Abstract
Introduction
Angiotensin II Receptor Blockers (ARB) are widely utilized in the treatment of hypertension and nephropathy. In March 2019, a mass recall of selected brands of an ARB, losartan due to reported elevated levels of potentially carcinogenic nitrosamine impurities was implemented in a Singapore primary care institution.
Objective
To describe the crisis management of the mass losartan recall in a public primary healthcare institution in Singapore.
Methods
The crisis management framework was adapted from the Gonzalez-Herrero and Pratt model. This crisis was managed over three phases: identification of impending crisis based on risk reports by local health authorities, formation of a task force to develop turnaround strategies and measures, implementation and monitoring of the remedial measures. Data to plan and chart implementation was retrieved from the institution databases, including the Electronic Health Intelligence System for patient medical and prescription records, Outpatient Administrative System for reimbursement of medications and other expenditures incurred. An exigency scheme, Losartas® Review Service (LRS), was established to expedite the replacement of the affected medication with another ARB or another anti-hypertensive medication.
Results
SingHealth Polyclinics completed the recall of 29,794 (92.1%) patients within 14 weeks. The majority of the patients (76.2%) were attended to by doctors, while 16.5% and 7.3% were managed using the LRS and pharmacy respectively. The institution incurred a cost of SGD2,824,760 (estimated SGD95 per patient) to manage the crisis.
Conclusion
The successful implementation of a mass medication recall hinged on timely intervention, cross-departmental coordination, effective communication and judicious resource allocation.
Background
Angiotensin II receptor blockers (ARBs) and angiotensin-converting enzyme inhibitors (ACEIs) are medications that decrease blood pressure via the inhibition of the vasoconstrictive effect of the renin-angiotensin system (RAS). These agents also reduce proteinuria and retard the progression to subsequent renal impairment. 1 These agents are also prescribed to individuals with type-2 diabetes mellitus (T2DM), hence ACEIs and ARBs are widely prescribed globally. 2
Starting from June 2018, several ARBs have been recalled overseas due to the detection of nitrosamine contaminants above acceptable limits. Commonly implicated contaminants include N-Nitroso-N-methyl-4-aminobutyric acid (NMBA), N- Nitrosodimethylamine (NDMA) and N-Nitrosodiethylamine (NDEA). While these nitrosamines are known environmental contaminants found in water and foods, studies conducted in animals have shown that prolonged consumption of these impurities may increase the occurrence of cancers, such as hepatocellular carcinoma. 3
On 28th March 2019, the Singapore Health Sciences Authority (HSA) held a national press conference to announce that elevated NMBA levels exceeding the acceptable threshold had been detected in three brands of losartan products (Hyperten®, Losagen®, and Losartas®). 4 This resulted in an unprecedented crisis in SingHealth Polyclinics, one of three large public primary healthcare institutions, as over 32,000 patients were on Losartas®.
Crisis management is the process through which an institution deals with a disruptive and unexpected event that threatens to harm the institution and its stakeholders. With the sudden public announcement of the contaminated medications, the institution had to face the imminent threat of massive patient attendances for review or exchange of the affected medications. Its existing clinical services and manpower would potentially be overwhelmed. Moreover, the stock of replacement medications would be at risk of being rapidly depleted. Affected patients were also at risk of morbidity and mortality as their blood pressure control could be adversely affected by a change in their pharmacological therapy. The potential carcinogenic risks of continuous consumption of the contaminated medications and the associated litigation risks were other major challenges to the institution.
This paper aims to describe the crisis management of the mass losartan recall in a public primary healthcare institution in Singapore. The objectives of the recall were to contain the fallout from this unexpected event and to sustain the trust of the patients towards the healthcare institution. Recognizing the lapses and issues that emerged during the execution of these remedial measures are critical real-time lessons in crisis management. Developing a crisis management plan based on these learning points will enable the institution to better tackle a similar crisis in the future.
Methods
Gonzalez-Herrero and Pratt described a four-phase crisis management model that includes: issues management, planning-prevention, the crisis and the post-crisis. 5 The crisis management model has been adapted from the Gonzalez-Herrero model and is divided into three phases: (1) danger signs leading to the diagnosis of the impending crisis, (2) implementing appropriate turnaround strategies and measures, (3) implementation of the change process and its monitoring. This adapted framework provides a structure to systematize the processes and workflow during the crisis.
I. Danger signs leading to the diagnosis of the impending crisis
Following the overseas recall of ARBs in June 2018, HSA convened an Expert Panel comprising specialists in the fields of toxicology, pharmacology, oncology and cardiology to advise on the regulatory approach for products detected with these impurities. The presence of carcinogenic impurities is generally unacceptable in medications, unless in unavoidable circumstances. In these instances, stringent limits are set according to harmonised international guidelines. The globally acceptable daily intake level was based on a cancer risk of 1 in 100,000 for exposure over a lifetime (i.e. over 70 years). 6 The levels of NMBA detected in the three brands of losartan products were in trace amounts which exceeded the acceptable limit of 96 ng/day. 7 The panel recommended that such products be recalled from the market.
On 28th March 2019, the HSA reported that testing of ten brands of the single-ingredient losartan products and five brands of the combination losartan products were completed. 4 Three brands of losartan products and all their associated batches were found to contain NMBA that exceeded the acceptable levels. One of the tainted ARBs was Losartas®, a brand widely prescribed across Singapore. Concurrently, all other locally available brands of ARBs, including valsartan, irbesartan and telmisartan were tested negative for impurities by HSA. 4
The Ministry of Health Singapore (MOH) conducted a press conference to inform the public of the safety, medical review, medication supply and financial concerns relating to the affected brands of losartan. The public was strongly encouraged to continue their medications until they were provided with a replacement of the affected losartan or were seen by their doctors, as the sudden discontinuation of their treatment could pose greater health risks. All healthcare professionals in Singapore received a circular from HSA stating the decision and reason for the recall of the affected brands of losartan. 4 As a major primary healthcare provider in Singapore, SingHealth Polyclinics (SHP) had to respond expeditiously to this event.
Singapore has a dual public-private primary healthcare system. The twenty local public primary care polyclinics provide 20% of the primary healthcare services to its 5.6 million multi-ethnic Asian population but manage 40% of the non-communicable diseases (NCDs) in the community. 8 Hypertension and T2DM are prevalent NCDs in the community, which rank amongst the top four medical conditions managed in the polyclinics. 8 Nationwide, an estimated 137,000 patients were affected by this crisis. 9
SingHealth Polyclinics has a network of eight polyclinics that managed over 2.5 million patient attendances in 2019. 10 In SHP, a total of 52,986 patients were prescribed ARBs with an estimated 33,527 (63.3%) taking losartan-containing medications in the preceding year before the recall.
II. Implementing appropriate turnaround strategies and measures
Formation of a cross-departmental crisis management team
Roles & responsibilities of crisis management team.
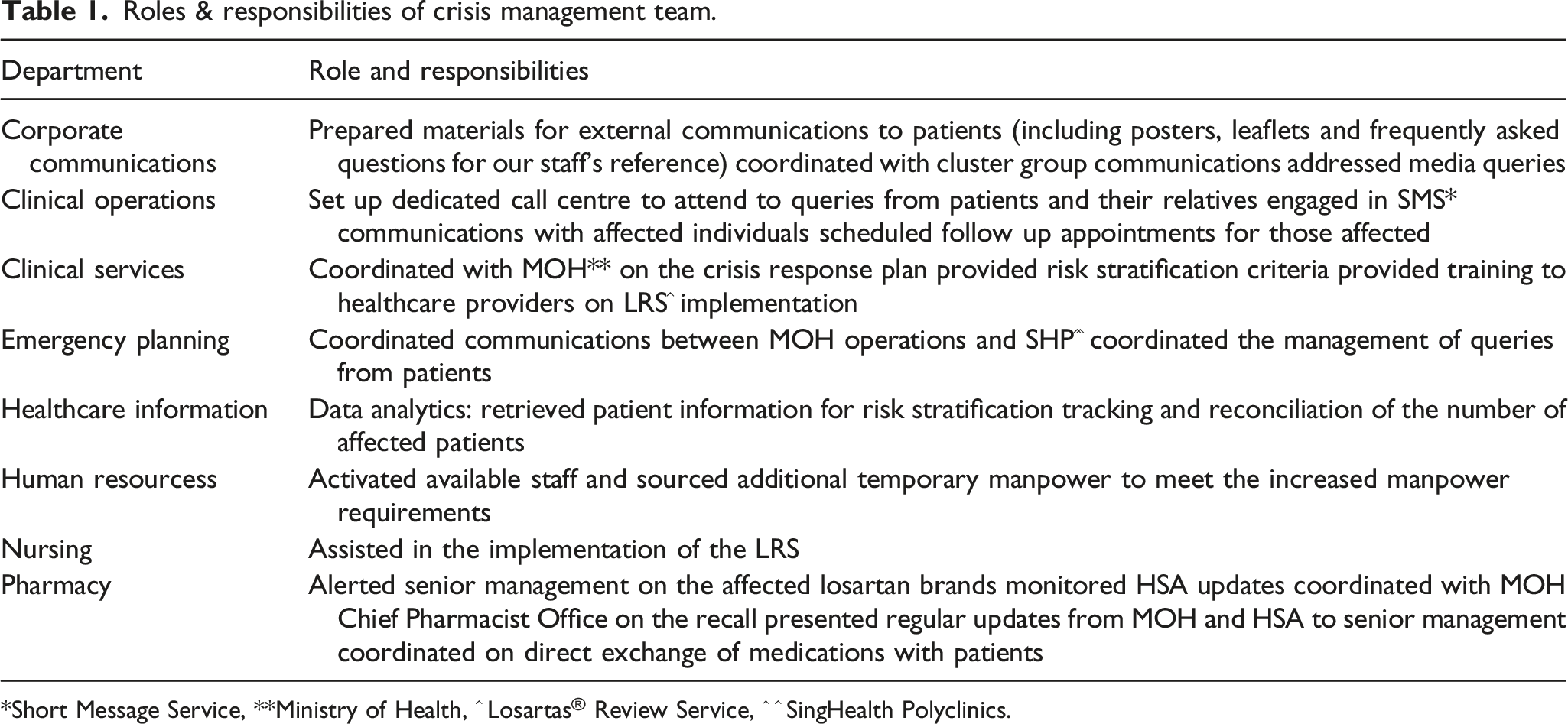
*Short Message Service, **Ministry of Health, ^ Losartas® Review Service, ^ ^ SingHealth Polyclinics.
Setting up the Losartas® Review Service (LRS): risk stratification & recall planning
Given the vast numbers of affected patients, a crisis management plan named the Losartas® Review Service (LRS) was swiftly developed by the multidisciplinary team. LRS was launched within 24 h of the public recall announcement. The intent was to stagger the recall of patients over the subsequent 14 weeks.
Following the HSA announcement of the affected losartan brands on 28th March 2019, a generic short message service, SMS 1 (Appendix I), was dispatched to all SHP patients by 6 p.m. on the same day. A follow-up SMS 2 (Appendix I) was sent on 29th March 2019 to inform patients on the recall plans based on whether their existing appointment was before 1st July 2019, or on or after 1st July 2019. Separately, patients who received the unaffected losartan on 27th March 2019 were also informed via SMS 3b (Appendix I) to reassure them.
Critical data management
Patient healthcare data is routinely extracted from the databases and transformed to its desired actionable format. The transformed data is channelled from multiple healthcare transactional systems into a single enterprise data repository known as Electronic Health lntelligence System (eHINTS). In SHP, doctors prescribe medications, including Losartas®, via the electronic prescription platform (Sunrise Clinical Manager). This data, including the date and dose of prescription, was extracted by the Healthcare Information Department.
Data on reimbursement and payment of medications was extracted from the institution’s Outpatient Administrative System (OAS). The Finance Department leveraged OAS to compute the overall expenditure incurred to implement the entire LRS. Additionally, OAS was also the platform to schedule appointments for patients.
For the affected patients, information on their upcoming appointments, history of neoplasm, chronic kidney disease (CKD) status, types of blood pressure lowering medications and dose of losartan were extracted from eHINTS and OAS. The data was used for risk stratification and recall planning.
Risk stratification of affected patients
For recall planning, affected patients were categorized into groups A, B and C based on their upcoming appointment date. A summary of the risk stratification and recall planning process is depicted in Figure 1. Risk stratification of SHP patients.
Group A comprised of patients with an appointment date before 1st July 2019, they were informed to keep their scheduled appointment for medical review. Group B comprised of patients with follow-up appointments on or after the 1st of July 2019. This group was further divided into groups B1 and B2, based on their risk factors. Patients with any of the following conditions were categorised as “high risk” and placed in group B1:
1. Current or previous history of malignant neoplasm
2. Chronic kidney disease stage 4 or 5: eGFR ≤30 mL/min
3. On spironolactone
4. Losartan dose ≥100 mg/day
SMS 3c (Appendix I), was sent to patients in group B1 on 3rd April 2019 to return by 1st May 2019 for direct exchange of Losartas® with an unaffected losartan brand. Patients in group B2 had their appointment rescheduled to before 1st July and were informed of the new appointment details via SMS 3a between 30th March and 5th April 2019 (Appendix I).
Group C were patients without an upcoming appointment, SMS 1a (Appendix I) was sent to determine if they were on active follow-up with SHP or at other public healthcare institutions on 4th April. If the response indicated that they were on follow-up at SHP, a follow-up appointment on or before 1st July would be scheduled. Finally, a letter was dispatched to the homes of individuals without a valid working contact number. The letter dispatched included instructions for rescheduling of appointments if they were still on active follow-up at SHP (Appendix 2). All the communication was completed within 2 weeks.
LRS provider training and design of manual LRS form
Medical services were expected to be stretched with the massive recall of products. Advanced practice nurses (APNs), nurse clinicians (NCs) and clinic pharmacy managers (CPMs) were recruited to assist in the crisis. Family physicians conducted compulsory training for all nurses and pharmacists involved in the LRS. These providers were inducted on eligibility criteria, conversion of losartan to other ACEI and ARB using an evidence-based conversion table and to formulate follow-up plans.
A manual LRS form was developed to detail the decision-making process by the healthcare providers (Appendix III). It also served as a prescription form for non-physicians. In compliance with the Singapore Medical Council regulation on prescription rights, physicians were required to file a parallel prescription for all the medications issued based on the LRS protocol. Patients who did not fulfil the eligibility criteria would be referred to a physician.
Managing demand and supply of replacement medications
To balance the demand and supply of the existing ARB stock across the nation, the HSA recommended these medications were dispensed for a maximum of 1 month. Patients were notified at the polyclinic pharmacies that this temporary measure would continue for the next 6 months.
Minimizing additional patients’ expenditure
A refund was given to the patients if they returned the affected brands of losartan. Replaced medications were charged at an equivalent or lower price. Patients whose medications were switched had the fees for the additional consultation and laboratory tests waived.
III. Implementation of the change process and its monitoring
Implementation of the Losartas® Review Service (LRS)
A direct medication exchange was performed at the pharmacy for patients with the following risk factors: 1. History of previous or current malignant neoplasm 2. CKD G4/5 3. On spironolactone 4. Losartan ≥100 mg/day
The remaining patients were screened by LRS providers for the following additional risk factors: 1. Age 80 years and above 2. Heart failure 3. Last serum potassium >5.0 mml/L 4. Systolic blood pressure 5. Home: <100 mmHg or >160 mmHg 6. Clinic: <110 mmHg or >160 mmHg 7. Other medical concerns
Patients with any of these risk factors were reviewed by a physician and encouraged to switch to an ACEI or a non-losartan ARB in the absence of contraindications. The LRS form included an ACEI and ARB conversion table to assist in the accurate calculation of equivalent doses.
Communication, coordination and collation of returned medications
Communication with affected patients was delivered via multiple platforms. A centralized call center was set up to inform patients by phone call. Call agents were trained and equipped with a set of “Frequently Asked Questions” to respond to patients’ queries. Complex queries were escalated to representatives from the Emergency Planning team.
For patients who were uncontactable by phone, SMS was used (Appendix I). A message was delivered to raise their awareness of the situation and a subsequent SMS notified them of their review schedule at the polyclinic.
Dedicated information counters were situated near the clinic’s entrance to address enquiries of walk-in patients and schedule review appointments. Posters featuring photos of the affected Losartas® were displayed to provide information to patients. The collaterals also served as visual aids for frontline counter staff to communicate with the patients among whom many were elderly.
Manpower mobilization
Manpower was critical in the implementation of this crisis management plan. The Human Resource (HR) team, with approval from institution leadership, introduced ad-hoc incentives to staff to manage the additional workload during this period. Staff could claim overtime pay and transportation costs for travel beyond office hours. Part-time workers were hired to assist with the additional work at the call center and pharmacy.
Direct and indirect costs
Direct cost refers to the following: 1. Cost of acquiring the various medications to replace those under recall 2. Cost of writing off those drugs under recall 3. Cost of pharmacy staff directly involved in the exchange exercise 4. Cost of clinicians providing additional consults to patients who need to be prescribed with alternative medications
Indirect costs refer to HQ costs incurred in providing the support for the recall exercise.
Results
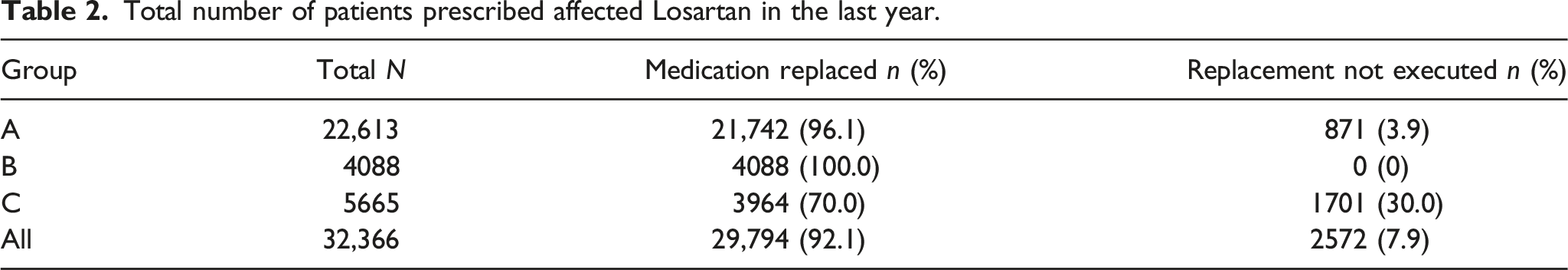
The Healthcare Information Department generated the list of patients who were prescribed Losartas® within the past year from the EMR.
Total number of patients prescribed affected Losartan in the last year.
Group B1: patients with high risk factors.

aNumbers do not add tally as patients may satisfy more than 1 high risk factor.
Service points and number of medication exchanges during the crisis.

aLosartas® review service.
The rate of medication exchange was projected based on the number of patients in each group. As depicted in Figure 2, by the end of 14 weeks, 29,794 (92.1%) had returned to the clinic and had their medication issues addressed. Actual and projected weekly number of SHP patients converted from affected Losartas between 1st April 2019 and 1st July 2019.
SHP incurred a total cost of SGD2,824,760 (USD2,093,880) over the span of 14 weeks to replace the affected medications for 29,794 patients. The direct cost was SGD 2,488,289 (USD 1,844,469) while indirect cost amounted to SGD336,471 (USD 249,412). This translates to about SGD95 (USD70) per patient.
Discussion
Within 14 weeks of the HSA notification, SHP completed the recall of 29,794 (92.1%) affected patients. The challenges encountered in the implementation of the crisis management plan provided invaluable lessons. We examined the crisis management using implementation science approaches, focusing on its reach to target patients, adoption and evaluation of the remedial measures, in addition to addressing the economic and reputation impact on the institution.
>A key element in the crisis management process was the alertness of institution leaders leading to the early formation of a multidisciplinary task force. The primary objective was to ensure that the contaminated medication was replaced within 14 weeks. It was deemed a reasonable duration given the large numbers of affected patients.
Ideally, the affected medication should be replaced for every patient. In this crisis, at least nine out of ten patients received a medication replacement. The local healthcare system allows patients to access any healthcare provider without any geographical restriction. No official patient registry is implemented throughout the entire primary healthcare system. Nonetheless, the Healthcare Information Department in the institution was able to generate a list of patients prescribed losartan at a prior polyclinic visit. Hence, patients who did not receive any medication review at the institution could have sought medical attention from other healthcare providers, who could have similarly facilitated an exchange.
The strategy to group the patients based on their risks and upcoming appointment dates allowed for prioritization and load levelling at the polyclinics. Data analysis revealed a majority (69.9%) had a scheduled appointment in the next 3 months, increasing the pressure to ensure that replacement medications and other resources were available to meet this need. Of note, 3.9% of patients in Group A (Table 2) did not turn up for their appointment despite receiving SMS reminders. This could be related to a failure in receiving the message due to telecommunication or mobile phone issues, the message being misinterpreted or ignored. In retrospect, the phrase in the message “no further action needed” might have misled the patients, resulting in the missed appointments. Messages have to be written clearly and tested on volunteers to exclude any ambiguity in future mass communication endeavors.
Overall, the use of various communication modalities to inform patients proved to be effective. The delivery of SMS and letters led to the 100% recall of patients in Group B (Table 2).
Patients without any scheduled appointment fared the worst in the recall (Table 2) with 17.5% not having any scheduled appointment at their respective polyclinic (Group C). Every effort was made to contact this group of patients, starting with sending SMS1a to individuals with a working handphone number. The majority of patients who were dispatched the SMS did not respond and this prompted SingHealth Polyclinics staff to call them. Each patient was called three times over 2 days; a letter was dispatched to the residences of those patients who remained uncontactable. Despite these difficulties, 70% of patients in Group C received a medication exchange. This group of patients could have multiple healthcare providers or passive health-seeking behavior. The national broadcast by the health authority and easy accessibility of the polyclinic could have collectively impacted their decision to seek medication review within the stipulated period.
The close uptake rate between the planned and actualized medication replacement alluded to the effectiveness of the strategies employed as depicted in (Figure 2). The peak uptake was between weeks 4 and 10, as the messages would have reached most patients. The timely introduction of additional service points allowed clinics to cope with the additional influx of patients.
The institution incurred a cost of over SGD2.8 million (USD2.09 million) for the LRS or about SGD95 (USD70) for each patient affected by the contaminated medication. While the cost incurred was significant, prioritizing timely interventions to reduce the risk of our patients developing any potential adverse events was the institution’s priority. This is in keeping with SingHealth’s philosophy of care: “Patients. At the heart of all we do”.
Beyond the potential cost and serious threat to life associated with the development of complications due to the consumption of contaminated medication, the repercussions on the healthcare provider’s reputation can be immense. The expeditious call to action has preserved the institution’s reputation and maintained the patients' trust. This further highlights the importance of effective and timely crisis management, as adverse events can detrimentally erode the patient’s confidence in the healthcare system.
Conclusion
The recall of the tainted losartan was a national healthcare crisis. The crisis management plan underpins the framework of the LRS. The early decision of institution leaders to form the crisis management task force was crucial in the successful formulation and coordinated implementation of the crisis management plan. The data generated via the various platforms allowed the progress of the LRS to be monitored. Regular reviews by the task force resulted in timely decision-making and focused interventions to address the barriers. Multi-media communication was also helpful in reaching out to affected patients and directing them to the appropriate channels to review their medications.
Supplemental Material
Supplemental Material - Crisis management in the community mass recall of angiotensin II receptor blocker losartan due to nitrosamine impurities
Supplemental Material for Crisis management in the community mass recall of angiotensin II receptor blocker losartan due to nitrosamine impurities by Ng Lok Pui, Prawira Oka, Lim Jit Fan Christina, Aau Wai Keong, Koh Yi Ling Eileen, Ee Guan Liang Adrian and Tan Ngiap Chuan in Proceedings of Singapore Healthcare.
Footnotes
Acknowledgements
The authors would like to thank the entire polyclinic workforce for their dedication to ensure patient safety amidst the crisis.
Author contributions
LPN, PO and NCT conceptualized the study. WKA and EYLK analysed the data. LPN, PO, CJFL and NCT interpreted the results. LPN, PO, CJFL, AGLE and NCT drafted the manuscript and all authors reviewed and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
SINGHEALTH CENTRALISED INSTITUTIONAL REVIEW BOARD (CIRB). CIRB Ref: 2020/2672. The SingHealth CIRB has reviewed and determined that the application does not require further ethical deliberation because this application is a Service Evaluation project to describe an effective and efficient crisis management framework that can be used by a healthcare institution in response to a medication recall.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
