Abstract
Background
The concept of a pharmacist-led collaborative gout clinic is relatively new in Singapore.
Objective
This study examined the impact of this pilot shared care model on providing dose titration of urate lowering therapy, gout education and patient support to achieve target serum uric acid (sUA) levels.
Method
A retrospective pre-post study was undertaken to compare outcomes in patients who were started on either allopurinol or febuxostat in the 24 months prior to (Group A: Pre implementation) or 33 months following launch of the collaborative gout clinic (Group B: Post implementation). The collaborative gout clinic comprises of a clinical pharmacist under the supervision of a rheumatologist.
Results
Of 98 eligible subjects enrolled in our study, there were 50 patients (all prescribed allopurinol) for Group A and 48 patients for Group B (allopurinol n = 29, febuxostat n = 19). Among patients who achieved target sUA level of 360 μmol/L or less at 1 year of drug initiation with use of allopurinol, the median [interquartile range, IQR] time taken to attain target sUA was shorter in Group B than Group A (111 [82–308] days vs. 293 [265–414] days, p = 0.016). As compared to Group A, Group B had lesser patients experiencing gout flare (41.3% vs. 70.0%, p = 0.018) and more patients achieving target sUA (75.9% vs. 22.0%, p < 0.001). Mean ± [standard deviation, SD] daily allopurinol dose to achieve target sUA levels was 276 mg ± [138 mg].
Conclusion
Our results demonstrate the use of a pharmacist-led collaborative gout clinic may help to achieve better clinical outcomes in gout management.
Introduction
Gout is the most common inflammatory arthritis in men with prevalence increasing in many developed countries including Singapore. 1 Poorly controlled gout is not only associated with pain, disability and reduced quality of life, it is also linked with lower productivity, increased healthcare costs, and is an independent risk factor for cardiovascular disease and mortality. 2 Studies have also reported that gout is associated with increased risk of developing diabetes 2 and hypertension. 3
The treat-to-target serum uric acid (sUA) levels is a widely accepted treatment approach in the management of gout. The American College of Rheumatology 4 and the European League against Rheumatism 5 recommend treatment of gout with urate lowering therapy (ULT) to achieve and maintain sUA levels to 360 μmol/L or lesser for most patients. Unfortunately, international studies have shown that less than one third of patients on ULT attained treatment goals.6,7 Similarly in Singapore, an audit of the rheumatology clinic in a restructured hospital found that only 25% of patients started on allopurinol had achieved their target sUA levels at 1 year follow up. 8
Possible reasons 9 for this gap between optimal gout management and actual clinical practice include lack of patient awareness of the disease condition which possibly translates to poor adherence to gout pharmacotherapy and diet. In addition, physician factors including non-familiarity with current guidelines for gout, fear of escalation of allopurinol dose in some patients, particularly those with chronic kidney disease and lack of appropriate monitoring could contribute to not attaining target sUAs.
In Singapore, we also face a challenge of practicing this intensified treat-to-target approach in a busy, restructured hospital with a high patient-to-doctor ratio, hence, the low proportion of patients achieving target. To address this problem, a pilot collaborative gout clinic was developed in 2014 comprising a clinical pharmacist under the supervision of a rheumatologist. Following a written protocol, the pharmacist monitored and titrated doses of ULT for patients referred by the rheumatologist for gout management. The pharmacist also provided education on gout disease, medications and diet to the patients. Pharmacist-based gout management had been implemented with good outcomes in other countries 10 but was a relatively new treatment initiative in Singapore. Therefore, the aim of this study was to evaluate the impact of a pilot pharmacist-led collaborative gout clinic on providing dose titration of ULT, gout education and patient support to achieve target sUA levels. We looked at the time taken to achieve target sUA, proportion of patients experiencing gout flare as well as the proportion of patients who achieved target sUA of 360 μmol/L or less at 1 year of drug initiation.
Methods
Design
A pilot retrospective pre-post observational case series study comparing ULT outcomes in patients managed by a rheumatologist only and another group managed by a rheumatologist and the pharmacist-led collaborative gout clinic was performed at Changi General Hospital, Singapore. The protocol for this research project was approved by the SingHealth Centralised Institutional Review Board.
Patient selection and referral
Potential study subjects were initially identified by generating a list of patients who received a prescription for allopurinol or febuxostat using the hospital’s pharmacy computer dispensing system from March 2012 to November 2016.
Inclusion criteria required patients with a confirmed clinical diagnosis of gout (based on clinical presentation and raised sUA of more than 360 μmol/L) by a rheumatologist, who were started on ULT (Allopurinol or Febuxostat) by a rheumatologist in the 24 months (from March 2012 to Feb 2014) prior to the launch of the collaborative gout clinic (Group A) and the 33 months (from March 2014 to November 2016) following the launch of the collaborative gout clinic (Group B). As this was a pilot study, sample size was determined according to rules of thumb. 11 A general rule was to take 30 subjects or greater to estimate a parameter 12 which in our study, time to achieve sUA goal. A sample size of 50 patients for each group was used. Due to the low acceptance rate of the pharmacist-led collaborative gout clinic by patients, it took a longer duration to recruit sufficient patients for Group B.
Patients were excluded from the study if they have an estimated glomerular filtration rate (eGFR) of less than 15 mL/min (immediately prior to commencement of ULT), those with incomplete data (for example, defaulted their appointments) and those who required change in medications halfway through the study.
A total of 2 rheumatologists and 3 pharmacists participated in this study. The pharmacists were trained by the rheumatologists using an in-house training competency framework which included 40 hours of didactics and 30 patient cases of clerkship. Clinic prescribing privileges would be granted to the pharmacist subsequently after passing a competency test.
Treatment protocol
For both groups, baseline laboratory assessment performed on all patients consisted of a sUA level, alanine aminotransferase (ALT), aspartate aminotransferase (AST), serum creatinine (sCr), eGFR and full blood count (FBC). This same panel of laboratory tests was repeated as needed to monitor the progress of the patient. A schematic diagram of the assessment and treatment protocol is shown in Figure 1. Assessment and treatment protocol that patients in Group A and B underwent respectively. This study was conducted in patients managed by a rheumatologist only (Group A: Pre implementation of the collaborative gout clinic) and in patients managed by the pharmacist-led collaborative gout clinic (Group B: Post implementation of the collaborative gout clinic).
For both groups, once a baseline laboratory assessment was available, the rheumatologist would start or adjust the ULT if the serum uric acid level was above 360 μmol/L. After starting ULT or change in dose, the patient was instructed to return for laboratory assessment (sUA, FBC, ALT, AST, SCr, eGFR) and report any adverse drug reactions (ADRs) or gout symptoms. Dose of ULT was adjusted and this process was repeated until an initial target sUA of ≤360 μmol/L was achieved or until the trial ended 1 year after drug initiation.
Group A (Pre implementation of the pharmacist-led collaborative gout clinic)
In this group, the patients were required to return to the rheumatologist in 12–14 weeks after initiation of ULT for review. Each consultation session with the rheumatologist lasted approximately 15 min.
Group B (Post implementation of the pharmacist-led collaborative gout clinic)
The decision and action process of the collaborative gout clinic required: (1) The rheumatologist ordered the ULT and referred the patient to the pharmacist with sUA target specified. Referring criteria for patients include patient safely initiated on ULT, did not require clinical examination and agreeable to review with the pharmacist with a rheumatologist follow up. (2) During the initial pharmacist consultation, the pharmacist would counsel and provide patients with written educational material regarding gout treatment that included, disease, dietary and drug information (administration, adverse effects) for allopurinol or febuxostat. The pharmacist, under an agreement approved by the hospital’s medical board, was also authorized to order relevant laboratory tests and initiate or change orders for the medications used to manage serum uric acid levels, flare prophylaxis and acute flares. Abnormal laboratory results, ADRs or gout flares were managed by the pharmacist, usually in consultation with the supervising rheumatologist (see Appendix 1 and 2). Each consultation session with pharmacist lasted approximately 20 min. For patients who did not achieve target sUA, they were required to return to the pharmacist-led clinic in 4 to 6 weeks for review. (3) For patients who achieved target sUA, they were kept on the same dose of ULT with reinforcement on medication adherence and other counselling advice. These patients were subsequently discharged from the pharmacist service and referred back to the rheumatologist.
Pharmacological treatments
ULT was initiated with either allopurinol or febuxostat. The starting dose for most allopurinol naïve patients was 100 mg daily (some patients were prescribed higher doses at the time of referral while some patients were started on lower doses as per their renal function 13 ). Doses for allopurinol range from 50 mg every other day to 200 mg/day. The starting dose for febuxostat naïve patients was 40 mg daily. Dose titration for patients not at target sUA was performed using 50–100 mg/day increments for allopurinol and 40 mg/day increments for febuxostat as per the hospital’s in-house guidelines (Appendix 1) that were approved by the hospital’s pharmacy and therapeutics committee. As part of the protocol of commencing ULT, gout prophylaxis was used in most patients and colchicine was the choice of prophylaxis.
Acute gout flares were managed by the pharmacist, in consultation with the rheumatologist on duty, using oral non-steroidal anti-inflammatory drugs (NSAIDs), prednisolone or colchicine.
ADRs to medications, incident gout flares and abnormal laboratory parameters were recorded and managed by the reviewing pharmacist at each clinic session in consultation with the supervising rheumatologist in accordance to in-house guidelines (Appendix 2).
Outcome variables
The primary efficacy end-point was time taken to initially attain goal sUA levels of 360 μmol/L or less in patients who achieved target sUA, with a comparison between Group A and B. Secondary outcomes included the proportion of patients who experienced a gout flare in the year after initiation of ULT, the mean dose of ULT required to achieve target sUA levels, and the proportion of patients that achieved target sUA of 360 μmol/L or less at 1 year of drug initiation. In addition, we performed a survival analysis looking at the proportion of patients achieving target at 3, 6 and 12 months respectively, with a comparison between the two groups.
Number of patients who selected the respective level of agreement and the average score on the pharmacist-led collaborative gout clinic survey.

Statistical analyses
All analyses of categorical variables were conducted with Chi squared test and all analyses of continuous variables were analyzed with either the Mann–Whitney U Test or Student T-test. The primary efficacy end-point of time taken to initially attain goal sUA levels of 360 μmol/L or less in patients who achieved target sUA between both groups was analysed with the Mann–Whitney U Test as the data was not normally distributed. Analyses were performed using SPSS Version 19, using a 0.05 alpha level.
Results
Initial screening identified 153 potentially eligible patients for Group A and 73 patients for Group B from the hospital electronic medication records. The lower number of potentially eligible patients for Group B was attributed to the lower acceptance rate of patients to see the pharmacist. The records were then reviewed for eligibility by a rheumatologist and a pharmacist to validate the inclusion and exclusion criteria. The number of patients who met the inclusion criteria and who were initiated on ULT therapy comprised 50 patients (all prescribed allopurinol) for Group A and 48 patients for Group B (allopurinol n = 29, febuxostat n = 19) as depicted in Figure 2. Febuxostat was only available for prescribing after the start of the collaborative gout clinic as it was only approved for inclusion in the hospital formulary from 2014. Since febuxostat was only available to patients who attended the collaborative clinic, for the analysis, only patients who were prescribed allopurinol in Group B were compared with Group A. Outcome on febuxostat, i.e. time taken to achieve target sUA, was reported without having an in-between group comparison. Flow diagram depicting the recruitment process of patients. This study was conducted in patients managed by a rheumatologist only (Group A: Pre implementation of the collaborative gout clinic) and in patients managed by the pharmacist-led collaborative gout clinic (Group B: Post implementation of the collaborative gout clinic).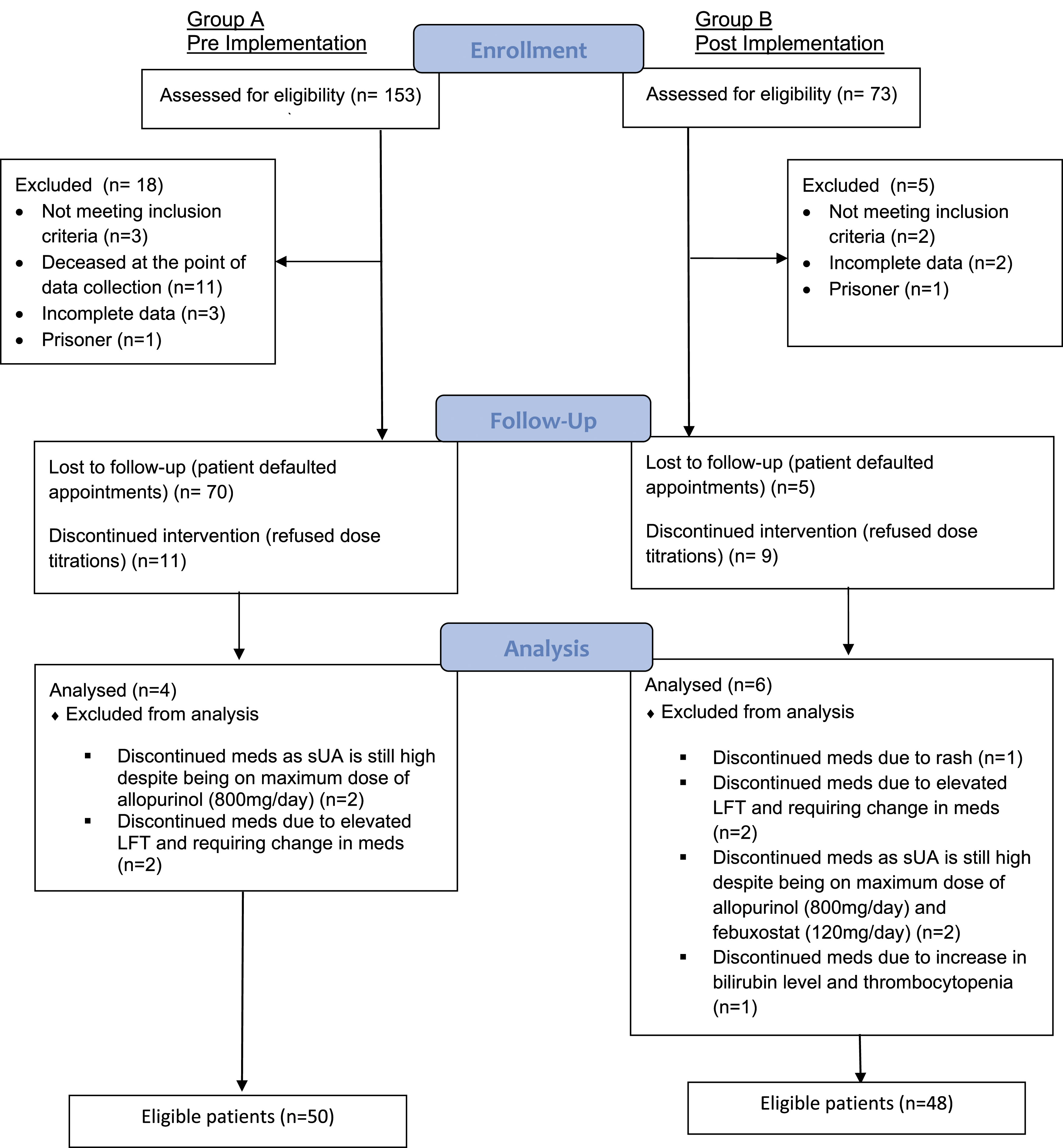
Patient demographics for Group A (patients managed by a rheumatologist only [pre implementation of the collaborative gout clinic]) and Group B (patients managed by the pharmacist-led collaborative gout clinic [post implementation of the collaborative gout clinic]).

*Performed using Chi squared test, **Performed using Student T-test.
aAs defined by eGFR prior to commencement of ULT and chronic kidney disease (CKD) criteria.
Summary of outcomes between Group A (patients managed by a rheumatologist only [pre implementation of the collaborative gout clinic]) and Group B (patients managed by the pharmacist-led collaborative gout clinic [post implementation of the collaborative gout clinic]).

*Performed using Mann–Whitney U Test, **Performed using Chi squared Test.
The proportion of patients who experienced at least one gout flare at 1 year of drug initiation was significantly lower in Group B as compared to Group A (41.3% vs. 70.0%, p = 0.012) as shown in Table 3.
The mean ± [standard deviation, SD] daily allopurinol dose to achieve target sUA levels was 276 mg ± [138 mg]. There were 79% of patients on allopurinol who achieved the target on 300 mg/day or less. Only two patients achieved the goal on the starting dose of 100 mg daily, one patient achieved target sUA with dose of 150 mg/day, three patients required 400 mg/day and one patient required 700 mg/day.
A higher proportion of patients achieved sUA of 360 μmol/L or less at 1 year of drug initiation in group B compared to group A (75.9% vs. 22.0%, p < 0.001) as shown in Table 3. To provide a more detailed view of the proportion of patients achieving sUA target over the year, the percentage of patients achieving target at 3-, 6- and 12-months of starting a ULT is detailed in Figure 3. Percentage (Number) of patients achieving target sUA of 360 μmol/L or less. A higher proportion of patients achieved sUA of 360 μmol/L or less in Group B compared to Group A at 3-, 6- and 12-month of starting a ULT. This study was conducted in patients managed by a rheumatologist only (Group A: Pre implementation of the collaborative gout clinic) and in patients managed by the pharmacist-led collaborative gout clinic (Group B: Post implementation of the collaborative gout clinic). Statistical analysis by Chi-squared test, **p = 0.01, ****p < 0.001.
A separate analysis on the patients who commenced febuxostat noted that the mean ± [SD] time taken to achieve target sUA was 120 ± [56] days. This result differed from other studies 15 that reported the use of febuxostat achieving serum uric acid target at an average of 86 days. However, our result should be interpreted with caution due to the small sample size.
Two patients (2%) experienced a rash with allopurinol. No patient required treatment other than discontinuation of the drug. One patient was switched to febuxostat while the other was re-challenged with no issues. Elevation of ALT was observed during the treatment of allopurinol in 10 patients. Of these, 4 patients required a change in medication to either probenecid or febuxostat. The rest of the patients stabilized or returned to normal with continued treatment and monitoring.
The questionnaire response rate was 39.6% (19/48). The results revealed that the collaborative gout clinic received positive feedback from the patients, with an overall high mean score of 4.53 out of 5 as shown in Table 1. The patients felt that the pharmacist provided clear information on the medications, the possible side effects of medications and non-pharmacological measures (mean score of 4.53) and they had a better understanding of their condition and how to manage it (mean score of 4.42). Most patients expressed that they were more likely to adhere to treatment (mean score of 4.42) and they were willing to come back for follow-up with the collaborative gout clinic (mean score of 4.42).
Discussion
Gout is a well-understood disease with effective treatment options readily available and what should be a straightforward recommended treatment approach to achieve sUA levels of 360 μmol/L or less. In addition, achieving treatment target has been associated with lower incidence of gout flares.16,17 Many reasons for poor management of gout have been identified with the important basis of our study being the failure to implement an intensified treat-to-target gout management in a busy restructured hospital with high doctor-patient ratio.
In our study, patients were seen by the pharmacist every 4 to 6 weeks compared to the usual care follow up interval of 12 to 14 weeks with the rheumatologist. As a result, due to the more frequent access and review by the pharmacist, not only a higher percentage of patients achieved target sUA level, the target was achieved at a faster rate, leading to less gout flares. In addition, with pharmacists providing gout education, dose titration, and patient support, there had been lesser patients lost to follow-up compared to usual care group, translating to more patients achieving target sUA in a shorter time. This is also reflected in similar pharmacist-based programs implemented in other countries,10,18 allowing patients to effectively and safely achieve treatment target.
There is ample evidence that fear of allopurinol dose escalation in certain patient populations contributes to inadequate management of gout. 19 Our program and protocol were designed to overcome this problem by providing close monitoring of repeated measurements of sUA and other relevant blood tests so that timely interventions could be done to help patients achieve target sUAs safely, including those with impaired renal functions. Our protocol did not limit the dose of allopurinol according to renal function as per Hande criteria, 20 which was one of the several impediments to successful gout treatment as noted in literature.21–23 Our observations confirmed this: only 2 (7%) patients and 1 (3%) patient, out of those who were taking allopurinol in the post group, achieved target with dose of 100 mg/day and 150 mg/day respectively.
Although not formally assessed and recorded, part of the clinic interaction and education involved general discussion regarding adherence. Through the monitoring of sUA levels, informally, there were many instances where the patient admitted to being not, or only partially compliant with medication. The pharmacist was then able to identify causes of medication non-adherence and render counselling and interventions to improve the patients’ medication adherence. We were unsure if our intervention would ultimately result in long-term control of sUA in the patients as they were discharged from the pharmacist-led clinic once target sUA was achieved. Nonetheless, we were able to detect medication non-adherence during the first few months of dose titration and we were able to counsel and reinforce the importance of compliance to long term treatment. This was also reflected in our patients’ satisfaction survey that they were more likely to adhere to treatment after counselling by the pharmacist. This finding was consistent with other studies10,24 that evaluated the impact and usefulness of non-physician-led programs in improving adherence.
In Singapore, the use of collaborative care to help achieve better clinical outcomes in gout management is not entirely new with nurse-led interventions8,25 as the usual standard of care. These include having a dedicated nurse to perform medication and diet counselling, management of gout flares and adverse effects with nurse-led, doctor-approved refill and dose escalation of ULT. However, in some institutions including ours, specialist nurse services may not be immediately available. To our knowledge, our study is the first in Singapore to demonstrate that pharmacists can potentially be trained up to provide similar monitoring and rapid titration programs for gout. Pharmacists are ideal for this venture given their training and experience with medications.
A major limitation of our study was the small sample size. Hence, the results may be difficult to interpret and generalize. In addition, some demographic data such as weight, severity of gout, number of patients with tophaceous gout were unavailable at the point of data collection and the retrospective collection of some data may be subjected to selection bias. Time-dependent variables such as the number of allopurinol titrations, frequency of clinic visits and the duration of colchicine prophylaxis were not evaluated. Furthermore, starting dose of allopurinol, the rate of dose escalation, and the frequency of SUA measurements were individualized according to patient’s preferences and physicians’ practice in the ‘real world’, a similar difficulty that other studies done previously have faced. 8 Patient’s medication adherence data was not collected and the patient satisfaction questionnaire was also administered by the pharmacist who saw the patient at the collaborative gout clinic, thus, there may be potential bias. Future studies would be needed to address the above limitations as well as the cost effectiveness of a pharmacist-based model in gout management.
Conclusion
Gout is a growing public health problem in Singapore. The use of a pharmacist-led collaborative gout clinic may help to achieve better clinical outcomes in gout management, even in the setting of renal impairment, by providing safe, step-wise increments of ULT with sUA monitoring and reinforcement of adherence through frequent initial visits.
Footnotes
Acknowledgements
This paper and the research behind it would not have been possible without the exceptional support of many people. We would like to specially express our appreciation for the following individuals: (1) Dr Archana Vasudevan, MBBS, Board Cert (Int Med), Board Cert (Rheumatology), for her tremendous support in setting up the pharmacist-led collaborative gout clinic, (2) Ms Serene Yeow for her diligent work in setting up the pharmacist-led collaborative gout clinic, (3) Ms Prista Ramskay Segar for dedicating much time and effort in collecting the data for this paper, (4) Mr Stefan Kowalski, B. Pharm, M App Sci, BCGP, of The University of South Australia for his extremely valuable pointers and suggestions in the writing of this manuscript.
Author Contributions
KMC and EL researched literature and conceived the study. KMC was involved in protocol development, gaining ethical approval, patient recruitment and data analysis. KMC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
SingHealth Centralised Institutional Review Board Trial Registration 2016/2428.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author.
Appendix 1
Dosing guideline for Allopurinol
No.
Drug Dosage
Pre-treatment
Monitoring
Side effects & action to be taken
Remarks/Notable drug Interactions
FBC
Cr
LFTs
Others
FBC
Cr
LFTs
Others
Side effect
Action
1
Allopurinol
Adults: 100 mg/day, titrate gradually every 2–5 weeks to achieve target serum uric acid levels. Maximum: 800 mg/day.
Total Daily Dose can be given either as single dose or in divided dose (2–3 times daily)
Patients with renal impairment will require further dose adjustment.Y
Y
Y
Serum uric acid
Serum uric acid every 2–5 weeks.
When serum urate of <5–6 mg/dL (300–360 μmol/L) is achieved, to monitor serum uric acid every 6 months.
FBC, Cr, LFTs with each visit.Fever
Refer to Rheumatologist immediately.
Chills
Headache
Aching muscles (flu-like syndrome)
Generally feeling unwell
Ulcers (mouth, throat, nose, genitals)
Eye problems
Skin rash (Itching, redness of skin)
Blisters or peeling in skin/mouth)
Platelets < 150x103/μL
Eosinophils > 0.4x103/μL
WBC < 4.0x103/μL
WBC > 10.0 ×103/μL
AST, ALT > 2xULN
Cr rises >30% from baseline
Proposed safe starting dose of PO allopurinol based on 1.5 mg per estimated GFR
Estimated GFR (ml/minute/1.73 m2)
Allopurinol starting dosage
<5
50 mg/week
5–15
50 mg twice weekly
16–30
50 mg every 2 days
31–45
50 mg/day
46–60
50 mg and 100 mg on alternate days
61–90
100 mg/day
91–130
150 mg/day
>130
200 mg/day
Dosing guideline for Febuxostat
No.
Drug Dosage
Pre-treatment
Monitoring
Side effects & action to be taken
Remarks/Notable drug Interactions
FBC
Cr
LFTs
Others
FBC
Cr
LFTs
Others
Side effect
Action
2
Febuxostat
Adults: 40 mg once daily; may increase to 80 mg once daily if serum uric acid level <300–360 μmol/L is not achieved after 2–4 weeks.
Max FDA-approved dose: 80 mg/day
Dose may be increased further to 120 mg once daily if clinically indicated
In CrCl<30 mL/min: use with caution (safety and efficacy has not been evaluated in this group of patients)
Mild hepatic impairment: 80 mg/day
In severe hepatic impairment (Child Pugh Class C): use with caution (efficacy and safety of febuxostat has not been studied)Y
Y
Y
Serum uric acid
LFT: Monitoring at 2 months and 4 months after initiation, then periodically thereafter
Signs & symptoms of MI or strokeNausea, diarrhoea
Refer back to Rheumatologist immediately.
Can be substitute for allopurinol or vice versa in the event of intolerance and adverse events
Avoid combination with:
Mercaptopurine, Azathioprine
Use with caution in patients with theophylline
Use in patients with ischemic heart disease or congestive heart failure is not recommended
Appendix 2
Guidelines on management of medication ADRs, incident gout flares and abnormal laboratory parameters Management of minor changes in clinical condition Patient may require an early review. If in doubt, contact the referring physician.
Minor changes
Management
Mild Flare of Disease
1. Patient could be prescribed with a higher dose of prednisolone that has been previously prescribed for a short duration.
Rheumatic Disease and Gout
2. Patient could be given analgesics or medication for rheumatic disease or gout flares. For example,
For example,
• PO Paracetamol 1g TDS/QDS PRN for a short course
• More joint involvement but little change in ambulatory disability
• PO NSAIDs e.g. PO Naproxen 550 mg BD PRN
• Acute gout flare
• Topical NSAIDs e.g. Ketoprofen gel
• PO colchicine 500mcg TDS PRN for a short course
Non-red flag changes
1. To look for other causes e.g. viral infection, URTI, alcohol use
If patient experiences a non-red flag changes, for example,
2. Do not need to effect a change in DMARD, immunomodulator or gout medication.
• Drop in Hb but <1.5 g/dL
3. Patient may require an early review. If in doubt, contact the referring physician.
• Raised AST/ALT but <2 ULN
