Abstract
Intravenous drug use, central catheters and intracardiac devices are known predispositions to right-sided infective endocarditis (IE). We report a case of tricuspid IE caused by Acinetobacter seifertii and Enterobacter bugandensis as a result of intravenous use of skin-whitening products bought online. Clinical implications and pharmaceutical regulations are briefly discussed.
Case report
A 26-year-old nulliparous female with no significant past medical history presented to the emergency department (ED) with productive cough for 3 months, worsening shortness of breath associated with orthopnoea and reduced effort tolerance for 2 months and fever for 1 day. There were no complaints of chest pain, haemoptysis, constitutional symptoms or symptoms to suggest autoimmune disease. COVID-19 tests by polymerase chain reaction (PCR) done prior to presentation were negative. She made multiple visits to the general practitioner during her course of illness and was prescribed a few courses of oral antibiotics but remained unwell. On presentation to ED, she was confused, dyspnoeic at a rate of 30 breaths/minute, tachycardic at 145 bpm, febrile at 37.9 C, saturating at 95% on room air, normotensive and pink. Systemic examination including a complete cardiovascular examination was unremarkable. She became increasingly dyspnoeic despite high-flow oxygen and was subsequently intubated for ventilatory support. Shortly after intubation, she desaturated and was pulseless, requiring cardio-pulmonary resuscitation (CPR) for 2 min.
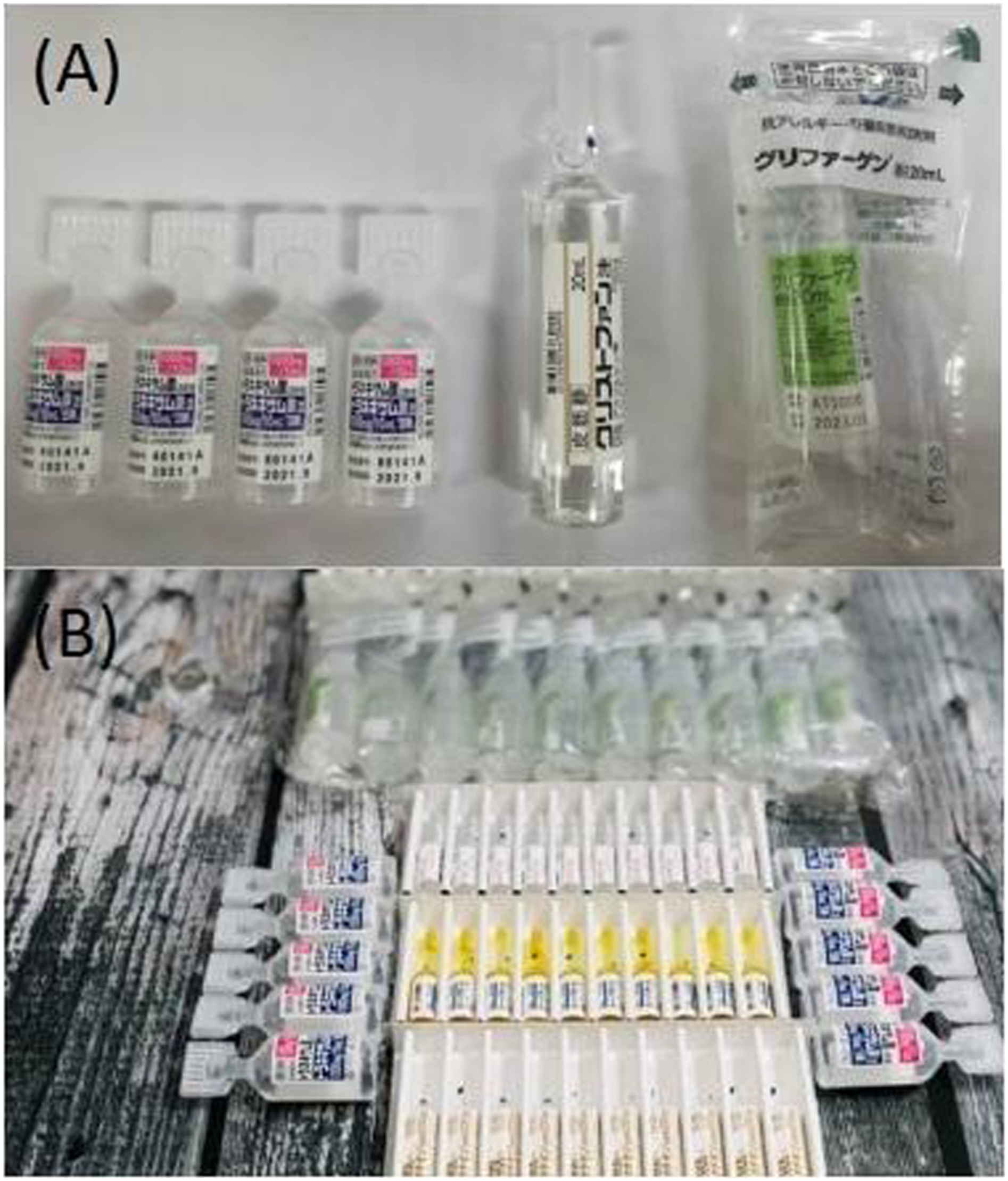
Laboratory investigations taken prior to resuscitation revealed severe leucocytosis with a C-reactive protein of 16 mg/dL and severe metabolic acidosis with a plasma lactate of 6.0 mmol/L. Bedside echocardiogram revealed a mobile, oscillating mass attached to the tricuspid valve with low reflectance (Figure 1), and computerised tomography (CT) pulmonary angiogram was in keeping with multiple septic pulmonary emboli with widespread pneumonic changes (Figure 2). Serology testing for HIV, hepatitis B and hepatitis C were negative, and investigations for connective tissue disease including antinuclear antibody, antidouble-stranded DNA antibody, complements C3 and C4, and anti-neutrophil cytoplasmic antibody later returned negative results. She did not show features of connective tissue or autoimmune disease nor did she exhibit any high-risk behaviour. Family history was of no significance. She was not on any prescribed medications but had recently started using intravenous beauty products purchased online for the purpose of skin whitening. Examination and cross-checking with the online platform from where the products were purchased estimated a 6-month usage. Three different types of vials were identified and found to contain 1000 mg tranexamic acid, 1000 mg ascorbic acid and 400 mg undisclosed amino acids, respectively (Figure 3). It was later revealed that she had the products administered by her friend on a weekly basis. The injection sites were usually cleaned with 70% isopropyl alcohol, and needles were discarded after single use. Echocardiogram showing a vegetation measuring 1.6 × 1.2 cm at the tricuspid valve. CT pulmonary angiogram showing bilateral extensive pneumonia. (A) Vials of beauty products consisting of tranexamic acid, ascorbic acid and amino acids used for skin-whitening purposes. (B) Similar set of intravenous beauty products advertised on an online shopping platform.

Intravenous meropenem and cloxacillin were initiated and continued after three cultured blood samples grew Acinetobacter seifertii and Enterobacter bugandensis. Whilst her inflammatory biomarkers, blood pressure and fever improved with antibiotics, the patient developed acute tubular necrosis requiring continuous veno-venous hemodiafiltration and acute liver failure with coagulopathy secondary to ischaemic hepatitis. Her Glasgow Coma Scale remained at E1VTM1 (2T/15) despite having ceased sedation, instigating a brain magnetic resonance imaging (MRI) which reported severe hypoxic ischaemic encephalopathy. She subsequently succumbed on week three of admission.
Discussion
Right-sided infective endocarditis (IE) is less common than left-sided IE, accounting for only 5–10% of all IE cases. However, with the increasing use of indwelling central catheters, intravenous drugs and intracardiac devices, its prevalence is expected to rise. 1 Intravenous drug use (IVDU) is the commonest cause of right-sided IE and while it usually refers to recreational drugs or drugs of abuse, the increasingly popular use of intravenous traditional and aesthetic medications administered by non-medical personnel deserves attention. Recently, a case of right-sided infective endocarditis following intravenous administration of traditional Chinese medicine was reported in literature. 2
Staphylococcus aureus accounts for up to 90% of IE affecting right-sided native valves. 1 Less implicated organisms include coagulase-negative Staphylococcus, Streptococcus, Enterococcus, gram-negative bacilli and fungi.1,3 In our case, cultures from central and peripheral bloods grew Acinetobacter seifertii and Enterobacter bugandensis – both novel gram-negative bacteria associated with catheter-related bloodstream infections and rare causes of endocarditis.4,5 The former organism is sensitive to most antimicrobials, whilst the latter is a virulent extended-spectrum beta-lactamase (ESBL) producer, thus warranting the continued use of meropenem in this patient. Cloxacillin was administered to treat possible staphylococcus infection of cutaneous origin caused by intravenous drug usage.
Transthoracic echocardiography offers high diagnostic specificity in right-sided IE given the valves’ proximity to the chest wall. Nonetheless, differentiating a thrombus from an infective vegetation can be challenging, requiring clinical correlation and additional imaging modalities such as cardiac MRI.1,6 In this case, infective vegetation is more likely than a thrombus in view of the patient’s intravenous drug use, positive blood cultures, pulmonary septic changes on CT imaging, raised inflammatory biomarkers such as C-reactive protein and echocardiographic findings (high mobility, appendment to the valvular leaflet and low reflectance). 7 By using the Modified Duke Criteria, our patient fulfilled one major criterion (presence of valvular vegetation) and three minor criteria (intravenous drug use, pulmonary septic emboli and atypical causative microorganisms on blood culture), confirming the diagnosis of infective endocarditis.
Tranexamic acid (TXA) in both oral and intravenous injection forms are licensed and approved for use in fibrinolysis-associated haemorrhage. 8 Under the Poisons Act 1952, it falls under Group B, First Schedule Poison and can only be sold by licensed personnel with a valid prescription from a registered healthcare professional. 9 In this case, TXA was bought online without a prescription and administered without supervision of a healthcare professional, with off-label indication.
Fair complexion has long been a cultural benchmark of beauty and youth amongst Asian women. In a survey involving female students in Malaysia, 60% of respondents used skin-lightening products, quoting reasonable pricing, accessibility and ingredients as purchasing factors despite being aware of possible adverse effects. 10 The desire to achieve societal beauty standards plus the lack of regulation by authorities and accessibility via online shopping platforms contribute to the rampant use of unregulated medicinal products.
Tranexamic acid works by inhibiting fibrinolysis by displacing plasminogen from fibrin in the coagulation cascade. 11 It also decreases tyrosinase expression and subsequently inhibits melanogenesis, making it an increasingly popular treatment for melasma. 12 However, only the oral, topical and intradermal routes of administration have been supported by clinical studies for melasma treatment. 13 Not only does intravenous TXA gather poor literature support on skin-depigmenting efficacy, it also raises concerns on increased risks of bloodstream infections, cardiac overload and thromboembolic events.13,14 Although evidence associating TXA with thromboembolic events is contradictory,14,15 a recent cohort study concluded that venous thromboembolism is an independent risk with use of TXA in trauma patients. 14
Conclusion
With the increasing social pressures to achieve beauty standards and availability of unlicensed drugs from online platforms, intravenous beauty products should be explored in the workup of right-sided IE. This case report also highlights the dire consequences of unregulated medicinal use and thus urgently calls for governing bodies to closely regulate unlicensed pharmaceutical trading.
Footnotes
Acknowledgments
None.
Author contributions
Huzairi Sani: Data acquisition and write-up of the manuscript
Nada Syazana Zulkufli: Write-up of the manuscript
Gan Yi Lung: Write-up of the manuscript
Ainur Fadlina Nadzir: Review of manuscript and revision of manuscript
Sazzli Kasim: Review of manuscript and revision of manuscript
Availability of data
Yes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable
Informed consent
Not applicable
Trial registration
Not applicable
