Abstract
Background
Recently, thoracoscopic resection of pulmonary sequestration has become more common, since resection of an aberrant artery using an end-stapler is a safe maneuver in many cases. However, injury of the vessels can lead to major hemorrhage. We reported our surgical experience based on thoracoscopic surgery, with five cases of interlobar pulmonary sequestration, focusing on precautions for aberrant arterial vessels.
Object and methods
We performed pulmonary resections for five patients with interlobar pulmonary sequestration in a lower lobe (left, n = 4; right, n = 1) between April 2004 and May 2020. All aberrant vessels were derived from the lower thoracic artery. Two patients had a single aberrant artery and three had multiple. In four patients, these vessels were detected before surgery, and pulmonary sequestration was diagnosed in four. In one elderly patient, the aberrant vessel was overlooked, and lung cancer was suspected before surgery. Angiography or multidetector-row computed tomography was subsequently performed in four cases. The surgical plan was determined according to the location and size of the pulmonary lesion and three-dimensional images of aberrant vessels.
Result
In all patients, approaches were made thoracoscopically. Hemorrhage from an anomalous vessel was encountered in one case. Pulmonary resections included two lobectomies and three limited resections. Angioplasty for the root of anomalous branches was performed following pulmonary resections under converted minimal lateral thoracotomy in two cases.
Conclusion
Preoperative assessment of the anatomical variations in abnormal vessels is essential to achieve safe surgical procedures. According to the situation of the aberrant vessels, selecting surgical procedures with consideration of potential subsequent complications arising over a long period of time is important.
Introduction
Pulmonary sequestration (PS) is a congenital anomaly of the lung, involving anomalous non-communication of parenchyma with normal bronchial trees, and aberrant arteries derived from systemic arteries. PS is sometimes treated by surgical resection, for reasons such as recurrent infection or bleeding.1,2 Since these lesions show a nodular or cystic formation, differentiation of PS from tumor or granulomatous nodules is also important.1,2 Detection of abnormal vessels derived from a systemic artery is essential to confirm PS. Recently, thoracoscopic resection of PS has become more common since resection of an aberrant artery using an end-stapler is a safe maneuver in many cases. However, injury of the vessels can lead to major hemorrhage. Preoperative assessment of the anatomical variations in abnormal vessels using multidetector-row computed tomography (MDCT) is essential to achieve safe surgical procedures. In this study, we evaluate our thoracoscopic procedures dealing with aberrant arterial vessels in 5 cases of PS from a single institution
Patients and Methods
Between April 2004 and May 2020, pulmonary resection was performed for five patients with PS in our institution. All cases underwent thoracic computed tomography (CT). Angiography or MDCT was used to obtain further information and assess the structure of aberrant vessels. Based on the preservation of lung parenchyma, surgical plans were determined according to the location and size of the sequestered area, and the diameter of aberrant vessels. Surgery was started under a thoracoscopic approach, with a focus on the management of aberrant vessels.
Results
Patient Characteristics.

PN: pulmonary nodule, PS: pulmonary sequestration, LC: lung cancer.
areexamining image findings

Surgical Result and Outcome.

ILPS: interlobar pulmonary sequestration

The key image of an overlooked aberrant vessel from the descending aorta in Case 3.
In this study, aberrant arteries arose from the thoracic aorta in all cases, as single vessels in three cases, and multiple in two. The draining vein was a systemic draining vein (azygos system) in two. As for management of aberrant vessels and draining veins to the hemi-azygos vein, stapling resection was safely completed in four cases. In a large-diameter case (Case 2), as a temporally maneuver, the distal branches of aberrant vessels were ligated using the knot pusher and divided (Figure 1(a)). Pulmonary resections included two lobectomies and three limited resections. In the case with limited resection, the margin of sequestered parenchyma between normal tissues was clear. Wide-wedge resection of sequestered lung was easily achieved using the end-stapler. On the other hand, in the lobectomy cases (Case 3 and 4), massive adhesion made the boundary between normal and sequestered lung unclear.
After limited pulmonary resection, angioplasty was performed in two cases. One case (Case 1) was the case with injury of the proximal branch, while the second (Case 2) involved a stump of large-diameter branch (24 mm). Following conversion to minimal lateral thoracotomy, lateral mattress suturing of the vessel stump at its origin with pledgets, under lateral clamping of the thoracic aorta, was performed under direct vision. Reasons for choosing this technique were that injury to the intima of the proximal branch (Case 1) or a remnant large aberrant vessel stump (Case 2) may subsequently result in delayed complications, such as aneurysmal formation. The pathological diagnosis was interlobar sequestration in all cases. All patients displayed an uneventful postoperative course. Follow-up for patients ranged from 6 to 57 months. Follow-up CT did not detect abnormal findings such as aneurysmal formation at the vascular stump. During the follow-up period, left pneumothorax developed 3 months after surgery in Case 5, and thoracoscopic bullectomy was performed. Looking at the site where the abnormal vessels were divided, the stump of the aberrant vessels was shortened and the surrounding area was organized (Figure 3). (A) Thoracoscopic findings showing the surgical stamps of the aberrant vessels at a distance from the branching point and (B) the surgical stamps 3 months after first surgery in Case 5.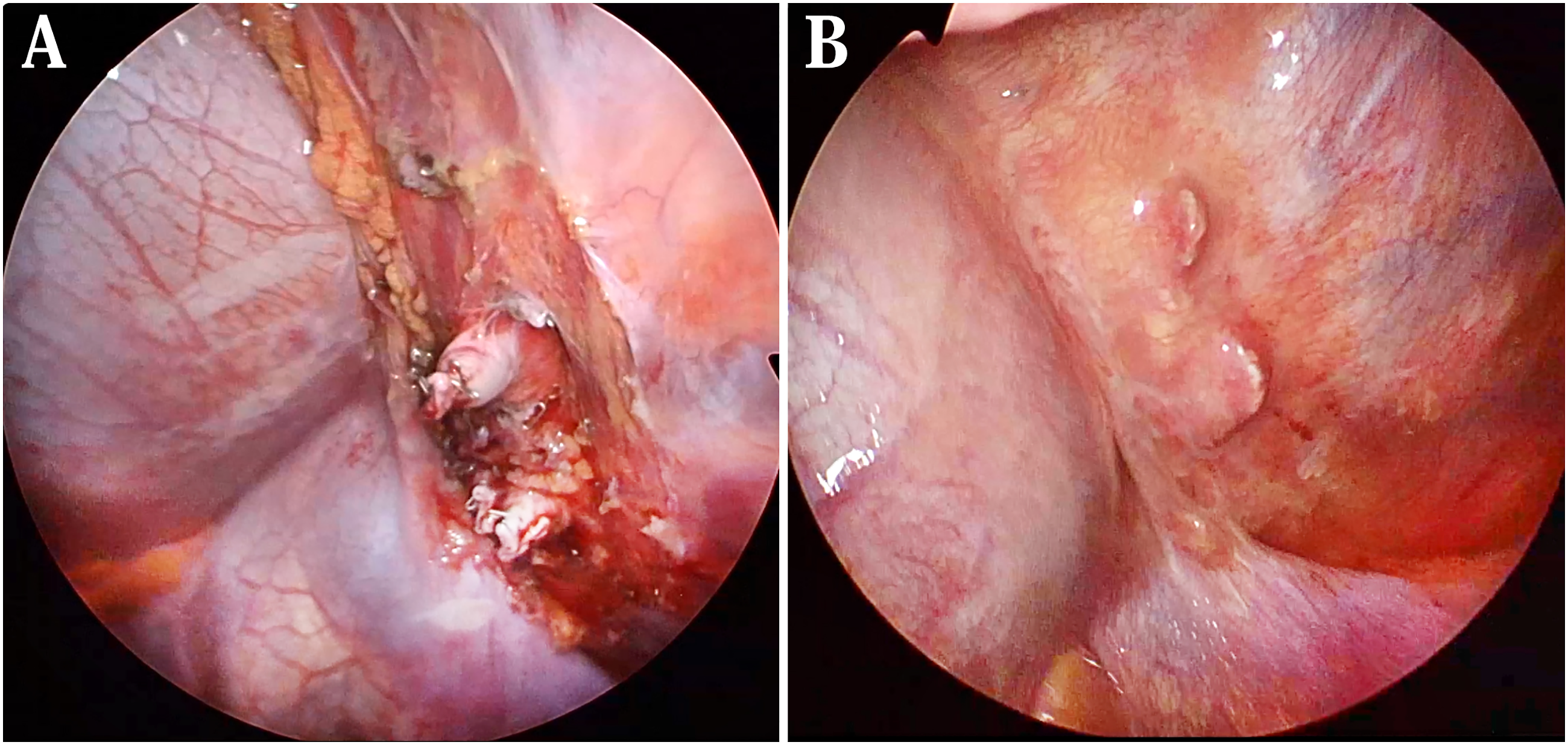
Discussion
Thoracoscopic surgical resection for PS has recently been becoming more common. Stapling resection of aberrant vessels using an end-stapler has recently been considered a safe maneuver in many cases. Generally, the majority of aberrant vessels arise from the descending thoracic aorta (76%) or abdominal aorta (21%). 2 Most cases involved a single supply (79%), with a smaller proportion showing two or more supplies (21%).1,2 In our experience, two of five cases (40%) showed multiple branches. These results impressed that the PS with multiple aberrant systemic supplies were relatively common. The surgical problems in vascular management are as follows. First, the vessels are direct branches from the aorta, and are thus constantly exposed to high intra-luminal blood pressure. Second, these vessels are more fragile than normal arteries of similar size. 1 An aberrant vessel wall histologically shows numerous elastic fibers and few smooth muscle layers, more closely resembling the pulmonary artery than the bronchial artery. As experienced in Case 1, such vessels are easily damaged by excessive tension or traction. Dissection of sufficient vessels and gentle maneuvering are required, especially when inserting an end-stapler for division of these vessels. Third, anatomically, most of these vessels enter the lung through the pulmonary ligament, which is deep below in the thorax. The pulmonary ligament is sometimes thickened and hemorrhagic due to chronic inflammation of sequestrated lung. These make surgical operability for aberrant vessels difficult. Intraoperative injury of these vessels, especially at the root of vessel, might lead to life-threatening hemorrhage, even during thoracotomy. Undiagnosed PS in right lower lobe, as in our Case 3, always carries a surgical risk of unexpected aberrant vessels.2,4 Especially, the right side approach in the left lateral decubitus position makes it anatomically difficult to secure the descending aorta as a last resort. Interlobar sequestration is diagnosed at or before 20 years old in approximately 50–60% of cases. 5 Discovery in patients over 70 years old (as in Case 3) is rare. As a result, abnormal pulmonary vessels were overlooked in consideration of lung cancer since lung cancer is more prevalent than PS. As PS is predominantly seen in the posterior basal segment,1,5 care must be taken to avoid overlooking aberrant vessels associated with undiagnosed mass lesions located in the posterior basal segment. In this study, we determined the surgical plan according to the anatomy of aberrant vessels using MDCT-angiography. In our experience, two of five cases (40%) showed multiple branches. Most preoperative images were consistent with surgical findings, but two thin-walled small vessels in Case 1 were not visible on preoperative MDCT imaging. Assessment of the anatomical variations of the aberrant arterial vessels before surgery is important. Management of aberrant vessels depends on the size of the vessels. In our experience from this study and the literature,6,7 division of aberrant arterial vessels using an end-stapler is safe and suitable for vessels up to 20 mm in diameter. Since the mean diameter of vessels associated with PS in the literature is 6.3–6.6 mm, 1 most aberrant vessels can be managed using an end-stapler. Theoretically, cutting immediately after the aberrant vessel branches from the descending thoracic aorta is optimal. Advances in the technology of linear stapling devices have led to a narrower anvil and greater flexibility, allowing easier access beyond strictures in the thoracic cavity. However, in our experience, maneuvering a stapling device to perpendicular to the proximal aberrant vessel branch in the thoracoscopic procedure is difficult. The device is instead often inserted diagonally into the vessel, and the tip contacts the mediastinal surface. A medical accident report suggested that a risk of injury to the inner layer of the main artery appears immediately after grasping the branching artery using the end-stapler, resulting in the potential for serious bleeding consequences. 8 The stapling site of an aberrant vessel using an end-stapler should thus be a certain distance from the branching point (Figure 3(a)). In such situations, we are concerned about subsequent complications at the cut-edge of fragile tissues. No reports have described aneurysm formation from remnant aberrant vessels after surgery. In Case 5, left pneumothorax surgery was able to reconfirm the stumps of three aberrant vessels (maximum diameter = 8 mm) 3 months after the initial surgery. The stumps of these vessels (Figure 3(a)) were shortened, and the surrounding area was organized (Figure 3(b)). The healing process for divided vascular stumps appeared good. On the other hand, aneurysmal dilatation with a diameter of 25 mm was reportedly seen in 0.25% of PS. 1 A few cases have been reported of PS with aneurysmal formation of an aberrant artery.9,10 The question thus remains as to whether a large vessel diameter or injury to the root of a remnant vessel represents problems. The main point of concern is that vessel stumps in either of the above situations may result in subsequent aneurysmal formation under long-term exposure to high blood pressure.
In addition, after pulmonary resection, angioplasty 11 was performed in two cases under minimum lateral thoracotomy. There are interesting surgical reports of PS with aneurysmal dilatation at the origin of the aberrant vessel. 9 The lumen of the aberrant vessel was closed by engraft, which involves application of aortic aneurysm treatment (thoracic endovascular aortic repair; TEVAR) in advance, and the aberrant vessel was divided using a stapling device. This method seems suitable for management in cases of dilated or aneurysmal aberrant vessels, as long as the endograft does not involve important blood vessels. The purpose of this surgery is in some way similar to two of our cases. The difference is whether to close the vessel of origin from the inside or outside, and this method also seems to be effective as an alternative treatment for a late-onset aneurysmal disorder of the vascular stamp.
Conclusion
We reported on our surgical experience with 5 cases of PS that we encountered at our hospital, focusing on the division of aberrant vessels. Multidetector-row computed tomography was used to provide further information to assess the structure of aberrant vessels. Almost all cases of aberrant vessels are ≤ 2 cm in diameter and could be managed using an end-stapler. However, the surgical stumps of these vessels are fragile and exist in a hypertensive environment. According to the situation of the aberrant vessels, selecting surgical procedures with consideration of potential surgical risk including subsequent complications arising over a long period of time is important.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
