Abstract
Multifocal and multicentric glioblastoma (GBM) or collectively, m-GBM, is an imaging diagnosis present in up to 34% of patients with GBM. Compared to unifocal disease, patients with m-GBM have worse outcomes owing to the enhanced aggressive nature of the disease and its resistance to currently available treatments. To improve the understanding of its complex behavior, many associations have been established between the radiologic findings of m-GBM and its gross histology, genetic composition, and patterns of spread. Additionally, the holistic knowledge of the exact mechanisms of m-GBM genesis and progression is crucial for identifying potential targets permitting enhanced diagnosis and treatment. In this review, we aim to provide a comprehensive summary of the cumulative knowledge of the unique molecular biology and behavior of m-GBM and the association of these features with neuroimaging.
Keywords
Introduction
Glioblastoma (GBM) is the most frequently occurring uniformly fatal primary adult central nervous system (CNS) malignancy.1,2 According to the fifth edition of the World Health Organization Classification of CNS tumors grading (WHO CNS 5/WHO 2021), GBM is defined as a grade 4 isocitrate dehydrogenase wildtype (IDH-wt) diffuse adult-type astrocytomas.3,4 GBMs are presumed to arise from the subventricular zone (SVZ) multipotent neuroglial stem cells, resulting in a broad range of genotypic and phenotypic tumor heterogeneity.3,5 Despite the increased understanding of the disease and optimized therapeutic approaches, the median overall survival of patients remains less than 17 months. 6
Multifocal and multicentric GBM constitute more aggressive and migratory subtypes of GBM, presenting with multiple tumor foci and occurring in up to 34% of patients with GBM.7,8 Patients diagnosed with these entities have a median overall survival of 6-8 months, which is significantly shorter compared to patients with solitary GBM.9,10 Yet there is no unique approach offered to these patients, and the adopted treatment standard, similar to unifocal GBM, is mostly localized surgical resection followed by chemoradiation.11–13 Developing systemic and/or targeted treatment options requires a solid understanding of the microscopic biology and macroscopic behavior of multifocal and multicentric GBMs.14,15
Although similar in their clinical course and prognosis, multifocal GBM is defined as multiple distinct foci of contrast enhancement that are connected, whereas multicentric GBM describes spatially distant tumor foci that lack a communication route.16–18 While these definitions are still valid since first identified in 1963, diagnosis has evolved with advancing histopathologic and radiologic techniques. Currently, the diagnosis of multifocal and multicentric GBM is primarily based on conventional contrast-enhanced magnetic resonance (MR) and T2 FLAIR imaging.17,19–21 Multifocal GBMs present with multiple distinct contrast-enhancing lesions on T1-weighted MRI that are connected with T2-weighted fluid-attenuated inversion recovery (T2 FLAIR) hyperintense signal abnormality representing non-enhancing malignant infiltration (Figure 1). Multicentric GBM describes the presence of multiple enhancing lesions without a T2 FLAIR hyperintense connection between enhancing foci (Figure 2).19,20,22 Since there is no significant relevance to distinguishing multifocal and multicentric GBM, we will refer to both entities collectively as multiple GBMs (m-GBMs).
23
We aim to provide a comprehensive overview of the unique molecular biology and behavior of m-GBM and their correlation to imaging. Baseline imaging of multifocal IDH-wildtype glioblastoma in a 66-year-old patient. (a), (b) Axial and (c) coronal contrast-enhanced T1-WI showing multiple distinct enhancing lesions in the left frontal lobe (arrows). (d) Coronal T2 FLAIR image showing hyperintense signal connecting the two enhancing foci (arrow).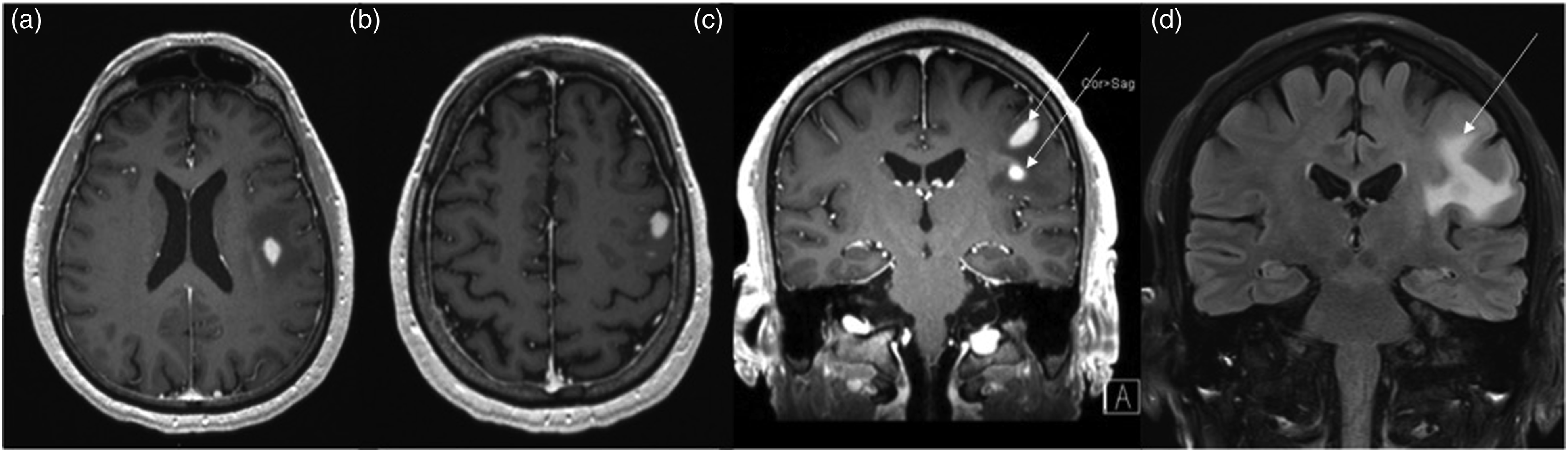
Discussion
Imaging reflects phenotypic heterogeneity
M-GBMs are heterogeneous in their genetic and epigenetic landscapes, protein expression levels, structural compositions, and biochemistry of their microenvironments. The mixture of these various changes is reflected as abnormalities observed in gross histology, tumor morphology, and radiographic imaging appearance (Figure 2).16,24 This multilevel heterogeneity may vary spatially within any single tumor focus, as well as across m-GBM foci in different brain regions in the same patient, reflecting the degree of genetic aberrations in the disease and regional epigenetic adaptations of the tumor to surrounding microenvironmental signals, respectively. Thus, m-GBM lesions of similar genomic alterations may express morphologic variabilities in their composition, including the degree of vascularity, color, texture, bulk, cellularity, and the presence of hemorrhagic or cystic components.16,24 Baseline imaging of pathology-proven multicentric IDH-wildtype glioblastoma in a 69-year-old patient. (a) Axial contrast-enhanced T1-WI showing a left frontal peripherally and heterogeneously enhancing mass. (b) Axial T2 FLAIR showing hyperintense signal surrounding the enhancing mass and two distinct hyperintense foci. (c) DWI and (d) ADC maps show diffusion restriction in the enhancing and the anterior non-enhancing lesion (arrows) concerning for high cellularity. (e) T2 star showing microhemorrhage in the enhancing lesion.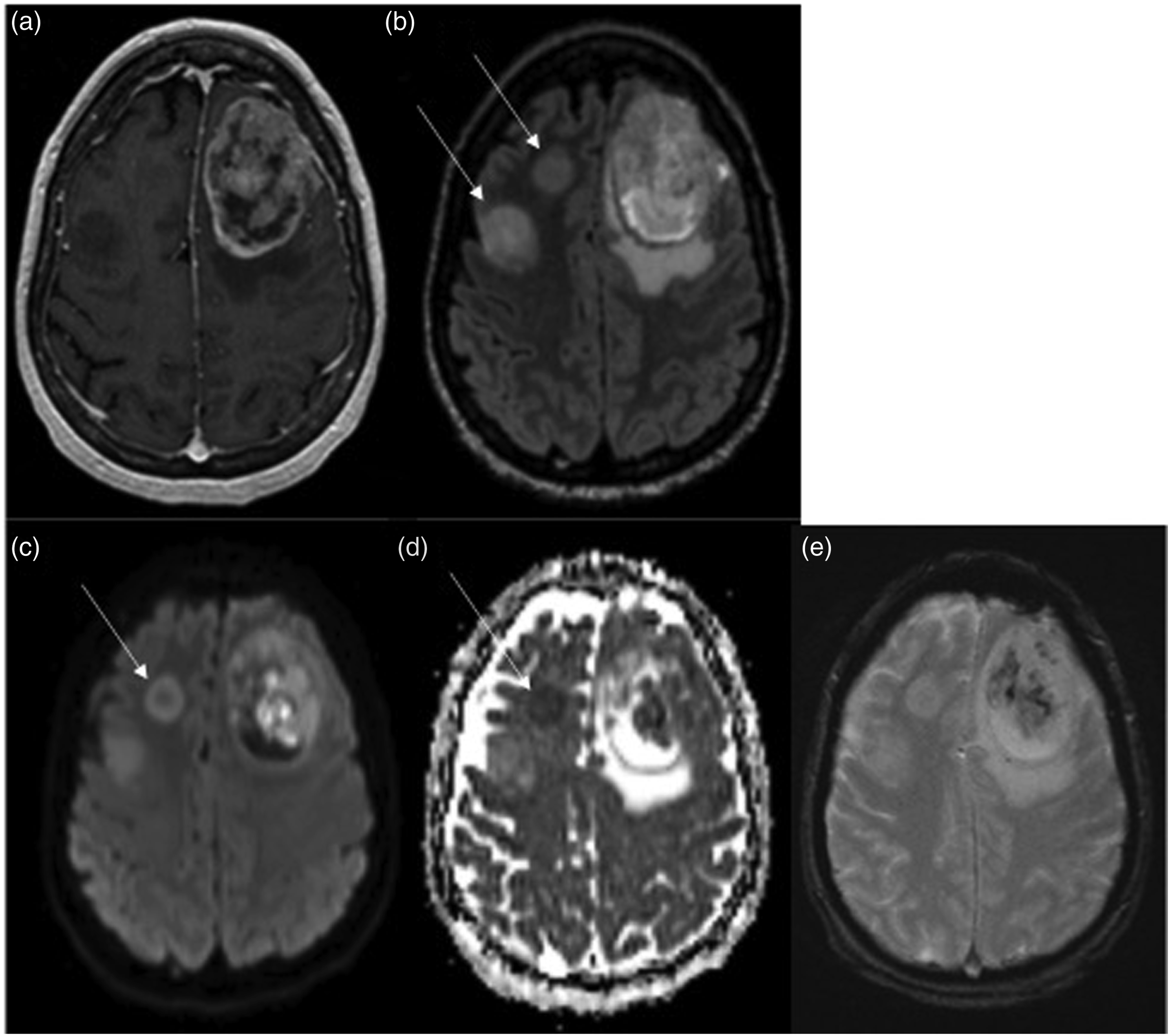
Conventional and advanced MR imaging provide non-invasive tools that reflect the described morphologic variations, uncovering the necessary molecular regional adaptations of the different GBM lesions.
25
On contrast-enhanced T1-weighted imaging (T1-WI), the different m-GBM lesions may demonstrate different sizes, growth kinetics, and composite intensities with varying degrees and patterns of contrast enhancement, reflecting the underlying heterogeneity in the tumor bulk, texture, and angiogenesis. Each enhancing lesion is associated with a heterogenous hyperintense signal abnormality on T2 FLAIR demonstrating an increased water content that represents the non-enhancing tumor component with indistinct margins and vasogenic edema.
26
There are yet no definitive imaging features permitting the separation of the two components. In multifocal GBM, the T2 FLAIR sequence is assumed to represent an infiltrative non-enhancing GBM component connecting two or more distinct enhancing lesions.1,20,27 Although the diagnosis of both multifocal and multicentric GBM requires the presence of contrast enhancement, the presence of T2 FLAIR hyperintense foci in a distinct brain region may represent an early appearance of m-GBM (Figures 2 and 3).
28
The variability in cellular density within the different GBM lesions may present as differences in diffusion restriction, or may be quantitatively estimated using apparent diffusion coefficient (ADC) derived from diffusion imaging. Finally, qualitative and quantitative measurements of different perfusion metrics may provide more specific reflection of vascular proliferation, permeability, and blood flow within the GBM foci.26,29 Baseline MRI of multicentric IDH-wildtype glioblastoma in a 74-year-old patient. (a) Contrast-enhanced T1-WI showing a ring-enhancing lesion in the left temporal lobe involving the ependymal surface of the left atrium. (b) T2 FLAIR image showing hyperintense signal surrounding the enhancing lesion. (c) Contrast-enhanced T1-WI showing a second non-enhancing tumor with only (d) T2 FLAIR hyperintense abnormality (arrow) in the left frontal opercular region that is not connected to the first one described. Follow-up imaging showed contrast enhancement within the originally non-enhancing region.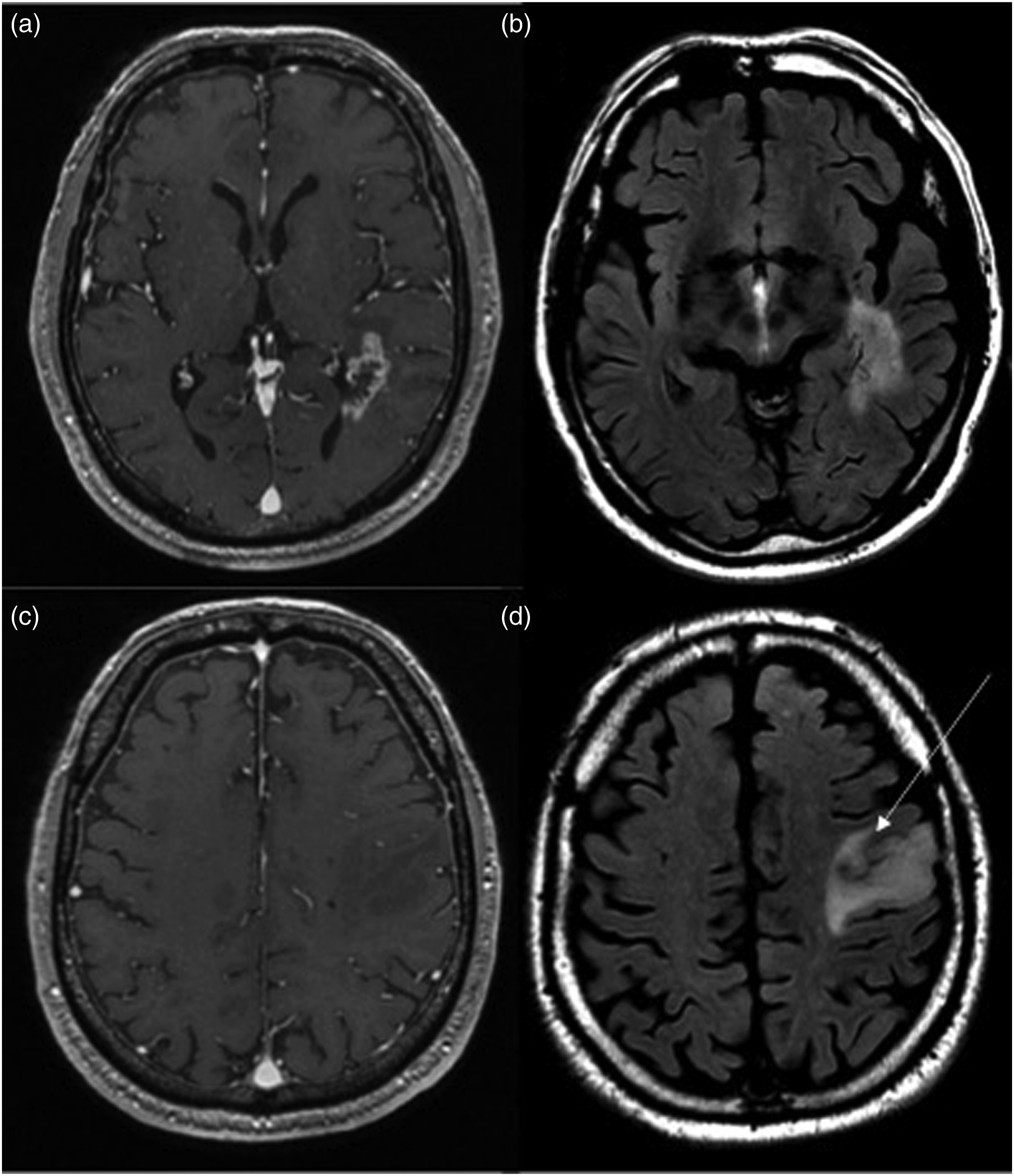
Genetic and epigenetic landscape of m-GBM
Genetic and epigenetic landscapes inform the radiologic phenotype of m-GBM.23,30–33 Thus, identifying correlations between different imaging characteristics and their underlying molecular correlates is indispensable to enhance the diagnostic and prognostic value of imaging.29,34 Genomic analyses comparing solitary to multiple high-grade gliomas concluded that the presence of multiple enhancing foci on MRI imaging almost exclusively predicts IDH-wildtype genotype.15,23,35,36 Additionally, m-GBMs were found predominantly to express the mesenchymal molecular signature, and none were proneural, with the latter being predominantly IDH-mutant tumors. This is of great interest because the cancer genome atlas (TCGA) mesenchymal subtype classification is defined by genetic and epigenetic alterations that are predictive of the most aggressive GBM phenotype and associated with the worst prognostic category. 37 Significantly, from an imaging perspective, mesenchymal subtype GBM exhibits a ratio of T2 FLAIR hyperintense signal to contrast enhancement (including necrotic volume) of less than 2.3 (sensitivity 82%, specificity 87%).23,35,37,38
Compared to unifocal GBMs, m-GBM uniformly harbors higher frequencies of epidermal growth factor receptor (EGFR) amplifications and concurrent alterations of EGFR with phosphatase and tensin homolog/mutated in multiple advanced cancers (PTEN/MMAC) gene, as well as the reverse transcriptase (TERT) promoter.31,35 On imaging, EGFR amplified tumors have a larger volume of contrast enhancement and T2 FLAIR hyperintensity compared to EGFR non-amplified tumors. These tumors exhibit a regional predilection to grow towards the posterior lateral ventricles, which is the anatomic location of the neural stem cells.29,39 These findings recapitulate other studies which have demonstrated the relationship between the proximity of m-GBM to the ventricular-subventricular zone. 40
In any specific patient, different tumor foci consistently shared distinct unique genetic aberration events, especially those involving chromosome 10 and the TERT promoter. These findings imply a monoclonal origin of m-GBM and highlight the role of chromosome 10 and TERT promoter alterations as founder events in glioblastoma genesis. 35 Similar to solitary GBM, the different m-GBM lesions in any patient consistently had different aberrations in the three principal pathways of GBM development, comprising: the receptor tyrosine kinase (RTK)/phosphatidylinositol 3′-kinase (PI3K)/AKT axis, p53 signaling, and retinoblastoma mediated control of cell cycle progression.35,41 However, the tumor foci expressed various degrees of heterogeneity in the affected genetic events, which suggests parallel evolution and possible early occurrences in the disease. 35 Distant recurrent and m-GBM were associated with higher degrees of genetic divergence compared to the initially treated tumor signifying that microscopic GBM migration occurs early in tumorigenesis. 30 Indeed, not only the presence of microscopic infiltration but also microscopic multicentricity has been reported, and detection of these events are current limitations of diagnostic imaging. 33
In addition to the genetic alterations identified in m-GBM, idiosyncratic epigenetic changes are often observed, and these carry valuable diagnostic, prognostic, and therapeutic implications.23,42 The pathways involved in the immune response, cytoskeletal remodeling, mitochondrial respiration, collagen decomposition, lipid kinase activity, and tumor necrosis factor presence were found to be differentially enriched in m-GBM. 23 The most significant differences in methylation were seen in the most differentially expressed genes in m-GBM compared to unifocal GBM. Among the 45 identified genes, cytochrome b5 reductase (CYB5R2) exhibited the most significant differences in gene expression, promoter methylation, and copy number alteration. The methylation level of the CpG islands of the CYB5R2 promoter, negatively associated with CYB5R2 expression, was significantly lower in the m-GBM group. CYB5R2 is involved in collagen degradation, immune regulation, and tumor invasion mechanisms, and its high expression is associated with a shorter overall survival duration. 23
Imaging correlates to pattern of spread
A comprehensive knowledge of the different patterns of malignant spread in m-GBM can help to optimize therapeutic approaches to improve patient outcomes. Currently, there is no consensus as to the origin, triggers, and routes of spread in m-GBM. However, the growth of m-GBM is speculated to originate from multipotent neural stem/progenitor cells (NSPCs) that possess migratory potential. M-GBM growth may be triggered by neural activity and supported by CNS cytoskeletal structures. Other routes of proposed tumor dissemination include which include neuronal communication, and conveyance by white matter tracts, the venous system, and/or the glymphatic system.16,43,44
Lateral ventricles and m-GBM malignancy
Emerging data suggest that m-GBM arises from NSPCs in the subventricular zone (SVZ) that layers the lateral ventricles, providing tumors with multipotent, self-renewing, and migratory potential.45,46 NSPCs have increased affinity for GBM and are involved in cross-communication with the tumor.
39
GBMs in closer proximity and/or in contact with the ventricular-SVZ have poorer overall outcomes and prognoses and overall outcome. The presence of the contrast-enhancing GBM in contact with the SVZ preoperatively significantly correlates with the development of m-GBM and/or distant recurrences (Figure 4).
47
Conversely, the absence of SVZ and/or cortical contact predicts localized disease in 100% of the cases.
47
This correlation between the spatial contact with ventricles and m-GBM may be also partly due to the presence of CSF and its potential to support the GBM spread. Axial MRI sequences showing multicentric glioblastoma in a 45-year-old patient. (a) and (c) Contrast-enhanced T1-WI showing bilateral cerebral and brainstem contrast-enhancing lesions, with the largest one abutting the left lateral ventricle (arrow). (b) and (d) T2 FLAIR MRI showing bilateral hyperintense abnormalities without intervening FLAIR signal.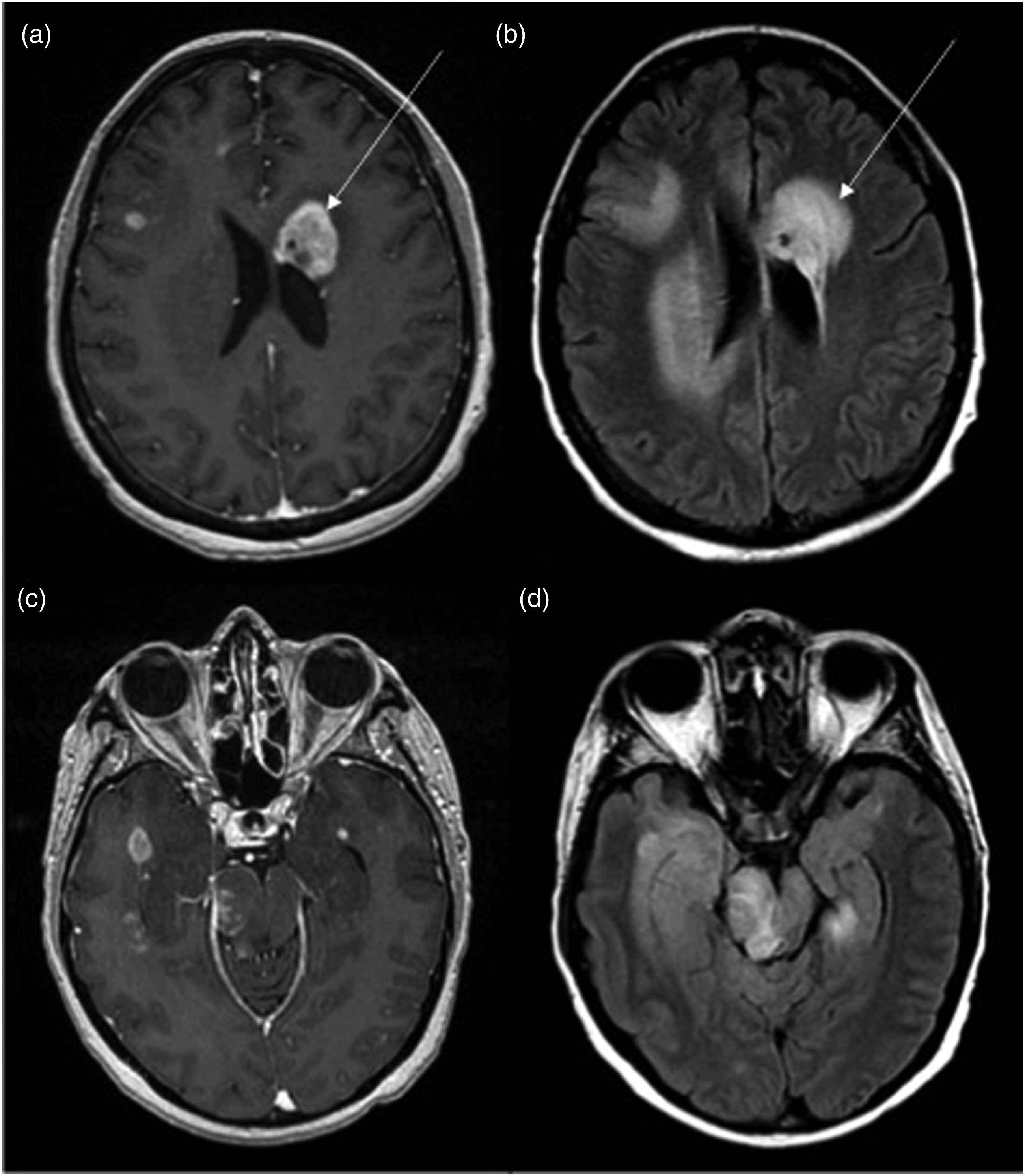
Cortical invasion and progression
The enhanced malignant potential and increased incidence of m-GBM seen in gliomas infiltrating the cortex may be due to activity-related neuronal triggers. Conversely, contrast-enhancing lesions confined to the white matter without cortical involvement are unlikely to develop distant m-GBM lesions. In vitro and animal studies have shown a positive electrochemical feedback loop between activity-related GBM progression and GBM-induced neuronal excitability. The synaptic protein neuroglien-3 (NLGN-3) and other neurotrophins are identified as major mitogens of GBM proliferation that may be used as future molecular diagnostic and therapeutic targets.48,49
White matter and spread
GBMs are known to spread within the white matter along the axonal tracts and perivascular spaces. In many m-GBM cases, infiltrative malignant cells are seen within the white matter areas connecting two macroscopic enhancing GBM foci. However, as discussed previously tumors exclusively present in the white matter are observed to have a lower tendency to form m-GBM (Figure 5).
47
This preferential demyelinating growth pattern along white matter is mediated by the attraction of soluble factors that activate the positive feedback loop in GBM stem cells. Radiographically, abnormal T2 FLAIR signal hyperintensities are found to correlate with the presence of focally diffuse and infiltrative tumor cells between two m-GBM lesions on histopathologic studies.
50
Using T2 FLAIR to identify spread through the white matter may be suboptimal because the hyperintensity seen may be only related to vasogenic edema, and normal appearing tissue on T2 FLAIR may contain microscopic GBM infiltration. When microscopic infiltration is invisible on conventional MRI, advanced diffusion tensor imaging (DTI) with probabilistic fiber tracking may be used to identify white matter dissemination between two m-GBM foci (Figure 6).
44
Although the white matter tracts seem to be the most common route of tumor growth seen in around 40% of cases, many tumors spread in a pattern that is perpendicular to the direction of white matter tracts.
44
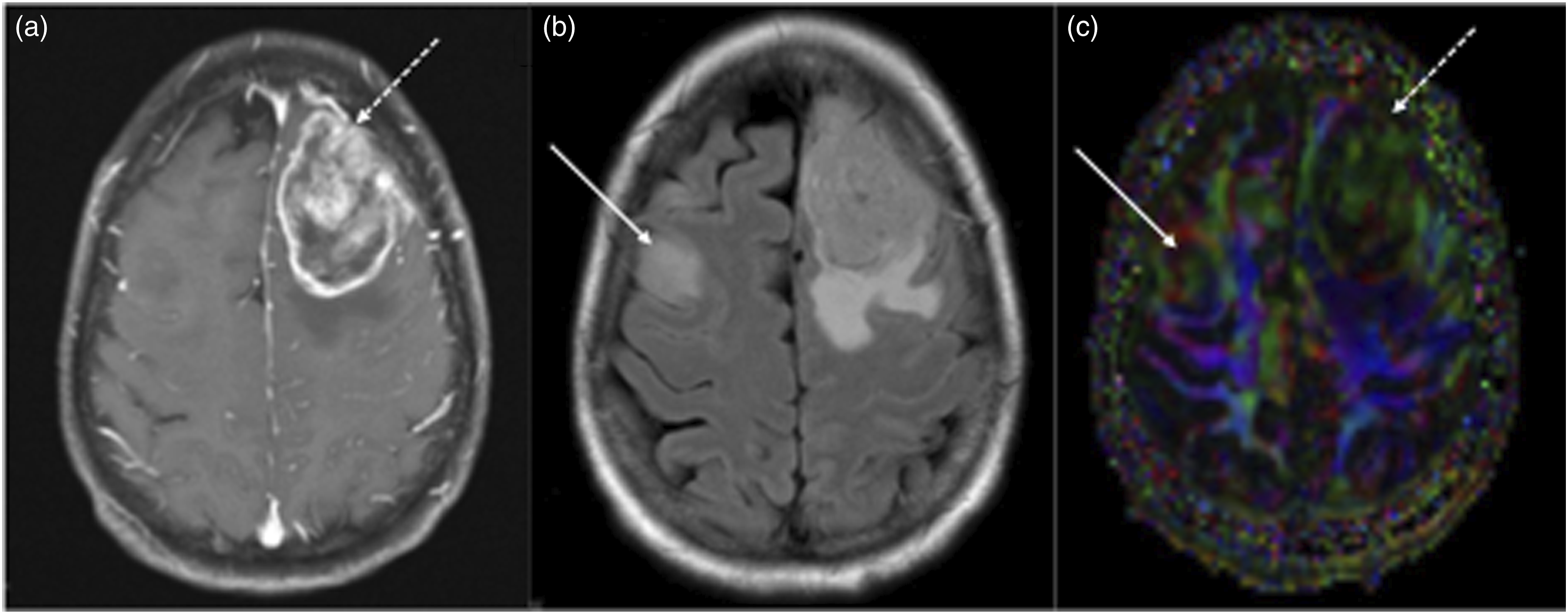
Baseline imaging of white matter IDH-wildtype glioblastoma in a 60-year-old patient. (a) Contrast-enhanced T1-WI showing a ring-enhancing lesion in the right corona radiata. (b) T2 FLAIR image showing hyperintense signal extending into the cortex causing a mass effect on the adjacent lateral ventricle. (c) DWI and (d) ADC maps showing peripheral diffusion restriction. The patient had positive outcomes after receiving tumor resection and chemoradiation with overall survival of 13 years after diagnosis. Baseline imaging of pathology-proven multicentric glioblastoma in a 69-year-old patient. (a) Contrast-enhanced axial T1-WI showing a left frontal heterogeneously enhancing mass (dotted arrow). (b) Axial T2 FLAIR image reveals an additional right frontal non-enhancing T2/FLAIR hyperintense lesion. (c) DTI Tractography shows white matter tract disruption and decreased fractional anisotropy (FA) at the site of the non-enhancing infiltrating right frontal lesion (arrow). At the site of the contrast enhancement left frontal mass, there is decreased FA as expected (dotted arrow).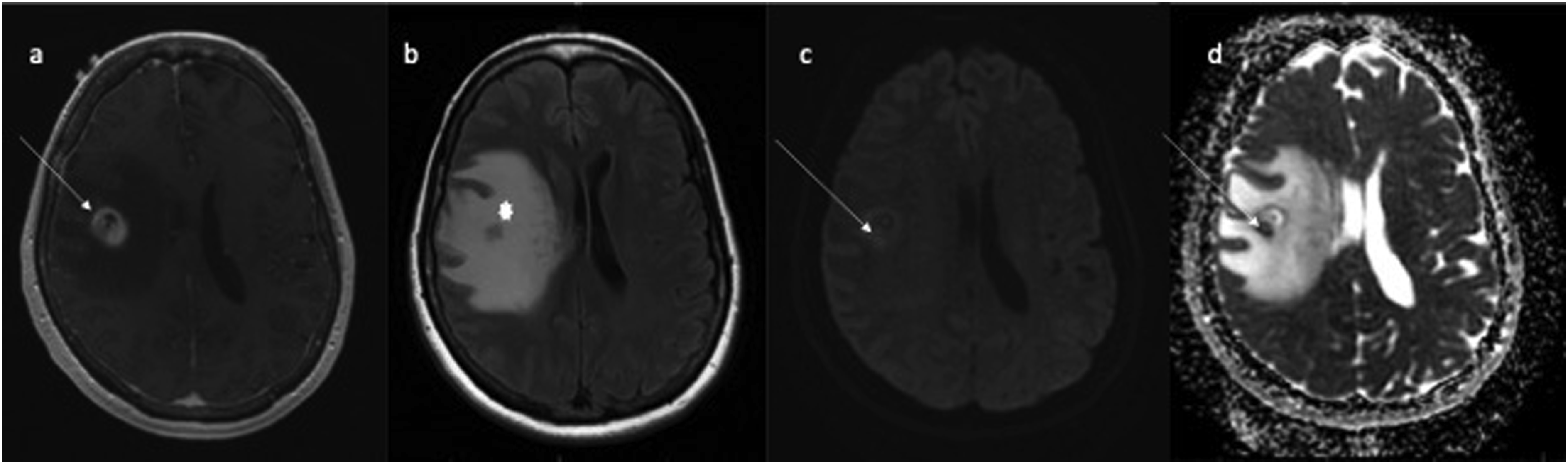
Fluid exchange and glymphatic regulation
As early as 1997, primary brain tumors were thought to spread through the interstitial fluid (ISF) along white matter tracts (“glymphatic”) to distant locations in the brain. 51 The ISF communicates with the CSF through a brain-wide paravascular flow regulated by aquaporin channels expressed on the long arm of astrocytic cells. The glymphatic system is considered a pathway for CSF influx and CSF/ISF efflux that interconnects the meningeal lymphatic vessel, paravascular spaces, vascular structures, subarachnoid space, and the ventricular system with the brain parenchyma.43,52 These paravascular routes were proposed to act as avenues for tumor cell migration and serve as candidate pathways for signaling molecules that guide GBM progression. 53 Additionally, aquaporin-4 channels were found to be upregulated in gliomas that were thought to be responsible for the increased ISF flow and contribute to the tumor-associated edema. The role of this relatively recently discovered system is currently under investigation with respect to the diagnosis, treatment, and treatment delivery systems for primary brain malignancies.54,55
Current treatment of m-GBM
Despite the enhanced understanding of biology of m-GBM and its therapeutic resilience, the treatment approach has remained unchanged over the past decade. Currently, the standard of clinical care of m-GBM is multimodal, comprising maximal safe resection of the tumor bulk followed by concurrent adjuvant chemotherapy with radiation as per the Stupp protocol. 20 Gross total resection (GTR) of the enhancing solid tumor is considered an ideal goal of surgery. However, in patients presenting with m-GBM involving multiple brain regions, frequently including eloquent areas, a conservative approach is usually adopted, resulting in the majority of patients receiving only biopsy or subtotal resection (STR).9,22,56 In m-GBM, the extent of tumor resection positively correlates with prolonged progression free and overall survival. Even when partial resection is performed, the resulting tumor cytoreduction reduces tumor-induced hypoxia mediated vascular proliferation and facilitates the diffusion of chemotherapeutic agents.13,57
Adjuvant irradiation of large target volumes in m-GBM is considered an additional treatment challenge in this patient population, primarily due to increased risk of radiation-induced neurotoxicity.5,12 Target definition, dosing and fractionation have not been investigated specifically for m-GBM, and the standard used for solitary GBM is applied to m-GBM.5,58 For instance, the clinical tumor volume (CTV) is generally defined as 2 cm increase in border beyond the gross tumor volume (GTV) (surgical cavity and contrast-enhancing portion), and intended to target microscopic non-enhancing disease based on the theory that most tumors recur locally, which might not be the case for m-GBM. 12 Additionally, although controversial, some studies have shown that irradiation of the tumor-adjacent SVZ results in improved outcome in patients with GBM with ventricular contact. Thus, rethinking target definition in m-GBM with regard to the increased likelihood of the presence of microscopic disease in the T2 FLAIR signal hyperintensity zone may be worthwhile. 50 For treatment fractionation, the gold standard is for an otherwise healthy patient to receive a dose of 60Gy delivered in 30 fractions with daily TMZ. However, in older patients or those with poor performance, hypofractionation may be the optimal alternative such as 40 Gy and 34 Gy delivered in 15 and 10 fractions, respectively.58,59 Although 3-dimensional (3D) conformal radiotherapy remains the most widely used standard for the treatment of GBM, intensity-modulated radiotherapy (IMRT) and volumetric intensity-modulated arc therapy (VMAT) may constitute superior approaches to target more tumors in more challenging locations which could be more probable in m-GBM. 12
Due to the extensive involvement of m-GBM where localized surgical and radiotherapy treatments may not be feasible, systemic treatment approaches might be optimally developed. Currently, TMZ is the sole approved standard chemotherapy drug proven to impact the progression of GBM.5,12 With recent discoveries in the field of immune access to the brain tumor microenvironment and advancements in genetic and epigenetic characterization of primary brain tumors, there has been an explosion of studies developing and investigating novel targeted and immunotherapeutic options for GBM. 60 Ongoing clinical trials are focused on the effects of various modes of drug delivery and combinations with other modalities such as radiotherapy. Although none is yet approved by the U.S. Food and Drug Administration, these treatment options provide promise for superior treatment approaches for m-GBM, a widely infiltrative brain malignancy.
Conclusion
Differences Between m-GBM and unifocal GBM.

Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
