Abstract
Background and Purpose
Cone-beam CT (CBCT) and high-resolution multislice CT (HR-MSCT) are the mainstay postoperative imaging modalities following cochlear implant (CI) surgery, with CBCT often preferred due to lower susceptibility to metallic artifacts. However, CBCT is more prone to motion artifacts due to longer acquisition times. Recent advancements in accelerated flat panel CT (Acc-FPCT) available with latest generation angiography systems addressed traditional limitations of CBCT by significantly decreasing scan time. This study evaluates the diagnostic performance and radiation dose of Acc-FPCT compared to HR-MSCT in postoperative CI evaluation.
Methods
Five cadaveric whole-head specimens (ten temporal bones) were operated on by an ENT surgeon. Ten CIs were inserted via cochleostomy. Post-operatively, specimens were scanned using six Acc-FPCT protocols and HR-MSCT. Three neuroradiologists assessed the image quality of FPCT protocols in comparison to HR-MSCT using a 5-point Likert scale. Seven electrode characteristics including scalar position and discernibility of individual contacts were evaluated. Radiation dose parameters (CTDIvol and DLP) were compared among protocols.
Results
Two high-resolution Acc-FPCT (HR-FPCT) protocols were rated superior to HR-MSCT (p < .01). There were no significant differences between these two protocols (p = .25). The remaining Acc-FPCT protocols were rated inferior to HR-MSCT (p < .05). Inter-rater reliability was excellent (ICC (2,k) = 0.908; CI [0.85–0.94]). DLP was significantly lower in all Acc-FPCT protocols compared to HR-MSCT.
Conclusions
The results of this study underscore the utility of Acc-FPCT protocols as a feasible alternative to HR-MSCT in postoperative CI evaluation, allowing for better visualization of electrode array while significantly reducing scan duration and radiation exposure.
Keywords
Introduction
Cochlear implants (CIs) are the standard of care treatment for children and adults with severe to profound sensorineural hearing loss. An external speech and sound processor worn behind the patient’s ear captures and encodes sounds into electric signal. The encoded stimuli are then transmitted to the spiral ganglion cells through the CI electrode array, surgically inserted into the cochlea, bypassing the damaged parts of the auditory pathway. 1
Postoperative imaging of CI is important to confirm the intracochlear position of the CI electrode or to document any electrode misinsertion, intracochlear malposition, scalar dislocation, and electrode tip fold-over.2,3
In this setting, various imaging techniques have been employed, with multislice computed tomography (CT) gradually replacing cochlear view radiography as the mainstay modality from the early 2000s.4–6
Shortly after its clinical introduction in the late 1990s, cone-beam CT (CBCT) emerged a viable alternative to multislice CT (MSCT) in maxillofacial, dental, and temporal bone imaging applications. CBCT’s single-rotation volumetric acquisition not only offered a lower radiation dose compared to MSCT, but also proved advantageous in ENT imaging, providing superior spatial resolution, better contrast, and fewer metal-induced artifacts.7–9 A significant leap forward in CBCT technology came with the development of flat detectors, which further improved spatial resolution and contrast over the earlier intensifier-based systems. 10 Over the past two decades, there have been substantial advancements in digital flat panel technology, particularly in detector size, pixel resolution, frame rate, and bit-depth. 11
The introduction of C-arm-mounted flat panel CT (FPCT) scanners in modern interventional suites has expanded the use of FPCT in diagnostic imaging. In many settings, FPCT has become the preferred imaging modality for postoperative cochlear imaging, benefiting from CBCT’s advantages.12,13 However, one major limitation of this technique is the relatively long acquisition time, which increases its vulnerability to motion artifacts. Recent advancements, including the development of accelerated FPCT (Acc-FPCT) acquisition protocols in the latest generation C-arm angiography systems, have addressed this issue by reducing scan times by up to 65% through faster C-arm rotation. 13
This cadaveric study aims to evaluate the potential role of these new techniques in postoperative CI imaging and to assess associated changes in radiation exposure. Specifically, we compare the diagnostic accuracy of Acc-FPCT protocols with the latest generation high-resolution MSCT (HR-MSCT) in evaluation of CI electrode characteristics.
Methods
Cadaveric specimens
For the purposes of this study and a previous related project, we obtained five intact cadaveric whole-head specimens, pre-designated for the institutional body donation program of the Institute of Anatomy of the Otto-Von-Guericke-University, Magdeburg. All donors of the present study have individually given written informed consent, which explicitly included the post-mortem use of the body for scientific research. The requirement for ethics approval was deemed unnecessary according to relevant regulations for this research project by the institutional ethics committee.
All specimens had intact inner ear structures. Except for one specimen with partial mastoidectomy, all other heads had intact mastoids and ossicular chains. Due to data protection regulations in Germany, the Institute of Anatomy has no access to the patient data of the body donors. Medical histories of the cadavers were not disclosed.
CI Implantation
Mastoidectomy and posterior tympanotomy were performed on all cadaveric heads to access to the cochlea. A cochleostomy was created at the basal turn of the cochlea using a fine drill, providing access to the scala tympani. An electrode array of the type Contour Advance™ Electrode 14 (Cochlear, Sydney, Australia) with perimodiolar design, which features 22 electrode contacts arranged in non-uniform spacing from 0.4 to 0.8 mm, was then inserted through the cochleostomy, ensuring proper placement within the scala tympani and advancing it to the desired depth. The electrode array was positioned in the standard manner within the mastoid.
MSCT Imaging
MSCT imaging was performed using a latest generation CT scanner (Somatom X.Cite, Siemens Healthineers, Forchheim, Germany). 15 The specimens were positioned in the gantry isocenter in supine orientation.
A high-dose temporal bone protocol was applied with a higher tube current than that used in our clinical setting with the purpose of obtaining highest image quality possible. The protocol parameters were as follow: Voltage, 120 kV; current, 384 mA; acquisition field of view (FOV), 25 × 5 cm; single collimation width, 0.6 mm; pitch factor, 0.85; slice thickness, 0.5 mm; and scan duration, 0.9 second.
Standardized reconstructions using a bone kernel (kernel J70h) were acquired with 0.5 mm slice thickness with a predetermined window level (WL) at 560 HU and window width (WW) of 3620 HU. Changing the windowing setting was allowed during rating.
FPCT Imaging
A total of six FPCT protocols were acquired on the specimens using an ICONO biplane angiography system (Siemens Healthineers, Forchheim, Germany) 16 :
Two 8 s circular FPCT protocols (cFPCT) using 200° rotation angle and 496 frames with tube voltage of 70 kV and 109 kV, respectively. Two sinusoidal FPCT (sFPCT) protocols with a duration of 7 s and 9 s, respectively, using a sinusoidal curve rotation of the C-arm around the isocenter resulting in 10% higher angular coverage (220° rotation angle) and 50 additional frames in comparison to conventional cFPCT. 17
Technical parameters of the tested scanning protocols.
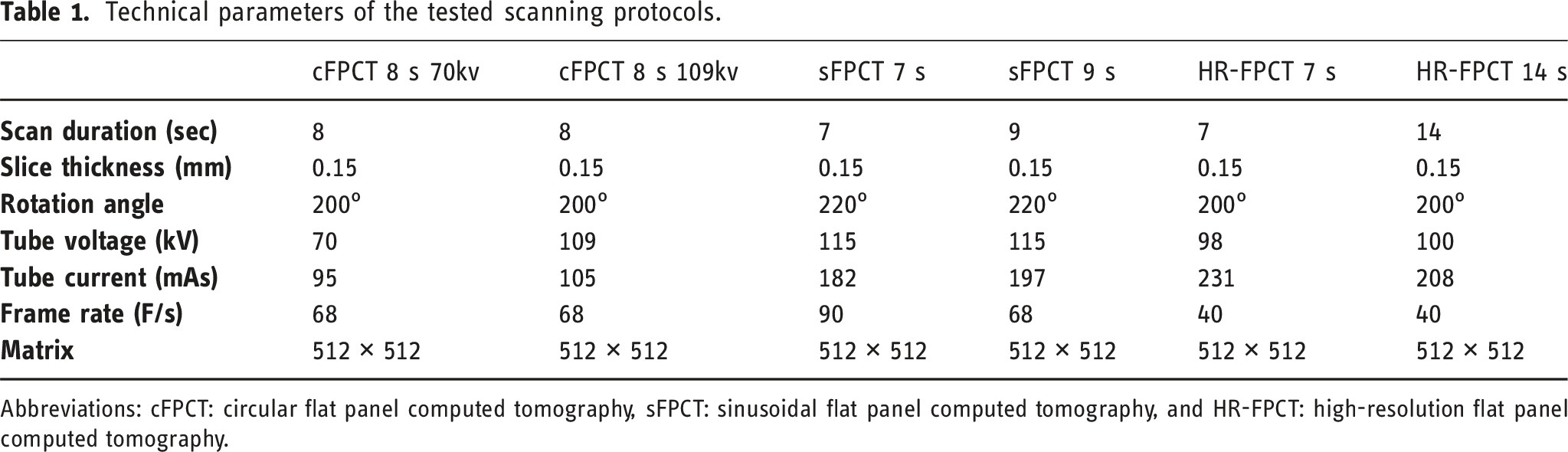
Abbreviations: cFPCT: circular flat panel computed tomography, sFPCT: sinusoidal flat panel computed tomography, and HR-FPCT: high-resolution flat panel computed tomography.
FPCT data were reconstructed using the Artis Icono post-processing platform (Siemens Healthineers, Forchheim, Germany). Standardized reconstructions of all protocols using kernel type “HU” and image characteristic “sharp” were acquired with 512 × 512 matrices and isotropic voxel size of 0.15 mm. Changing the windowing setting was allowed during rating.
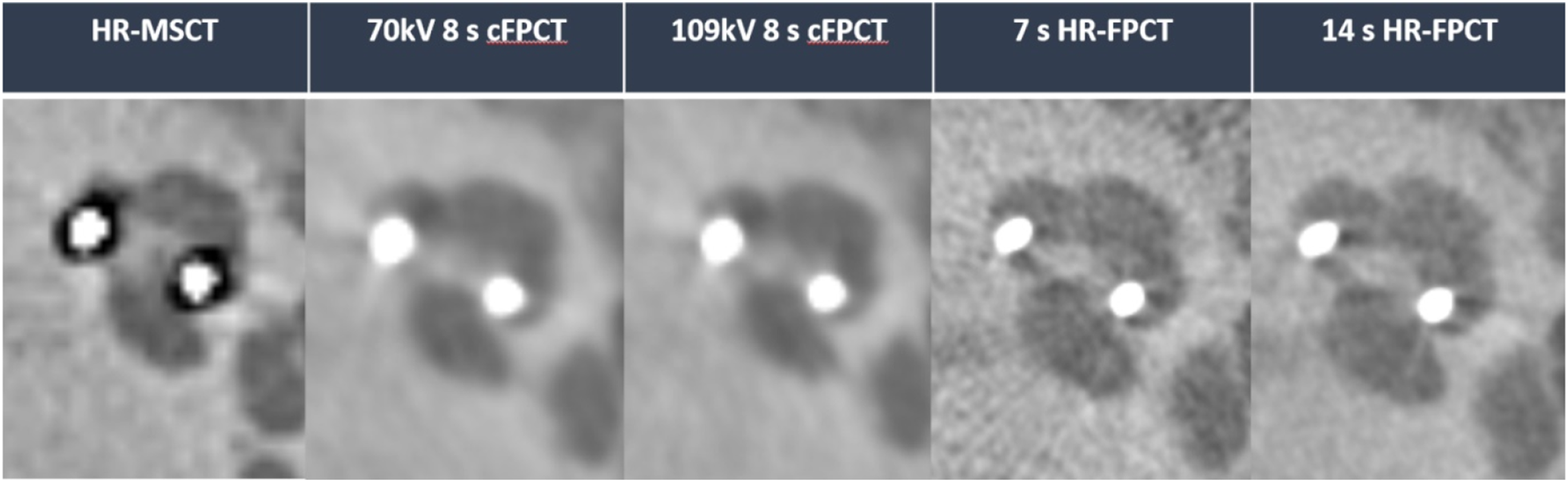
Illustrative examples of HR-MSCT and the different Acc-FPCT protocols are provided in Figures 1 and 2. Illustrative example of different protocols in the evaluation of electrode array, axial view (top row) and paracoronar view (lower row) of the cochlea. Only HR-FPCT protocols allowed for superior discernability of individual electrode contacts and inter-contact spaces. Illustrative example of different protocols in the evaluation of electrode array, axial view of the cochlea. Intrascalar position within scala tympani and contact between the modiolar hugging electrode and the inner wall of the cochlear canal were clearly demonstrated in all Acc-FPCT protocols. Notice the degraded image quality of HR-MSCT due to beam-hardening artifacts.
Imaging evaluation
Three neuroradiologists experienced in temporal bone imaging assessed the image quality of each FPCT protocol for seven electrode array characteristics including beam-hardening artifacts.
The image quality for individual characteristics in all FPCT protocols was compared to that of HR-MSCT using a 5-point Likert scale questionnaire (5 = superior with improved IQ, 4 = slightly superior IQ, 3 = equivalent to HR-MSCT, 2 = slightly inferior IQ, 1 = inferior with degraded IQ). Although the three raters were blinded to the protocol names, the protocols were in part inherently recognizable.
A commercially available PACS viewer (Infinitt PACS, Infinitt Healthcare, Seoul, South Korea) was used to analyze all datasets.
Dosimetry
Radiation dose parameters were recorded in dosimetry reports. For FPCT protocols, computed tomography dose index (CTDIvol) values in mGy and dose length products (DLP) values in mGy.cm were extracted from the individual reports. For MSCT, the DLP values were calculated by multiplying CTDIvol to scan length.
Statistical analysis
Statistical analysis was performed using Python (3.9.18).
The statistical differences of cumulative protocol scores and scores for individual electrode characteristics were compared between each FPCT protocol and MSCT using Wilkoxon signed-rank test.
Inter-rater reliability was assessed using intra-class correlation (ICC). A two-way random effect ICC form based on absolute agreement among multiple raters (ICC 2,k) was calculated. 18
Results
Image quality
The overall performance of the two HR-FPCT protocols was rated superior to HR-MSCT by all raters (p < .01) with cumulative rating scores of 27.8 and 27.5 for the 7s and 14s protocol, respectively. Regarding the ratings of individual electrode characteristics, the HR-FPCT consistently outperformed HR-MSCT. When compared to one another, the two HR-FPCT protocols, the image quality score was slightly higher in the 7s HR-FPCT than in the 14s HR-FPCT with overall mean values of 3.97 and 3.93, respectively. The difference was not statistically significant (p = .25). The rating of the beam-hardening artifacts were slightly superior in the 7s protocol compared to the 14 s protocol with mean scores of 3.43 and 3.23, respectively, however without reaching statistical significance (p = .23).
The four non-HR-FPCT protocols were rated inferior to HR-MSCT (p < .05) with cumulative scores ranging from 17.85 for the 70 kV 8 s cFPCT to 18.27 for the 109 kV 8 s cFPCT.
In terms of ratings of individual electrode characteristics, they consistently underperformed HR-MSCT except for beam-hardening artefacts where both cFPCT protocols and the 7 s sFPCT protocol yielded superior results (p < .05).
A graphic representation of rating results for all electrode characteristics is provided in Figure 3. Graphic representation of rating results. Abbreviations: cFPCT: circular flat panel computed tomography, sFPCT: sinusoidal flat panel computed tomography, and HR-FPCT: high-resolution flat panel computed tomography.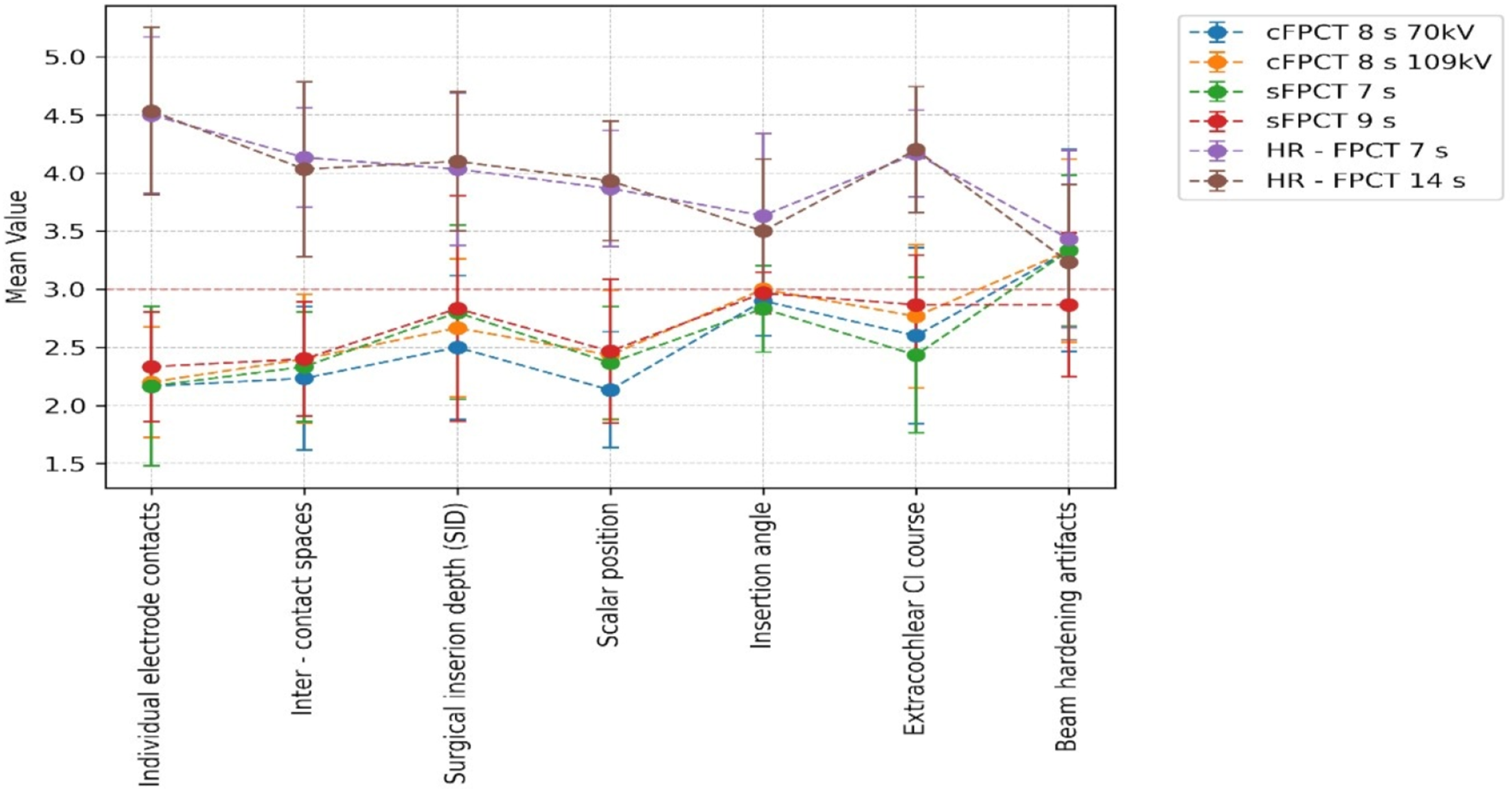
Overview of image quality results, including mean scores for individual electrode characteristics.
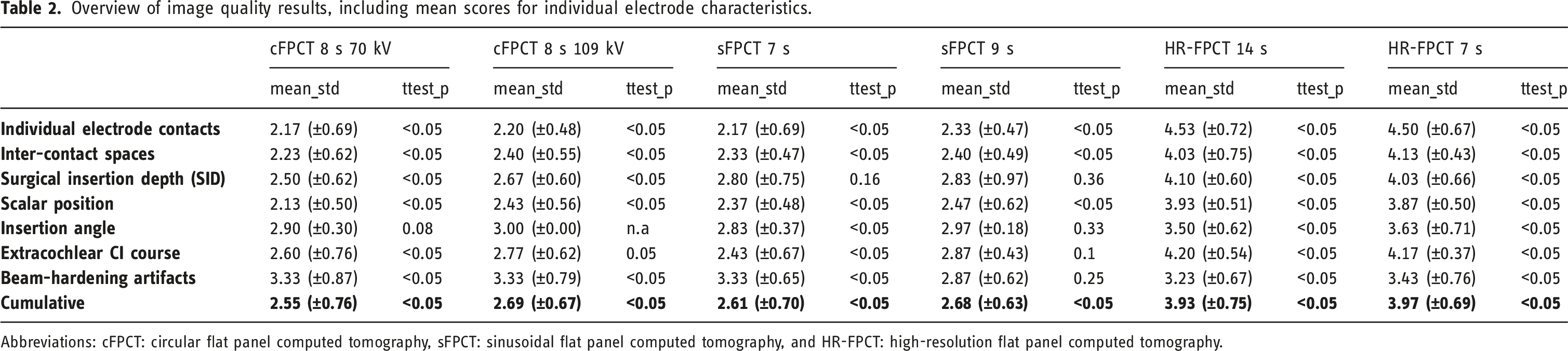
Abbreviations: cFPCT: circular flat panel computed tomography, sFPCT: sinusoidal flat panel computed tomography, and HR-FPCT: high-resolution flat panel computed tomography.
Inter-rater reliability
The inter-rater reliability was excellent ICC (2,k) = 0.908; confidence interval [0.85–0.94].
Dosimetry
The HR-MSCT yielded the highest dose parameters with a mean DLP of 311 mGy.cm (290.2–321.1). The 70 kV 8 s cFPCT had the lowest mean DLP value among the non-HR-FPCT protocols at 146 mGy.cm, whereas the 14 s HR-FPCT had the highest at 261 mGy.cm.
Due to their increased rotation angle, the sFPCT protocols with higher mean DLP values at 229 and 242 mGy.cm, respectively, exceed that of 7 s HR-FPCT protocol (212 mGy.cm).
Dose parameters of HR-MSCT and FPCT protocols.
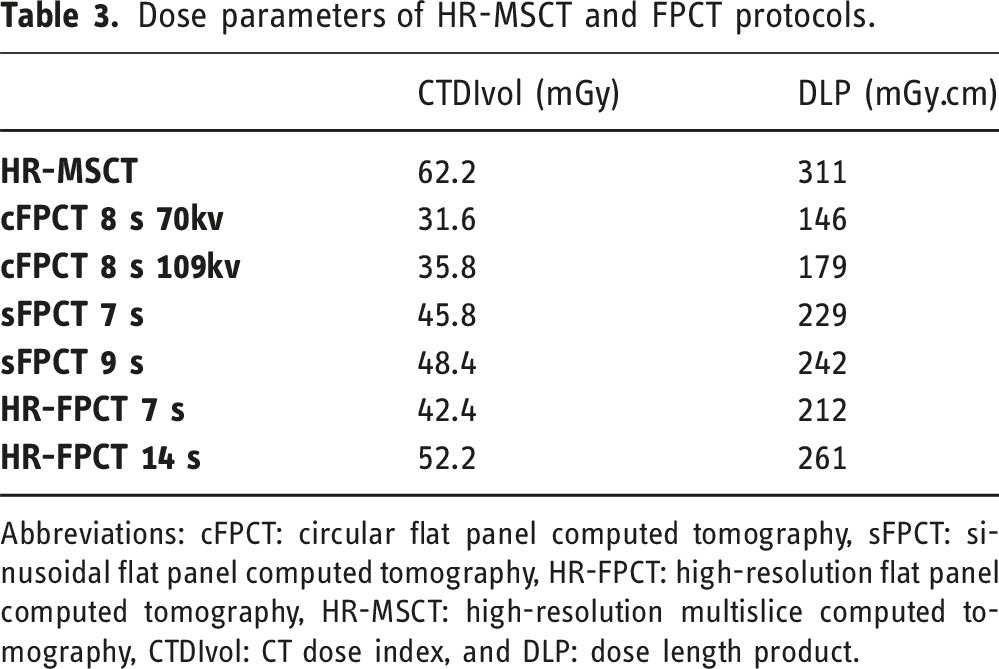
Abbreviations: cFPCT: circular flat panel computed tomography, sFPCT: sinusoidal flat panel computed tomography, HR-FPCT: high-resolution flat panel computed tomography, HR-MSCT: high-resolution multislice computed tomography, CTDIvol: CT dose index, and DLP: dose length product.
Discussions
Over the past two decades, numerous studies have shown CBCT is at least as effective as MSCT for evaluating the inner ear following cochlear implant surgery.4,19–22 While most of this research has focused on specialized digital volume tomography (DVT) CBCT systems, similar findings have been reported using multipurpose, commercially available C-arm-mounted flat panel CT (FPCT) systems.13,23
Use of accelerated FPCT protocols in postoperative temporal bone imaging
A cadaveric study by Eisenhut et al 13 first investigated the utility of using the accelerated flat panel FPCT protocols available with the latest generation of C-arm angiography systems in CI evaluation.
The group observed superior image quality in all Acc-FPCT protocols tested compared to a 128-row MSCT scanner (Somatom Definition AS+, Siemens Healthcare GmbH, Erlangen, Germany).
In our study, only HR-FPCT protocols were demonstrated to be superior to HR-MSCT. HR-MSCT outperformed all other FPCT protocols with superior image quality for all CI electrode characteristics. This could be attributed to the improved quality of MSCT images conducted on a latest generation scanner (Somatom X.cite, Siemens Healthcare GmbH, Erlangen, Germany) with optimized scan parameters, set to obtain the highest image quality possible.
The HR-FPCT protocols not only delivered superior image quality but also required only 68% and 84% of the dose in the 7 s and 14 s scans, respectively, compared to HR-MSCT.
Additionally, we found no particular benefit in using sinusoidal rotation protocols (sFPCT), which showed comparable results to other non-HR-FPCT protocols with significantly increased radiation dose. These protocols were reported to be useful in reducing beam-hardening artefacts in stroke imaging17,24,25; however, to our best knowledge, they haven’t been investigated in temporal bone imaging yet.
With regard to beam-hardening artifacts, all but one Acc-FPCT protocol demonstrated superior artifact profile compared to HR-MSCT in line with literature.4,9,26
Limitations
Beyond the inherent limitations of cadaveric bone imaging studies such as changes in tissue consistency and altered bone mineral density, the main limitation to the applicability of our findings in in-vivo setting is the lack of motion-related artefacts in cadaveric imaging. Although the acquisition times of Acc-FPCT protocols were shortened to 7 s, the vulnerability to motion-related artefacts is still higher than in MSCT, which involves significantly faster acquisition time of less than one second. Such artefacts could degrade the diagnostic performance of FPCT imaging favoring MSCT in clinical setting. Hence, further in-vivo studies are warranted to validate the utility of Acc-FPCT protocols in inner ear imaging.
Methodological limitations to our study include the reliance on CTDIvol and DLP values for dose comparisons as no phantom measurements were applied. Second, HR-MSCT acquisitions with higher tube currents resulted in DLP values exceeding those used in our clinical setting by a factor of 1.35. The dose values of Acc-FPCT protocols (except for the 14 s HR-FPCT) were still lower when compared to our “real-world” values. Third, there was a difference in size of FOV between HR-MSCT and FPCT. Limited FOV was applied in FPCT to image temporal bones separately, while a wider FOV encompassing both temporal bones was used in HR-MSCT, which partially compromises the comparability of dosimetry between the two modalities. The same methodological issue was encountered in similar studies comparing CBCT to MSCT in CI evaluation.7,23,27,28 However, these discrepancies reflect common imaging practices in clinical setting. Moreover, the sample size was relatively small (5 heads with 10 temporal bones), limiting the generalizability of our findings.
Conclusion
The outcomes of our cadaveric-based study underscore the utility of using Acc-FPCT available with latest generation of C-arm angiography systems in postoperative CI evaluation.
The HR-FPCT protocols demonstrated superior image quality and less susceptibility to metallic artifacts while maintaining lower radiation doses compared to HR-MSCT.
Their fast acquisition times could be effective in reducing motion artifacts usually encountered in CBCT imaging. Nevertheless, further in-vivo studies are warranted to validate these findings. As the latest generation of C-arm mounted angiography systems becomes increasingly accessible, there is potential for a significant shift in postoperative temporal bone imaging practices. This advancement could lead to a more integrated workflow, enhancing the versatility and efficiency of imaging options available to clinicians.
Footnotes
Acknowledgments
We acknowledge support by the Open Access Publication fund of medical faculty of the Otto-von-Guericke-University Magdeburg.
Authors' contributions
DB, MD, and ED: conceptualization and design. DB and MD: project administration. MD: cochlear implant insertions. ED, SM, and EK: imaging assessment, rating, and data analysis. ED: writing of the article. SK: image acquisition. SK: methodological and technical support and supervision of data analysis. AA: storage and use of cadaveric specimens. All authors reviewed and contributed to the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
