Abstract
The diagnosis and classification of primary adenocarcinoma of the vulva is a complicated and understudied area, as this is a rare histological subtype of vulvar cancers. The differential diagnoses include extramammary Paget disease with invasion, adenocarcinoma arising from mammary-like glands, skin adnexal structures as well as Bartholin’s glands in the vicinity. We report a case of extramammary Paget disease which first presented as primary vulvar adenocarcinoma. The patient was a 71-year-old woman who presented with a swelling at the external genitalia. Clinical examination revealed a right vulvar lump, with eczematous features on the overlying skin. The lump was biopsied and was called Paget disease with underlying adenocarcinoma. Subsequently, the patient underwent radical vulvectomy and bilateral groin and pelvis dissection. The resection specimen was reported as extramammary Paget disease, with dermal invasion forming a poorly differentiated adenocarcinoma showing strong HER-2 expression on immunohistochemistry. This case illustrates the diagnostic challenge in the classification of primary vulvar adenocarcinoma. Positivity for HER-2 has been reported in Paget-associated vulvar adenocarcinoma and might offer potentially actionable targets on the selection of therapy, especially in advanced metastatic disease.
Introduction
Vulvar cancer is the fourth most common gynecological cancer and constitutes 5% of all malignancies of the female genital tract, after cancer of the uterine corpus, ovary and cervix. 1 There are several histological types, with squamous cell carcinoma of the vulva the most common category. Primary adenocarcinoma of the vulva is relatively rare, and represents a heterogenous group of tumours with a wide range of differential diagnoses, including extramammary Paget disease (EMPD) with invasion, as well as adenocarcinoma arising from mammary-like glands, skin adnexal structures or Bartholin’s glands. Each of these tumours has a different prognosis. Therefore, it is clinically relevant to classify them accurately. A resection specimen is often required to make the definitive diagnosis. We report a case of primary vulvar adenocarcinoma which was diagnosed as EMPD with dermal invasion on the resection specimen.
Case report
The patient was a 71-year-old nulliparous woman who presented with a lump at the external genitalia. There was no family history of malignancies. Clinical examination revealed a palpable lump measuring 3.5 × 3 cm, located at the right side of the vulva (see Figure 1(a)). The overlying skin showed eczematous-like changes. No inguinal lymph nodes were palpable. Clinical examination of the breasts was unremarkable. Magnetic resonance imaging of the pelvis confirmed the presence of a soft tissue mass in the right vulva with restricted diffusion and heterogenous enhancement, suspicious of a vulvar neoplasm. There were enlarged right inguinal lymph nodes. No tumour was visualised in the pelvic organs.

(a) Clinical examination showing a tumour with an erythematous to scaly surface centred at the right labia majora, measuring 3.5 cm in maximal dimension; (b) Cut sections of the tumour in a radical vulvectomy specimen showing a whitish, lobulated appearance with ill-defined margins; (c) Tumour nests in the dermis with areas of central necrosis. Haematoxylin and eosin stain; 2× magnification; (d) Higher magnification view showing nests of tumour cells with ample amphophilic cytoplasm, concentrated in the basal layer of the epidermis with pagetoid spread. Focal glandular formation was noted (arrow). Haematoxylin and eosin stain; 10× maginification.
An incision biopsy was performed. On haematoxylin and eosin (H&E) section, the epidermis showed large atypical cells with abundant pale amphophilic cytoplasm, disposed singly or in clusters, concentrated in the basal layer of the epidermis and extending to the skin adnexa. There was a tumour nodule within the deep dermis, composed of irregular nests of malignant cells with brisk mitotic activity and areas of necrosis. On immunohistochemistry, the atypical cells in the epidermis and tumour nodule showed strong cytoplasmic staining for CK7 and polyclonal carcinoembryonic antigen (pCEA), while being negative for CK20, CK5/6, HMB45, S100, CDX2, PAX8 and p16. GATA 3 showed focal positive nuclear staining. The initial interpretation of this vulva biopsy was Paget disease and underlying adenocarcinoma, with a caveat stating that the origin of adenocarcinoma will be best determined by examination of the resection specimen.
Subsequently, the patient underwent radical vulvectomy and bilateral groin and pelvis dissection.
Radical vulvectomy specimen (see Figure 1(b) to (d) and Figure 2)
Macroscopically, the skin of the vulva appeared whitish and hyperkeratotic, with a tumour centred at the right labia majora, measuring 3.5 × 3.0 × 1.7 cm. Microscopic examination showed an extensive involvement of the skin by Paget disease, extending to the right lateral and inferior resection margins. Occasional small clusters of invasive tumour in the papillary dermis were identified. In addition, there was a tumour in the dermis, composed of irregular nests and trabeculae of malignant cells connected to the overlying epidermis involved by Paget disease. The tumour cells showed moderate to high grade cytological atypia with moderate amounts of eosinophilic cytoplasm. Increased mitotic activity and areas of necrosis were observed within the tumour nodule. No ectopic breast or mammary type duct glands were seen. The tumour showed the same immunoprofile as the previous biopsy. In addition, cytoplasmic positive staining of gross cystic disease fluid protein 15 (GCDFP-15) was observed in the tumour nodule. Oesterogen receptor (ER) and progesterone receptor (PR) were negative in both tumour nodule and Paget cells. Of note, the tumour nodule showed strong membranous staining of Her- 2 (3+) while the Paget cells showed weak to moderate staining (2+). Based on the histomorphological features and immunoprofile, a diagnosis of EMPD with underlying poorly differentiated adenocarcinoma was rendered (Figure 2). Two of the right inguinal lymph nodes were positive for metastatic carcinoma, with the largest focus measuring 35 mm in maximal dimension.
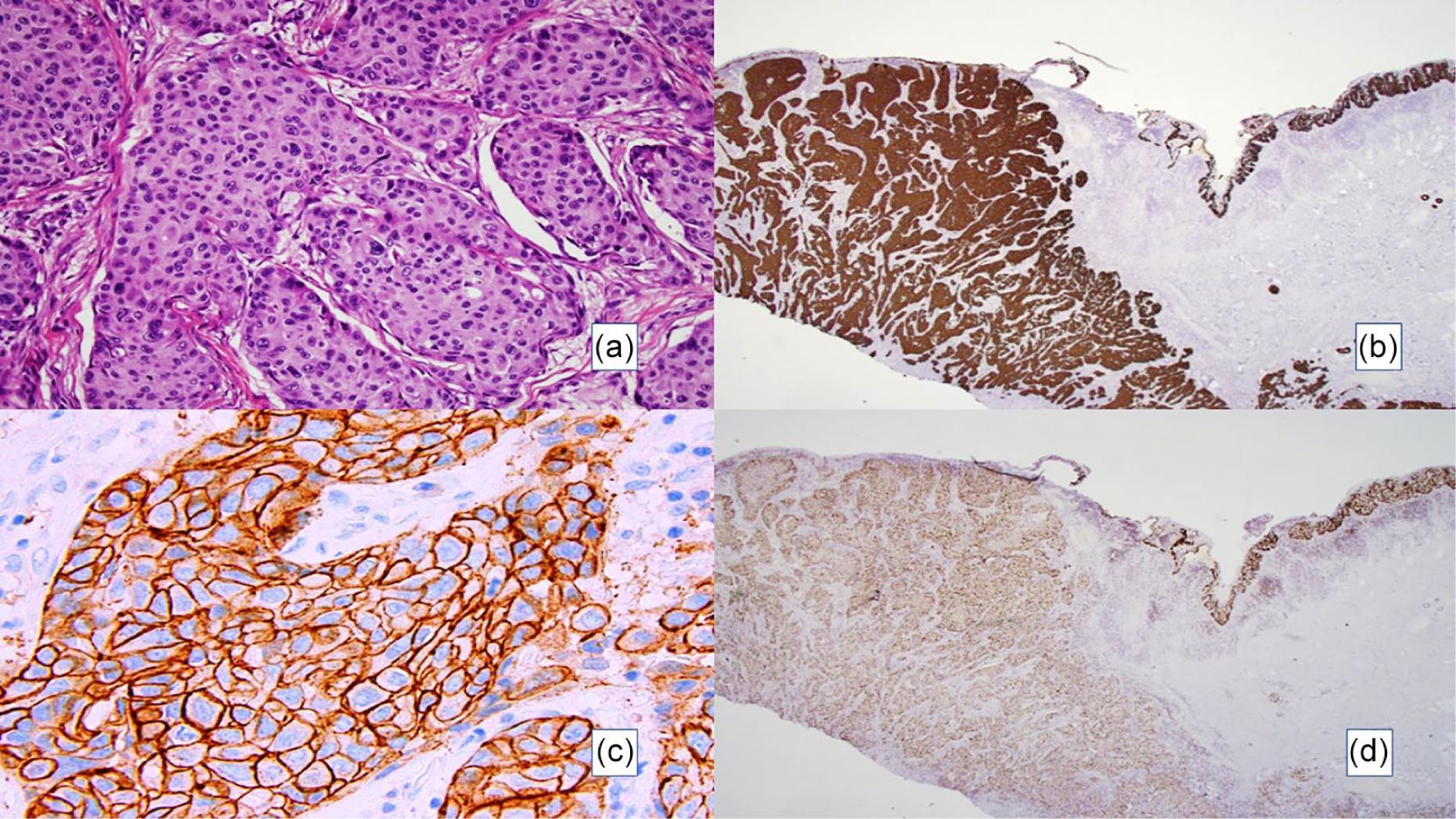
(a) Higher magnification view showing tumour cell nests within the dermis featuring pleomorphism and malignant cytological features. Haematoxylin and eosin stain; 20× magnification; (b) Strong CK7 cytoplasmic staining in both tumour nodule and Paget cells. CK7 immunohistochemical stain; 2× magnification; (c) Diffuse and strong Her-2 membranous staining (3+) in the tumour nodule. Her-2 immunohistochemical stain; 40× magnification; (d) Stronger GATA 3 nuclear staining was observed in Paget’s cells compared to the tumour nodule. GATA 3 immunohistochemical stain; 2× magnification.
The operation was uneventful, and the patient made a good recovery. She was managed as stage IIIB vulvar adenocarcinoma and received post-adjuvant brachytherapy. No chemotherapy or hormonal therapy was given in her case. She was under follow-up for more than one year and there was no recurrence to date.
Discussion
The diagnosis and classification of primary adenocarcinoma of the vulva is complicated and often overlooked due to the rarity of these tumours. In our case, the patient presented with a discrete vulvar lump, with eczematous and scaling rashes on the overlying skin. In this clinical scenario, an incisional biopsy is crucial in establishing the diagnosis, as the list of differential diagnoses of a vulvar lump is long, ranging from benign entities such as Bartholin abscess, to vulvar mesenchymal neoplasm and malignant tumours such as squamous cell carcinoma. The initial vulvar biopsy of our case was reported as Paget disease with underlying adenocarcinoma, based on the presence of characteristic tumour cells featuring abundant pale cytoplasm and large vesicular nuclei within the epidermis, forming nests and spreading in a pagetoid pattern. The immunoprofile of the tumour cells was in keeping with Paget disease (CK7 and pCEA positive). The melanocytic and squamous markers were negative. Nevertheless, the limited biopsy material precluded further classification of adenocarcinoma present in the dermis. In order to determine the origin of an adenocarcinoma, demonstration of the relationship between the invasive tumour and the overlying Paget disease is critical. The subsequent vulvectomy specimen is able to address this issue by adequate sampling of the tumour. In our case, the diagnosis of EMPD with invasive adenocarcinoma was clinched when continuity of the adenocarcinoma to overlying Paget disease was demonstrated.
On the basis of histomorphology, the top differential diagnosis in this case is mammary-like adenocarcinoma (MLA) of the vulva, which is perceived to have a poorer prognosis. MLAs of the vulva are rare and aggressive tumours that are thought to arise from vulvar mammary-like glands. These tumours arise in the subcutaneous tissue and share similar histomorphology and immunoprofiles with invasive breast carcinoma. The presence of mammary-like glands adjacent to the invasive carcinoma is a useful clue to diagnosis. 2 In our case, features that go against the diagnosis of MLA are extensive pagetoid spread of tumour cells and neither mammary-like glands nor intraductal carcinoma were demonstrated in the resection specimen. The second differential diagnosis is adenocarcinoma originating from Bartholin’s glands, which typically occurs in the labia minora. These tumours histologically demonstrate squamous differentiation or papillary architecture with mucinous differentiation, 3 both were not seen in our case. Also, these tumours typically do not have cutaneous involvement. Other considerations include primary vulvar sweat gland carcinomas arising from the labia majora. These tumour are rare and are reported to have a diverse histopathological appearance, including ductal, acinar, papillary, solid, etc. They are generally considered as a diagnosis of exclusion. 4 It is important to be aware of this entity as these tumours usually have an infiltrative growth pattern with a higher risk of metastatic disease.
Immunohistochemical studies are critical in the classification of primary vulvar adenocarcinoma.
Paget cells are known to be positive for apocrine and eccrine markers, such as low molecular weight cytokeratins, GCDFP-15 and pCEA. 5 Therefore, positive cytoplasmic staining of all these three markers for adenocarcinoma in our case, supports the diagnosis of it arising from EMPD. Strong positive staining of pCEA in tumour cells is suggestive of primary EMPD, rather than secondary EMPD with an associated carcinoma. 6 ER and PR are negative in both adenocarcinoma and Paget cells, this finding is in keeping with the result described by Diaz de Leon et al., whose study showed that the immunophenotype characteristic of apocrine carcinomas (androgen receptor-positive, ER negative, PR negative) was seen in a substantial proportion of EMPD cases. 7 In a series of 72 cases of EMPD, Zhao et al. indicated that GATA 3 is a very sensitive marker for primary genital EMPDs and is more sensitive than GCDFP15. 8 The high sensitivity of GATA 3 not only presents in intraepithelial disease but also in invasive adenocarcinomas and metastatic adenocarcinomas, as seen in our case. Of note, a positive GATA 3 staining pattern can be seen in urothelial carcinoma with secondary Paget disease, which can present with similar clinical pictures. This highlights the importance of clinical correlation as 5% of Paget disease represents secondary vulvar involvement by a regional internal cancer. 5 This diagnostic issue can easily be resolved by checking a patient’s history of primary urothelial cancer, and performing a panel of other specific eccrine or apocrine markers. In addition, a systemic work-up would be necessary to exclude a tumour in the breast, as breast carcinoma has been known to metastasise to the vulva, which could well be GATA 3 positive. 9 Patchy positive staining of GATA 3 can be observed in squamous cell carcinoma in situ, which is a differential diagnosis of EMPD especially when the tumour shows a pagetoid growth pattern. Therefore, it is important to do additional markers and not to rely solely on GATA 3 in this setting. Ber-EP4 is reported to differentiate EMPD reliably from pagetoid squamous cell carcinoma in situ and pagetoid melanoma in situ. This useful immunohistochemical stain can be included along with a panel of other markers when evaluating for pagetoid cutaneous neoplasms. 10
In most cases, EMPD is an intraepidermal adenocarcinoma in which tumour cells infiltrate the epidermis and skin appendages, showing glandular differentiation. However, this tumour possesses the potential for dermal invasion and distant metastasis to the lymph nodes. 11 In a study of 31 cases of EMPD by Masuguchi et al., 19 cases (61%) of EMPD were positive for HER-2. 12 That study also demonstrated that there was significant correlation between the presence of invasion and strong positivity (3+) for HER-2. It suggested that patients with EMPD strongly positive for HER-2 may have a high risk of lymph node metastasis and should be followed up carefully. The findings in our case support the conclusion of that study, as the tumour showed strong HER-2 positivity (3+ staining), associated with aggressive behaviour as evident by dermal invasion and lymph node metastasis. Due to its multifocal nature, the extent of EMPD is often difficult to assess clinically. As a result, surgical margins of the resection specimen are frequently positive, as seen in our case. A study by Stacy et al. showed that intraoperative frozen sections of margins in patients with EMPD of the vulva reduce the need for subsequent operations. 13 Surgeons can use this technique to evaluate margins and to define the extent of surgery necessary, thus minimising disease recurrences. Nevertheless, regardless of the margin status, patients with Paget disease of the vulva often develop multiple recurrences. 14
As targeted therapy with trastuzumab is well established in breast cancer with HER-2 overexpression, patients affected by HER-2 positive EMPD might benefit from similar therapy, particularly those with invasive or metastatic disease. 15 There have been several cases of advanced EMPD reported in the literature that have exhibited a good response to trastuzumab, alone or combined with chemotherapy. 16 More data are required to prove the efficacy of this therapy, which hopefully will lead to better treatments for patients with metastatic EMPD in the future.
Conclusions
In conclusion, we report a case of primary vulvar adenocarcinoma which was classified as EMPD with dermal invasion based on demonstration of the continuity of the adenocarcinoma with overlying Paget disease. Immunohistochemical studies are critical in the classification of this group of tumours. We wish to highlight the importance of accurate histological classification of primary vulvar adenocarcinoma, as the prognosis varies. Testing of HER-2 is important in EMPD as it might offer potentially actionable targets in the selection of therapy, especially in the setting of advanced metastatic disease.
Footnotes
Acknowledgements
None.
Authors’ contributions
CYC, YCY and GM researched the literature and conceived the study. RPN was involved in gaining consent and clinical input. CYC wrote the first draft of the paper. All authors reviewed and edited the paper and approved the final version of the paper.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Ethical approval
Singapore General Hospital and KK Women’s and Children’s Hospital do not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
