Abstract
Spinal epidural arteriovenous fistulas (SEAVFs) with intradural reflux are rare but important vascular lesions that may cause progressive myelopathy due to spinal cord venous hypertension. Although traditionally managed by means of arterial embolization or surgical disconnection, these approaches can pose risks, particularly when critical vascular structures, such as the artery of Adamkiewicz, originate in close proximity to the fistulous site. We report the case of a patient in their 60s who presented with progressive paraparesis over approximately 1 year, ultimately attributed to an SEAVF located adjacent to the right L1-L2 neural foramen, with radiculo–perimedullary reflux. The artery of Adamkiewicz was visualized with its origin near the shunt site, rendering arterial embolization unsafe and prompting selection of a purely transvenous endovascular approach. Venous access was obtained via the azygos system, and a microcatheter was navigated through the epidural venous pouch to reach a cranially directed draining vein extending toward the perimedullary venous system, which was then embolized using detachable platinum coils. The procedure achieved complete occlusion of the targeted vein, with early neurological improvement and significant reduction in spinal cord edema on follow-up imaging. This case supports the feasibility, safety, and efficacy of transvenous embolization for SEAVFs and underscores its expanding role in the treatment of complex spinal vascular lesions.
Keywords
Introduction
Spinal epidural arteriovenous fistulas (SEAVFs), also historically referred to as paravertebral arteriovenous fistulas or paravertebral arteriovenous malformations, 1 are rare vascular anomalies characterized by pathological shunting between branches of the segmental arteries and the epidural venous plexus.2,3 Among them, type A lesions are distinguished by the presence of an arterialized epidural plexus with radicular venous reflux toward the perimedullary venous system, often resulting in congestive myelopathy. 4 In contrast, types B1 and B2 are confined to drainage into the epidural venous plexus and subsequently outward to the paraspinal venous system. Type B1 is characterized by mass effect on the thecal sac and/or nerve roots due to dilated epidural veins, whereas type B2 lacks such mass effect. 4
The pathophysiology of type A SEAVFs is thought to be driven by arterialization of the epidural venous plexus, resulting in venous hypertension, which may propagate retrogradely into the intradural compartment when protective mechanisms fail.1,5,6 These mechanisms, including angulated venous junctions and focal anatomical narrowings at dural entry points, constitute an antireflux mechanism and are thought to limit perimedullary venous reflux. 7 However, when intradural drainage occurs, this may lead to venous congestion, spinal cord edema, and progressive myelopathy. In type B1 SEAVFs, neurological symptoms are presumed to arise primarily from direct mass effect by dilated epidural veins. In contrast, type B2 SEAVFs are often asymptomatic. 4
Magnetic resonance imaging (MRI) typically reveals T2-weighted hyperintensity of the spinal cord and prominent perimedullary flow voids, 4 and digital subtraction angiography (DSA) remains essential for definitive diagnosis. 8 Treatment has traditionally relied on surgical disconnection 8 or arterial embolization using agents such as n-BCA 4 or Onyx.4,9 However, when the shunt is located near the artery of Adamkiewicz, arterial embolization may carry an unacceptably high risk of spinal cord ischemia.
In recent years, transvenous embolization has emerged as a promising alternative, especially in cases where arterial access is limited or unsafe. This approach reflects broader advances in neurointervention, including the successful endovascular treatment of cerebrospinal fluid—venous fistulas via the azygos venous system. 10
We present a case of an SEAVF with radicular-to-perimedullary venous reflux, in which symptomatic intradural venous drainage was successfully occluded via purely transvenous embolization. Technical considerations and the evolving role of venous approaches in the management of spinal vascular pathology are highlighted.
Case description
A patient in their 60s presented with progressive bilateral leg weakness, paresthesias, and burning dysesthesia that had worsened over the previous year. In recent months, her condition further deteriorated, progressing from cane-assisted ambulation to complete wheelchair dependency. Neurological examination revealed spastic paraparesis, more pronounced on the right, with symmetric diminished sensation in both legs. There were no sphincter disturbances or cranial nerve deficits.
MRI of the spine demonstrated extensive T2-weighted hyperintensity and mild cord expansion from T6 to the conus medullaris, with prominent perimedullary flow voids (Figure 1). Time-resolved contrast-enhanced MR angiography revealed prominent vascular structures in the right L1-L2 periforaminal region, including an endocanalar epidural vascular pouch. Evidence of intradural venous reflux was also present, with early contrast filling of perimedullary veins. Altogether, these findings were highly suggestive of an SEAVF shunt. Pre-treatment spinal MRI: (a) Sagittal T2-weighted spin-echo image shows spinal cord hyperintensity from T6 to the conus medullaris, along with mild expansion of the cord, consistent with edema. Multiple perimedullary flow voids are visible (white arrowheads). (b) Sagittal T2-weighted image demonstrates a prominent periforaminal signal void at the right L1–L2 level (white arrowhead). (c–f) Time-resolved contrast-enhanced MR angiography reveals early venous enhancement indicative of arteriovenous shunting. (c) A prominent right L1 segmental artery is visualized (red arrow). (d) A possible fistulous network is seen in the right L1-L2 foraminal region (green arrow). (e) An arterialized radiculomedullary vein (blue arrow) emerges cranially from an epidural venous pouch adjacent to the right L1-L2 foramen. (f) Early enhancing perimedullary veins are identified (blue arrows).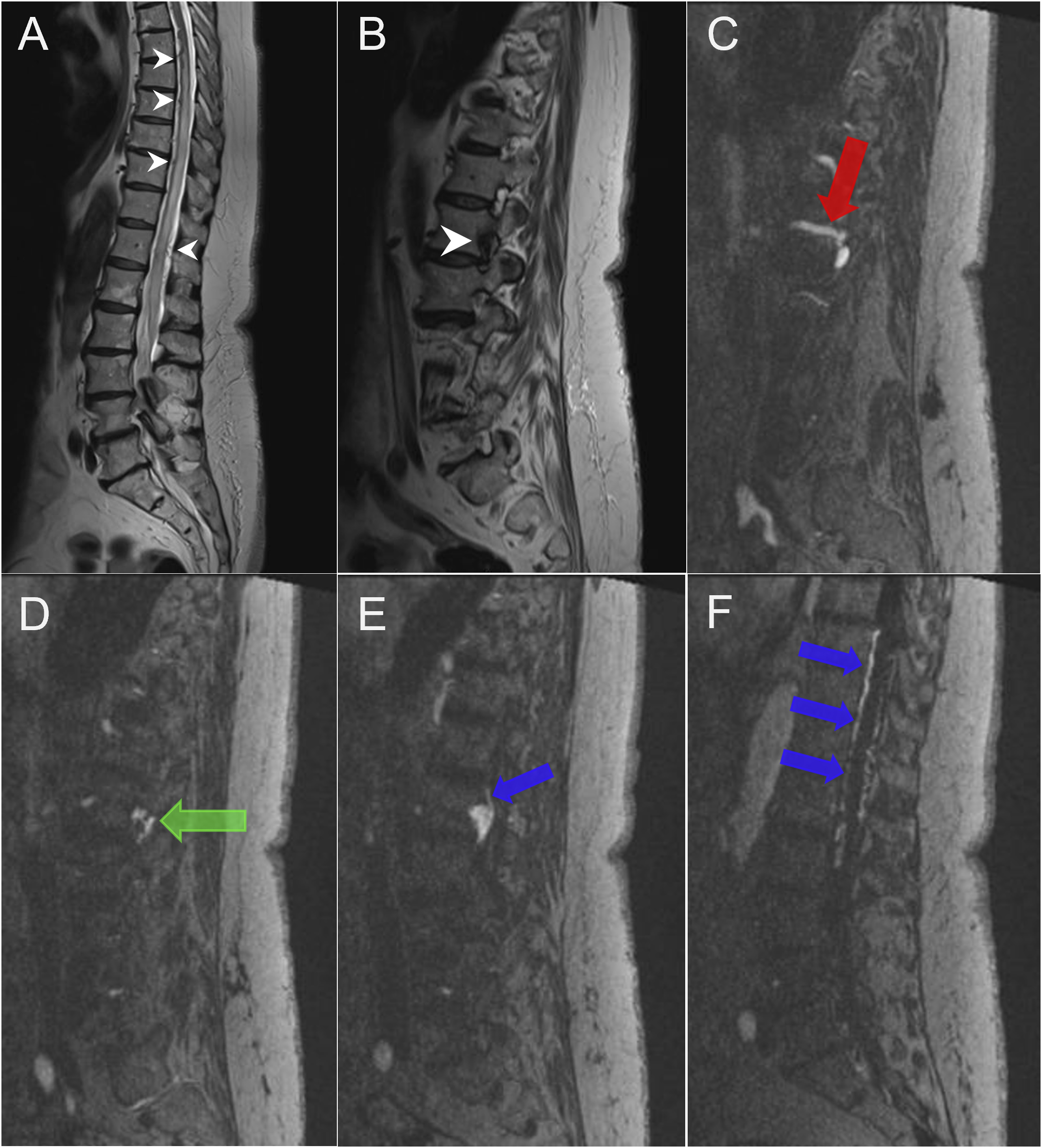
Spinal DSA demonstrated an arteriovenous fistula with early opacification of an epidural venous pouch located adjacent to the right L1-L2 neural foramen, with venous reflux into a cranially directed radicular vein extending toward perimedullary veins (Figure 2(a)–(f)). The dominant arterial supply originated from the right L1 segmental artery, with contributing feeders from the left L1 and L2 segmental arteries. The artery of Adamkiewicz was visualized, with an origin in close proximity to the point of venous reflux into the intradural compartment (Figure 2(f)). Spinal DSA and transvenous embolization: (a)–(c) Right L1 segmental artery injection through a 5F C2 catheter, showing sequential phases of contrast progression. (a) Early arterial phase reveals opacification of a prominent radicular artery and a portion of the retro-corporal fistulous epidural network. (b) Intermediate phase demonstrates the epidural venous pouch, a tortuous, ectatic radicular vein ascending toward the perimedullary venous system, and early opacification of the azygos vein. (c) Late phase shows continued venous drainage, including persistent opacification of epidural and perimedullary components. (d)–(f) Annotated versions of panels (a)–(c). (d) Red box highlights the prominent radicular artery. Green box indicates part of the fistulous network. (e) Blue box outlines the epidural venous pouch. Dashed blue line marks an arterialized radiculomedullary vein extending cranially from the pouch. Blue arrow points to the azygos vein. (f) Dashed red line outlines the course of the artery of Adamkiewicz, seen in continuity with the anterior spinal artery. (g) Smart mask image acquired from a right L1 segmental artery injection. A 6 F guiding catheter is seen positioned in the azygos system, and a microcatheter (SL-10) was advanced through the loop of the epidural venous pouch. The tip of the microcatheter is visible (black arrow) at the site of embolization. (h) Same projection as (g), without the mask overlay. (i) Post-embolization injection into the right L1 segmental artery shows persistent opacification of the fistulous network, the epidural venous pouch and the azygos vein, but no residual intradural venous reflux. The previously visualized radiculomedullary vein is no longer opacified following embolization with 15 detachable coils. The artery of Adamkiewicz and the anterior spinal artery (red arrow) are now more clearly delineated. Coils are not clearly visible in this subtracted image but are well seen on the corresponding unsubtracted acquisition (not shown).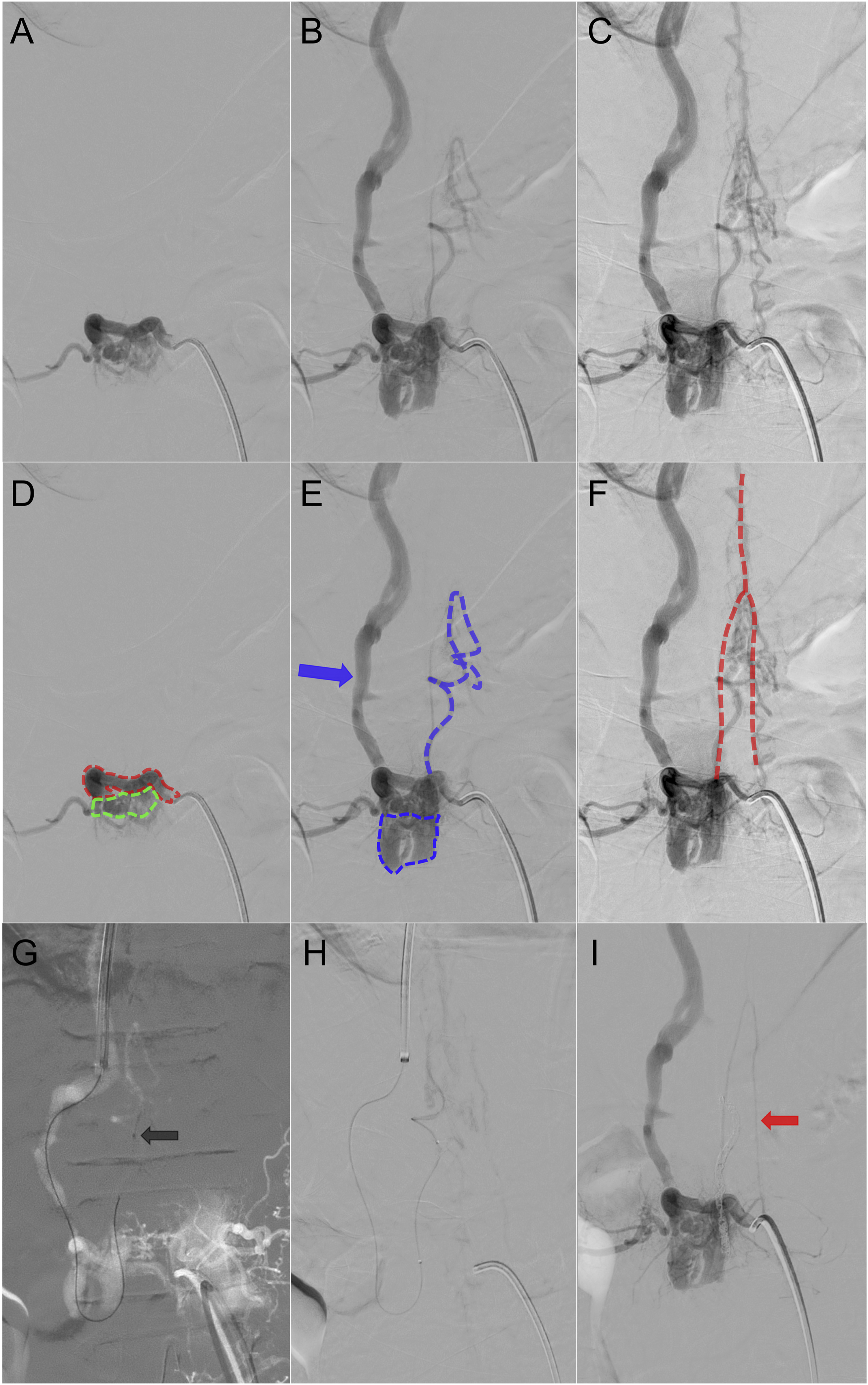
Given the complexity of the vascular anatomy and the risks associated with both endovascular arterial and surgical approaches, an exclusive endovascular transvenous approach was selected for treatment. In particular, the artery of Adamkiewicz was visualized with an apparent origin near the fistulous zone, raising concerns about the safety of arterial embolization due to the risk of anterior spinal artery compromise. Surgical disconnection was also considered less favorable, due to the intricate vascular architecture and the nearby presence of critical spinal cord supply. Notably, the close and parallel course of the arterialized radiculomedullary vein and the dominant radiculomedullary artery posed a significant surgical risk, further supporting the selection of a transvenous approach. Venous access was obtained via the right basilic vein, and a 6F guiding catheter was advanced into the azygos system. An SL-10 microcatheter (Stryker Neurovascular, Portage, USA) was navigated through the epidural venous loop of the pouch to reach the cranially directed radicular vein draining into the perimedullary venous system (Figure 2(g) and (h)). Embolization was performed with a total of 15 detachable platinum coils, deployed distally within the intradural segment and progressing proximally to the vicinity of the presumed epidural shunt point. The coils included a combination of Target 360 Ultra, Target 360 Nano, Target Helical Nano, Target Helical Ultra, and Target 360 Soft coils, ranging from 1 to 2 mm in diameter and 1 to 8 cm in length (Stryker Neurovascular, Portage, USA). The embolization achieved complete occlusion of the targeted draining vein, with no further intradural venous reflux (Figure 2(i)).
The procedure was uneventful, and the patient awoke neurologically stable. Prophylactic low-molecular-weight heparin (enoxaparin) was initiated post-procedure to reduce the risk of thrombosis propagation. Clinically, the patient reported improved strength and sensation in both lower limbs within 24 hours. Follow-up MRI performed the day after treatment showed persistent spinal cord hyperintensity and mild expansion, with a marked reduction in perimedullary flow voids (Figure 3a1 and a2). At 8-week follow-up, the patient was walking independently with a walker and climbing stairs, without sensory complaints or incontinence. MRI performed at that time demonstrated substantial improvement of the spinal cord edema and no appreciable residual flow voids (Figure 3(b)). Post-treatment spinal MRI studies: (a1), (a2) Sagittal T2-weighted spin-echo images obtained on the day following embolization show persistent spinal cord hyperintensity and mild expansion from T6 to the conus medullaris, consistent with ongoing venous congestive edema, similar to the pre-treatment study. Perimedullary flow voids are now barely perceptible. (b) Sagittal T2-weighted image acquired approximately 2 months post-treatment demonstrates substantial reduction in spinal cord hyperintensity and expansion, compatible with partial resolution of venous congestive edema. No appreciable perimedullary flow voids are identified.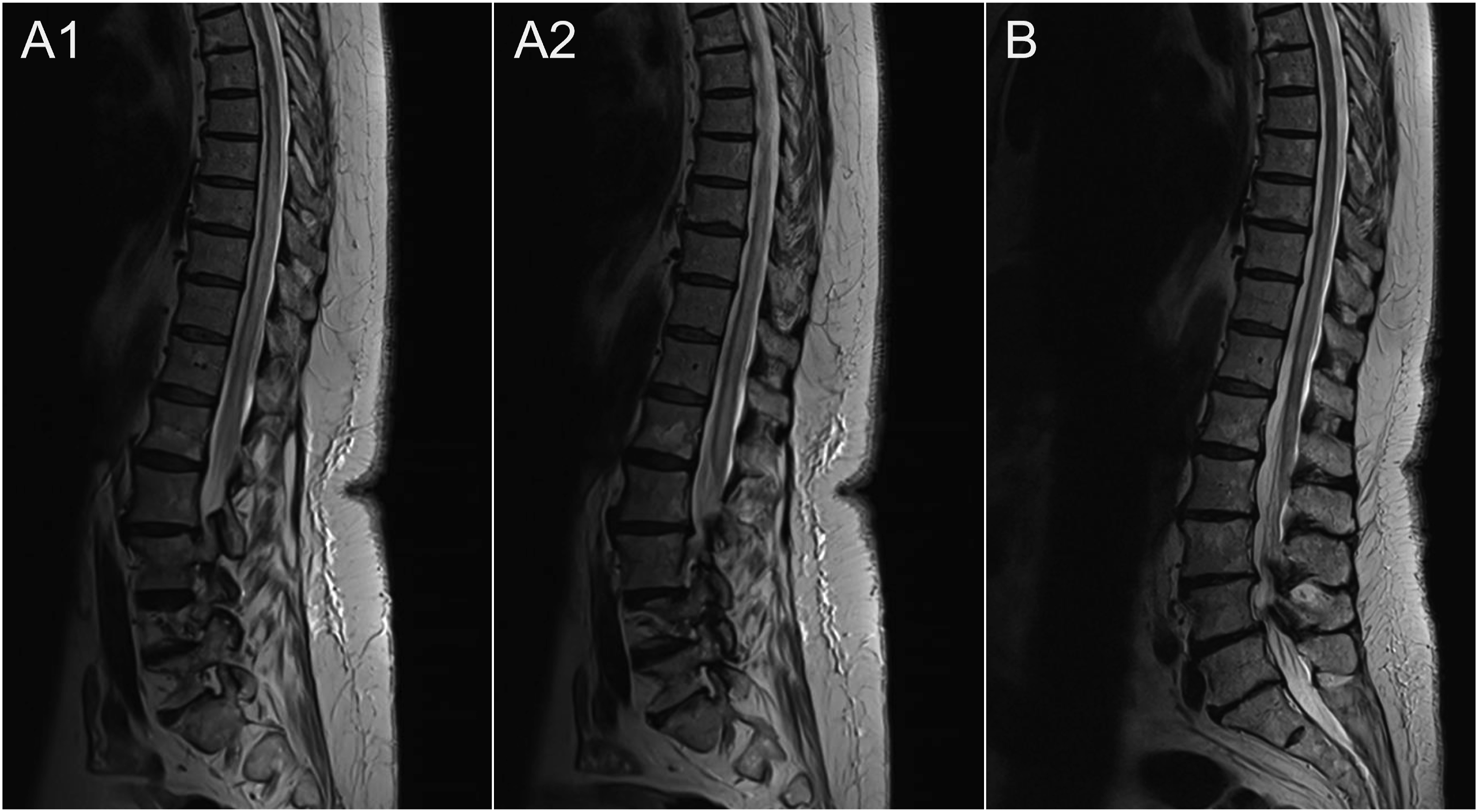
Discussion
Spinal arteriovenous shunts comprise a heterogeneous group of vascular lesions, classified primarily according to their anatomical location and angioarchitecture.3–5,11–13 Paraspinal arteriovenous shunts are fed by segmental arteries and may drain solely into the paraspinal venous system or, in some cases, reflux retrogradely into the epidural venous plexus, with or without secondary drainage into the intradural compartment. In contrast, SEAVFs represent direct connections between segmental arteries and the epidural venous system, with or without associated intradural venous reflux. Spinal dural arteriovenous fistulas are acquired lesions characterized by arteriovenous shunting within the dura mater, most often located within the nerve root sheath, and typically draining directly into intradural radiculomedullary veins. Spinal pial arteriovenous fistulas are intradural superficial vascular lesions that consist of a direct shunt between spinal pial arteries and veins. Spinal arteriovenous malformations are typically congenital lesions with a nidus, most commonly located in a perimedullary position or embedded within the spinal cord, and less frequently along the dorsal or ventral nerve roots. Additionally, these lesions may also manifest as metameric lesions, often extending across multiple tissue planes, reflecting their embryologic segmental origin.
Spinal arteriovenous shunts, despite their diverse angioarchitectures, often share overlapping clinical and imaging manifestations.13,14 They typically present with progressive myelopathy resulting from venous congestion or mass effect from engorged draining veins. Hemorrhagic presentation has been reported, most frequently in arteriovenous malformations. On MRI, findings such as spinal cord T2 hyperintensity, cord swelling, and prominent perimedullary flow voids are common across different shunt types. Time-resolved contrast-enhanced MR angiography, as used in this case, can provide preliminary dynamic information about arteriovenous shunting, although with lower spatial and temporal resolution compared to DSA. DSA remains indispensable for high-resolution, real-time assessment of arterial feeders, fistula location, venous drainage pathways, and possible intradural reflux. A multimodal imaging strategy combining MRI with DSA is therefore essential for accurate lesion classification and for guiding an appropriate therapeutic approach.
This case illustrates the importance of detailed pre-procedural characterization in selecting the safest and most effective treatment strategy for SEAVFs. Published data indicate that both surgical and endovascular techniques can achieve obliteration rates approaching 90%, with clinical improvement in most patients and no significant difference in outcomes between the two approaches. 14 The feasibility of each technique, however, depends on lesion-specific anatomical and hemodynamic factors.14,15 Transarterial embolization is generally preferred in the presence of a single accessible feeder. However, it may be technically limited by small or tortuous arteries, the risk of inadvertent embolization of critical radiculomedullary vessels, or by large venous pouches that prevent sufficient penetration of embolic material beyond the shunt point, which may allow for de novo recruitment of arterial feeders. Surgical disconnection, although effective, is often favored when the fistula is near the anterior spinal artery or when multiple feeders supply a high-flow shunt, conditions that increase the complexity and risk of arterial embolization. In our case, the artery of Adamkiewicz was visualized in close proximity to the arterial feeders, raising concern for unintentional embolization. This finding was a key factor in favoring a transvenous approach and is particularly relevant in fistulas supplied between T8 and L2, the most common segmental levels for origin of the artery of Adamkiewicz. 16 Although transvenous access can be technically challenging due to venous valves and complex metameric connections in the azygos system, the venous approach in this case provided direct access to the symptomatic draining vein, allowing targeted occlusion without jeopardizing spinal cord perfusion.
Our embolization strategy involved coil occlusion of a cranially directed radiculomedullary vein, from its intradural segment, with intentional extension down to the presumed site of epidural arteriovenous shunting. This approach aims primarily to reduce intradural venous hypertension and, consequently, the risk of venous congestive myelopathy or subarachnoid hemorrhage. While the shunt itself was not directly occluded, the strategy focused on interrupting the symptomatic intradural drainage, which represented the most clinically relevant component of the lesion, as supported by the patient’s neurological improvement. Nevertheless, clinical and imaging follow-up is recommended due to persistence of the epidural arteriovenous connection, which may remain asymptomatic or, over time, become significant because of a changed venous drainage pattern.
While surgical or arterial endovascular techniques remain the most established approaches, transvenous embolization provides a complementary alternative, either as a stand-alone or combined approach, 14 in anatomically complex or high-risk cases, particularly when arterial treatment carries unacceptable risk due to the proximity of critical spinal arteries, such as the artery of Adamkiewicz. Beyond simple coil occlusion, other strategies include direct transvenous injection of n-BCA glue and Lipiodol, especially in refractory cases after failed arterial embolization and surgery. 15 Furthermore, coil embolization via the venous route can be combined with subsequent Onyx injection to enable retrograde occlusion of small arterial feeders through the venous pouch. 17 In our case, the configuration of the arterialized radiculomedullary vein and its close anatomical relationship with the artery of Adamkiewicz raised particular concerns regarding the control and safety of liquid embolic agents. Their irreversible nature and potential for unpredictable reflux were considered less suitable in this context. Coils enabled precise, segmental occlusion from the intradural segment up to the epidural shunt zone, allowing real-time control of deployment and the ability to halt or adjust the procedure at any time. For these reasons, exclusive coil embolization was deemed the safest and most controllable option in this case. This case also reflects a broader trend in neurointervention: the increasing use of transvenous approaches in the management of complex spinal vascular lesions. 10 As experience with transvenous access increases, this route may become a first-line strategy in selected SEAVFs.
Notably, there are currently no formal guidelines for the management of SEAVFs, and decisions regarding treatment strategy—particularly the choice between surgical disconnection, endovascular arterial embolization, or transvenous embolization—are largely based on institutional experience and expert consensus. In this context, a personalized, anatomy-driven approach becomes essential, taking into account not only the angioarchitecture and hemodynamics of the lesion but also the patient’s clinical status, and the accessibility and safety of each route. This case underscores the importance of tailoring the endovascular strategy to each patient’s unique vascular anatomy and clinical profile.
Conclusion
This report highlights the feasibility and safety of transvenous coiling embolization for the management of type A SEAVFs. When arterial or surgical approaches are limited by anatomical constraints, transvenous access can offer a safe and effective means of occluding the symptomatic venous drainage associated with the fistula. Careful angiographic planning and an individualized treatment strategy are essential to optimize outcomes in these complex lesions.
Footnotes
Acknowledgements
DV and EJH acknowledge the support of University Medical Imaging Toronto.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DV and EJH acknowledge the support of University Medical Imaging Toronto.
Consent to participate
Written informed consent to undergo endovascular treatment was obtained from the patient.
Consent for publication
Written informed consent for publication of this case, including anonymized images, was obtained from the patient.
Ethical considerations
This case report was conducted in accordance with institutional guidelines. Ethical approval was not required for this type of study, in line with local policy at the University Health Network (Toronto, Canada). Written informed consent for the procedure and for publication of anonymized data and images was obtained from the patient.
Data Availability Statement
All data supporting the findings of this case report are contained within the manuscript. Additional imaging data are available from the corresponding author upon reasonable request.
