Abstract
Background
Geriatric assessment (GA) has been shown to be pivotal to the prognosis of elderly cancer patients. A G8 screening tool could act as a potential surrogate to identify patients who would benefit from further GA in a busy oncology clinic and a digital version would allow for a wider application.
Objectives
To develop a digital self-reported version of G8, and to compare its outcomes with in-person G8 administered by a healthcare professional.
Methods
A total of 483 cancer patients aged 70 years and older who received an in-person G8 assessment at the National Cancer Centre Singapore (NCCS) clinic, and another 544 who completed the digital self-reported G8 were analysed. Total G8 score ≤14 was defined as positive screen.
Results
Response rate of the digital self-reported G8 was 50%. Median G8 total score was lower among online screeners than in-person screeners (11 vs 12.5, p < 0.001). The odds of a G8 positive screen among online screeners was higher than that of in-person screeners on multivariable logistic regression analysis (odds ratio = 1.56, 95% CI 1.08-2.26). Some 20 in-person screeners had also completed the digital self-reported G8, and the agreement between their in-person and online G8 total scores was high (concordance correlation coefficient = 0.798, 95% CI 0.635-0.962).
Conclusions
A digital self-reported G8 is feasible. However, given the higher positive screen rate among the online screeners, replacement of the in-person G8 with the digital self-reported G8 should be implemented only after more conclusive evidence on the agreement between in-person and online G8 score is available.
Introduction
The risk of malignancy increases with ageing. Almost 54% of new cases and 70% of mortality occur in patients aged 65 years and older. 1 The Singapore, Cancer Registry has reported a shift towards a greater proportion of cancer diagnosis in the older age groups. The proportion of cancer patients aged ≥70 increased from 15.7% to 42.3% in males and from 17% to 32.6% in females over the last 50 years. 2 To tackle the complexities of an older cancer patient, the Geriatric Assessment (GA) has been recommended by multiple international advisories such as the International Society of Geriatric Oncology (SIOG), 3 the American Society of Clinical Oncology (ASCO) 4 and the National Comprehensive Cancer Network. 5
The GA provides an armamentarium of information that aids in navigating the complexities of the elderly cancer patients to provide for individualized care plan. GA domains include functional status, comorbidities, psychological state, social status and support, comorbidities, medication review, nutrition as well as cognitive function. Numerous studies using the GA in older cancer patients have shown that it can predict survival and it helps decide on treatment dosage and selection.6–8 Information gathered is applied to evaluate overall survival, chemotherapy adverse events predictions as well as detect geriatric-associated comorbidities. 9 This allows for clinicians to develop a well-integrated and coordinated plan for treatment.
Despite the effectiveness of CGA, it is time consuming and is not applicable to every patient. 10 Some studies suggest that healthy older patients could be excluded. 11 Thus, screening tools such as G8 could act as a potential surrogate to identify patients who would truly benefit from further assessment with a GA.9,10,12 G8 is a screening tool that consists of eight items, including seven items from the Mini Nutritional Assessment, where patients are required to score the following domains – nutritional status, weight loss during the last 3 months, mobility, psychological status, body mass index, drug history, self-perception of health as well as age 13 (Appendix A). A score of ≤14 suggests a need for further assessment with a GA. 14
The G8 screening tool is currently administered by a healthcare professional prior to oncology consultations. A recent study analysed the challenges of implementation of G8 in clinical practice and established that healthcare staff prefer to complete other higher priority duties than G8 implementation. 15 With the increasing number of older cancer patients and man-hours required for the manual administration of this screening tool, the advent of an online self-reported G8 can be a solution. 13 This can be further optimized by sending it digitally via a short messaging system message prior to the oncology consultation. This study aimed to develop a digital self-reported G8, and to compare its outcomes against in-person G8 administered by a healthcare professional.
Methods
Digital self-reported G8: ZEDOC
A digital version of the G8 was developed with the aid of a digital health platform developed locally called ZEDOC. Paper-administered G8 was replaced with an integrated digital platform that automates data capture. ZEDOC sends patients a customized text message to their mobile device with secure links to G8 screening. The text message is sent 3 days before their clinic visit and a reminder text message is sent in the morning of their clinic appointment if no earlier response was received. This allows patients to complete the G8 on their own devices at their convenience in their preferred language of English or Mandarin.
Study participants
The study population was comprised of two cohorts of patients aged 70 years and older with a diagnosis of cancer at any stage. The first cohort consisted of 483 consecutive new patients who attended the outpatient geriatric oncology clinic at National Cancer Centre Singapore (NCCS) from July 2014 to November 2021. These patients received an in-person G8 assessment by a clinical research assistant trained in GA assessment during their first visit to the clinic prior to seeing the oncologist. The second cohort consisted of 544 consecutive patients who completed the digital self-reported G8 from ZEDOC prior to their attendance at one of the medical oncology outpatient clinics at NCCS from December 2021 to December 2022.
All patients signed informed consent prior to their study participation. The Singhealth Centralised Institutional Review Board reviewed and approved the study (CIRB Ref No.: 2019/2314).
Clinical and treatment data
Clinical characteristics and treatment details of each study participant were extracted from electronic medical records. Cancer diagnoses were coded based on the 9th revision of the International Classification of Disease (ICD-9) before 2012, and the 10th revision of the ICD (Australian Modification) (ICD-10-AM) from 2012 onwards. Diagnoses before 2012 were mapped from ICD-9 into ICD-10-AM for comparability with diagnoses after 2012. Tumour histology were coded based on the 3rd edition of the ICD for Oncology.
Statistical analysis
Comparing G8 scores and positive screen rate between in-person and online screeners
To compare the G8 scores and positive screen rate between patients who did in-person screening and patients who did the online screening, categorical and continuous characteristics between two groups of patients who did the in-person and online G8 screening were compared using Fisher’s exact test and Mann Whitney U test, respectively. Logistic regression models were fitted to assess the association between G8 positive screen and screening modality (in-person vs online). Goodness of fit of each fitted model was assessed based on the Hosmer-Lemeshow test.
Assessment of agreement in G8 scores among patients who did both in-person and online screening
A group of 20 patients who received the in-person G8 assessment had also completed the digital self-reported G8 when they were contacted by ZEDOC. To assess the agreement in G8 scores among patients who did both in-person and online screening, the percentage of patients who had the same online and in-person response to each question in the G8 questionnaire was used as an accuracy measure. The extent of agreement between the online and in-person ordinal responses was assessed based on weighted Kappa statistic. This statistic measured the degree of agreement between the two responses above what was expected by chance alone. It has a maximum of one when agreement was perfect, 0 when agreement was no better than chance, and negative values when agreement was worse than chance. Landis and Koch suggested this interpretation for the Kappa coefficient: <0: poor agreement, 0-0.2: slight agreement, 0.21-0.4: fair agreement, 0.41-0.6: moderate agreement, 0.61-0.8: substantial agreement and 0.8-1.0: almost perfect agreement. 16
The extent of agreement between the online and in-person G8 total score was assessed based on the concordance correlation coefficient. The concordance correlation coefficient ranged from −1 to 1, with −1 denoting perfect negative agreement, 0 denoting no agreement and one denoting perfect agreement. Additional assessments were made graphically via a Bland-Altman plot which was a scatter plot of the difference between the two scores against the average of the two scores, with lines indicating the absolute bias (i.e. mean difference) and 95% limits of agreement between the two scores. Agreement was deemed good if most of the points in this plot were within the 95% limits of agreement with no observable distinct pattern. A scatter plot of online score against in-person score was also made, and agreement was good if most of the points in this plot fell along the line of identity which denoted perfect agreement between the two scores.
SAS version 9.4 (SAS Institute, Cary, NC) was used for all statistical analyses, and a two-sided p < 0.05 was considered statistically significant.
Results
Comparing G8 scores and positive screen rate between in-person and online screeners
Baseline and treatment characteristics.

IQR, interquartile range; NOS, not otherwise specified.
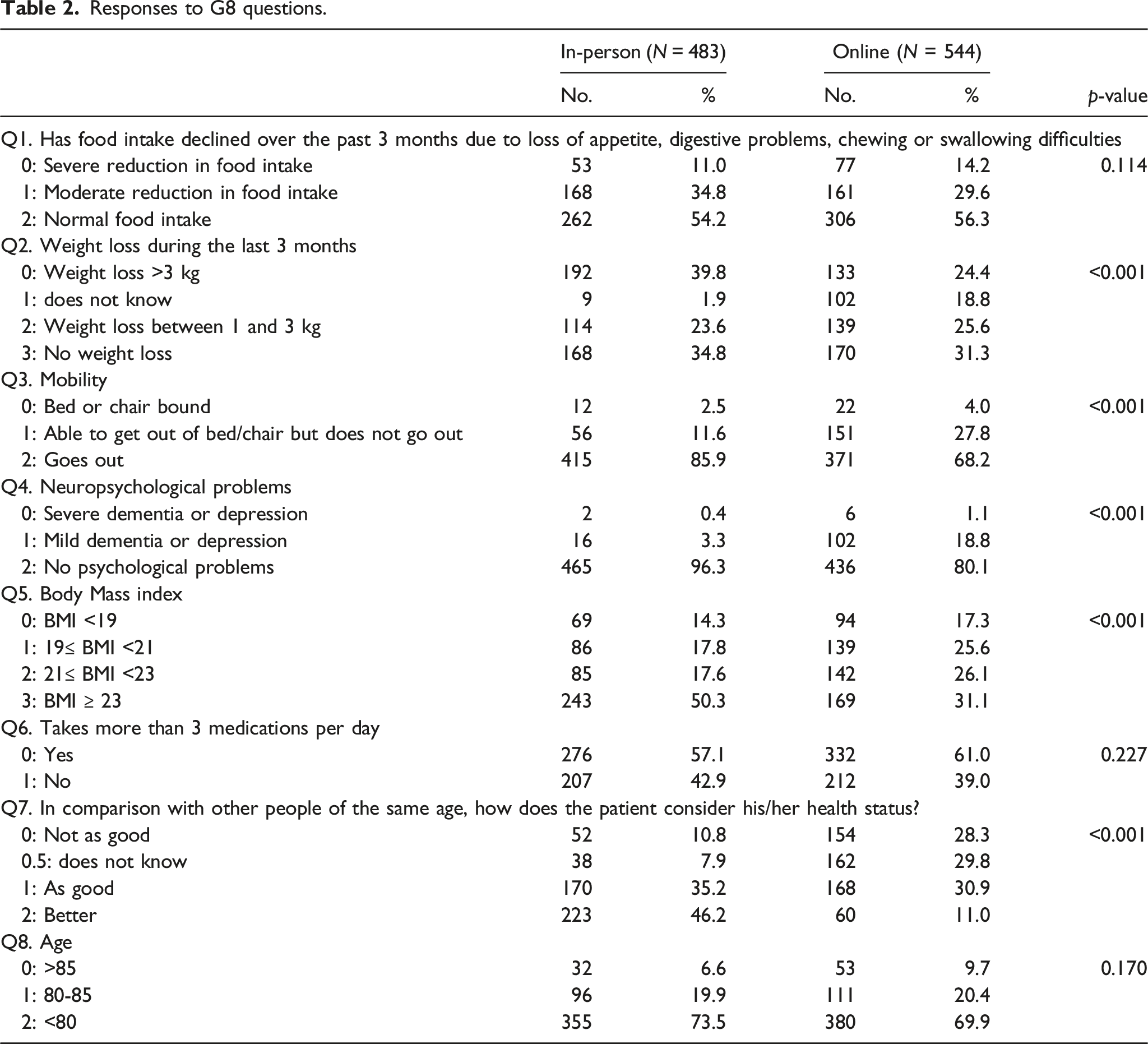
Responses to G8 questions.
G8 total scores.

IQR, interquartile range.
Logistic regression of G8 positive screen.
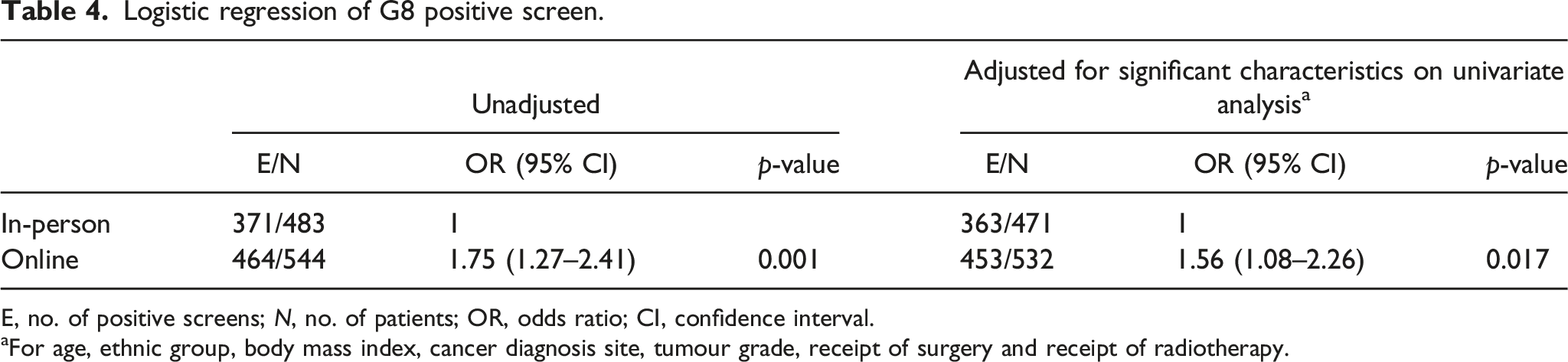
E, no. of positive screens; N, no. of patients; OR, odds ratio; CI, confidence interval.
aFor age, ethnic group, body mass index, cancer diagnosis site, tumour grade, receipt of surgery and receipt of radiotherapy.
Assessment of agreement in G8 scores among patients who did both in-person and online screening
Agreement between online and in-person response to G8 questions.

Concordance correlation coefficient between the online and in-person G8 total scores was high at 0.798 (95% CI, 0.635 - 0.962). There was good agreement between the two sets of scores as seen from the plots in Figure 1. Agreement between online and in-person G8 total scores: (a) Scatter plot of online score against in-person score, with red line denoting the line of identity or perfect agreement. (b) Bland-Altman plot of the difference between online and in-person score against the average of online and in-person score, with the red dotted line denoting the mean difference between scores and the two red dashed lines denoting 95% limits of agreement.
Discussion
There was encouraging response rate among elderly cancer patients to the newly developed self-reported G8 tool in this study. Out of 1088 patients who were invited to complete G8 screening by ZEDOC between December 2001 and December 2002, a total of 544 (50%) completed the online form. It has been reported that survey response rates in the older adults ranging from 26.5% to over 50% from economic studies from different countries. 17 In a local study, a 52% response rates to online surveys were reported. 18 For patients who responded to the online G8, it may have been due to a greater likelihood of having more complaints of concern. For those patients who did not respond to the online G8, it could be attributed to not having a smart phone to assess the online form, not being familiar with the online technology or unable to understand language used. Data was not collected of the non-responders’ reasons for not responding, this could help provide feedback on the improvement of the online G8. As the older adult population become more savvy with the use of smart phones, we expect higher response rates in the future.
There were differences in the G8 scores between the online and in-person screeners. The online screeners had lower G8 total scores than in-person screeners, and had reported lower mobility, more neuropsychological problems, lower body mass index and lower self-assessment of health status in comparison with other people of same age. The lower scores on questions related to more personal questions such as mobility, neuropsychological problems, body mass index and self-assessment of health status could be due to individuals having more privacy when answering these questions, hence, providing more insight to their personal thoughts. Another possibility that we could not preclude is that some online-screeners might have provided lower scores to the various G8 questions intentionally so as to secure a full GA assessment.
Many of the current geriatric screening tools for older patients with cancer require a healthcare worker for the assessment. There are limited studies that compare the agreement of a patient completed screening tool against a healthcare completed tool. 19 In this study, there was a small group of 20 patients who did both mode of G8 screening. While there was substantial agreement between most of the online and in-person responses amongst these patients, this analysis was limited by sample size. As all the 20 patients completed their online G8 screening after they had in-person G8 assessment, they were thus more likely to self-report similar responses in the online G8 form as those they had previously provided to the clinical research assistant during their in-person G8 assessment. The relatively lower agreement in the weight loss and BMI question among the 20 patients suggest that these definitive parameters may be better assessed at clinic rather than depending on subjective reporting by patients. Given this, more research is required to properly assess the agreement between online and in-person responses to G8 screening and to confirm whether online G8 form can replace the in-person assessment.
The mode of screening was an independent predictor of the G8 positive screen in this study, with the odds of G8 positive screen among online screeners 1.56 times that of in-person screeners. This implies that the number of patients identified to require GA would be larger when based on online screening than in-person screening. As the time required for GA is much higher than G8 screening, it is possible that the time and effort saved with the use of an online G8 screening may ultimately be washed out by time and effort required to administer GA to a bigger group of patients. Given this, the replacement of in-person G8 assessment with digital self-reported G8 should be implemented only after more conclusive evidence on the agreement between in-person and online G8 score is available. With the increasing trends of incorporating artificial intelligence (AI) to improve healthcare provision, research can also explore using AI to augment or replace standard online-portal communication. With an ongoing trial that has utilised a Chatbot to facilitate its G8 screening, 20 the use of real-time communication when patients are waiting for their clinic appointments could be the next upgrade.
To the best of our knowledge, we are the first group to compare a digital self-reported G8 against a healthcare professional administrated G8. There are some limitations in this study. First, the study was conducted in a single academic centre in a public health care system, and therefore the results may not be generalizable to other healthcare settings. Second, the information on the number of patients who required help from their caregiver with filling up the online G8 was not available. Therefore, we are not able to assess the acceptability of online screening mode amongst our elderly patients. Feedback on the use of the online form will be collected in future.
In conclusion, we have developed a new digital self-reported G8 that has an encouraging response rate among older adults with cancer. However, given the higher positive screen rate among the online screeners, replacement of the in-person G8 assessment with the digital self-reported G8 should be implemented only after more conclusive evidence on the agreement between in-person and online G8 score is available.
Footnotes
Acknowledgements
The authors would like to thank ZEDOC for providing the online platform
Author contributions
AT and RK researched the literature and conceived the study. AT and OWS study design, data analysis, writing. YLJC data analysis, writing. ACLY study design, data collection. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Centre Singapore Research Fund (NCCSRF).
Ethical statement
Data Availability Statement
The datasets generated and/or analysed during the current study are available from the corresponding authors.
