Abstract
Amniotic fluid embolism (AFE) is a rare and potentially catastrophic condition unique to pregnancy. Presentation may range from subtle clinical events to sudden and fatal maternal cardiorespiratory arrest, with a reported incidence of 2–6 in 100,000 deliveries and mortality of 15–60%. Management should include immediate high-grade resuscitation for AFE presenting as cardiac arrest, involvement of multidisciplinary team, provision of adequate ventilator and circulatory support, treatment of coagulopathy precipitated by AFE, and immediate delivery of the foetus in cases of cardiac arrest. The following case report involves a patient with suspected AFE post-induction of labour with favourable maternal and neonatal outcome in our local setting. The management of coagulopathy in AFE is further discussed, including use of massive transfusion protocols, pharmacological agents and recombinant factor VIIa.
Introduction
Amniotic fluid embolism (AFE) is a rare and potentially catastrophic condition unique to pregnancy. It is currently a leading cause of maternal mortality in many developed countries. AFE may occur from onset of labour up to 48 h post-partum.1,2 The pathophysiology of AFE is now attributed to immune activation with presentation of an anaphylactoid process rather than as an embolic obstruction of the pulmonary vasculature by components of amniotic fluid. 1 The presentation may range from subtle clinical events to sudden and fatal maternal cardiorespiratory arrest. It has a reported incidence of 2–6 in 100,000 deliveries and mortality of 15–60%.3,4
The Society for Fetal Maternal Medicine in 2016 suggested evidence-based recommendations for the management of suspected AFE. 5 These recommendations included immediate high-quality resuscitation for AFE, adopting a multidisciplinary approach, early ventilation and haemodynamic support, management of coagulopathy, and immediate foetal delivery in cases of cardiac arrest.
The following case report involves a patient with suspected AFE with favourable maternal and neonatal outcome in our local setting. The management of coagulopathy in AFE will be further discussed.
Case report
A 25-year-old gravida 2 parous 0 with body mass index of 38.8 and gestational diabetes mellitus on diet control was electively admitted at gestational age 38 weeks 1 day for induction of labour in view of macrosomia.
Four hours following vaginal Prostin insertion, the patient had spontaneous rupture of the amniotic membrane and subsequently developed acute respiratory distress and drowsiness.
Initial assessment
Initial assessment revealed sinus tachycardia with pulse rate of 140 beats per minute. Peripheral capillary oxygen saturations were poorly detected despite oxygen supplementation with 100% inspired oxygen. Heart and lungs examination was unremarkable. Bedside foetal ultrasound revealed foetal bradycardia.
The patient was emergently intubated and transferred to the operating theatre for emergency caesarean section. A provisional diagnosis of AFE was made in view of her acute presentation after spontaneous rupture of amniotic membrane.
The patient had poor lung compliance and low oxygen saturation (spO2) of around 70% despite FiO2 1.0. Ventilator settings were adjusted to an acceptable peak pressure of 27cm H2O, with aim of adequate ventilation and oxygenation on blood gas analysis.
Massive transfusion protocol was activated soon after arrival to the operating theatre as the patient had poor uterine tone with oozy surgical wound. Multiple boluses of phenylephrine (total 4000 mcg) and fluids (Hartmann’s 500 ml) were given to support her haemodynamics. A radial arterial line was inserted for beat-to-beat blood pressure monitoring and an internal jugular central vascular catheter for additional intravenous access and potential need for inotropic support
The patient had hypotension with systolic BP ranging between 60 and 80 mmHg which improved with blood product transfusion; however, she remained persistently tachycardic with heart rate of 160–170 bpm. Blood products given intra-operatively included 4 units of packed red blood cells, 500 ml of fresh frozen plasma (FFP), 1 unit of pooled platelet concentrate (PC), cryoprecipitate 10 units given over the first hour in operating theatre. Uterotonics included syntocinon bolus 3 units, IV duratocin 100 mcg and IM carboprost 500 mcg. Estimated blood loss was 3 l with abdominal drain and Bakri balloon drain left in-situ. The patient was kept intubated and sent to intensive care unit (ICU) post-op.
The delivered foetus had an APGAR score of 5 on the 1st and 5th minute, was intubated and sent to Neonatal ICU for further monitoring and haemodynamic support.
Multidisciplinary team involvement
The multidisciplinary approach adopted for our case included involvement of Cardiothoracic Surgery (CTS), Cardiovascular Medicine (CVM), Obstetrics, Haematology and Interventional Radiology. The patient was reviewed by CTS to explore possibility of extracorporeal membrane oxygenation (ECMO); however, she was not a suitable candidate in view of coagulopathy and use of heparin for priming of circuit. CVM performed a bedside 2-dimensional echocardiography (2DE) and assessment; significant findings included normal ejection fraction estimated at 70–75% with underfilled left ventricle, dilated right ventricle (RV), McConnell’s sign negative. Electrocardiogram (ECG) showed ST elevation with RV strain (S1Q3T3) (Figure 1).

ECG showed ST elevation with right ventricular strain pattern (S1Q3T3).
Resuscitation was continued with blood products in ICU. Arterial blood gas (Figure 2) showed high anion gap metabolic acidosis (HAGMA) and persistent lactic acidosis with lowest Hb of 9.5.

Arterial blood gas showing high anion gap metabolic acidosis (HAGMA) with lowest Hb of 9.5.
Rotational thromboelastometry (ROTEM) (Figure 3) results revealed deranged clot firmness test, reduction in Alpha angle and increased clotting time on EXTEM and INTEM.
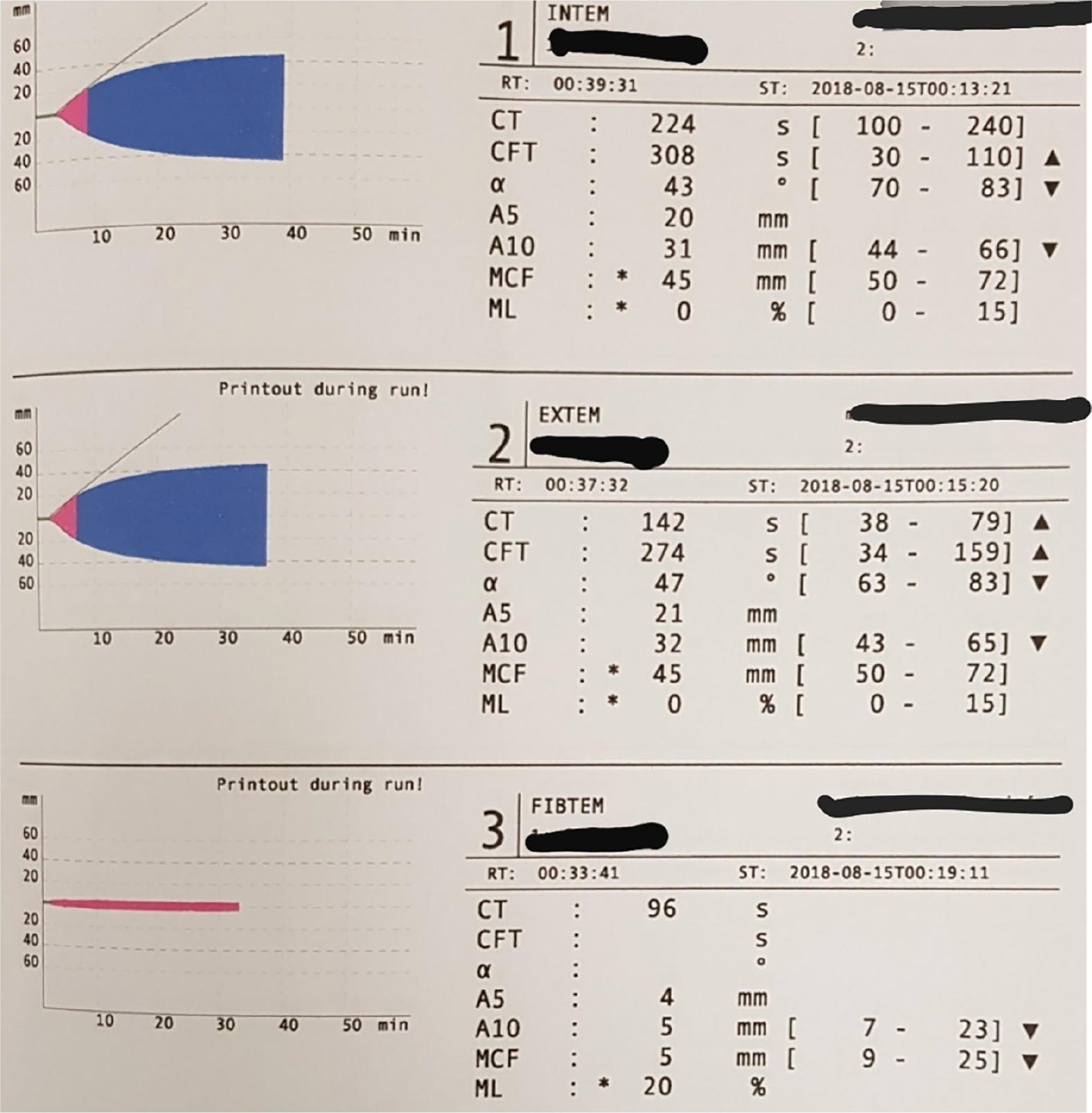
Rotational thromboelastometry (ROTEM) showing deranged clot firmness test, reduction in Alpha angle and increased clotting time on EXTEM and INTEM.
Pharmacologically, two doses of recombinant factor VII were given based on recommendation by Haematology with subsequent redosing of uterotonics; IV Ergometrine 1 ml, IV Duratocin 100 mcg with intramuscular one dose of carboprost 250 mg. Bedside B Lynch suture was applied to the uterus to further arrest bleeding surgically.
The patient subsequently underwent interventional radiology guided angioembolization of bilateral internal iliac artery.
The total amount of blood products transfused was 19 units PCT, 12 units FFP, 3 units CSP, 20 units Cryoppt, and two doses of Novo7 were given.
Outcome
The patient had a favourable outcome with subsequent extubation on day 3, transfer out of ICU on day 4 and discharge to home on day 7 of admission. She had no documented neurological deficits and repeat 2DE prior to discharge showed return to normal heart function. Her baby experienced neonatal encephalopathy secondary to perinatal depression and seizure requiring phenobarbitone and was nursed in Special Care Nursing, but was subsequently discharged with ongoing follow-up and normal developmental milestones.
Discussion
Clark et al.’s proposed model to diagnose AFE comprising four diagnostic criteria (Table 1), 4 acute hypotension or cardiac arrest, acute hypoxia, coagulopathy or severe haemorrhage in the absence of other explanations and all symptoms presenting during labour, delivery or within 30 min post-partum has been widely adopted since 1995, and our patient’s presentation met every diagnostic criteria.
Diagnostic criteria of AFE adapted from Clark SL et al. 4 .

Of note, specific markers including squamous cells in maternal pulmonary circulation, foetal antimucine TKH2 monoclonal antibody and zinc coproporphyrin may also be helpful in establishing the diagnosis of AFE. 7 However, these markers may not be rapidly available in clinical practice. It must be emphasized that achieving a diagnosis of AFE is not necessary for appropriate treatment because management is primarily supportive and based on observable pathophysiology.
In terms of hypotension, blood pressure should be supported with fluids and pressor agents when necessary adhering to BCLS and ACLS protocols. Oxygen administration should be guided by pulse oximetry and arterial blood gas. Patients may require intubation for cases of hypoxia/ dyspnoea.
Assessment of cardiovascular compromise with echocardiography may accurately guide haemodynamic therapy, while transfusion requirements may be substantial and should be anticipated. Coagulopathy should be aggressively replaced with blood and components replacement, with low threshold of activation of massive transfusion protocol. 6 The use of rapid infusers, such as the Belmont or Level 1 system, should be considered to infuse warm blood and fluids to replenish intravascular blood volume. In our case, there were noted technical difficulties with the use of rapid infusers and we had to resort to manually syringing blood products and fluids for our patient. This process did achieve a similar improved effect on the haemodynamics, albeit rather labour intensive.
There are isolated reports of survival associated with ECMO, intra-aortic balloon counterpulsation, cardiopulmonary bypass, nitric oxide, and right ventricular assist device; however, evidence is still scant and mainly experimental in nature.4,7-11 ECMO was considered for our patient; however, in view of potentially worsening of coagulopathy with use of heparin, we opted to not proceed with as discussed with our CTS surgeons.
There is now a shift from the use of Recombinant VIIa (VIIa) for treatment of coagulopathy, as there are studies implicating poorer outcomes than cohorts receiving component replacement alone. 12 It has been postulated that VIIa could interact with circulating tissue factor to enhance intravascular clot formation. 12 Therefore VIIa should probably be reserved for women with severe coagulopathy with poorly controlled bleeding despite adequate component therapy, as highlighted in our case. Delivery of the foetus should be ensured if undelivered and of viable age for survival of foetus.
The mortality associated with AFE was initially reported to be very high, with case fatality rates of 67–86 % reported in some of the first case series of AFE.13,14 However there has been improvement in recent years, where mortality of less than 20% has been observed in developed countries such as Australia,15,16 the UK, 17 Netherlands, 18 Canada 19 and the USA. 20 Factors that may contribute to lower fatality rates include an improved awareness of the condition so that women with less severe AFE are included in registries, in addition to improvements in the resuscitation of pregnant women and advances in intensive care. Considerations should be given to establish a local registry for patients with documented AFE to identify such patients presenting for repeat deliveries to anticipate potential reoccurrence and for research.
Conclusion
As AFE presentation can be variable and non-specific, a high index of suspicion is required for its diagnosis. Setting up a local registry and incorporating use of known specific markers such as maternal squamous cells, TKH2 monoclonal antibody and zinc coproporphyrin may aid in the overall management of AFE. Irrefutably, early recognition with immediate high-grade resuscitation, involvement of multidisciplinary team, provision of adequate ventilator and circulatory support, aggressive treatment of coagulopathy including use of recombinant factor VII and immediate delivery of the foetus is still mainstay management, which led to a successful maternal and foetal outcome for our patient.
Footnotes
Acknowledgements
None.
Author’s contributions
JHT wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Ethical approval
KK Women’s and Children’s Hospital does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for her anonymized information to be published in this article.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
Not applicable.
