by such beliefs would be of interest.
Conclusions
The consumption of Coke Zero or regular Coke did not result in significant differences in inpatient course and mortality during on-call duties for doctors. Interestingly, the consumption of Coke resulted in increased Karolinska Sleepiness scale scores but similar Epworth Sleepiness scale scores, compared to participants who took Coke Zero. Our study showed significant sleep deprivation and increased situational sleepiness among junior doctors during their night studies in the hospital. There is a need for policy changes to improve working hours for junior doctors given the important ramifications of sleep deprivation on potential patient and physician outcomes, such as considering the implementation of diurnal and nocturnal shift work systems, revision of the limit of working hours, or improvement of concrete welfare benefits for junior doctors.
Footnotes
Acknowledgments
The Coca-Cola beverages in the study were sponsored by Lee Haur Yueh.
Author contributions
YHK conceived the study with input from BJJS, TO, SSWG, HYL, and WF. YHK wrote the protocol with support from HT. HT led recruitment from participants with support from LHQ, AFHWY, LY, YXL, AZEL, ASWF, YKC, FYL, and CWYS. HT drafted the first version of the manuscript with input from YHK. HT conducted the statistical analysis with input from YHK. All authors contributed to the review and interpretation of results and approved the final version of the manuscript. The corresponding author confirms that all listed authors meet authorship criteria and nobody else has been omitted in the process.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the ICMJE uniform disclosure form at ![]() . Otherwise, there was no support from any organization for the submitted work, no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years, and no other relationships or activities that could appear to have influenced the submitted.
. Otherwise, there was no support from any organization for the submitted work, no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years, and no other relationships or activities that could appear to have influenced the submitted.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
