Abstract
Cauda Equina Neuroendocrine Tumors (CE-NET), previously referred to as paragangliomas are a rare subset of spinal tumors, with limited data on imaging. Herein, we present a retrospective review of clinical and imaging findings of CE-NETs in ten patients who were evaluated at our institution over the past two decades. All patients had well-defined intradural lesions in the lumbar spine which demonstrated slow growth. A review of imaging findings revealed the presence of an eccentric vascular pedicle along the dorsal aspect of the tumor in 8 of the 10 patients (eccentric vessel sign), a distinctive finding that has not previously been reported with this tumor and may help improve the accuracy of imaging-based diagnosis. In all cases, a gross-total resection was performed, with resolution of symptoms in most of the cases.
Keywords
Introduction
CE-NETs, previously referred to as paragangliomas, were recently classified under “paraganglioma-like neuroendocrine neoplasms” under the 2022 World Health Organization tumor classification. 1 This change was prompted given the differences in underlying molecular profile, pathogenesis, and biologic behavior of these tumors when compared with phaeochromocytomas and paragangliomas (PPGLs) outside the CNS. 2
CE-NETs are thought to arise from the specialized neural crest cells in the cauda equina/filum terminale region and are histogenetically and molecularly different from extra-CNS PPGLs. CE-NETs overexpress transcription factor HOXB13, unlike hereditary PPGLs which often show mutations in Succinate dehydrogenase (SDH) subunit genes.1,2 The former is also expressed in myxopapillary ependymomas, and developmentally expressed in caudal spinal cord and urogenital sinus and may provide clues to cell of origin. CE-NETs are also invariably sporadic, unlike extra-CNS PPGLs which are often inherited, especially in children. 1 The chromosomal copy number profiles and DNA methylation profiles of CE-NETs are also different from extra-spinal paragangliomas. 3 Finally, in terms of biologic behavior, only 4% of CE-NETs recur following gross-total resection, while extra-CNS metastasis has only been reported once, unlike 10–20% of extra-CNS paragangliomas which have metastatic potential.1,4,5
CE-NETs are rare, accounting for <3% of spinal intradural tumors, with an estimated annual incidence rate of 3 cases per million.6–8 Since their initial description in 1970, nearly 300 cases have been reported, mostly as sporadic case reports and small series. 9 CE-NETs generally occur in adults, peaking in incidence between the fourth and sixth decades (age range: 9–75 years; mean 46–47 years) and have a slight male predominance (M:F::1.5:1).9,10
Most CE-NETs are non-functional, often presenting with low back pain and sciatica, followed by numbness, paraparesis, or sphincter dysfunction symptoms. Presentation as a full-blown cauda equina syndrome or subarachnoid hemorrhage is uncommon. Endocrinologically active CE-NETs are rare, but may manifest with episodic or sustained hypertension, headache, or palpitations.1,11,12
Due to the rarity of their occurrence, the radiological features of CE-NETs are not well delineated. Previously described imaging features include the “cap sign” (T2 hypo-intensity along tumor margin reflecting hemosiderin deposition), “salt-and-pepper” appearance (similar to head and neck paragangliomas) and “tadpole” sign (tumor and dilated vessel representing the head and tail of the tadpole, respectively).13,14 However, accurate pre-operative distinction remains challenging. Currently, the diagnosis of CE-NET relies on histopathological findings, as specific clinical and radiological features are lacking. 15 Herein, we present our institutional experience on the imaging of CE-NETs on multi-modality cross-sectional imaging.
Methodology
The study was approved by the institutional IRB and informed patient consent was waived, given the retrospective nature. The institutional medical and pathology database archives were searched for patients presenting between January 2000, and April 2023 using search terms paraganglioma [OR] neuroendocrine [AND] spine [OR] central nervous system [OR] cauda equina. Inclusion criteria included age >18 years, presence of a CE-NET/spinal paraganglioma with proven tissue diagnosis and availability of pre-treatment imaging. Exclusion criteria were metastatic paragangliomas, extra-spinal paragangliomas, and significant motion artifacts on the index scan. A total of 10 unique cases of CE-NET in 10 patients were identified (out of the 128 screened cases) and subsequently evaluated. An additional case of a primary vertebral paraganglioma in the thoracic spine in a young patient was also identified, but subsequently excluded from current study given the recent change in WHO tumor classification which considers CE-NETs as a separate category. Figure 1 illustrates selection of eligible cases in the current study. Flowchart depicting patient search and selection of current cohort.
For the eligible cases, clinical, demographic, imaging variables, as well as details regarding the surgical procedure and post-operative follow up were collected. The patient's imaging studies were reviewed in consensus by two staff neuroradiologists, both of whom were fellowship trained, hold certificate of added qualification in neuroradiology and have a combined subspecialty experience of greater than fifteen years. Images were reviewed to assess the size, shape, location, margins, MRI signal characteristics and enhancement patterns, presence or absence of any underlying cysts, calcification or hemorrhage, presence or absence of bony erosion/remodeling, as well as existence of any of the previously described imaging signs (cap sign, salt and pepper sign, and tadpole sign). In cases where serial pre-operative imaging was available (n = 4), lesion volumes were also calculated using standard 3D-region of interest tool on the picture archiving and communication system to ascertain tumor growth over time (Visage imaging, version 7.1.18). The reviewers also focused on identifying any additional imaging findings which may help improve accurate pre-operative identification of CE-NETs.
Results
Demographic, clinical, and post-operative outcome details of the patients with CE-NETs in the current cohort.
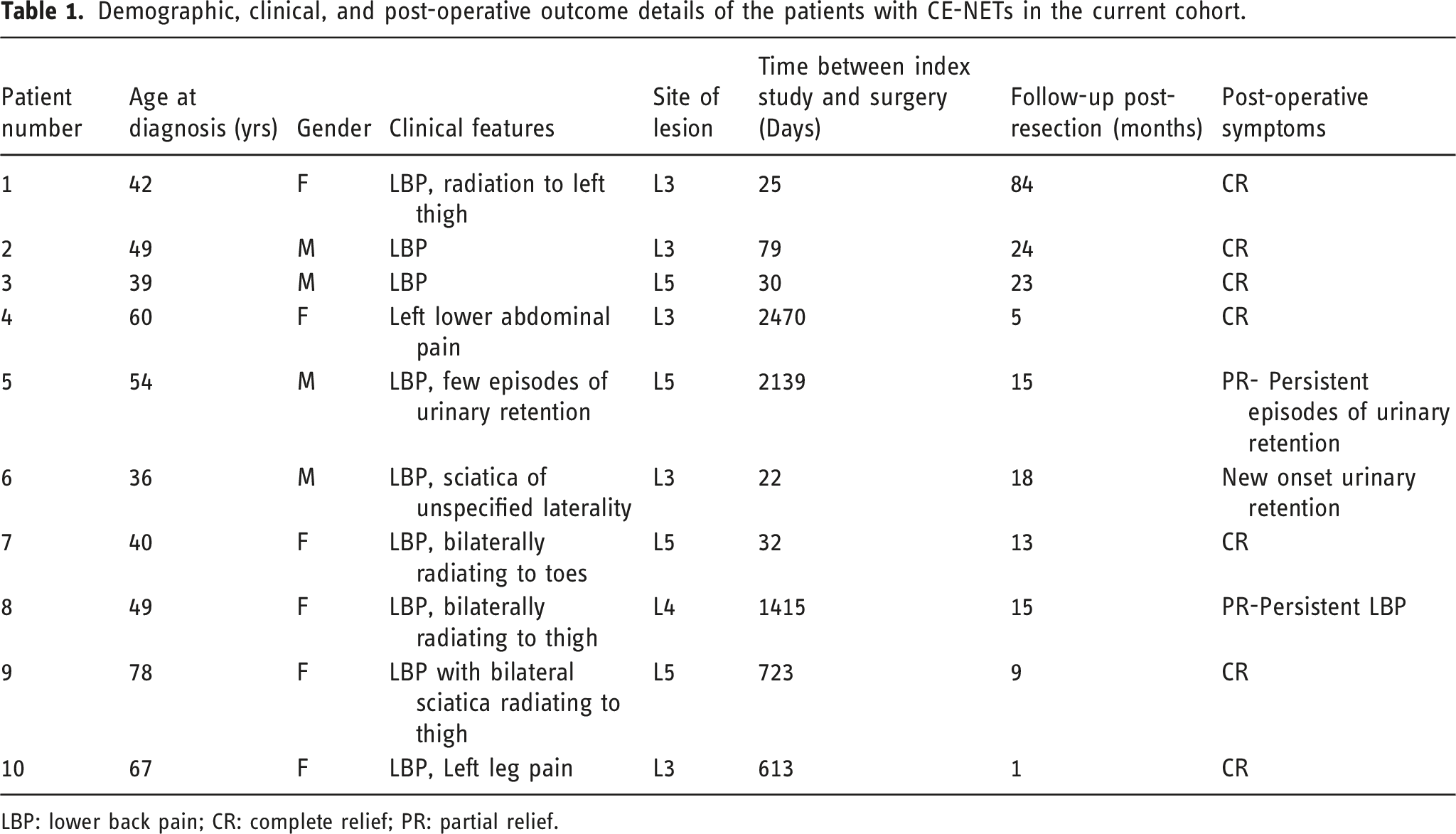
LBP: lower back pain; CR: complete relief; PR: partial relief.
Imaging performed comprised MR imaging in 8 and CT imaging in 2 patients (Figures 1 and 2). One patient (P-10) also underwent a pre-operative CT myelogram (Figure 2(c)). A total of 8/10 patients also underwent an intraoperative ultrasound based on the operative note. However, intra-operative ultrasound images were only available in the medical archive in one patient (Figure 2(d)). T2W axial MR images of the six patients (a-c, e-g) depicting the intradural cauda equina neuroendocrine tumors with an eccentric flow void (white arrows) located dorsally within the lesion. Axial (d) and coronal MPR (h) CT images in 2 different patients who only had CT imaging show the hyperattenuating lesion with a linear eccentric vessel cranially (black arrows).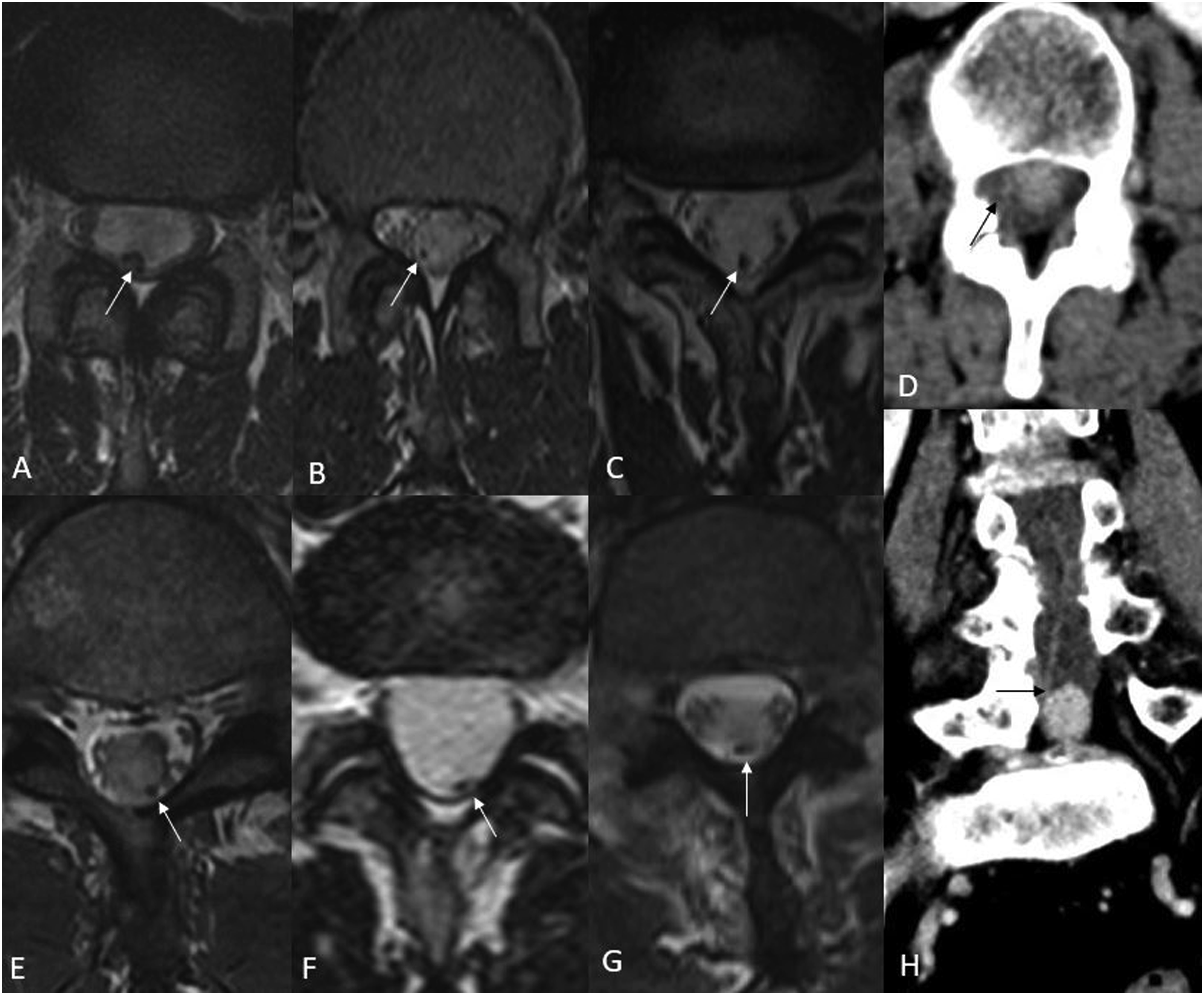
MR Imaging characteristics of the CE-NET patients.
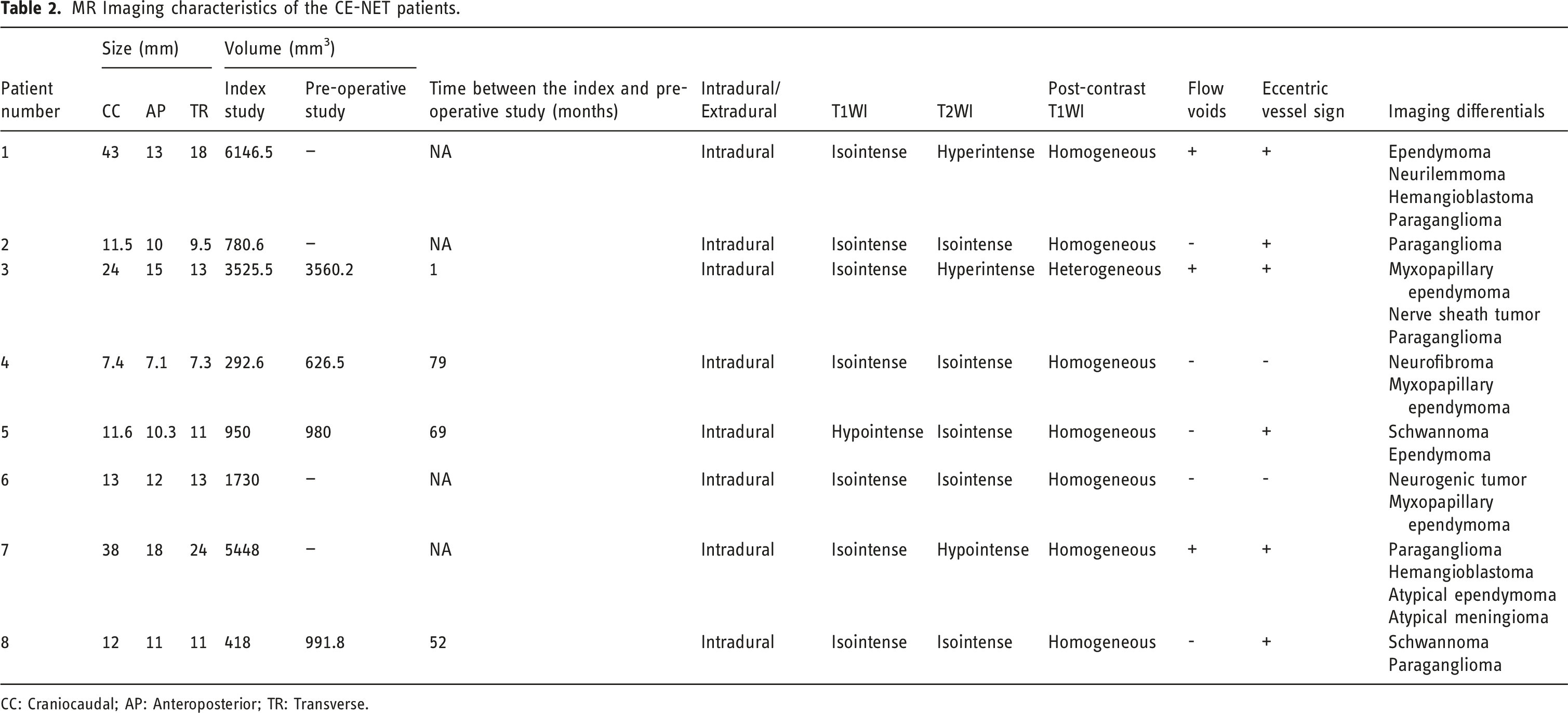
CC: Craniocaudal; AP: Anteroposterior; TR: Transverse.
CT imaging characteristics of the CE-NET patients.

CC: Craniocaudal; AP: Anteroposterior; TR: Transverse.
Immunophenotyping details in the current CE-NET cohort.

NR: not reported; P: present.
There was considerable scanner heterogeneity, and the MRI studies were performed on a GE (GE Healthcare, Milwaukee, Wisconsin) [3], Siemens (Siemens, Erlangen, Germany) [2], Hitachi (Hitachi Ltd, Japan) [2], and a Philips (Philips Healthcare, Best, the Netherlands) [1] scanner with magnet strength of <1.5 [2], 1.5 [5] and 3T [1]. Most studies had axial and sagittal T1WI, T2WI, and post-contrast images, while 5/8 images had sagittal short-tau inversion recovery [STIR] images. On MRI, the lesions were isointense on T1WI in 7/8 patients and hypointense in 1/8 case. On T2WI, lesions were hyper-, hypo-, and isointense in 2, 1, and 5 cases, respectively. All lesions showed homogenous enhancement post-contrast, except one case which had small associated cysts. None of the lesions demonstrated any hemorrhage, necrosis, or calcification. The average lesion size of the 8 cases was 19.7 mm in the craniocaudal axis, 12.55 in the anteroposterior axis, and 12.1 in the transverse axis. Intradural flow voids were seen in 3/8 cases, all of which had maximal craniocaudal dimension >2 cm. In two patients with only CT imaging available, both studies were performed on a Siemens scanner, the lesions were ovoid and demonstrated well-defined margins. The average lesion size was 23 mm in the craniocaudal axis, 17.5 mm in the anteroposterior axis, and 23 mm in the transverse axis (Figure 3). Sagittal T1-contrast enhanced MR image (a) shows an enhancing ovoid intradural lesion within the spinal canal with a prominent vessel along the postero-superior aspect (white arrow). Sagittal CT image (b) in another patient demonstrates a hyper enhancing homogeneous intradural mass at the L5 level (black arrow). Sagittal CT myelogram image (c) of the same patient in (b) reconfirms the intradural location (black arrow). Intra-operative ultrasound image (d) in another CE-NET patient demonstrates the hyperechoic characteristics of the lesion (black arrow).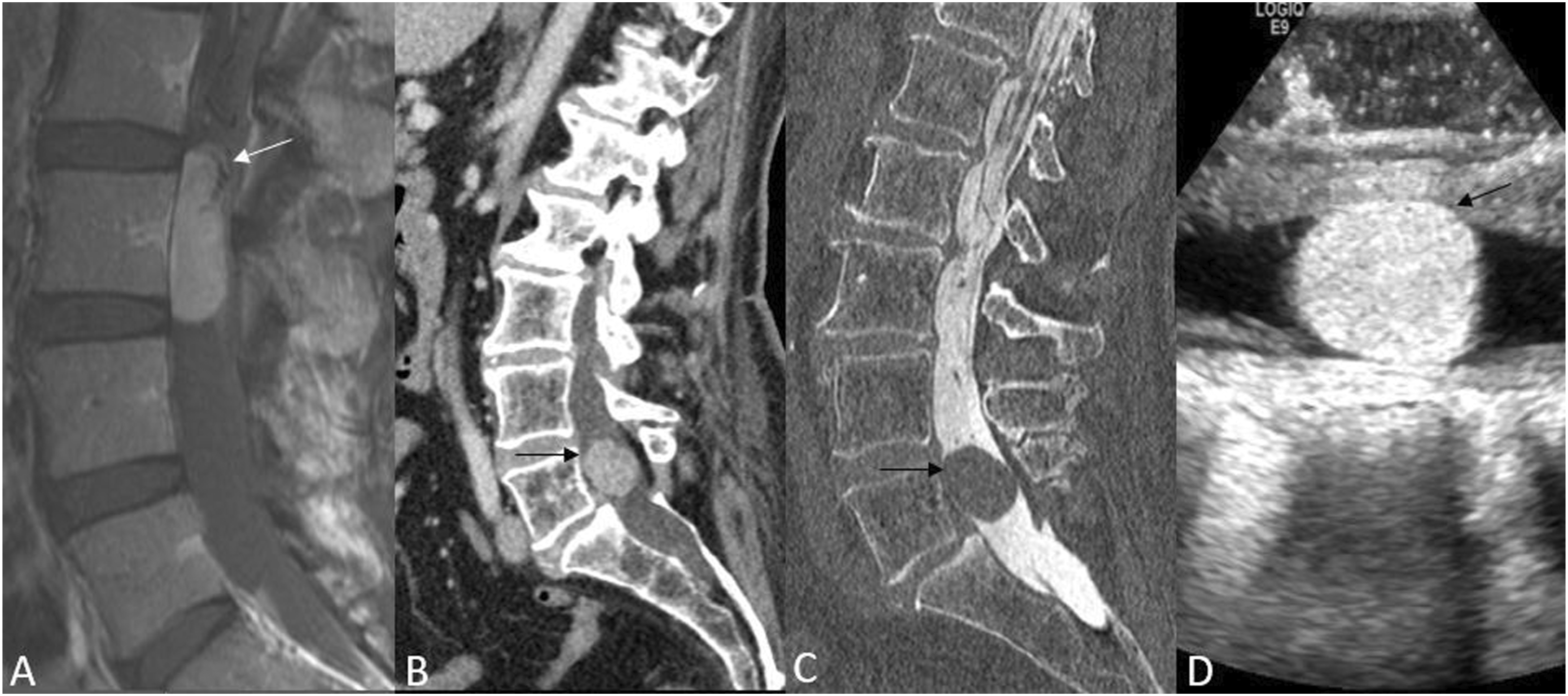
None of the lesions had imaging findings suggestive of a “salt and pepper” or “cap sign” on MR imaging. The “tadpole sign,” on either CT or MR imaging was seen in 5/10 cases. Interestingly, axial T2WI images in 6/8 cases showed the presence of an eccentric hypo intensity along the cranial aspect and in two cases along the caudal aspect of the tumor. The hypo intensity was felt to represent a vascular flow void and showed a branching pattern within the tumor in a few cases. Unlike the cauda equina nerve roots, which were displaced by the tumor, the flow void appeared to extend into the tumor itself, but was often located eccentrically to the lesion, either dorsally or laterally (eccentric vessel sign). A small vessel along the cranial aspect of the tumor was also noted in 2/2 cases and along the caudal aspect of the tumor in one case of CE-NET where only a contrast-enhanced CT study was performed. Intraoperative details of tumor vascularity were provided in 5 cases, describing a correlative vascular pedicle along the superior aspect of the tumor in all cases. Overall, a prominent vascular pedicle along the cranial aspect of the tumor was seen in 8/10 cases (eccentric vessel sign), as compared to the tadpole sign which was seen in 5/10 cases.
Serial follow-up MR imaging was available in 4/10 patients, allowing for assessment of volumetric changes in tumor volume over time. The mean lesion size in the index study for these 4 cases was 1296.5 mm3, and in immediate pre-operative study was 1539.6 mm3 over a mean follow-up of 4.2 years, suggestive of indolent course and consistent with the natural history of CE-NETs (Table 2).
None of the cases showed any local invasion or evidence of systemic metastasis. Osseous remodeling was seen only in one patient (P-7). None of the cases demonstrated any additional synchronous/metachronous paraganglioma. In patients with contrast enhanced MR imaging (8/10), none showed any leptomeningeal seeding. It is pertinent to note that two patients had CT-only imaging (P-9, 10), which is not optimal for identifying smaller metastatic CNS lesions. However, no evidence of CNS metastatic lesions was noted between the index scan and surgery (mean 668 days) in both patients. The primary differential considerations on imaging reports included ependymoma, schwannoma, and neurofibroma, with paraganglioma being prospectively considered in 4/11 cases. PET scan was performed in one patient (P-7) and revealed somatostatin uptake (Krenner stage-4). While lumbar radiographs were available in the majority of cases, they were unrevealing for an underlying mass lesion.
Discussion
The cell of origin of CE-NETs is not definitely established. These were previously felt to originate from the sympathetic or heterotopic neurons in the thoracic and lumbar lateral horns of the spinal cord, from non-neural crest tissue or vestigial fetal paraganglia tissue that incompletely regressed and persisted into adulthood.11,16 However, CE-NETs are not seen in patients with hereditary PPGLs secondary to SDH subunit gene mutations and also do not express zinc finger transcription factor GATA3 (seen in paragangliomas of the autonomic system). Furthermore, CE-NETs express HOXB13, a transcription factor developmentally expressed in the progenitor cells in the caudal spinal cord and in myxopapillary ependymomas. In light of these findings, CE-NETs are currently believed to arise from specialized neural crest cells in the cauda equina/filum terminale region. 1
The pre-operative diagnosis of CE-NETs is challenging primarily given the rarity of the lesion, but also because of lack of any additional imaging signs that may allow for a more accurate diagnosis. Certain imaging findings have been described but are again not common. For example, the salt and pepper appearance, typically described in head and neck paragangliomas is less commonly described in CE-NETs. 13 Yang et al. however, noted presence of salt and pepper sign in 8/19 patients in their cohort, although it is unclear if the authors also considered prominent intradural vessels as salt and pepper sign. 10 Notably, the mean tumor size along the maximum axis in their cohort was 49.3 mm, compared to about 2 cm in our cohort. Similarly, the cap sign, which refers to the T2-hypointensity along the tumor margin and felt to reflect hemosiderin deposition, is only seen in 5% of cases. 13 In the current series, none of the lesions showed “salt and pepper” or “cap sign,” which may be due to the smaller tumor size and number of cases in the current cohort, respectively. The tadpole sign, which describes the tumor lesion as the head of the tadpole and the tortuous vessels as the tail of the tadpole, was previously described by Yi et al. in their institutional review series. 14 They noted the tadpole sign in 9/13 (69%) of cases, while it was only seen in 5/10 (50%) of cases in the current series. One possible explanation might be that mean tumor size in their series was larger (2.6 cm craniocaudal) as compared to our study (2 cm craniocaudal). Additionally, the tadpole sign is only seen on the sagittal images and the prominent vessel as well as the tumor need to be in the same sagittal plane for the vessel to be seen extending into the tumor. Following the tumor vascular pedicle on the axial images, as in our series, allows for a more consistent assessment of its relation to the tumor and is therefore likely more sensitive.
Presence of prominent intradural vessels has been described previously, 13 but was only seen in 3/8 cases (37.5%) in the current series and only present in tumors larger than 2 cm in maximal dimension. On the other hand, a vascular pedicle along the cranial aspect of the tumor, possibly a draining vein, was seen in 8/10 cases as described above. This vessel was generally observed along the dorsal aspect of the mass and was invariably eccentrically located in relation to the mass. The presence of the eccentric vessel sign, as described here, was noted in 80% of CE-NET cases, and may be a useful imaging clue, given that it was present even in smaller tumors. However, further large-scale studies should be performed in the future to evaluate the prevalence of this sign in cases of CE-NET vis a vis other intradural tumors.
Our findings are similar to the previously reported literature on CE-NETs in terms of age at presentation and clinical symptomatology.8,17 None of the tumors in our series showed any hemorrhage, which has been variably reported in 10% (2/19) of cases by Yang et al. and 15% (2/13) of cases by Fiorini et al. in their series.10,17 This may at least be partly due to the larger tumors encountered in those series, with a mean craniocaudal dimension of 4.9 and 3.4 cm, respectively, in contrast to 2 cm in our series. Similarly, Fiorini et al. noted cystic degeneration in 5/13 (38%) cases as compared to 1/10 (10%) of cases in our series.
On imaging, diagnosing a CE-NET poses significant challenges partly due to the rarity of their occurrence and overlap with other more common tumors such as ependymoma (primarily myxopapillary variant) and schwannoma.13,18 Myxopapillary ependymoma (WHO grade-2 tumor) accounts for nearly a quarter of ependymoma cases and has a predilection for the conus medullaris. Demographically, it tends to afflict the population in the 4th and 5th decade of life with no gender predilection. On MRI, they are usually heterogeneous due to variable amounts of soft tissue, mucin, cysts and hemorrhage. In our case series none of the CE-NETs showed any hemorrhage. Similarly, intratumoral cyst was seen in only 1/10 cases, unlike ependymomas where they may be seen in up to 50% of cases. 18
Spinal schwannomas may also involve the cauda equina. They are typically small, well encapsulated, with a small minority reported to be extradural and with malignant potential. Demographically, they have a similar profile to ependymoma, presenting in the fourth and fifth decades and with no gender predilection. They tend to occur predominantly in the cervical and the lumbar spine.19,20 On MRI, they are also well-circumscribed and appear iso-/hypointense on T1WI and generally hyperintense on T2WI. With continued growth, they may show cystic degeneration and correspondingly a heterogeneous appearance. Tumors are generally oriented along the axis of the nerve root and may have a characteristic dumbbell shape as they exit through the neural foramen. 13
Finally, hemangioblastoma may involve the spinal canal and can present in the absence of underlying Von Hippel Lindau syndrome. The latter, however, are extremely rare, and a recent study noted only 20 previously reported cases in the literature. Like CE-NETs, the mean age of presentation was around 53 years. Lesions may show associated prominent vessels, and most commonly occur at L3 level. 21 Unlike spinal cord/intramedullary hemangioblastomas which usually tend to be associated with either a syrinx or edema in tumor vicinity, similar findings may not be seen with an extramedullary hemangioblastoma. 22 However, limited imaging literature from sporadic case reports suggests that intradural flow voids and peritumoral cysts may be seen. 23 Additionally, since a vast majority of these are syndromic, co-existing lesions suggestive of underlying Von Hippel Lindau syndrome may provide additional imaging clues to the correct diagnosis.
Histopathologically, CE-NETs are well differentiated and categorized as WHO grade I tumors. They are composed of type I chief cells disposed in nests or lobules (Zellballen) and surrounded by an inconspicuous single layer of type II sustentacular cells. Degenerative nuclear pleomorphism may occur and is generally mild. Approximately 25% of CE-NETs have mature ganglion cells and a Schwann cell component (referred to as gangliocytic neuroendocrine tumors). Foci of hemorrhagic necrosis and scattered mitotic figures may be seen, although neither these, nor nuclear pleomorphism are of any prognostic significance. 1
The primary treatment for CE-NETs is complete surgical removal of the tumor, as it is well-contained.15,24 This was the case in all of our patients who underwent gross total resection. The prognosis is generally good for CE-NETs after total surgical resection, with a low local recurrence rate. However, during surgery, tumor manipulation can trigger sudden release of catecholamines, leading to fluctuations in blood pressure, or cerebrovascular accidents. 14 Intraoperative neuromonitoring and careful preoperative evaluation of imaging studies are essential for optimizing surgical outcomes. The role of adjuvant therapies in the management of CE-NETs, such as radiotherapy and chemotherapy, remains unclear and requires further investigation. 25
Conclusion
CE-NETs are rare, highly vascular tumors that can be diagnostically challenging. However, the presence of a well-defined, generally homogeneously enhancing soft tissue mass in a middle-aged patient with a prominent eccentric vessel sign and absence of calcification should arise suspicion for underlying CE-NETs. The eccentric vessel sign, which has not previously been reported to our knowledge, can provide a useful adjunct to other previously described signs for establishing an accurate pre-operative radiological diagnosis of CE-NET.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
