Abstract
Introduction
PCSK9 inhibitors demonstrated their effectiveness in reducing low-density lipoprotein cholesterol (LDL-C) and cardiovascular events in landmark trials. It remains unclear whether the results can be translated to Asian populations. This study was designed to assess the real-world safety, adherence and efficacy of PCSK9 inhibitors.
Methods
A retrospective review for patients newly initiated on PCSK9 inhibitors between 1st June 2017 and 31st July 2021 was conducted in a tertiary cardiology centre. Patients aged ≥ 21 years with a minimum one-month follow-up were included. Adverse drug reactions (ADRs), drug discontinuation, adherence patterns and efficacy between evolocumab and alirocumab groups were compared. Multivariable and propensity score adjusted Cox regression analyses were applied to analyse the outcomes.
Results
Of 87 patients screened, 80 (51 evolocumab; 29 alirocumab) were included. There were no significant differences between evolocumab and alirocumab groups in ADRs (11.8% vs 3.4%, adjusted HR, 2.97; 95% CI, 0.34 – 25.89 in multivariable analysis; adjusted HR, 3.24; 95% CI, 0.38–27.69 after propensity score adjustment) and discontinuation rates (27.5% vs 34.5%, adjusted HR, 0.89; 95% CI, 0.40–2.02 in multivariable analysis; adjusted HR, 0.88; 95% CI, 0.39–1.99 after propensity score adjustment). High medication cost was the main reason for discontinuation. One-third of patients had inadequate adherence to PCSK9 inhibitors. Both groups showed significant reductions of LDL-C compared to baseline.
Conclusions
PCSK9 inhibitors are efficacious, safe and well tolerated. Further studies are warranted to examine the cost-effectiveness of PCSK9 inhibitors to rationalise their sustainable use for cardiovascular prevention.
Introduction
Cardiovascular disease (CVD) is the leading cause of mortality and morbidity worldwide, 1 including Asia. In Singapore, CVD is the highest contributor to premature death and morbidity, accounting for 14.2% loss of total disability-adjusted life years. 2 Elevated levels of low-density lipoprotein cholesterol (LDL-C) are associated with increased CVD risk, 3 and LDL-C lowering with statin therapy to guideline recommended targets can avert adverse cardiovascular outcomes. Two proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors, alirocumab and evolocumab have been approved in Singapore for treatment of hyperlipidaemia in high-risk patients who are not at LDL-C targets despite maximally tolerated statin doses with or without adjunctive oral ezetimibe. 4 These are monoclonal antibodies that when administered parenterally bind to the proteolytic enzyme PCSK9, resulting in reduced degradation of LDL-C receptors in hepatocytes, increased number of cell surface LDL-C receptors and decreased blood LDL-C levels. In landmark studies, alirocumab and evolocumab reduce LDL-C levels by 62% at 24 weeks and 59% at 48 weeks, respectively, compared with baseline.5,6 Both are well tolerated with low rates of adverse drug reactions (ADRs), the most common being local injection site reaction.5,6
Singapore has a multi-racial population that comprises 75.9% Chinese, 15% Malay, 7.5% Indian and 1.6% other ethnicities. 7 There is relatively little by way of data on PCSK9 inhibitor use among local patients. Of note, only 13.2% and 9.9% of subjects in the ODYSSEY OUTCOMES and FOURIER trials, respectively, were Asian, and real-world studies of PCSK9 inhibitor use have largely focused on Western cohorts.8–15 While a few real-world studies have been conducted on Asian cohorts,16–21 Singapore’s unique multi-racial population and genetic differences may impact drug tolerability and efficacy; and social and culture factors, drug adherence and access. In this study, we aimed to study the safety and tolerability of PCSK9 inhibitors among Singaporean patients and determine factors, including method of drug payment, affecting their adherence in the real world. In addition, the study would report on the real-world efficacy of PCSK9 inhibitors.
Methods
Study design
We conducted a retrospective review of the electronic health records of patients being prescribed alirocumab or evolocumab for the first time during the period from 1st June 2017 to 31st July 2021 at a single tertiary cardiology centre. Inclusion criteria included age 21 years and above, administration of at least one dose of PCSK9 inhibitor, and minimum follow-up of 1 month. A minimum follow-up period of 1-month was imposed to ensure that follow-up information would be obtained. Written informed consent was obtained from patients to access their electronic health records, and patients who declined were excluded from analysis. The study had been approved by the centralised institutional review board.
The decision to initiate either PCSK9 inhibitor was at the discretion of individual patients’ treating physicians, who were allowed to adjust the doses as well as other medications, e.g., statins, throughout the study period. In addition, the PCSK9 inhibitor dose may be titrated, or the drug discontinued or switched from one to the other due to ADRs, suspected ADRs or inadequate or excessive LDL-C reduction, or other reasons. Included patients were classified into two groups based on alirocumab or evolocumab prescribed at initiation.
Outcomes
The primary outcome was the proportion of patients on PCSK9 inhibitors experiencing ADRs. Key secondary outcomes were incidence of ADRs while on PCSK9 inhibitors, PCSK9 inhibitor discontinuation rates and reasons for discontinuation, and patient adherence. We included all enrolled subjects who fulfilled the study entry criteria in the analyses of primary outcomes and key secondary outcomes to better evaluate real-world ADRs and patient adherence behaviours. Tertiary outcomes were percentage LDL-C reduction after PCSK9 inhibitor initiation and the proportion of patients who required lipid-lowering therapy adjustment after PCSK9 inhibitor initiation and reasons for adjustment. For analysis of LDL-C lowering, we excluded patients with poor adherence, i.e., medication possession threshold <40% (see below), or incomplete adherence data, as well as those with unmeasurable baseline LDL-C levels, e.g., due to high triglyceride levels.
Data collection
Patients’ demographic information at baseline (age, gender, race, body-mass index), past medical history, cardiovascular risk factors, PCSK9 inhibitor prescription and dose, reasons for PCSK9 inhibitor initiation and relevant laboratory investigations including creatinine kinase, lipid panel, liver enzymes and glycated haemoglobin were retrieved from their electronic health records. In addition, patients were contacted for more information if there was incomplete information documented in the electronic health records. We defined ADRs as those occurring while patients were on PCSK9 inhibitor that had been documented by the treating physicians in the electronic health records. PCSK9 inhibitor doses at initial and follow-up visits were collected. The ADRs events were observed until patient discontinued the PCSK9 inhibitor or was switched to the other PCSK9 inhibitor. Dates and reasons for discontinuation were collected. Changes in other lipid-lowering therapy and reasons for the changes were also recorded. Laboratory investigations results (blood levels of creatinine kinase, lipid panel, liver enzymes and glycated haemoglobin) through all follow-up visits were collected. Clinical outcomes events including cardiovascular death, stroke, myocardial infarction, hospitalization for heart failure or revascularization were censored at end-of-study follow up date 31st August 2021.
Patient adherence to PCSK9 inhibitors was determined by mapping the pharmacy dispensed records to prescribed drug orders. Medication possession percentage, 22 a surrogate measure of patient adherence, was defined as:
Medication possession (%) = 100 X Number of doses of PCSK9 inhibitor collected/Number of doses of PCSK9 inhibitor required from first prescription order date until censoring date on 31st August 2021 or the date when the PCSK9 inhibitor was discontinued or switched to another PCSK9 inhibitor.
Adequate, moderate and poor adherence were defined at medication possession thresholds of 80% or more, 40% to less than 80%, and less than 40%, respectively. 22 Patients who collected only one dose of PCSK9 inhibitor from the study site or one or more doses of PCSK9 inhibitor from other institutions were excluded from adherence assessment due to incomplete data. Data on patients’ payment methods were retrieved from the electronic billing system. Self-paying was defined as full payment fulfilled by patient; third-party paying, full payment fulfilled by insurance, employee benefits or social assistance; and partial paying, payment fulfilled by both patient and third-party payers.
Statistical methods
Demographic data and baseline characteristics were presented as descriptive statistics. Continuous variables were reported as mean and standard deviation if the data were normally distributed, and median and interquartile range (IQR) if the data were skewed or not normally distributed. Categorical variables were presented as the numbers and percentages. Normality of data was assessed by visually inspecting the distribution in normal plots, checking for skewness, and using the Shapiro-Wilk test. Differences between the two groups at baseline were analysed using Student’s t-test for normally distributed continuous variables, Mann-Whitney U test for continuous variables that were not normally distributed and Fisher’s exact test for categorical variables. The level of statistical significance was defined as p value less than 0.05.
The incidence of ADRs, reasons for PCSK9 inhibitor discontinuation, patient adherence and patients’ method of payment were compared using Fisher’s exact test. Cox regression analysis was applied to analyse the effects of covariates that affect the primary outcomes due to the different durations of follow-up visits in both groups. Univariable Cox regression analysis was applied to investigate the possible predictors or variables that were independently associated with ADRs and discontinuation of PCSK9 inhibitor. The hazard ratio (HR) was adjusted for possible confounding factors or clinically relevant risk factors using multivariable and propensity score adjusted Cox regression analysis. Variables with a p-value less than 0.20 in the univariable regression were entered into the multivariable Cox regression and were used for estimating the propensity score to control for the differences between patients initiated on evolocumab and alirocumab.
Wilcoxon signed-rank test was used to compare LDL-C levels between baseline and different follow-up time points within the same group. Follow-up time points were further groups into epochs: third month (from at least 1 month to less than 3 months post-initiation), sixth month (from at least 3 months to less than 6 months post-initiation), ninth month (from at least 6 months to less than 9 months post-initiation), and every subsequent 3 months until censoring date on 31st August 2021, PCSK9 inhibitor discontinuation or switching to another PCSK9 inhibitor. Mann-Whitney U test was used to evaluate the differences of LDL-C reduction between two groups. p value of less than 0.05 was considered statistically significant. All statistical analyses were performed using IBM SPSS Statistic Window Version 28.0 (IBM Corp., Armonk, NY, USA).
Results
Study disposition
Of 87 patients screened, 80 patients (51 and 29 from evolocumab to alirocumab groups, respectively) were included in the analysis. Reasons for exclusion are shown in Figure 1. In the LDL-C sub-analysis, 64 patients (42 and 22 from evolocumab and alirocumab groups, respectively) were included after excluding those patients with poor adherence, incomplete adherence data, and unmeasurable baseline LDL-C levels (Figure 1). Study disposition.
Baseline characteristics
Baseline characteristics.

PCSK9i: proprotein convertase subtilisin/kexin type 9 inhibitor; CABG: coronary-artery bypass grafting; PCI: percutaneous coronary intervention; FHx: family history; CHD: coronary heart disease; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Values were presented as mean ± standard deviation (SD), number (%) and median (Interquartile range).
aMissing data were not included in the analysis.
bHigh intensity statin = Rosuvastatin 20–40 mg daily or atorvastatin 40–80 mg daily.
cModerate intensity = Rosuvastatin 5–10 mg daily, atorvastatin 10–20 mg daily, simvastatin 20–40 mg daily, pravastatin 40–80 mg daily or lovastatin 40 mg daily.
dLow intensity = simvastatin 10 mg daily, pravastatin 10–20 mg daily, lovastatin 20 mg daily.
Study outcomes
Study outcomes by treatment groups.

ADRs: adverse drug reactions; URTI: upper respiratory tract infection; LFT: liver function test, LDL-C: low-density lipoprotein cholesterol.
aEach patient may have more than one incidence of ADRs (1 patient had both URTI symptoms and other ADRs, 1 patient had gastrointestinal symptoms and hand tingling).
bAlanine aminotransferase (ALT) >2 times upper limit of normal range.
cFlushing (1), increase blood sugar with history of uncontrolled blood sugar level (1).
dSuspected ADR such as albuminuria, mildly raised ALT (<1 time from upper limit of normal range), uncontrolled blood sugar level.
einconvenience, bulky and fridge item.
fUnsatisfactory of LDL cholesterol reduction despite maximum dose of PCSK9 inhibitor (LDL cholesterol from 3.33 mmol/L to 3.14 mmol/L; 3.81 mmol/L to 4.11 mmol/L).
gLDL cholesterol dropped from 3.21 mmol/L to 0.42 mmol/L.
hAdequate adherence was defined as medication possession threshold of 80% or more. Moderate adherence was defined as medication possession threshold of 40% to less than 80%. Poor adherence was defined as medication possession threshold of less than 40%. Unable to assess adherence was defined as collection of one dose of PCSK9 inhibitor or collection of PCSK9 inhibitors from other institutions.
iSelf-paying was defined as full payment fulfilled by patient. Third-party paying was defined as full payment fulfilled by insurance, employee benefits or social assistance. Partial paying was defined as payment fulfilled by both patient and third-party payers.
Cox regression analysis for variables associated with adverse drug reactions.
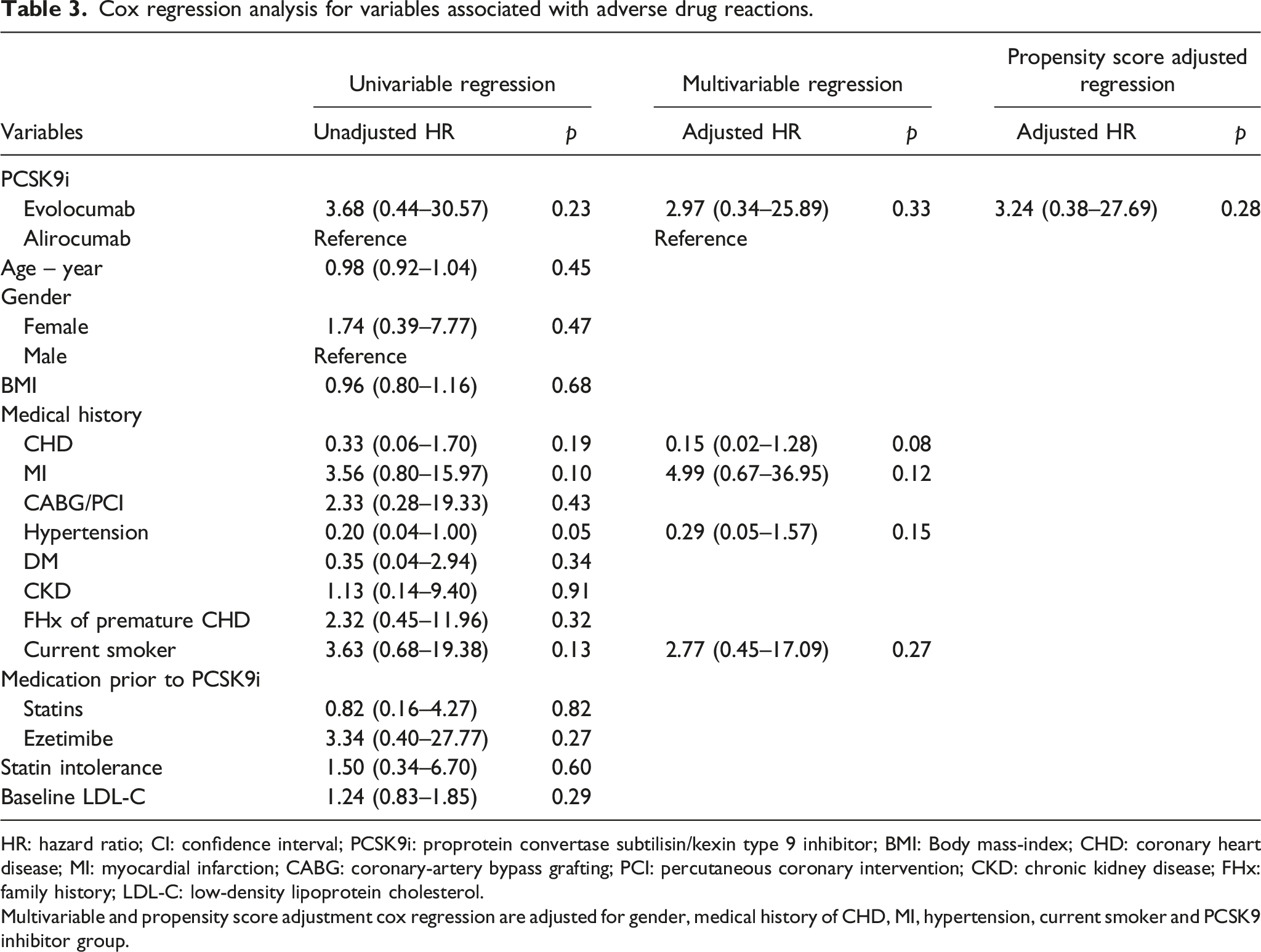
HR: hazard ratio; CI: confidence interval; PCSK9i: proprotein convertase subtilisin/kexin type 9 inhibitor; BMI: Body mass-index; CHD: coronary heart disease; MI: myocardial infarction; CABG: coronary-artery bypass grafting; PCI: percutaneous coronary intervention; CKD: chronic kidney disease; FHx: family history; LDL-C: low-density lipoprotein cholesterol.
Multivariable and propensity score adjustment cox regression are adjusted for gender, medical history of CHD, MI, hypertension, current smoker and PCSK9 inhibitor group.
Cox regression analysis for variables associated with discontinuation rate of PCSK9 inhibitors.
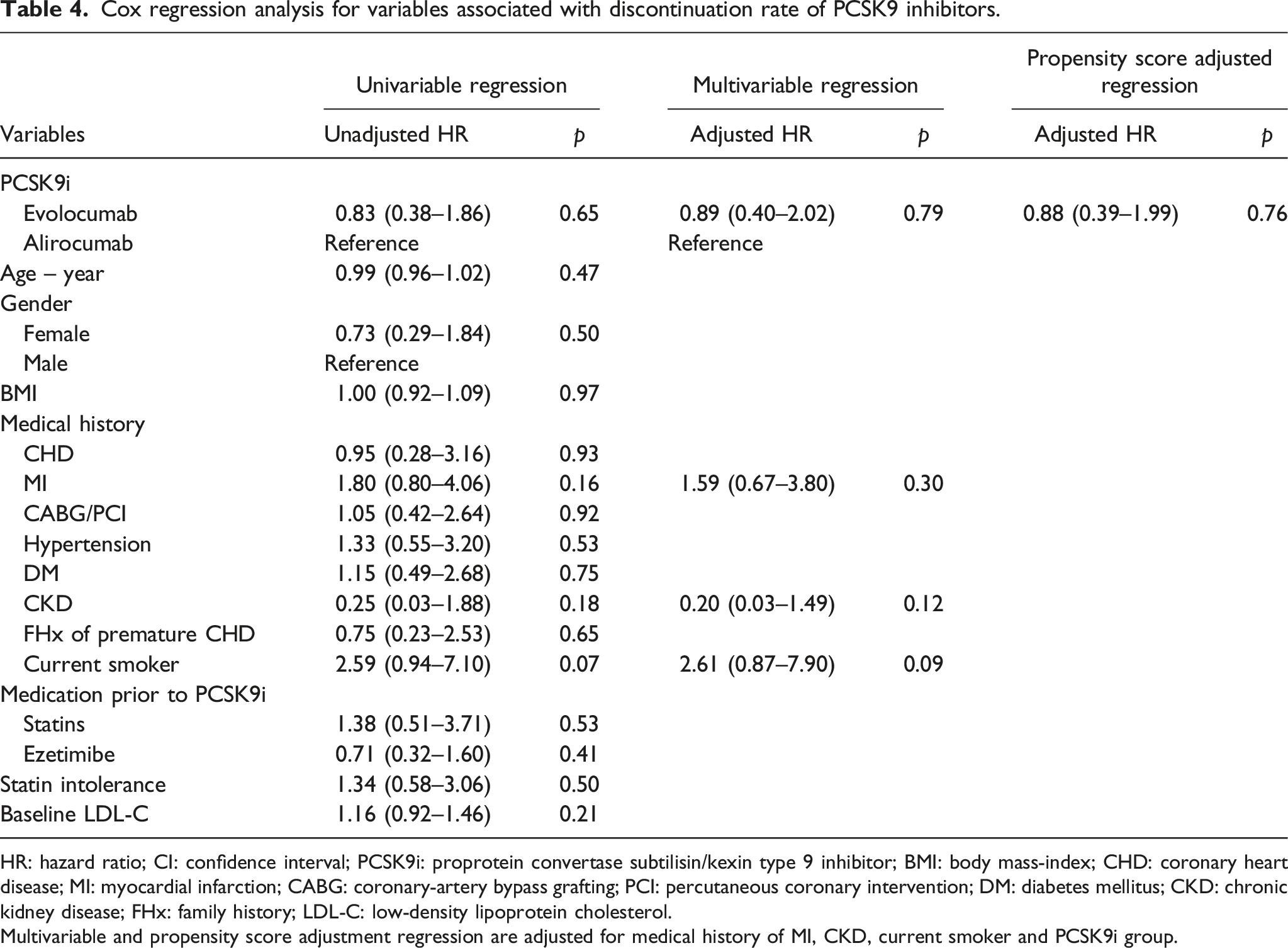
HR: hazard ratio; CI: confidence interval; PCSK9i: proprotein convertase subtilisin/kexin type 9 inhibitor; BMI: body mass-index; CHD: coronary heart disease; MI: myocardial infarction; CABG: coronary-artery bypass grafting; PCI: percutaneous coronary intervention; DM: diabetes mellitus; CKD: chronic kidney disease; FHx: family history; LDL-C: low-density lipoprotein cholesterol.
Multivariable and propensity score adjustment regression are adjusted for medical history of MI, CKD, current smoker and PCSK9i group.
64%, 19% and 15% of patients had adequate, moderate and poor adherence, respectively, to PCSK9 inhibitors (55.2%, 20.7% and 20.7%, respectively, in alirocumab group vs 68.8%, 17.6% and 11.8%, respectively, in evolocumab group) (Table 2). Among self-paying patients (n = 47), 26 (55%), 10 (21%), and 9 (19%) had adequate, moderate and poor adherence, respectively, whereas among patients whose medication bills were fully paid by third-party payers (n = 31), 24 (77%), 5 (16%) and only 2 (7%) had adequate, moderate and poor adherence, respectively.
LDL cholesterol sub-analysis.

LDL-C: low-density lipoprotein cholesterol; Median LDL reported in mmol/L.
3m was defined as follow-up epoch from at least 1 month to less than 3 months post-initiation.
6m was defined as follow-up epoch from at least 3 month to less than 6 months post-initiation.
9m was defined as follow-up epoch from at least 6 month to less than 9 months post-initiation.
12m was defined as follow-up epoch from at least 9 month to less than 12 months post-initiation.
15m was defined as follow-up epoch from at least 12 month to less than 15 months post-initiation.
18m was defined as follow-up epoch from at least 15 month to less than 18 months post-initiation.
21m was defined as follow-up epoch from at least 18 month to less than 21 months post-initiation.
24m was defined as follow-up epoch from at least 21 month to less than 24 months post-initiation.
Wilcoxon signed rank analysis was used to compare median LDL cholesterol between baseline and each follow-up time point.
aMann-Whitney U test was used to evaluate the differences of LDL cholesterol reduction in percentage between alirocumab and evolocumab.
Almost half of all patients in both groups had their lipid-lowering therapy adjusted after PCSK9 inhibitor initiation, mainly driven by very low attained LDL-C levels, patient preference and/or incident ADRs. Of 24 patients who discontinued PCSK9 inhibitors, 13 were permanently discontinued, 5 were restarted on same drug between 5 and 31 months after and 6 were switched to another PCSK9 inhibitor during the study period.
Eighteen patients were initiated with low dose alirocumab 75 mg, among whom 3 had the dose increased to 150 mg, 6 discontinued alirocumab and 9 remained on alirocumab 75 mg during follow-up. Eleven patients were started on alirocumab 150 mg, among whom 4 discontinued and 7 remained on alirocumab 150 mg. Among 41 patients started on evolocumab 140 mg every 2 weeks, 5 were switched to off-label longer dosing intervals (every 3 weeks or once monthly), 13 discontinued and 23 kept the same dosing regimen. Ten patients were initiated on off-label evolocumab 140 mg once a month, among whom 2 later switched to shorter dosing intervals (every 3 weeks or every 2 weeks), 1 discontinued and 7 were maintained on initial dosing regimen.
Discussion
PCSK9 inhibitors were efficacious, safe and well-tolerated in our study. These findings are consistent with those observed in outcome trials, real-world studies and meta-analyses.6,7,9,10,16–20,23,24 ADRs reported by study patients were mostly nonspecific (e.g., gastrointestinal side effects, dizziness, etc.). The relationship to PCSK9 inhibitor use is temporal and causation cannot be inferred. Discontinuation rates were higher in our real-world study (34.5% in alirocumab and 27.5% in evolocumab) compared to the 14.2% 5 and 12.5% 6 for alirocumab and evolocumab, respectively, in randomised controlled trials, which might be attributable to patients’ perception of high treatment costs (20.7% and 9.8% in the alirocumab and evolocumab groups, respectively, p = .193). This is consistent with another real-world study that found an inordinately high discontinuation rate (33.3%) of PCSK9 inhibitors at 60 days. 10
Both alirocumab and evolocumab lowered LDL-C reduction by up to 60%, similar to clinical trials and systematic reviews,6,7,23,24 despite having more than a third of patients being moderately or poorly adherent to PCSK9 inhibitors and 19.6% of patients in the evolocumab group being initiated on off-label longer dosing interval regimen (140 mg every 3 or 4 weeks). Patients on evolocumab had marginally better LDL-C reductions that was statistically significant at the ninth month follow-up epoch. This could have been a chance finding. Alternatively, this might be explained by the treating physicians’ tendency to initiate and maintain the lower dose of alirocumab 75 mg among patients. Three of 18 patients started on alirocumab 75 mg every 2 weeks had the dose increased to 150 mg every 2 weeks only at the ninth month follow-up epoch.
The adherence of our study patients was less than that observed in randomised clinical trials. In our study, the majority of our patients were self-paying, and we found that high medication costs adversely influenced patient adherence. Similar to other countries, high medication costs were associated with poor adherence of PCSK9 inhibitors and had great impact on medication persistence.14,15,18 While the price to patient for standard doses of both alirocumab and evolocumab are similar, more patients had poor adherence to alirocumab compared to evolocumab. This is likely due to the higher proportion of patients being initiated on off-label longer dosing intervals of evolocumab that resulted in cost savings, whereas 93.1% of patients initiated on alirocumab were prescribed the standard 2-weekly dosing regimen.
This study has several limitations. This is a single Asian centre study and the results may not be generalisable. Some patients declined consent and were excluded from the study. This might have introduced selection bias. The retrospective observational nature of this study increased the chance of missing data (e.g., laboratory data, reasons for PCSK9 inhibitor discontinuation, etc.). Although patients were contacted to supplement the data where applicable, recall bias might be present because ADRs or discontinuations could have happened a long time ago. The median follow-up durations of 16 and 10 months in the alirocumab and evolocumab groups, respectively, allowed us to observe common ADRs and assess medium-term adherence. Rare ADRs that manifest long after initiation and long-term adherence were not assessed in this study. The study size is small. While we did not specifically study factors related to drug initiation, high out-of-pocket drug costs (which we found to be a factor limiting patient adherence) is a likely factor that had deterred many doctors and patients from initiating the drug. Furthermore, there were disproportionately fewer patients on alirocumab versus evolocumab, which might lead to bias in the results of LDL-lowering efficacy by drug type. This imbalance might plausibly be explained by later drug listing into hospital formulary, individual doctor or patient preferences, etc. Regardless, selection bias in this retrospective review was mitigated by inclusion of all consecutive patients who fulfilled our study criteria. i.e., the distribution of PCSK9 inhibitors accurately reflected our real-world institutional practice. Patient adherence was determined using a surrogate measure: percentage of medication possession within the stipulated period. We could not verify whether patients actually took their PSCK9 inhibitor after collection. However, given that the majority of patients paid out-of-pocket for very expensive drugs, the probability of non-administration is low. Further, patients’ adherence patterns may fluctuate, e.g., motivation to adhere to prescribed regimen tend to slacken when target levels have been attained. Of note, medication possession percentage used in the study provides an indicator of the average adherence throughout the study follow-up, which might attenuate the effects of fluctuation. We did not control for multiple factors that might impact LDL-C control and adherence as well as their assessment, e.g., lifestyle modifications, variations in physician prescription behaviour (including the use of nonstandard off-label dosing regimen) and follow-up schedule. To mitigate the effects of variable follow-up intervals on LDL-C level comparisons, the analyses were performed on measurements taken at specific follow-up time epochs. Lastly, we did not include a control group of patients who were not prescribed PCSK9 inhibitors. Without a control group, nonspecific ADRs that were not related to parenteral injection could not be reliably distinguished from background noise.
Conclusion
Alirocumab and evolocumab are efficacious, safe and well-tolerated by real-world Asian patients. However, approximately one-third of patients discontinued PCSK9 inhibitors, primarily driven by high medication costs. Given that PSCK9 inhibitors have been proven to reduce cardiovascular events in randomised controlled trials, further studies are warranted to examine the cost-effectiveness of PCSK9 inhibitors to rationalise sustainable use of this efficacious but expensive therapeutic class of drugs for cardiovascular prevention.
Footnotes
Acknowledgements
We would like to thank Ms Thng Shu Hui for her initial involvement in protocol development and gaining ethical approval.
Author contributions
First 4 authors researched literature, conceived the study, involved in protocol development and patient recruitment. First 3 authors wrote the first draft of the manuscript. All authors involved in data analysis, edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data
The datasets generated and/or analysed during the current study are available from corresponding author.
Ethical approval
Ethical approval for this study was obtained from Singhealth Centralised Institution Review Board (CIRB), approval number: 2018/3175.
Informed consent
Written informed consent was obtained from the patients for data collection and their anonymized information to be published in this article.
