Abstract
Tumour lysis syndrome is common in haematological malignancies but is rarely reported in solid tumours. Peripheral blood lymphocytosis is an autoimmune feature of thymomas. We report a 63-year-old female who presented with a mediastinal mass, spontaneous tumour lysis syndrome and a leukoerythroblastic picture on peripheral blood film. Bone marrow aspiration and trephine biopsy ruled out haematological malignancy. Subsequent biopsy of the mediastinal mass confirmed thymoma. This is the first reported case of thymoma with peripheral blood lymphocytosis presenting with spontaneous tumour lysis syndrome. Clinicians are reminded that solid tumours may masquerade as haematological malignancies in the presence of peripheral blood lymphocytosis, hence careful clinical evaluation is needed to differentiate between the two diagnoses.
Introduction
Thymomas are rare epithelial solid tumours that may be associated with various autoimmune conditions such as myasthenia gravis (MG), pure red cell aplasia (PRCA), hypogammaglobulinaemia and peripheral blood T lymphocytosis (PBTL).1,2 PBTL itself is rare, with only 21 cases of PBTL were reported between 1997 and 2019. 1 Solid tumours rarely present with tumour lysis syndrome (TLS). We describe the first case of thymoma with peripheral blood lymphocytosis presenting with spontaneous TLS.
Case report
A 63-year-old indigenous Malaysian woman with prior co-morbids of hypertension and type 2 diabetes mellitus presented from the emergency department with abdominal distension and lethargy for the past 1 month, with recent difficulty breathing on lying flat for the past 1 week. She had a blood pressure of 130/76 mmHg, pulse rate of 101 beats per minute, oxygen saturation of 99% under room air when recumbent, respiratory rate of 22 breaths per minute and was not febrile.
Her complete blood count showed normocytic normochromic anaemia (haemoglobin 10.6 g/dL, mean corpuscular volume of 90 fL, mean corpuscular haemoglobin concentration of 31 pg), marked lymphocytosis (white blood cell count of 34 × 103/μL, neutrophils 55%, absolute neutrophil count of 18.71 × 103/μL, lymphocytes 25%, absolute lymphocyte count of 8.49 × 103/μL) and thrombocytopaenia (platelet count of 45 × 103/μL). Other haematological investigations include reticulocyte count of 2.31%, and a normal coagulation profile. Peripheral blood film (PBF) taken at initial presentation showed a leukoerythroblastic picture with 6% blasts (Figure 1).
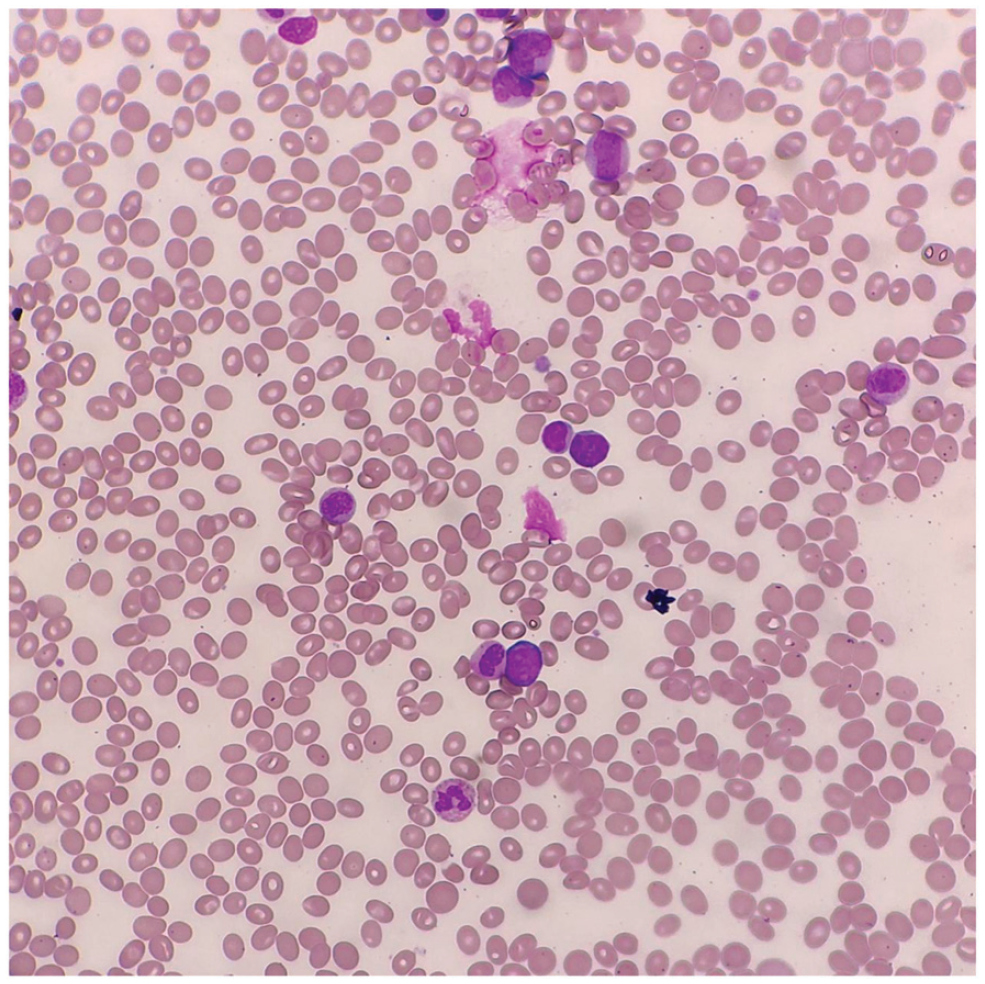
Peripheral Blood Film (PBF) showed lymphocytosis, with leukoerythroblastic picture. There are 6% small to moderate size blasts demonstrating round to slightly irregular nuclear margin, with open to clumped nuclear chromatin, and occasional prominent nucleoli.
Serum biochemistry was remarkable for markedly raised uric acid of 3,068 μmol/L, lactate dehydrogenase of 1,422 U/L, phosphate of 2.19 mmol/L, and raised potassium of 5.4 mmol/L. She had renal impairment with a urea of 42.9 mmol/L, and creatinine of 709 mmol/L. She fulfilled the Cairo–Bishop definition of laboratory and clinical tumour lysis syndrome (TLS). Venous blood gas examination revealed decompensated metabolic acidosis – pH of 7.30 and bicarbonate of 9.8 unit mmol/l. Calcium, globulin levels were normal. Serum beta-HCG and alpha fetoprotein were both within normal range. Chest radiograph revealed a widened mediastinum. Echocardiogram showed no pericardial effusion.
The patient was unable to lie flat, was tachypneic and required oxygen supplementation. She was then referred to the intensive care unit (ICU) for close monitoring from the emergency department. In view of TLS with acute kidney injury and decompensated metabolic acidosis, she was commenced on urgent renal replacement therapy (RRT). Intravenous dexamethasone 8 mg twice daily was commenced overnight on the day of admission to ICU due to presumed SVCO and the possibility of lymphoma as a differential diagnosis. She did not require mechanical ventilation and remained stable on nasal prong oxygen supplementation. With her clinical condition improving after dialysis, bone marrow aspiration (BMA) and trephine biopsy was performed the next day, in view of blasts seen in her PBF. BMA and trephine biopsy showed no excess of blasts or abnormal lymphoid population; hence no flow analysis was performed.
A contrasted computed tomography of the neck, thorax, abdomen and pelvis was performed subsequently, which showed a non-invasive anterior mediastinal mass measuring 4.9 × 9.0 × 8.7 cm, sub-centimetre cervical and axillary lymphadenopathy, and mild hepatosplenomegaly. There was no evidence of SVCO, no thrombus or abscesses. Ultrasound guided percutaneous biopsy of the mediastinal mass confirmed type B3 thymoma, Masaoka–Koga stage I (Figure 2). The patient was referred to the oncology team and started on chemotherapy. She made a good clinical and biochemical recovery and was transferred out of the ICU. She was subsequently referred to the cardiothoracic team for consideration of thymoma resection.

Histopathological examination and immunohistochemical staining of mediastinal tissue showed (a) epithelial cells diffusely reactive for CK AE1/AE3, 40× (b) sheets of epithelial cells exhibiting hyperchromatic, round to ovoid and also elongated nuclei with scant and moderate cytoplasm, haematoxylin and eosin stain, 200×. (c) immature T cells interspersed amongst the epithelial cells, Tdt stain, 200×. Overall, the findings were consistent with a type B3 thymoma.
Discussion
Thymomas are rare epithelial neoplasms which have the unique ability to generate mature T cells from immature precursors, 3 as evidenced by the presence of immature T cells interspersed amongst epithelial tissue in our patient’s histology sample. PTBL is rare, with only 21 cases reported in the past 42 years. 1 The exact mechanism of PTBL remains elusive, but is postulated to be due to a ‘spillover’ effect of thymic lymphocytes into the peripheral blood. 4 The presence of 6% blasts in PBF may have been due to reactive blast formation within thymic tumour tissue. As the BMA was performed the day after dexamethasone initiation, the absence of blasts could be attributed to glucocorticoid-related suppression of the ‘spillover’ effect previously described. Hence, we were not able to prove the T-cell lineage of the lymphocytosis at presentation. It is known that glucocorticoids may induce thymoma regression via apoptosis of thymic lymphocytes and suppression of tumour epithelial cell proliferation. 5 Steroid pulse therapy has been proposed as an induction therapy for thymomas prior to operative intervention which until today remains the mainstay of thymoma management. 5
TLS is commonly seen in haematological malignancies, 6 but in solid tumours is rare – with only 45 cases reported between 1977 and 2002. 7 Our patient fulfilled both the laboratory criterial for TLS as evidenced by elevated potassium, uric acid and phosphate levels and the clinical criteria for TLS, as evidenced by raised creatinine. 8 The majority of TLS occurs post-chemotherapy, while spontaneous TLS is much rarer and is characterised as TLS occurring in the absence of active chemotherapy. 6 Risk factors for TLS in solid tumours include high tumour volume with metastasis, liver metastasis, and high pre-treatment LDH, uric acid, and renal insufficiency. 9 The literature on thymoma-related TLS is sparse – it was first reported in 1997, and occurred in a patient post-chemotherapy, 10 and was followed by only two case reports since then.11,12 To our knowledge, this is the first report of spontaneous tumour lysis in thymoma with associated PTBL.
Conclusion
TLS has long been associated with haematological malignancy, but must not be forgotten in the setting of solid tumours. Clinicians should be aware of PTBL as one of the myriad autoimmune manifestations of thymoma, thus a leukoerythroblastic picture on PBF should not limit one’s differential diagnoses exclusively to haematological malignancy. Strong clinical suspicion for solid tumours should be heightened in the presence of obvious radiological abnormalities, and if proven, necessitates urgent tissue biopsy for a definitive diagnosis.
Footnotes
Acknowledgements
The authors would like to thank the Director General of Health Malaysia for the permission to publish this paper.
Author contributions
LN wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the manuscript. All authors were involved in the overall management of the patient.
Availability of data
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from National Medical Research Register Malaysia (ID NMRR-21-190-58634).
Informed consent
Written consent was obtained from the patient for their anonymised information to be published in this article.
