Abstract
Background and purpose
The anterior communicating artery (Acoma) aneurysms are difficult to understand because of their anatomical variations. This study reported a new classification of Acoma aneurysms on a basis of endovascular treatments.
Methods
This retrospective study enrolled consecutive 94 patients harbored 100 Acoma aneurysms treated with coil embolization. All Acoma aneurysms were classified into 5 orientations (straight, superior, inferior, anterior, and posterior) based on the axis connecting A1 origin and the base of the aneurysm. Aneurysm characteristics, endovascular treatments, angiographic, and clinical outcomes were evaluated using this classification.
Results
According to this classification, there were 64 (64%) straight, 16 (16%) inferior, 16 (16%) superior, 2 (2%) anterior, and 2 (2%) posterior projection aneurysms. There were significant differences between different groups in aneurysm height (p=0.009) and aneurysm neck size (p = 0.003), and in endovascular treatments (p=0.006). There was a significant difference in the proportion of rupture presentation among the three groups of straight, inferior, and superior (p = 0.019). There was no difference in the results of postoperative angiography (p = 0.090). An excellent functional outcome with an mRS ≤1 was achieved in 91 (96.8%) patients.
Conclusions
This classification of Acoma aneurysms based on the axis connecting A1 origin and the base of the aneurysm is associated with aneurysm height, aneurysm neck size, and in need for stent-assisted therapy. Endovascular embolization can provide the same effective treatment for aneurysms in all directions.
Background
The anterior communicating artery (Acoma), observed in 25% of cases, is the most common location of intracranial aneurysms. 1 And the Acoma aneurysm occurs at the bifurcation of the anterior cerebral artery and the Acoma.2,3 Therefore, the side of the Acoma aneurysm, for example, the base of the aneurysm located on the bifurcation of which side, is inclined to provide clinical reference for its treatment.
It is well known that the generation of Acoma aneurysm is related to the variation of Acoma complex, such as the presence of a dominant A1 on one side or the Acoma fenestration. 4 Acoma aneurysm with dominant A1 has showed less angiographic stability at follow-up than those with codominant A1, 1 but this association does not correlate with their presentations, geometry, and treatment modalities.
The location, size, orientation, and the neck of the Acoma aneurysms are more important factors determining the endovascular treatment approach in addition to judging the side of the Acoma aneurysm and dominant A1. Compared with other intracranial aneurysms, their morphology, anatomy, and surrounding vascular structure are the most difficult to understand because of anatomical variations and complex morphology of the Acoma.2,3 Endovascular treatment has become an alternative to surgical clipping and the primary method for Acoma aneurysms.5,6 Therefore, we provide a new classification of Acoma aneurysms based on their anatomical structures from the perspective of endovascular neurosurgery.
Materials and methods
Between January 2018 and January 2021, 94 consecutive patients (44 male patients and 50 female patients, and a mean age of 57 ± 11 years) with 100 coiled Acoma aneurysms at our hospital were retrospectively reviewed. The institutional research ethics board approved the study and granted a waiver of consent. All patients were diagnosed by CT scanning, cerebral angiography, three-dimensional rotational angiography, and three-dimensional reconstruction of angiography. In accordance with the grades of Hunt and Hess, the patients with unruptured aneurysms were of Grade 0 and the patients with ruptured aneurysms of grades 1–3 were of good preoperative grades. The patients with grade 4 or 5 SAH were of poor preoperative grades. All patients were treated endovascularly under general anesthesia. Clinical outcome was assessed using the modified Rankin Scale (mRS) and angiographic follow-up at 6 to 12 months.
Determination of aneurysm projection
All Acoma aneurysms were reclassified into straight, superior, anterior, inferior, and posterior projections based on the line connecting A1 origin and the base of the aneurysm as the axis (Figure 1). The projection of the aneurysm is determined by three-dimensional rotational angiography of the internal carotid artery on the side of the Acoma aneurysm and its reconstruction. The line connecting A1 origin and the base of the Acoma aneurysm on the side of the dominant A1 segment of the anterior cerebral artery was taken as the axis to judge the direction of the Acoma aneurysm (Figure 1). The line connecting the base and the dome of the Acoma aneurysm (the most distal dome in bilobed or multilobed Acoma aneurysm) is the projection, which is the straight projection (consistent with the axis), the anterior projection (in the front of the axis), the inferior projection (below the axis), the superior projection (above the axis), and the posterior projection (behind the axis). Giving five examples of different projecting Acoma aneurysms relative to the axis connecting A1 origin and the base of the aneurysm. The upper row is the preoperative angiograms, and the lower row is the control angiograms after endovascular treatment.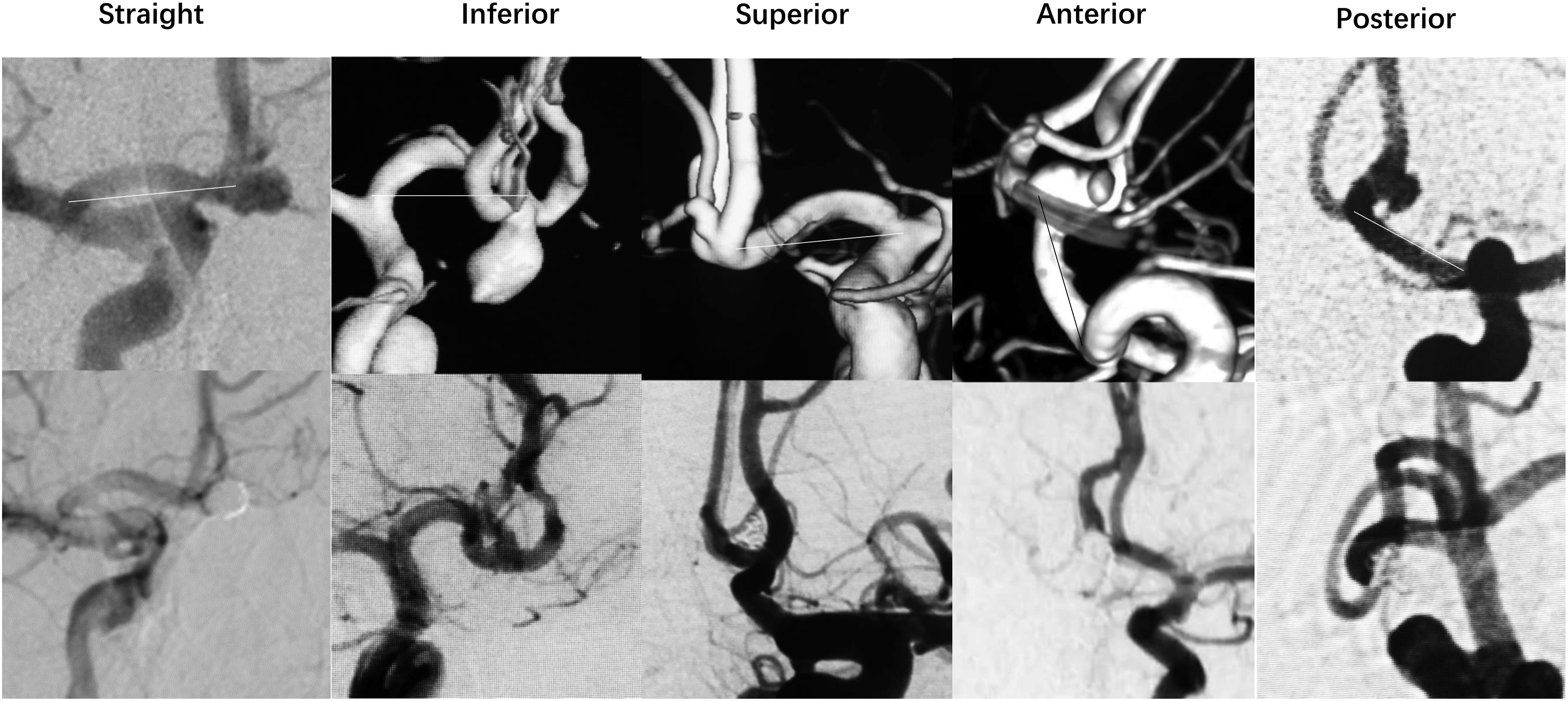
Determination of aneurysm size
The longest diameter of aneurysm is the measurement standard of height,<2 mm is micro, 2–5 mm is small, 6–9 mm is medium, and ≥10 mm is large.
Techniques of microcatheter and coil embolization
According to the projections, sizes, and aneurysm necks, the curvature of the microcatheter and sizes of coils were selected. For the straight and superior types, the microcatheter of straight head was chosen. The anterior and posterior types were of the most difficult for coil embolization because of their non-preferable working angle, and the tip of the microcatheter should be molded into a very small curve. The inferior type was the easiest for coil embolization, and the tip of the microcatheter was bent to 45°. For microaneurysms, the 1.6F Headway Duo microcatheter (MicroVention, Aliso Viejo, California) and Nano coils (Stryker Neurovascular, Fremont, CA) were routinely used.
Techniques of stents
For wide-necked aneurysms, intracranial stents were routinely used. According to market availability, Neuroform Atlas (Stryker Neurovascular, Fremont, CA), Leo Baby (Balt Extrusion; Montmorency, France), or LVIS Jr (MicroVention, Aliso Viejo, California) was preferably to choose. Dual antiplatelet therapy was necessary as previously reported.5,6 If intra-stent thrombosis was found during operation, tirofiban hydrochloride was given for treatment.5,6
Statistical analysis
The Fisher's exact test was used for comparison of proportions, and ANOVA test was used for comparison of continuous variables. A p-value of <0.05 was considered statistically significant. Data was analyzed with SPSS 26.0 (IBM SPSS Statistics).
Results
Summary of characteristics of 100 Acoma aneurysms.
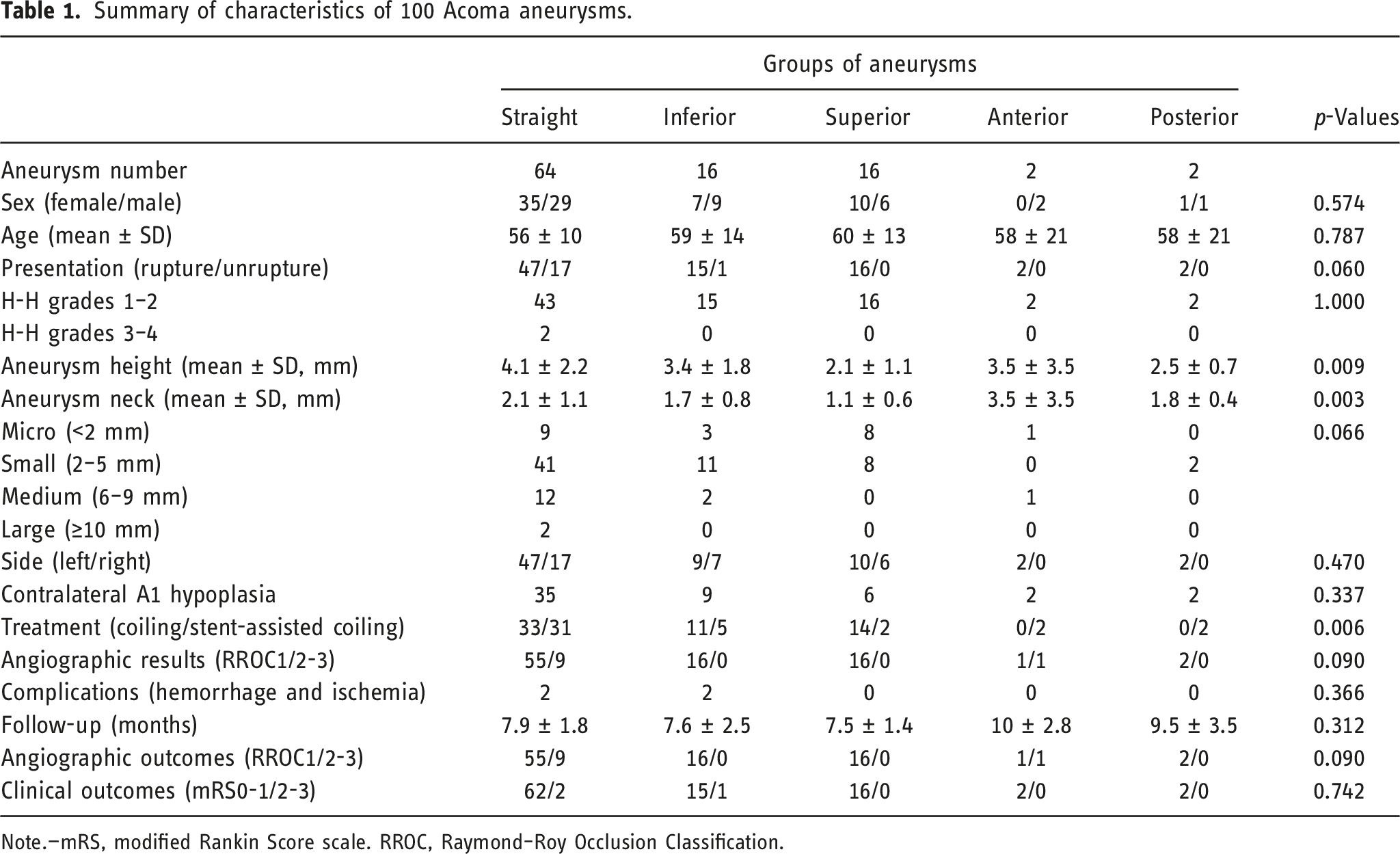
Note.—mRS, modified Rankin Score scale. RROC, Raymond–Roy Occlusion Classification.
Statistical results
There were significant differences in aneurysm heights (p = 0.009), neck sizes (p = 0.003), and stent-assisted therapy (p = 0.006) between different groups (Table 1). The mean aneurysm heights from the top to bottom were straight (4.1 ± 2.2 mm), anterior (3.5 ± 3.5 mm), inferior (3.4 ± 1.8 mm), posterior (2.5 ± 0.7 mm), and superior (2.1 ± 1.1 mm) (Figure 2). The anterior and posterior aneurysms were tend to be wide-necked aneurysms. The possibility of stent-assisted therapy was lower for superior aneurysms but higher for other aneurysms. Schematic diagram of the characteristics of Acoma aneurysms in different projections.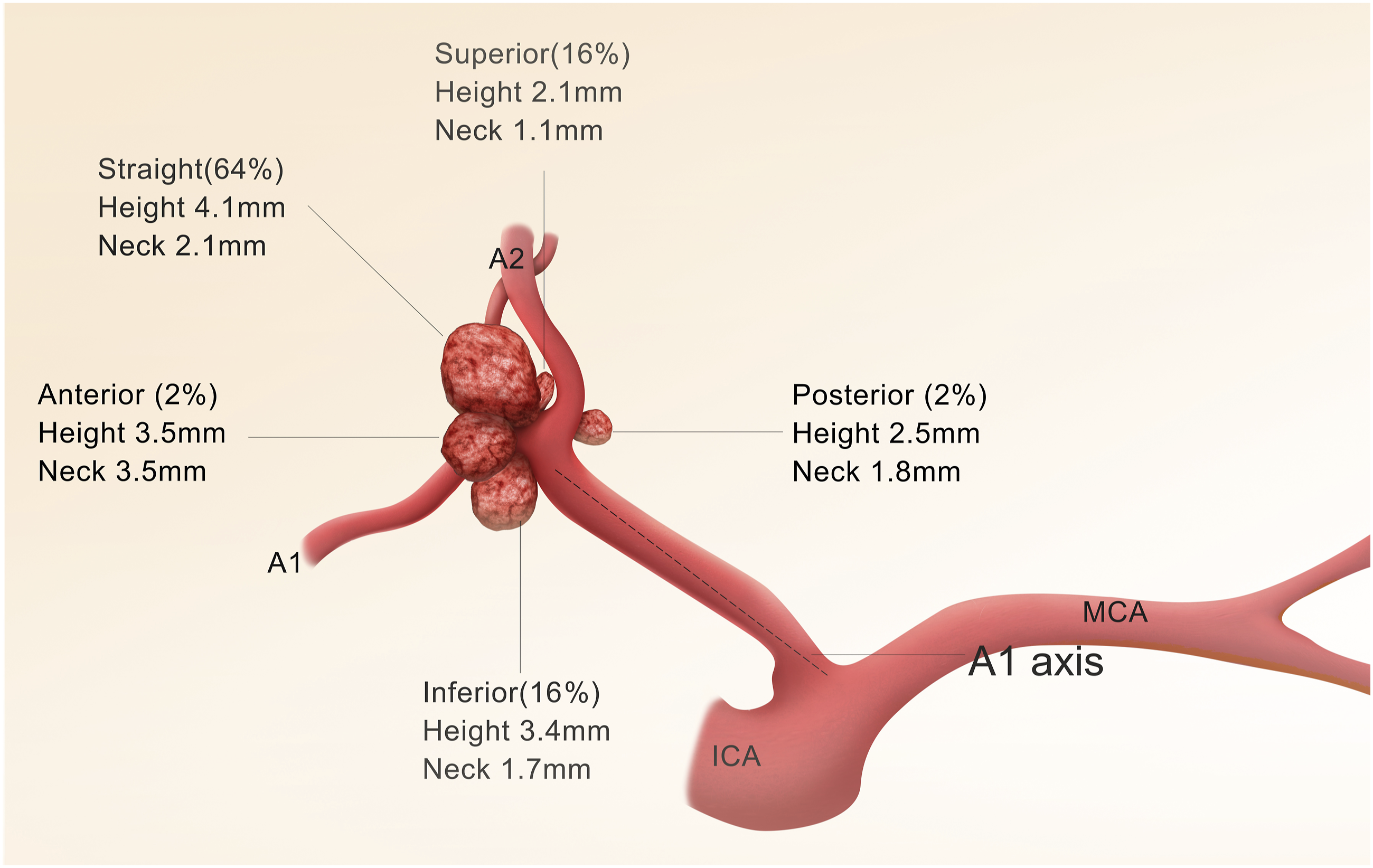
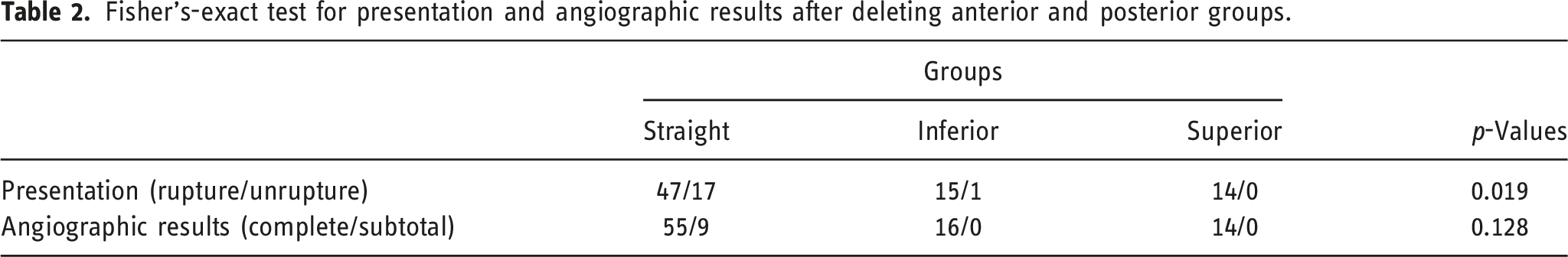
Fisher's-exact test for presentation and angiographic results after deleting anterior and posterior groups.
Discussion
Acoma aneurysms are the most common intracranial aneurysms and have a higher risk of rupture, which can lead to morbidity and mortality. They represent challenges to clinical understanding and treatment because of their anatomical variations of the Acoma complex. The classifications of Acoma aneurysms in previous studies were based on surgical findings, such as the relationship between the aneurysm projection and the axis defined by the pericallosal artery, 7 the relationship between the aneurysm projection and A2 segment plane, 8 the relationship between the aneurysm projection and A1 segment morphology, 9 and the relationship between the aneurysm and the parent artery.2,3
The previous classifications of Acoma aneurysms based on intraoperative observations are limited by the differences of their surgical approaches, intraoperative anatomical field, and lack of the standard position leading to the complex and divergent classifications. The visualization of vascular elements is also different in various autopsy studies. Therefore, these classifications are not enough to demonstrate the overall characteristics of Acoma aneurysms and are less to be used in clinical practice.
There are more and more data supporting endovascular embolization of Acoma aneurysms. 10 In a Propensity-Matched Study of 1332 patients in the United States, 11 patients with endovascular coiling of ruptured Acoma aneurysms had significantly less hydrocephalus within 1 year compared with the clipping cohort.
Until now, there is only one classification of Acoma aneurysms based on the flow diversion treatment, and its use needs to be further evaluated. 12 This classification comparing the diameters of both A1 segments is proposed: H1=same diameters; H2=<50% difference in diameters; H3=≥50% difference; and Y=no A1 segment. The most suitable anatomical configuration for flow diversion treatment seems to be the H1 configuration, which is suitable for flow diversion placement from an A1 segment to the ipsilateral A2. There is a tendency that the H3 configuration is not a good indication for flow diversion treatment.
Although there are successful reports on the flow diversion treatment of Acoma aneurysms, 5 its use in Acoma aneurysms is still controversial because the diameter of A1 is 2.4 mm to 2.6 mm and the length of A1 is 12.7 mm in adults. 12 Intrasaccular embolization is still the main treatment for Acoma aneurysms. Stent-assisted coiling provides a long-term efficacy of Acoma aneurysms with a low incidence of aneurysm regrowth after treatment. 13 Double-stent treatment with a Y-configured or X-configured stent-assisted coiling also provides an option for wide-necked and complex Acoma aneurysms.14,15 Many new techniques are still targeted to intrasaccular embolization for Acoma aneurysms, such as the intrasaccular flow-diverting device of Medina,16,17 the Woven EndoBridge (WEB) device,18,19 and PulseRider device. 20
The classification in this study was based on intrasaccular coil embolization and their different projections relative to the axis of A1 origin and the base of the Acoma aneurysm. This classification can reflect the anatomical and hemodynamic characteristics of Acoma aneurysms indirectly and can provide a guidance for endovascular techniques. It is easy to distinguish these types at the time of diagnosis and to take different endovascular techniques for treatment. Based on this concept, it is also possible to make a prediction at the time of computed tomography angiography (CTA) because the characteristics of Acoma aneurysms in different projections had been depicted (Figure 2). The classification may also reflect the factors initiating aneurysm formation and influencing its growth, hemodynamic factors, as well as the natural history of Acoma aneurysms. The association between A1 symmetry/asymmetry was not considered in this study because one previous study had confirmed that dominant A1 configuration is only related to the genesis of Acoma aneurysms but not to their clinical characteristics. 4
In one surgical study, the authors found that superiorly projecting Acoma aneurysms had a higher risk of intraoperative rupture (37.5%) than other Acoma aneurysms. 6 According to our classification, superiorly projecting Acoma aneurysms are usually ruptured microaneurysms, which can explain the surgical challenges of these Acoma aneurysms. 21 The Acoma location represents the most common one for ruptured microaneurysms, in which it is difficult to obtain a “stable” microcatheter position to deliver coils safely. 22 Appropriate selection of coils and use of sophisticated techniques can provide a good outcome for patients with very small aneurysms. 23 In our clinical practice, the 1.6F microcatheter and Nano coils are routinely used for these microaneurysms.
In another study, the authors proposed that microsurgical clipping should be the preferred option for anteriorly projecting Acoma aneurysms and that endovascular embolization should be selected for those lesions that are posteriorly projecting. 7 The authors' projection judgment is based on the axis defined by the pericallosal artery. According to our classification, both of them belong to our straight projection, which is the most common type of Acoma aneurysms and is easier to be treated with endovascular coil embolization. The authors may also have missed other rare types of Acoma aneurysms, such as the aneurysms that project laterally to the pericallosal artery.
The unruptured Acoma aneurysms should be treated aggressively though they are usually small in size. According to the ISUIA (International Study of Unruptured Intracranial Aneurysms), anterior circulation aneurysms of <7 mm in diameter have a minimal risk of rupture. 24 But, Acoma aneurysms show a risk of rupture twice as high as that of other intracranial aneurysms. 25 This has also been reported by Bijlenga et al. They found that anterior circulation aneurysms are not a homogeneous group. Acoma aneurysms and distal anterior cerebral artery aneurysms of 4 and 7 mm sizes presented a similar rupture risk to posterior circulation aneurysms. 26 They concluded that intervention should be recommended for this high-risk lesion group.
Our classification also indirectly reflects that blood flow may create different forces that impinge directly against the aneurysmal wall in different projections, eventually causing the different sizes, morphologies, and ruptures in Acoma aneurysms. Hemodynamic study in intracranial aneurysms with the use of Doppler guidewires found that flow velocities were higher at the neck than within the aneurysmal sac. 27 However, the pulsatility was clearly higher at the dome than at the neck or in the parent artery. The pulsatility is used to describe the increased flow resistance inside the aneurysm sac. 27 Castro et al found that small impact zones, high velocity of flow entering the aneurysm, and elevated maximum intra-aneurysmal wall shear stress were associated with a previous clinical history of rupture of Acoma aneurysms. 28
The limitations of this study are the lack of the measurement of the angles between A1 and the aneurysm, the small number of the posterior and anterior projecting aneurysms, and the missing of untreated Acoma aneurysms because it is a retrospective study. This study may have a selective bias because of lacking high SAH grade (Hunt and Hess) patients. Endovascular treatment is carefully considered in patients with high SAH grade with ruptured aneurysms because of their poor outcome after endovascular treatment. 29 Our classification is used for anatomical evaluation of Acoma aneurysms and guidance for endovascular treatment. Further prospective research is needed to evaluate whether this classification can predict the prognosis of patients.
Conclusion
The classification of Acoma aneurysms based on the axis connecting A1 origin and the base of the aneurysm is practical for understanding their characteristics, including the shape, location, orientation, size, probability of occurrence, and endovascular treatment. It is possible to establish an overall concept of Acoma aneurysms based on this classification. Endovascular embolization can provide effective treatment for aneurysms in all directions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Tsinghua Precision Medicine Foundation (20219990008), Tsinghua University, Beijing, China and Beijing Municipal Administration of Hospitals Incubating Program (PX2020039), Beijing, China.
