Abstract
Background
In the COVID-19 pandemic, ventilators vital to keeping infected patients alive, were in short supply globally. Our aim was to rapidly prototype and implement production of basic ventilators to serve the local and regional needs in this emergency situation.
Methods
We adopted a supply-to-design approach, estimating the potential demand for ventilator units and sourcing for common off-the-shelf components available in the estimated quantities, to assemble ventilator units which met the essential requirements for clinical use. We determined the minimum requirements of a basic ventilator based on published specifications and clinician input. Building the ventilator involved interdisciplinary collaboration (between clinicians, industry, hospital innovation engineers and government partners), prototyping and repeated iterations, bench testing, animal testing, regulatory processes, ISO13485 quality management processes, licensing and user acceptability testing.
Results
We prototyped a limited feature ventilator to supplement hospital ventilators which could be manufactured in sufficient numbers within a short span of time from easily available component parts. Developed with close attention to clinician user input with compliance to ISO standards and quality management processes where possible, this ventilator system was composed of coupled resuscitation bags, motor systems, and pressure and flow sensors capable of delivering ventilator breaths within safe and clinically important targets. This system is functional on ambient air with or without low pressure oxygen supplementation. User feedback cited size, alarms and intuitiveness of controls as potential areas for improvement.
Conclusions
Further modification based on user acceptability testing results are needed to refine the usability of this limited feature ventilator.
Keywords
Introduction
The COVID-19 pandemic resulted in millions of infected persons and deaths worldwide. 1 Acute respiratory distress syndrome (ARDS) is a major feature of severe infection, requiring intensive care and ventilation. Rapid spread of this disease overwhelmed the healthcare infrastructure in many countries, even in the developed world, particularly in the early phase of the pandemic. Various reports estimated 12.3–16.0% of hospitalized patients with COVID-19 required invasive mechanical ventilation2,3 leading to a worldwide shortage of ventilators. 4 Moreover, critically ill patients spent an average of 4–18 days on the ventilator.5–7 The estimated number of ventilators needed by U.S. patients with COVID-19 could be as high as a million, requiring mobilization of stockpile/storage units, non-invasive devices and limited-feature devices. 8 Ventilators were vital medical devices in this COVID-19 pandemic, keeping infected patients alive.
A conventional ventilator is highly sophisticated and contains hundreds of specialized parts, costing over SGD$35,000 and taking significant time and effort to make. Even with ramping up of conventional ventilator production, the maximum production speed was slow, limited by the complexity of the parts and need to obtain them from a variety of global sources amidst supply chain disruptions. Even where building ventilators under special licences, manufacturers have been challenged by the need to re-tool and revamp their production lines to meet the demand in the short time frame required to cope with a surge in infections. In addition to a shortage of qualified staff, a shortage of conventional ventilators and inability to obtain/manufacture them resulted in widespread shortages, triaging of patients for ventilators and even sharing of ventilators by several patients in some countries. 9 Hence, there was an urgent need to design, prototype and produce low-cost ventilators with the necessary minimum capabilities to support a surge in patients requiring ventilation. Media stories abounded with rapidly-prototyped ventilators. However, these bare-bones ventilators described on the internet suffered from many limitations such as only having a mandatory ventilation mode and not being suitable for spontaneously breathing and lightly sedated patients.
Our aim was thus to rapidly prototype and implement production of basic ventilators to serve local and regional needs in a respiratory pandemic or other emergency/crisis situation, which also met the clinical requirements to enable safe ventilation and weaning of respiratory support. We adopted a novel supply-to-design approach, estimating the potential demand for ventilator units and sourcing common off-the-shelf components available in the estimated quantities, to assemble ventilator units which met the essential requirements for clinical ventilation of patients and could be produced in quantities estimated to meet our worst-case scenario projections. User requirements were obtained from clinicians from four major hospitals, and included respiratory and critical care medicine specialists, adult and paediatric intensivists, respiratory therapists, and anaesthesiologists. Design and development were conducted in compliance with an ISO13485 quality management system where possible. This would enable us to ventilate patients at a low cost, avoid heavy sedation of patients to prevent ventilator dyssynchrony, minimise ventilator-associated complications from the latter and enable operation by non-expert users in a pandemic situation.
Methods
Design conception
Design specifications for minimum viable product.

Must-have specifications (M) and Good-to-have specifications (G).
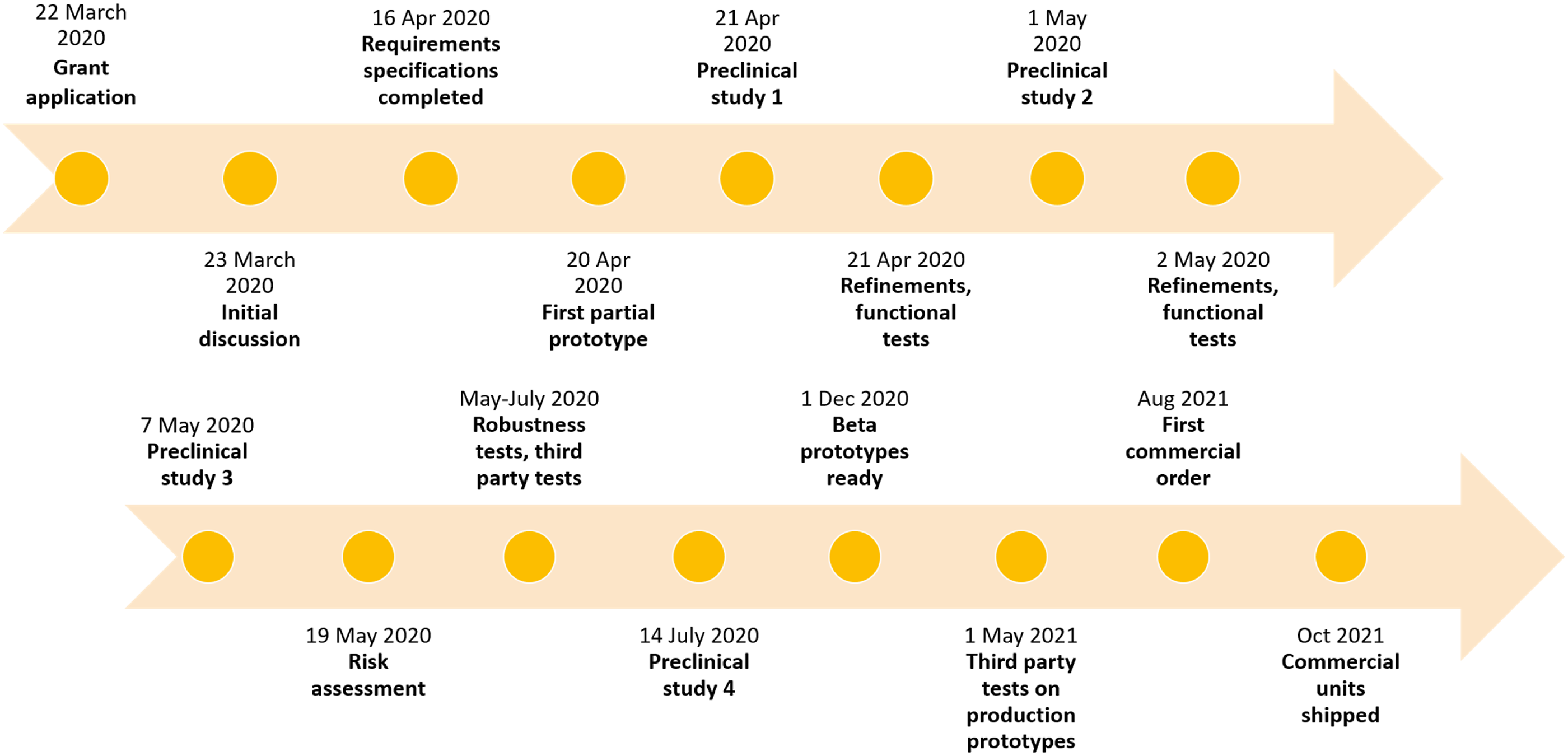
Accelerated development timeline.
Coordination and collaboration
Roles of interdisciplinary team members.

Applying the Stanford Biodesign (best practices for medicaltechnology innovation) rapid-iterative approach of “fail fast, fail early”, 11 our team engineers prototyped the first design within 3 weeks of the initial project discussion. Industry partners came on board and the design was largely finalized 2 months from the project start date. Thereafter, the prototype transitioned into the beta version over the following few months as further refinements were made to address specific issues identified in testing and the Bill of Materials was finalized. Prototype assembly raised learning points in the laboratory such as how to install, pre-configure and pre-test key components and parts, common mistakes and issues, which were passed on to the production team and recorded in the design documentation as well. A ventilator test protocol was developed and administered by our intensivist (JW), the ventilator’s performance limits quantified and the results detailed in the user’s manual.
Prototyping and iterations process
From the clinical requirements, the engineering specifications were determined and our team connected with local industry partners for the prototype build (Trilogy Technology Pte Ltd), component supply (Akribis Pte Ltd, IFM Electronic Pte Ltd and others) and software programming (Vixel Inc Pte Ltd). Frequent iterative discussions between engineers, clinicians and industry partners took place to iron out technical specifications. The team adopted a supply-to-design approach, which meant that the ventilator was designed with component parts that were available at targeted quantities (initial estimate of 500 units within 4–6 weeks). Local supply chain constraints meant only a few vendors could meet the lead time and minimum order requirement. In contrast to the conventional Biodesign model, which develops several concepts simultaneously after a wide-ranging brainstorm and selects those with the lowest commercial and regulatory risks to go to market, our team focused on only one design at a time. 11 The design was iterated as it was tested, optimizing performance at each iteration, and the lessons learned incorporated into the subsequent design. In total, there were five design iterations and four preclinical in vivo studies.
Bench testing
To ensure technical and clinical feasibility, pre-clinical studies were conducted. Detailed characterization of ventilator performance was tested using a gas flow analyzer (Fluke VT650, Medtronic) and an artificial lung simulating different resistance and compliance characteristics of the human lung (Smart Lung 2000, IMT Medical) in the Medical Technology Office facility and the SingHealth Simulation Centre.
Animal testing
The SGINSPIRE ventilator was tested in a physiological animal model to determine its 1) ability to deliver programmed settings in a normal respiratory system, 2) efficacy of ventilation and oxygenation, 3) safety in monitoring and alarms, 4) reproducibility of programmed settings, and 5) assessment of the trigger sensitivity in synchronized modes of ventilation. The testing protocol complies with the National Advisory Committee for laboratory Animal Research (NACLAR) guidelines and was approved by the Institutional Animal Care and Use Committee (IACUC). Four adult Yorkshire landrace pigs were used throughout the testing protocol. In brief, the pigs were anesthetized, intubated and ventilated using the SGINSPIRE ventilator (the full animal study protocol is available in the supplementary material). Prescribed ventilator settings on SGINSPIRE were independently checked. The delivered peak inspiratory pressure (PIP) and peak end expiratory pressure (PEEP) were obtained using an analogue manometer (Marshall Town). The delivered fraction of inspired oxygen (FiO2) was obtained using an oxygen analyser (Maxtec MaxO2+). The flow time curves were obtained by using a bidirectional flow sensor (Sensirion mass flow meter SFM3019). Inspiratory time (Ti) was then estimated based on the duration of positive deflection of these curves. The manometer, oxygen analyser and flow sensor were sequentially attached between the endotracheal tube and ventilator circuit to obtain measurements. Tidal volume (TV) was measured using a Wright’s respirometer (Anesthesia Associates Inc Haloscale Wright Respirometer) attached to the distal end of the expiratory limb of the circuit (near the endotracheal tube). Oxygenation was assessed continuously by pulse oximetry and correlated with intermittent partial pressure of arterial oxygen measurements. Ventilation was assessed continuously by side stream end tidal carbon dioxide (Medtronic Capnostream 35 Portable Respiratory Monitor) and correlated with intermittent partial pressure of carbon dioxide measurements. Point of care blood gases were analysed using (Siemens Healthineers epoc blood analysis system). Measured rate was obtained from the end-tidal rate. We also compared the performance of SGINSPIRE against a commercially available conventional ventilator – the full feature mechanical ventilator (Drager Fabius Plus) was used as the comparator.
Regulatory processes
We followed a quality management system (QMS) and risk management framework following ISO13485 and ISO14971 standards to generate a high-quality and safe medical device within the given time, cost and supply constraints. The risk assessments influenced design to ensure essential capabilities were fail-safe. For example, initial software prototyping was done using Arduino controllers and a Raspberry Pi microprocessor, which were more widely available and cheaper than industrial computers. The design team’s risk assessment of the reliability of these microprocessors and controllers was 90–95%. This was insufficiently robust for ventilators and was therefore mitigated by incorporating redundancy, using two master-and-slave sets, with heartbeat monitoring implemented and failover measures to automatically take over and sound an alarm if the master unit failed.
To align with the minimum requirements for emergency-use approval in an expedited timeframe, design documents and results of functional, benchtop and preclinical testing were submitted to the Singapore Health Science Authority (HSA) after completion of the initial design. Along the iterative process, further updates were given to HSA. HSA in turn provided prompt response and guidance on the regulatory process, further testing and recommended standards for medical devices.
To meet regulatory and safety requirements, electromagnetic compatibility (EMC) tests, dielectric (high potential) tests, burn-in tests (running the ventilator continuously over several weeks to ensure durability), leak tests and battery lifespan tests were conducted in parallel with other pre-clinical tests.
Licensing and manufacturing process
Traditionally innovation favours a concept that has potential for patentable intellectual property, which is valued by investors. However our approach avoided novel concepts which would have required prolonged development, carry a high risk of failure and introduce barriers to regulatory approval, opting instead to adopt and adapt tried and tested technology with lower risk for regulatory approval but lacking patent potential. Instead the team filed an invention disclosure to cover the know-how such as test protocols, results, and other technical and design information.
Our team worked closely with competent and ISO 13485-certified partners to create a high quality product with best-effort compliance to regulatory requirements despite the constraints imposed by the pandemic. Relevant design history documents were captured and handed over to the subsequent licensee/manufacturer and a set of three ventilators close to design-for-manufacture were produced to validate the manufacturing process. Approval was sought and obtained from the SingHealth pandemic taskforce for user acceptability testing and SGINSPIRE was deployed to local hospitals for comprehensive user acceptability testing.
User acceptability testing
User acceptability testing was performed by clinical staff including intensive care physicians, nurses and respiratory therapists from four local institutions. Staff, both trained and untrained in ICU care were involved in the user acceptability testing. Feedback on intuitiveness and usability was obtained with a standardised ventilator evaluation checklist. Major and minor comments will be considered in further iterations of the prototype. Deployment obstacles will also be identified and addressed. In so doing, the necessary processes will be streamlined for rapid implementation in the event of future surges in ventilator demand.
Results
We designed a limited feature ventilator composed of coupled resuscitation bags, motor systems and pressure and flow sensors, capable of delivering ventilator breaths within clinically important target parameters (Figure 2). Generic resuscitation bags were used in the design and build of the device. Mechanized compression of the resuscitation bag produce non-linear dynamics in pressure and air, which required tuning of the algorithm. At higher pressure settings the second coupled resuscitation bag connected in a parallel circuit activates to achieve the desired pressure, volume and flow characteristics. Integrated sensors enable precise adjustments of ventilator pressure, detection of harmful volume limits and support patient-triggered ventilation. SGINSPIRE is able to accommodate both pressurised (tank) and non-pressurised gas flow (ambient air), a key feature absent in conventional ventilators dependent on gas pressure to function. Use of disposable off-the-shelf components and an easy-to-use design mitigated the need for intense staff training. The first completed design is shown in Figure 2. With this prototype, our team proceeded to conduct independent third-party tests necessary for regulatory approval. SG-INSPIRE - High-level functional design layout.
Animal testing
PIP was titrated from 10 to 40 cmH20, PEEP 5 to 20 cmH20, rate 10–30 breaths per minute, and Ti 1.0–1.4 s. Set and measured PIP and PEEP were highly correlated and had good agreement within most of these clinically relevant ranges (Figure 3). End tidal carbon dioxide was seen to decrease with increasing driving pressure/tidal volume However, at high pressures (>30 cmH20), set and measured PIP did not have good agreement (Supplementary Figure 1). Set and measured Ti was also highly correlated and rate correlated perfectly. There was a stepwise increase in measured tidal volume (TV) as driving pressure was increased from 5 to 30 cmH20, however, this was not as high compared to the TV measured on the conventional full feature ventilator. Results of animal studies in titrating ventilator parameters.
User acceptability testing
User acceptability testing was performed at five sites and involved 86 members of staff. The results are listed in Figure 4. Additional comments identified size, alarms, intuitiveness of controls, oxygen sensor, noise, display format, lack of volume control mode, oxygen inlet connector and trigger sensitivity as considerations for further improvement. User acceptability testing results.
Commercialization and reimbursement
Development of the SGINSPIRE system was completed for under SGD$400,000. A limited quantity of SGINSPIRE ventilators were purchased by an overseas buyer, with the understanding that the emergency-use approval from the overseas regulatory agency was required prior to use. This sale included an option for remote maintenance for a duration of 6 months. As the project was undertaken on compassionate grounds, the ventilators were sold on a non-profit basis, and out-licensing to the manufacturer was also done without charge. At the time of manuscript review, there were no feedback, queries or requests for technical support raised by the buyers.
Discussion and conclusion
We designed a limited feature ventilator which could be manufactured in sufficient numbers within a short span of time from existing component parts to supplement hospital ventilators should a rise in demand occur due to the pandemic. This ventilator system was composed of coupled resuscitation bags, motor systems and pressure and flow sensors, capable of delivering ventilator breaths within clinically important target parameters. This is in contrast with full-feature commercial ventilators which may have superior performance and other capabilities, but in normal circumstances are available in small numbers, require specialized components, require guaranteed pressurized gas supply to function, need skilled staff to operate them and cannot be produced rapidly in a pandemic situation. Due to challenges associated with obtaining sufficient suitable parts, we engaged industry and hospital innovation units, and government partners to obtain the necessary components. The design team further adapted the design to match available components.
Singapore was in partial lock down through most of the development phase of this project. Apart from not being able to meet, the industry partners could not enter clinical settings to see how the SGINSPIRE would actually be used. Supply chain constraints also arose as many vendors were made to shut down operations during this period. The project lead clinicians involved had to write supporting letters to the Ministry of Trade and Industry to allow key vendors to stay open. As the work was carried out against a backdrop of safe distancing restrictions, the worry of rising local infection rates, with long hours and late nights over weekends and public holidays leading to exhaustion among the team, burn-out was a real concern. However, the team’s camaraderie, as well as, support from vendors eager to contribute beyond the call of duty, provided strong encouragement to persevere.
Multiple considerations were required to ensure the production of the single build prototype designed could be upscaled to meet surge conditions. When sourcing for component parts the team had to focus on components which were familiar to the existing staff and for which large quantities could be acquired in a short timeframe of weeks. Electronic messaging and video conferencing facilitated progress despite physical separation of team members across different sites in the country. Application for institutional funding and sourcing for components occurred concurrently. This process entailed close cooperation between the SingHealth Medical Technology Office, Office of Research, ALPS (the Singapore healthcare procurement and supply chain entity) and support from the SingHealth Office of Education.
The Medical Technology Office’s CIEs played a key role to propose and discuss key design decisions with all parties and incorporate clinical user feedback. The CIEs had to understand the limitations for clinical use of SGINSPIRE due to technical considerations. For example, the decision to use resuscitator bags in the design was made due to the disadvantages of longer development time, component supply lead time, and regulatory risk profile for non-resuscitator bag designs. The team therefore had to deal with the technical difficulty of non-linear pressure generation with a given compression by modifying the compression pattern. Use of resuscitator bags also raised concerns over the limited lifespan of this component, necessitating testing to ensure that the product life span met the clinical requirement. The team introduced a maintenance protocol to change the resuscitator bags, which aligned with the projected duration of a patient on assisted ventilation and elegantly also removed the risk of cross-infection between patients by removing most of the air circuit components exposed to the patient. For every key decision, the CIEs would need to communicate to clinicians the limitations in ventilator performance, while balancing against the availability of alternative parts and cost considerations. The CIEs also communicated with industry teams on engineering specifications required to achieve acceptable performance from the clinical perspective.
This project’s greatest strength was the interdisciplinary teamwork demonstrated by all participants. Valuable insights were provided by clinical end users who continuously engaged with industry partners during the development phase and user testing phase. The CIEs role in integrating clinical and technical requirements was key to completion of an acceptable product. Strict quality management system for the product development process incorporating the relevant ISO standards produced the highest possible quality product, with the necessary documentation, records and supporting third party test results, despite the constraints faced. Finally the UAT provided valuable feedback on users’ assumptions and expectations when using a ventilator. Fortunately, the pandemic in Singapore did not experience the grave trajectory of some other countries and the number of patients requiring ventilation did not exceed the capacity of the healthcare system. There were also several limitations. The use of this ventilator has not yet been tested in simulated scenarios of varying respiratory system compliance and resistance, nor in the clinical setting. In the current bench and animal model, the ventilator was not able to deliver airway pressures above 28 cmH20 and delivery of oxygen could not be accurately measured. Though the inspiratory trigger was set at 2 cmH20 below PEEP, a detailed assessment of trigger sensitivity has not been performed. As such, the use of this experimental ventilator cannot be recommended for any critically ill patients in whom a conventional ventilator is available.
We demonstrated that it is feasible to undertake an accelerated innovation journey which contrasts with traditional innovation development cycles. Starting with a clearly defined unmet need based on expert clinical opinion, translating into design specifications and producing multiple iterations, a functioning ventilator system suitable for pandemics and/or limited-resource settings was created at low development cost in an academic medical setting – SingHealth Duke-NUS.
Supplemental Material
Supplemental Material - Real-world considerations for the rapid prototyping and manufacture of a ventilator for the COVID-19 pandemic
Supplemental Material for Real-world considerations for the rapid prototyping and manufacture of a ventilator for the COVID-19 pandemic by Judith Ju Ming Wong, Fiona Wei Ling Loke, Herng Lee Tan, Jessica Lishan Quah, Wai Chye Cheong, Shariq Ali Khan, Biju Thomas, Ghee Chee Phua and Derrick Wei Shih Chan in Proceedings of Singapore Healthcare.
Footnotes
Author contributions
JJMW, FWLL, HLT, JLQ, WCC, SAK, BT, GCP and DWSC made substantial contributions to the conception and design of the work, data acquisition, analysis and interpretation. JJMW and FWLL drafted the manuscript. HLT, JLQ, WCC, SAK, BT, GCP and DWSC revised the manuscript critically for important intellectual content. All authors approve the final version of the manuscript to be published and agree to be accountable for all aspects of the work ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Commercial partners of SGINSPIRE include Trilogy Tech Pte Ltd, Akribis Pte Ltd, Vixel Inc Pte Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SGINSPIRE was funded by KK Women’s and Children’s Hospital Health Services Innovation and Development grant (PI: JJMW and PI: DWSC), SingHealth Duke-NUS Academic Medicine COVID-19 Grant (PI: DWSC) and SingHealth Duke-NUS Academic Medicine Special Request for Urgent COVID-19 Research Funding (PI: DWSC).
Ethical approval
(include full name of committee approving the research and if available mention reference number of that approval) This study does not involve human research subjects. The animal testing protocol complies with the National Advisory Committee for laboratory Animal Research (NACLAR) guidelines and was approved by the Institutional Animal Care and Use Committee (IACUC) reference number 202003-0012.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
