Abstract
High grade torcular DAVFs can be challenging to treat, particularly when there is retrograde flow in the straight sinus and deep venous system. We describe the technical steps to safely sacrifice the straight sinus, including preoperative MRI findings, arterial and venous cerebral flow assessment and key points for endovascular treatment.
Keywords
Introduction
Intracranial dural arteriovenous fistulas (DAVFs) are acquired and abnormal connections between meningeal arteries, shunting into dural venous sinus and/or cortical veins. Retrograde flow into cortical veins defines a DAVF as high-grade, with an annual risk of adverse events and mortality reaching 15.0% and 10.4%, respectively. 1 Various potential triggers such as venous thrombosis, trauma, or previous surgery, have been postulated to cause these acquired lesions by inducing angiogenesis in the presence of venous hypertension. 2
Four subtypes of shunting have been identified: 1 classical dural to fistulous supply from dural arterial branches (e.g. the middle meningeal arteries); 2 pre-existing dural branches of pial arteries supplying the shunt (e.g. the artery of Davidoff and Schechter); 3 de novo “pure” pial supply of the shunt (e.g. distal induced pial to dural branches of the anterior, middle and posterior cerebral arteries) 3 ; and 4 the more recently identified dural to pial supply (i.e. a dural branch supplying the DAVF with simultaneous supply to a significant area of brain parenchyma). 4
Endovascular treatment has become the mainstay of DAVF management, using transarterial, transvenous, or open ‘burr-hole’ assisted endovascular approaches. Surgery is less commonly used nowadays and reserved for certain locations that are difficult to treat endovascularly. 5 The method of endovascular treatment is dependent on the specific angiographic anatomy of the fistula, however, the vast majority of DAVFs lend themselves to treatment via transarterial embolization (TAE) after the introduction of modern liquid embolic agents such as Onyx (Medtronic, USA).6–8 In cases in which the fistulous site involves a highly stenotic, compartmentalized, or isolated sinus or a relatively small and tortuous cortical vein, TAE may be the only option.
Transvenous embolization (TVE) is suitable when the diseased sinus segment is not used by the brain, such as in DAVFs with an isolated sinus. This means that there is no normal venous drainage from the cerebral and cerebellar hemispheres into the affected sinus. Only if the brain does not use the segment to be treated can a dural sinus be safely occluded. 9 In addition, TVE can be successful even in difficult locations such as in inner petrosal ridge, ethmoidal 10 and hypoglossal canal DAVFs. 11 Lastly, TVE may be the preferred route when innumerable small feeders shunt into a widely dispersed segment of the dural sinus.
Transvenous sacrifice of the transverse, sigmoid 12 or superior sagittal sinus for the treatment of a high grade DAVF has been shown an effective treatment with durable outcomes, as long as the normal venous drainage of the brain (e.g. vein of Labbé or Trolard) is not compromised. Here, we report on the transvenous sacrifice of the torcular and straight sinus in a high-grade DAVF with numerous arterial feeders and a severely stenotic venous segment, causing significant reflux into the Galenic venous system. Key points are the preoperative imaging findings, technical steps of the procedure and key indicators that allow the proceduralist to safely take the straight sinus.
Case description
Clinical presentation and angioarchitecture
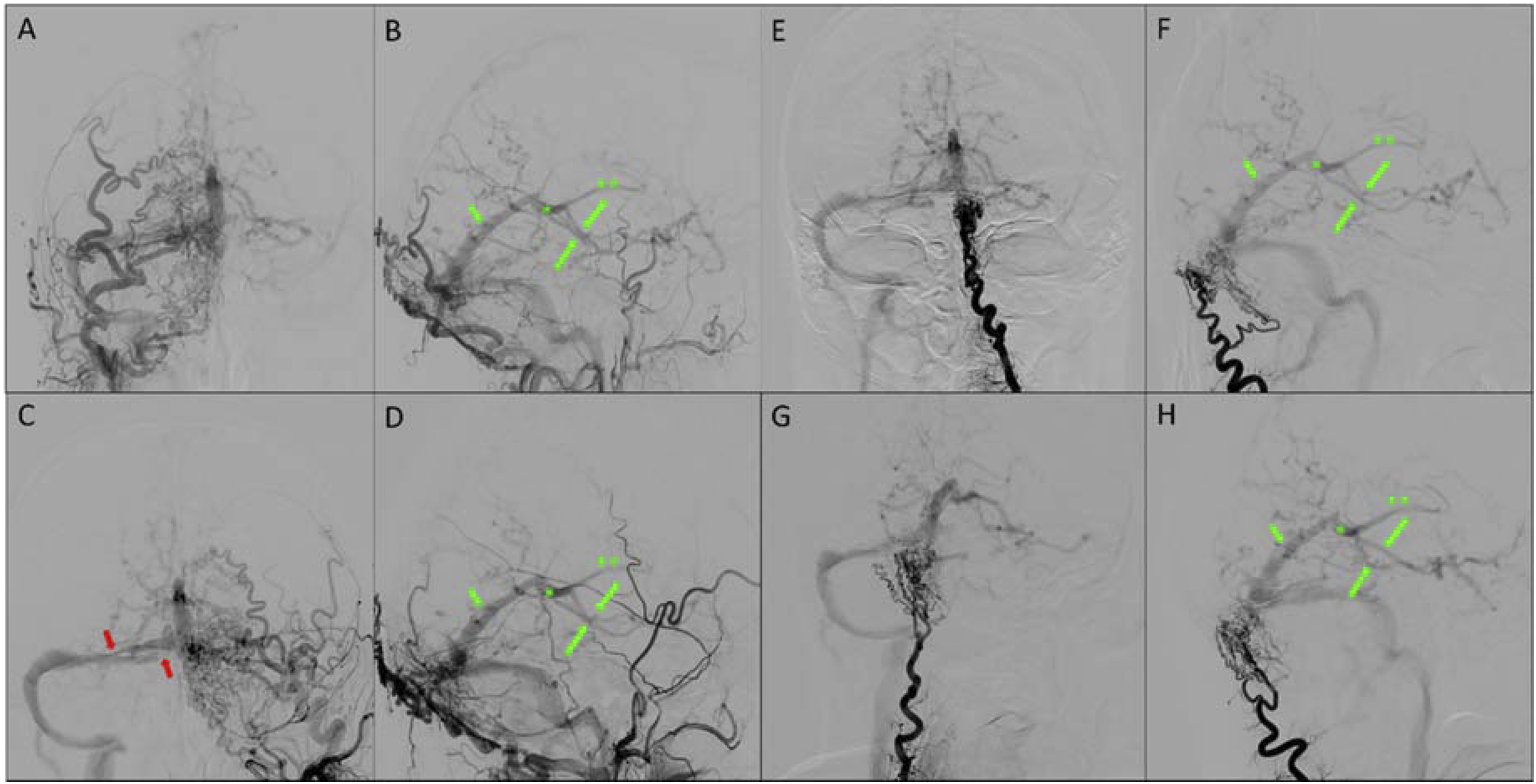
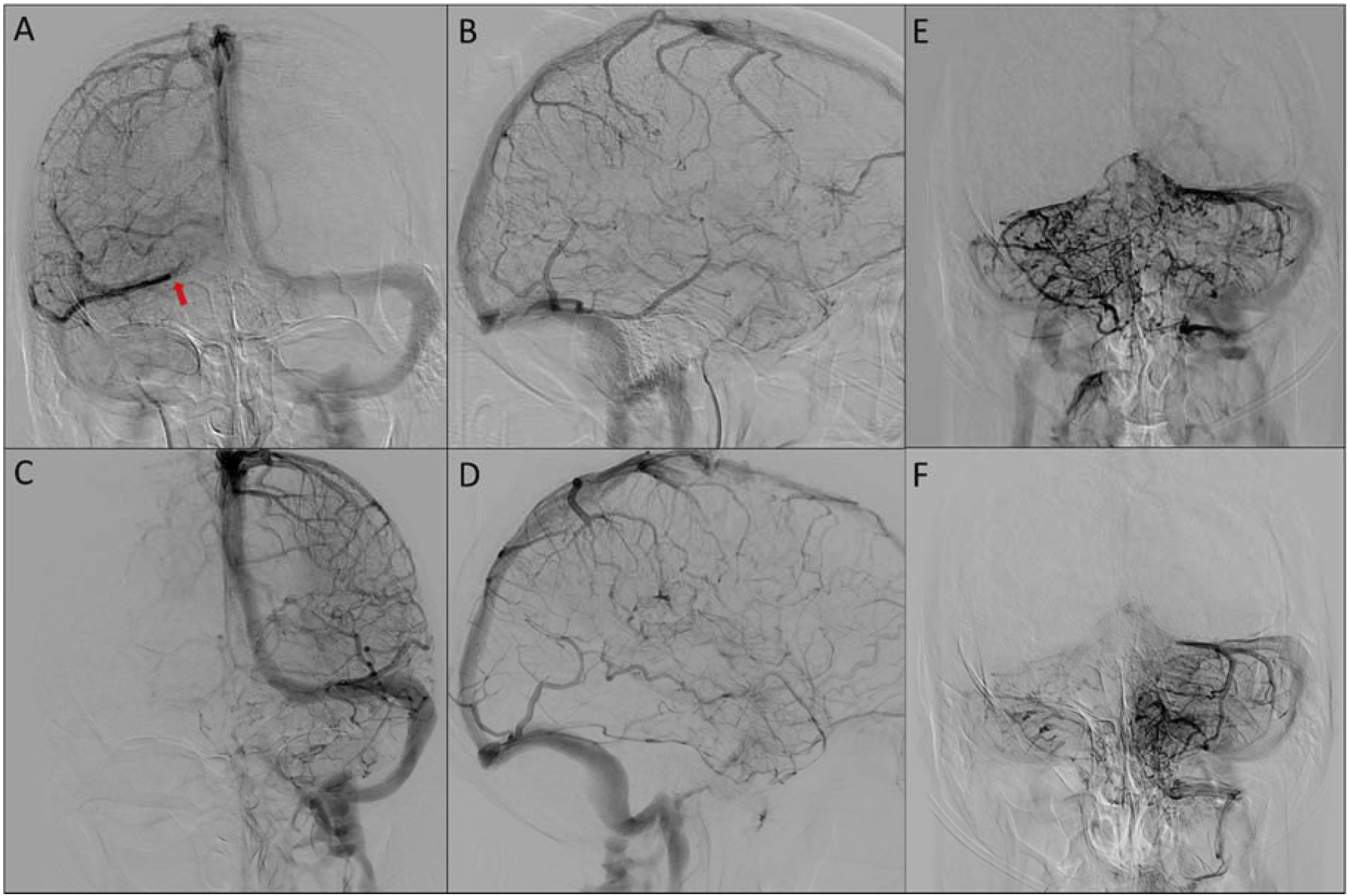
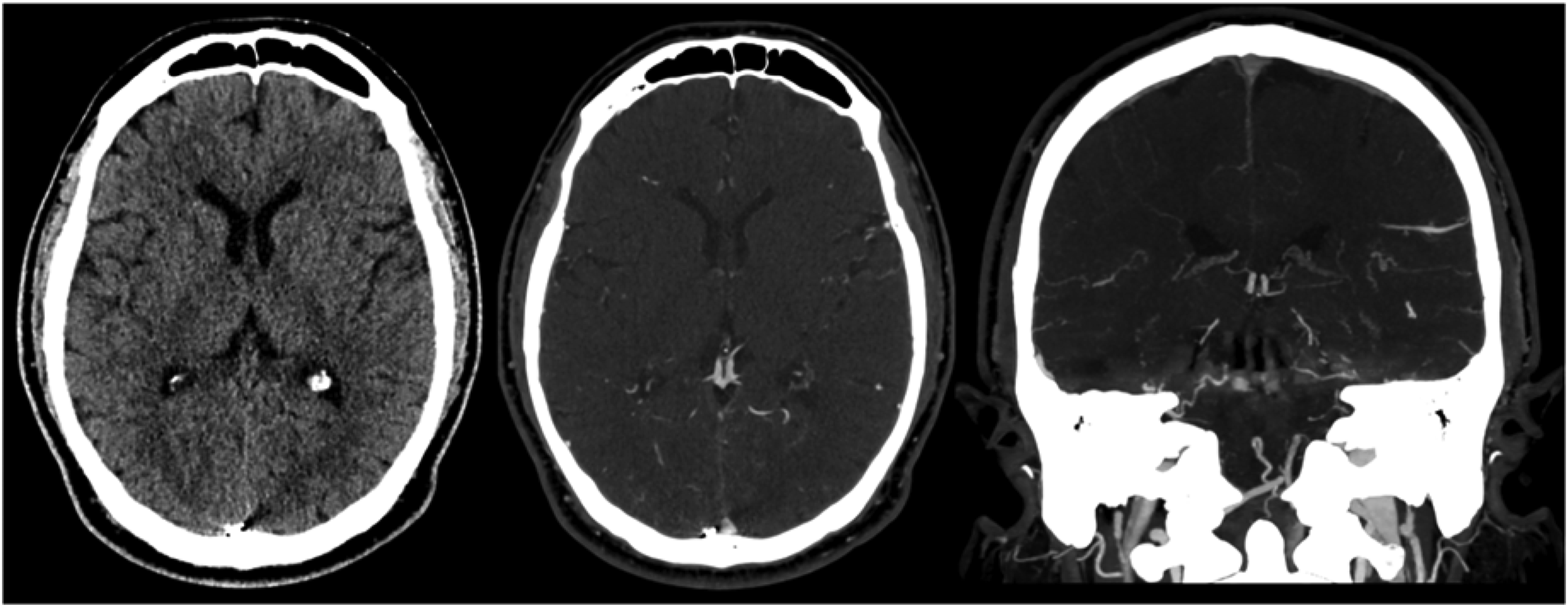
A patient in their 50s presented with cognitive decline and right-sided pulsatile tinnitus, superimposed on a history of long-standing left-sided pulsatile tinnitus. There was no known history of head trauma or cortical venous thrombosis. The past medical history was notable for atrial fibrillation and TIA managed with Apixaban. Cross-sectional imaging demonstrated suspicion on a high grade DAVF with arterialization of the Galenic venous system, including the straight sinus, internal cerebral veins and basal veins of Rosenthal. No vasogenic edema was noted in the thalami or elsewhere in the brain parenchyma (Figure 1). Diagnostic cerebral angiogram demonstrated a multihole high-grade DAVF located at the torcular herophili (Figure 2). There was classical dural supply from bilateral external carotid artery (ECA) branches: major transosseous supply from the left occipital arteries and minor from middle meningeal arteries, the posterior auricular artery and posterior meningeal artery. There was secondary supply from transosseous branches of the bilateral deep cervical arteries. There was retrograde drainage into the Galenic venous system, including the straight sinus, vein of Galen, internal cerebral veins and rerouting of flow via the bilateral basal veins of Rosenthal and deep middle cerebral veins into the superficial cortical venous system. Furthermore, there was parietal occipital and temporal cortical venous reflux as well as reflux into the posterior fossa. The affected torcular venous sinus was disconnected from the superior sagittal sinus and left transverse sinus, and exhibited high-grade stenosis at its junction with the right transverse sinus, in close proximity to the right vein of Labbe (Figure 3). The cerebral and cerebellar hemispheres demonstrated normal venous drainage via the superior sagittal sinus and left-sided dural venous system, and right vein of Labbe. The deep venous system was not involved in normal venous drainage. 3.0 T axial T2 FLAIR images (left) demonstrate absence of vasogenic edema in the thalami, basal ganglia and periventricular white matter, as well as a small area with vasculo-ischemic white matter changes in the left parietal deep white matter. Axial SWI images (right) show arterialized (hyperintense) signal in the deep venous system, including the internal cerebral veins (double asterisks), vein of Galen (asterisk) and basal veins of Rosenthal (arrows). Preoperative cerebral angiogram demonstrates a multihole high grade DAVF at the torcular. AP (A, C) and lateral injections (B, D) of the external carotid arteries demonstrate innumerable hypertrophic arterial feeders from the occipital arteries (transosseous branches), and minor supply from the middle meningeal arteries, posterior auricular and posterior meningeal arteries. AP (E, G) and lateral (F, H) injection of the deep cervical arteries show additional transosseous feeders. There is reflux into the straight sinus (small arrow), vein of Galen (asterisk), internal cerebral veins (asterisks), basal veins of Rosenthal (arrows) as cerebral and cerebellar cortical veins. The torcular is disconnected from the superior sagittal sinus and left transverse sinus and demonstrates high grade stenotic channels (red arrows) with the right transverse sinus. AP (A,C) and lateral (B,D) venograms of the right and left hemisphere, respectively and AP venograms of the right (E) and left (F) vertebral artery injections demonstrate normal patency of the left-sided dural venous system. The straight sinus is not used for normal venous drainage of the cerebral and cerebellar hemispheres. The cerebral hemispheres primarily demonstrate centrifugal venous drainage of the deep white matter towards the cortex. The right vein of Labbe demonstrates persistent drainage into the right proximal transverse sinus (arrow) close to the DAVF and deserves balloon protection during embolization.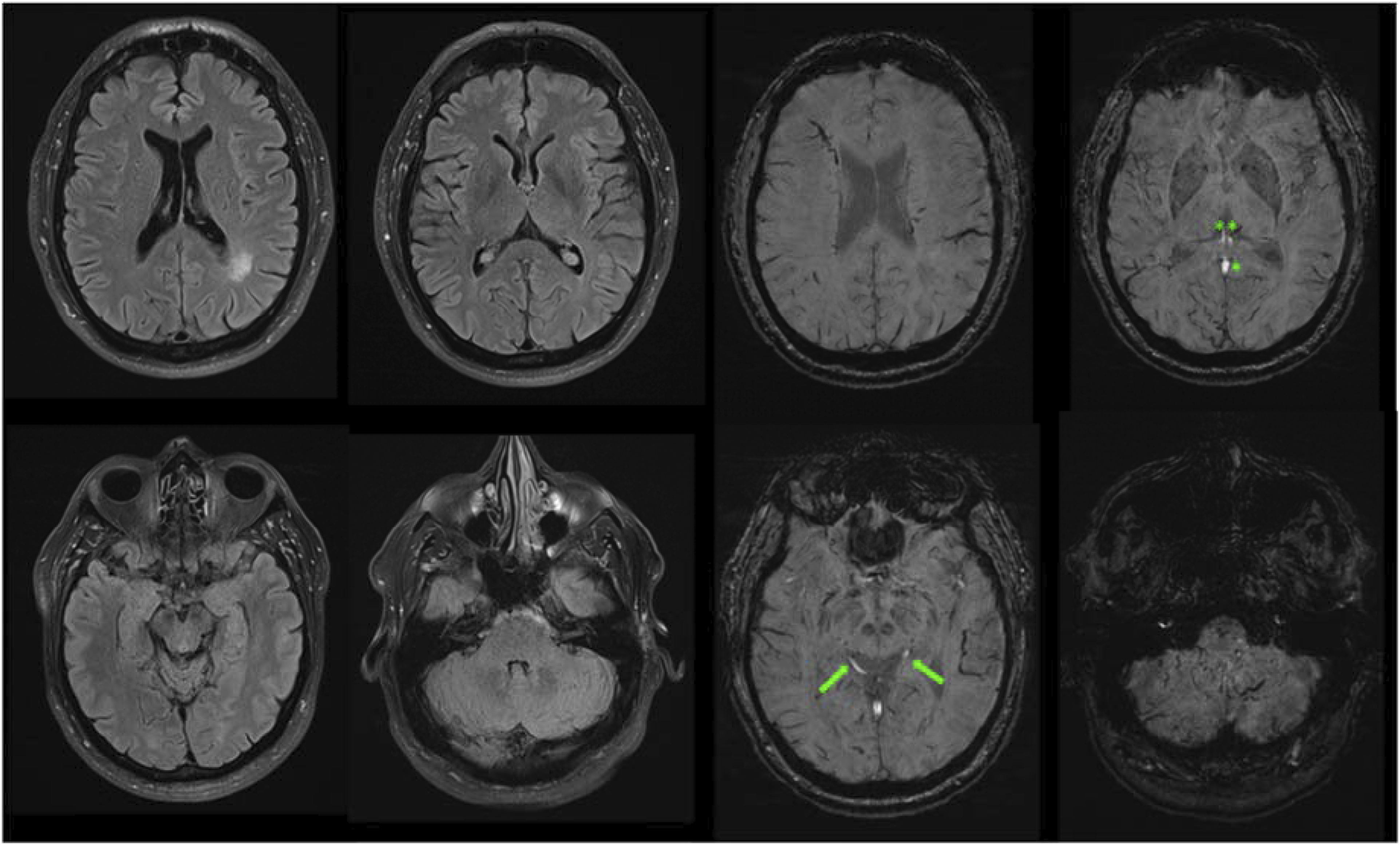
Treatment
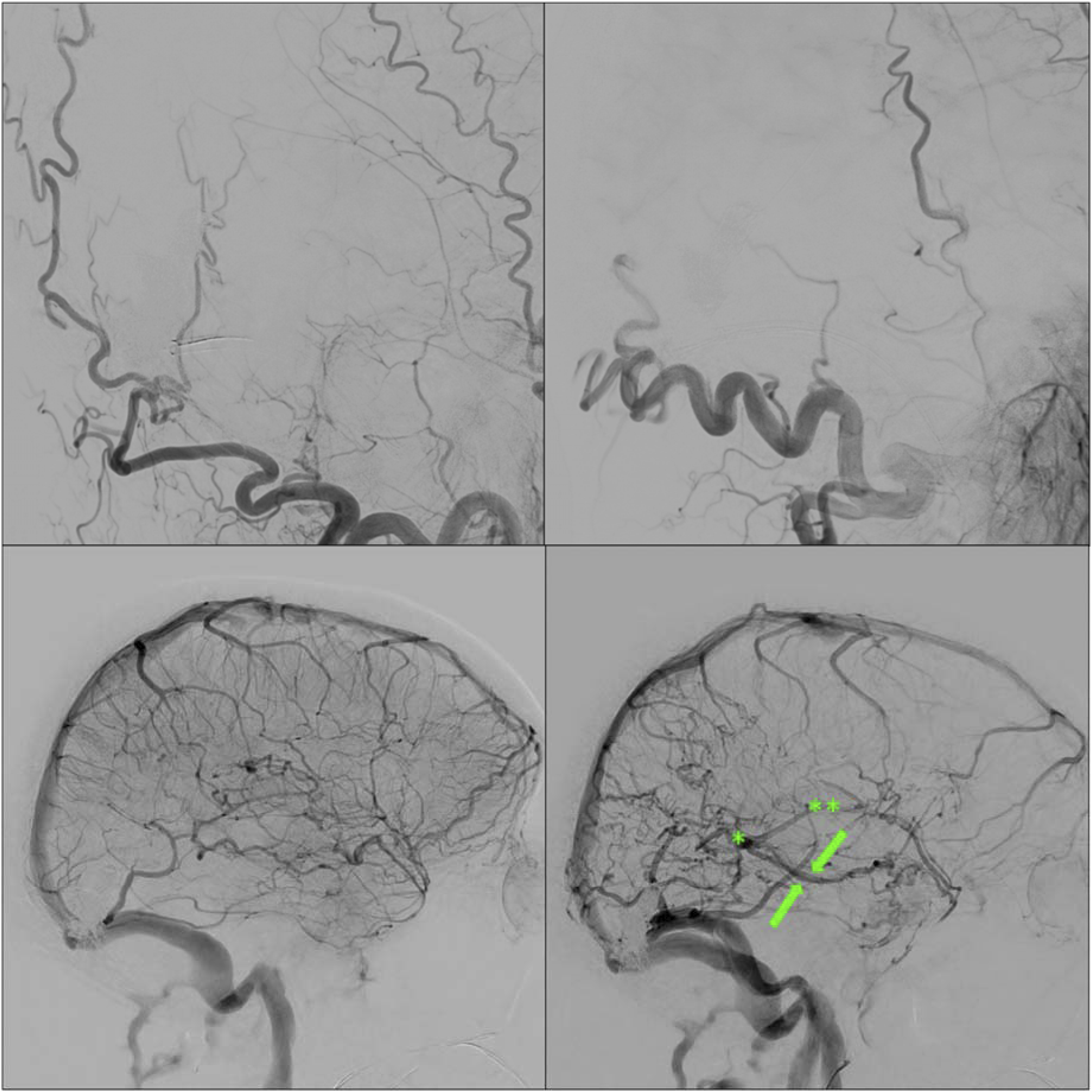
Transvenous embolization was the preferred endovascular treatment modality given: 1 the multihole fistulous segment of the torcular; 2 absence of cerebral and cerebellar venous drainage in the affected torcular sinus, straight sinus and deep venous system; 3 arterialization of the straight sinus and deep venous system on MRI and angiography, without vasogenic edema in the thalami, basal ganglia or other areas, and rerouting of drainage via the basal vein of Rosenthal. Transarterial embolization with a liquid embolic agent and balloon protection of the straight sinus was deemed infeasible, due to the high-grade stenosis of the right transverse sinus and the presence of innumerable arterial feeders. Therefore, transvenous coiling of the distal straight sinus and proximal right transverse sinus was performed, followed by Onyx embolization. Protection of the right vein of Labbe was achieved with an Eclipse 2L 6 × 20 mm balloon (Balt, France). Prior to transvenous coil embolization via an ipsilateral jugular approach, three Echelon 10 microcatheters (Medtronic, USA) were positioned in a staggered configuration within the straight sinus and torcular to allow controlled embolization, first with coils to reduce arterial flow, followed by Onyx to fill the remaining spaces within the coil matrix (Figure 4). Post embolization angiography demonstrated complete occlusion of the shunt, with normalization of deep venous drainage through the internal cerebral veins, vein of Galen and basal veins of Rosenthal (Figure 5). Apixaban was restarted immediately postoperatively, and delayed CT venogram confirmed continued patency of the deep venous system (Figure 6). There were no complications. Cognitive symptoms improved, and the pulsatile tinnitus resolved. A follow-up DSA performed 3 months after embolization showed persistent occlusion of the fistula. Technical steps of endovascular treatment. Working projections (1) demonstrate the 8 Fr guide catheter in the right distal transverse sinus via right internal jugular access for optimal support. Three Echelon 10 microcatheter were navigated via the inferior high grade stenotic lumen into the affected sinus segment (inferior red arrow). The microcatheters were positioned in staggered fashion (2) in the proximal straight sinus close to the vein of Galen, at the mid portion of the straight sinus and in the torcular (green arrows). A 6 × 20 mm Eclipse balloon was inflated protecting the outflow trajectory of the right vein of Labbe during Onyx embolization (3) resulting in closure of the interstices in between the coil mesh (4). Direct postoperative angiogram demonstrated complete closure of the DAVF on right ECA (left upper) and left ECA (right upper) injections. Venograms of the left ICA (left lower) and right ICA (right lower) demonstrate persistent non-opacification of the straight sinus. There is immediate normalization of venous drainage of the deep venous system, including the internal cerebral veins (asterisks), vein of Galen (asterisk) and basal veins of Rosenthal (arrows). Delayed CT venogram after 6 weeks demonstrates unremarkable aspect of the brain parenchyma and patency of the deep venous system.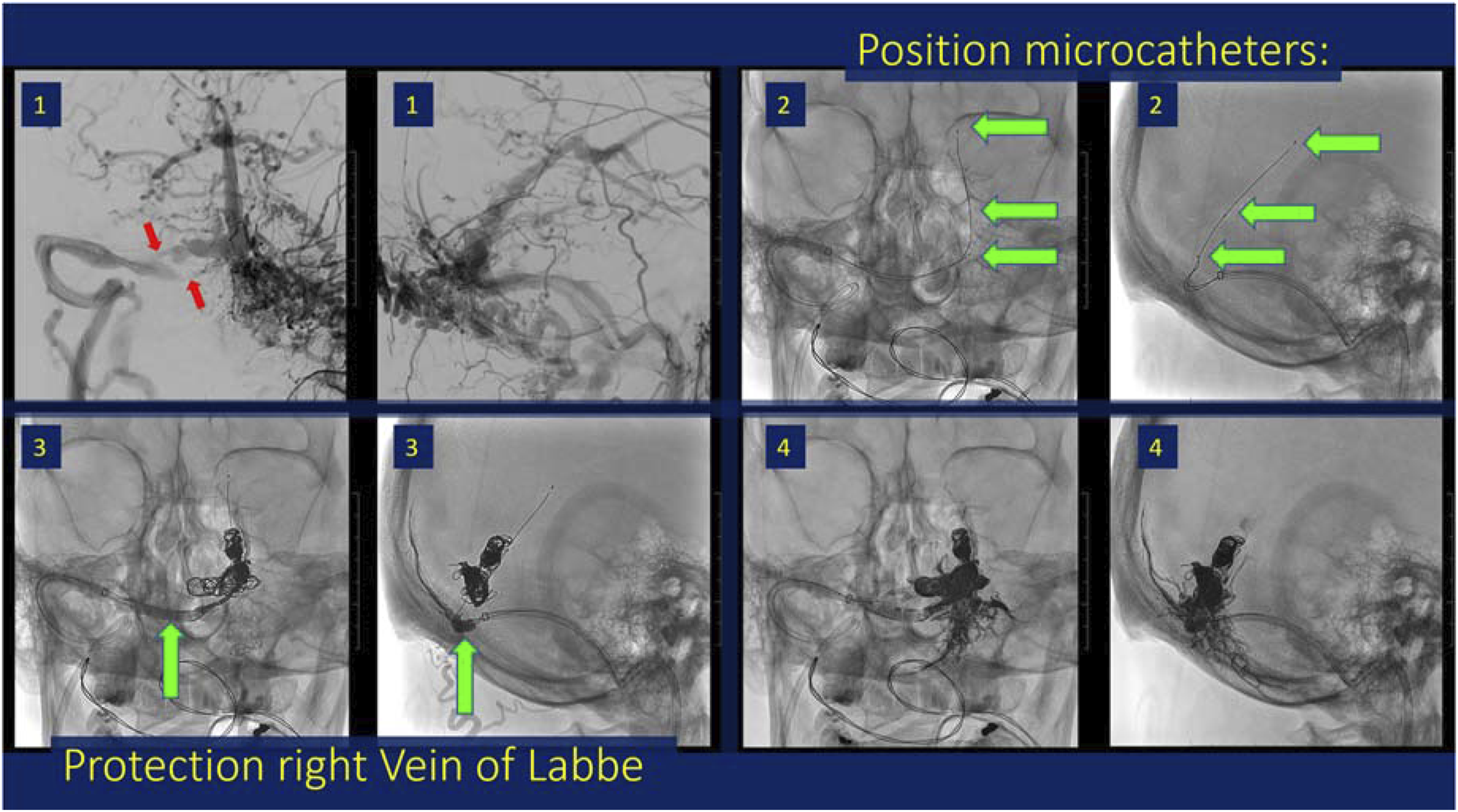
Discussion
We present a case of a high-grade torcular DAVF successfully treated via transvenous embolization and sacrifice of both the torcular and straight sinus. While the concept of this technique is straightforward in high grade DAVFs with pre-existing thrombosis of the transverse, sigmoid, or superior sagittal sinus and no adjacent normal venous drainage, 12 sacrificing the straight sinus should be reserved as a last resort option given the pivotal role of the straight sinus in normal drainage of the deep venous system.
In our case, straight sinus sacrifice was deemed safe based on: 1. Arterialization of the straight sinus and deep venous system with rerouting of the normal deep drainage via the basal vein of Rosenthal, 2. Absence of vasogenic edema in the thalami and basal ganglia, and 3. High-grade stenosis of the remaining transverse sinus, making balloon protection technically challenging. We utilized a staggered embolization strategy, placing three microcatheters in the proximal straight sinus (near the vein of Galen), mid-straight sinus and torcular. This approach allowed 1. Controlled reduction of arterial flow via progressive coiling 2. Complete shunt closure with Onyx embolization while 3. Maintenance of distal access to avoid undertreatment of remote compartments within the diseased sinus. Postoperative angiography confirmed normalization of sluggish venous outflow from the basal ganglia and thalami via the internal cerebral veins, vein of Galen and basal vein of Rosenthal.
Although the basal vein of Rosenthal (BvR) is typically considered part of the deep venous system, its anatomical and functional classification may vary. The BVR is located “superficially” over the undersurface of the brain in the quadrigeminal and ambient cisterns, and when completely developed merely functions as an anastomotic vein connecting the Vein of Galen posteriorly with the venous circle of Willis, deep middle cerebral vein and uncal vein anteriorly, 13 and drains multiple cortical “end” veins such as the inferior frontal vein, inferior striate vein, hippocampal and medial temporal veins, infratemporal vein, tegmental perforating vein and tectal perforating vein (listed from anterior to posterior). In case of incomplete embryological development, the BVR functions more as an anastomotic “end” vein, which has been associated with perimesencephalic subarachnoid hemorrhage. 14 Its recognition and evaluation are essential during preoperative planning in both neurosurgery 15 and interventional neuroradiology, 13 as emphasized by our case. Preoperative SWI-sequence demonstrated arterialized flow in the deep venous system and bilateral BvRs without vasogenic edema, supporting their capacity to tolerate retrograde high flow and validating our rationale for safely sacrificing the straight sinus. In cases with more favourable venous access to the straight sinus, an awake balloon test occlusion could be considered, as has been done prior to resecting meningiomas involving the straight sinus. 16
In conclusion, this technical report underscores key principles for treating high-grade DAVFs involving the straight sinus including: preoperative MR-imaging assessment of SWI signal arterialization, thorough analysis of both arterial and venous flow on cerebral angiography, evaluation of the basal vein of Rosenthal and a staggered microcatheter embolization technique enabling safe transvenous occlusion of a complex high grade DAVF.
Footnotes
Acknowledgments
DV and EH thank UMIT for their kind support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
