Abstract
Objectives
Histological studies have shown alterations of thalamic nuclei in patients with Down syndrome (DS). The correlation of these changes on MRI (magnetic resonance imaging) is unclear. Therefore, this study investigates volumetric differences of thalamic nuclei in children with DS compared to controls.
Methods
Patients were retrospectively identified between 01/2000 and 10/2021. Patient inclusion criteria were: (1) 0–18 years of age, (2) diagnosis of DS, and (3) availability of a brain MRI without parenchymal injury and a non-motion-degraded volumetric T1-weighted sequence. Whole thalamus and thalamic nuclei (n = 25) volumes were analyzed bilaterally relative to the total brain volume (TBV). Two-sided t-tests were used to evaluate differences between groups. Differences were considered significant if the adjusted p-value was <0.05 after correction for multiple hypothesis testing using the Holm-Bonferroni method.
Results
21 children with DS (11 females, 52.4%, mean age: 8.6 ± 4.3 years) and 63 age- and sex-matched controls (32 females, 50.8%, 8.6 ± 4.3 years) were studied using automated volumetric segmentation. Significantly smaller ratios were found for nine thalamic nuclei and the whole thalamus on the right and five thalamic nuclei on the left. TBV was significantly smaller in patients with DS (p < 0.001). No significant differences were found between the groups for age and sex.
Conclusions
In this exploratory volumetric analysis of the thalamus and thalamic nuclei, we observed statistically significant volumetric changes in children with DS. Our findings confirm prior neuroimaging and histological studies and extend the range of involved thalamic nuclei in pediatric DS.
Introduction
Down syndrome (DS) is one of the most common congenital anomalies occurring in one in 800 live births. 1 DS usually results in neurological impairments in childhood which involve several domains including hypotonia, cognition, language, and memory function.2–7 There is increasing evidence that phenotypic features of the brain in DS can be traced back to early developmental stages. 8 Several studies reported neuroanatomic alterations with reduced brain parenchymal volumes including reduced total brain volume (TBV), gray and white matter volumes, frontal and temporal lobe volumes, hippocampal and cerebellar volumes.2,7,9–11 Given its crucial function as the main relay station for motor, limbic, and sensory pathways 12 and that thalamus-related behavioral changes are present in infancy, 13 surprisingly little is known about volumetric changes of the thalamus and especially of its nuclei in pediatric patients with DS. There are conflicting studies regarding thalamic volume and structure in pediatric DS. Gunbey et al. assessed brain MRIs of 10 pediatric patients with DS (mean age: 2.6 ± 0.69 years) and found significantly lower volumes of the bilateral thalami compared to healthy controls. 7 Lee et al. used diffusion tensor imaging-based morphometry to analyze cerebellar afferent networks in 15 patients with DS (mean age: 17 years) and found significantly reduced thalamic volumes. 2 Fatma et al., on the other hand, did not report significant changes of the thalamus in their cohort of five children with DS (mean age: 2.6 ± 0.69 years). 11 Few histological studies have assessed thalamic nuclei in pediatric 13 and adult patients with DS. 14 In brain tissue sections of fetuses with DS, Stagni et al. found reduced cellularity of the mediodorsal, centromedian (CM), and parafascicular (Pf) nuclei and reduced density of the proliferating cells in the third ventricle. 13 Karlsen et al. assessed brain sections of four adult females with DS and found significantly reduced number of neurons in the mediodorsal nucleus. 14 To the best of our knowledge, neuroimaging-based assessment of the volume of thalamic nuclei in pediatric DS is lacking in the literature. Given the evidence of histological alterations in the thalamic nuclei in the literature, we aimed to volumetrically analyze thalamic nuclei based on magnetic resonance imaging (MRI) in pediatric patients with DS and compare them to an age- and sex-matched control cohort.
Material and methods
Study population
This retrospective study was approved by the research ethics board of the Hospital for Sick Children (SickKids) (# 1000078916). Because of the retrospective nature of the study, informed consent was waived by the local research ethics board. The study was performed in line with the principles of the Declaration of Helsinki. All patients were identified from the electronic health record data base from January 2000 to October 2021. Patient inclusion criteria were the following: (1) 0–18 years of age at the time of imaging, (2) diagnosis of Trisomy 21, and (3) availability of a brain MRI with a non-motion-degraded volumetric T1-weighted sequence. Patients with acute or chronic structural brain parenchymal injury visualized on MRI of the brain were excluded. Demographic patient data were collected by a review of the electronic health records. Age- and sex-matched controls were selected from the local institutional MRI database using the following criteria: (1) normal MR imaging examination of the brain, (2) absence of neurological disorders, and (3) availability of a non-motion-degraded volumetric T1-weighted sequence.
MRI acquisition
Patients with DS and healthy controls underwent MR imaging of the brain at 1.5T or 3T across various scanners (Signa HDxt, GE Healthcare, Waukesha, WI; Achieva, Philips Healthcare, Best, The Netherlands; Magnetom Skyra, Siemens Healthineers, Erlangen, Germany) using a dedicated head-coil. For each individual, high spatial resolution volumetric T1-weighted 3D gradient echo images were acquired either in the sagittal or the axial plane: TR/TE, 6–9/2–5 ms; flip angle: 10–15; resolution: 0.85–1.1 × 0.85–1.1 × 0.9–1.1 mm; field of view (FOV): 22–24 cm; 150–200 slices.
Analysis: Visual inspection and FreeSurfer analysis
FreeSurfer is an open-access software package for structural brain imaging analysis.15–17 It serves as an automatic reconstruction pipeline for the processing of brain images including skull stripping, motion artifact correction, B1 bias field correction, gray-white matter segmentation, and region labeling on the cortical surface using different atlases in the pediatric18,19 and adult age groups.20–22 Technical details are available from prior publications.23–25 De-identified 3D T1-weighted structural sequences were made available from our local picture archiving and communication system (PACS) and processed using FreeSurfer version 7.1.1 (Massachusetts General Hospital, Harvard Medical School; http://surfer.nmr.mgh.harvard.edu), 23 which aligns the input examination to several atlases including the Iglesias atlas 26 for thalamic nuclei. To manage image processing, we used an in-house developed web-based data management software running on a local server (Lenovo Legion C730 with i9 processor and 32 Gb of RAM (Lenovo, Quarry Bay, Hong Kong)).
Thalamic nuclei are categorized into relay nuclei (RN) including the lateral, medial, anterior and midline groups, the reticular nucleus, and intralaminar nuclei (IN) including the rostral and caudal nuclei according to Ref. [12]. The anatomical regions of interest (ROI) included all regions of the bilateral thalami that could be obtained using FreeSurfer version 7.1.1 with the Iglesias atlas.
26
Briefly, individual thalamic nuclei are automatically segmented using the segmentThalamicNuclei.sh function, which divides the thalamus ROIs based on a probabilistic atlas built using ex-vivo brain MRI and histological data.
26
The atlas showed excellent test-retest reliability and robustness to changes in input MRI contrast when applied to segmentation of in vivo scans and using Bayesian inference.
26
For the RN, these included the lateral nuclear group, namely, the ventral anterior (VA), ventral anterior magnocellular (VAmc), ventral lateral anterior (VLa), ventral lateral posterior (VLp), ventral posterolateral (VPL), laterodorsal (LD), lateral posterior (LP), lateral geniculate (LGN), medial geniculate (MGN), limitans suprageniculate (L-SG), pulvinar anterior (PuA), pulvinar medial (PuM), pulvinar lateral (PuL), and pulvinar inferior (PuI) nuclei. For the medial group, these were the mediodorsal lateral parvocellular (MDl), mediodorsal medial magnocellular (MDm), and the ventromedial (VM) nuclei. The anterior group consisted of the anteroventral (AV) nucleus. The midline group consisted of the paratenial (Pt) and medial ventral reuniens (MV-re) nuclei. The reticular nucleus could not be obtained using FreeSurfer version 7.1.1. For the IN, these included the rostral nuclei, namely, the central medial (CeM), paracentral (Pc), and central lateral (CL) nuclei. For the caudal group, these were the CM and Pf nuclei. In addition, the volume of the whole left and right thalamus (WT) and the TBV were obtained using FreeSurfer version 7.1.1. The segmented output was displayed with the respective label map overlay and images were visually inspected for quality of regional segmentation results (Figure 1). FreeSurfer results were excluded from further analysis if there was a failure to align parcellations and segmentations to the T1-weighted MRI sequence. The whole thalamus segmentation was assessed with respect to its anatomical borders against the surrounding white matter. Thalamic nuclei segmentations could not be inspected, since they are not visually discernible. Selected thalamic nuclei as per FreeSurfer atlas. Sagittal (a) and axial (c) reformats of T1-weighted sequence show thalamic nuclei. Three-dimensional (3D) reformat of thalamic nuclei in coronal perspective (b), smoothing factor 0.5. Thalamic nuclei: ventral anterior (1), ventral lateral anterior (2), ventral lateral posterior (3), anteroventral (4), mediodorsal medial magnocellular (5), ventral posterolateral (6), pulvinar medial (7), lateral geniculate (8), medial geniculate (9), pulvinar anterior (10), pulvinar lateral (11), and mediodorsal lateral parvocellular (12).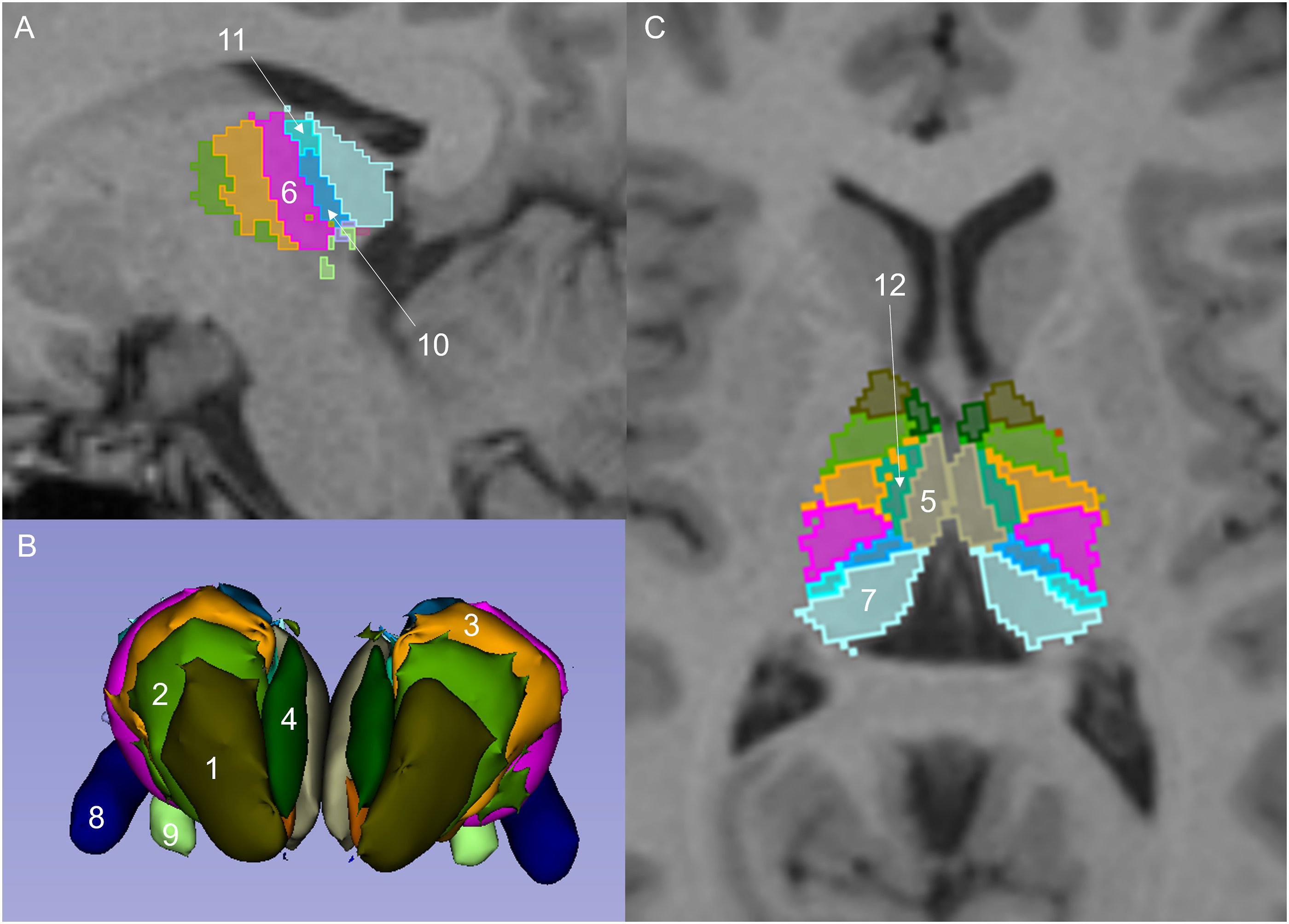
Statistical analysis
Statistical analyses were performed using Matlab (Mathworks, Nattick, MA). Two-sided t-tests were used to assess differences in age between patients and controls. Chi-square test was used to assess for differences in sex between patients and controls. The volumes of the thalamic nuclei and whole thalami volumes were compared between patients and controls using students t-test. All tests were two-sided and observed differences of thalamic nuclei and WT volumes were considered statistically significant if the adjusted p-value was <0.05 after correction for multiple hypothesis testing using the Holm-Bonferroni method. 27
Results
Study population
Demographic and clinical characteristics for the sample included in the analysis.
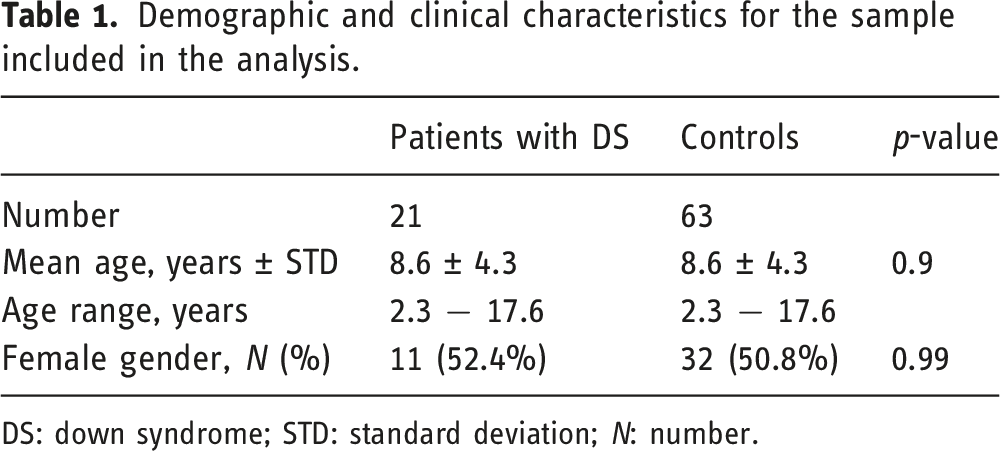
DS: down syndrome; STD: standard deviation; N: number.
Volumetric results
Mean volume (ml) of thalamic nuclei and regions of interest.
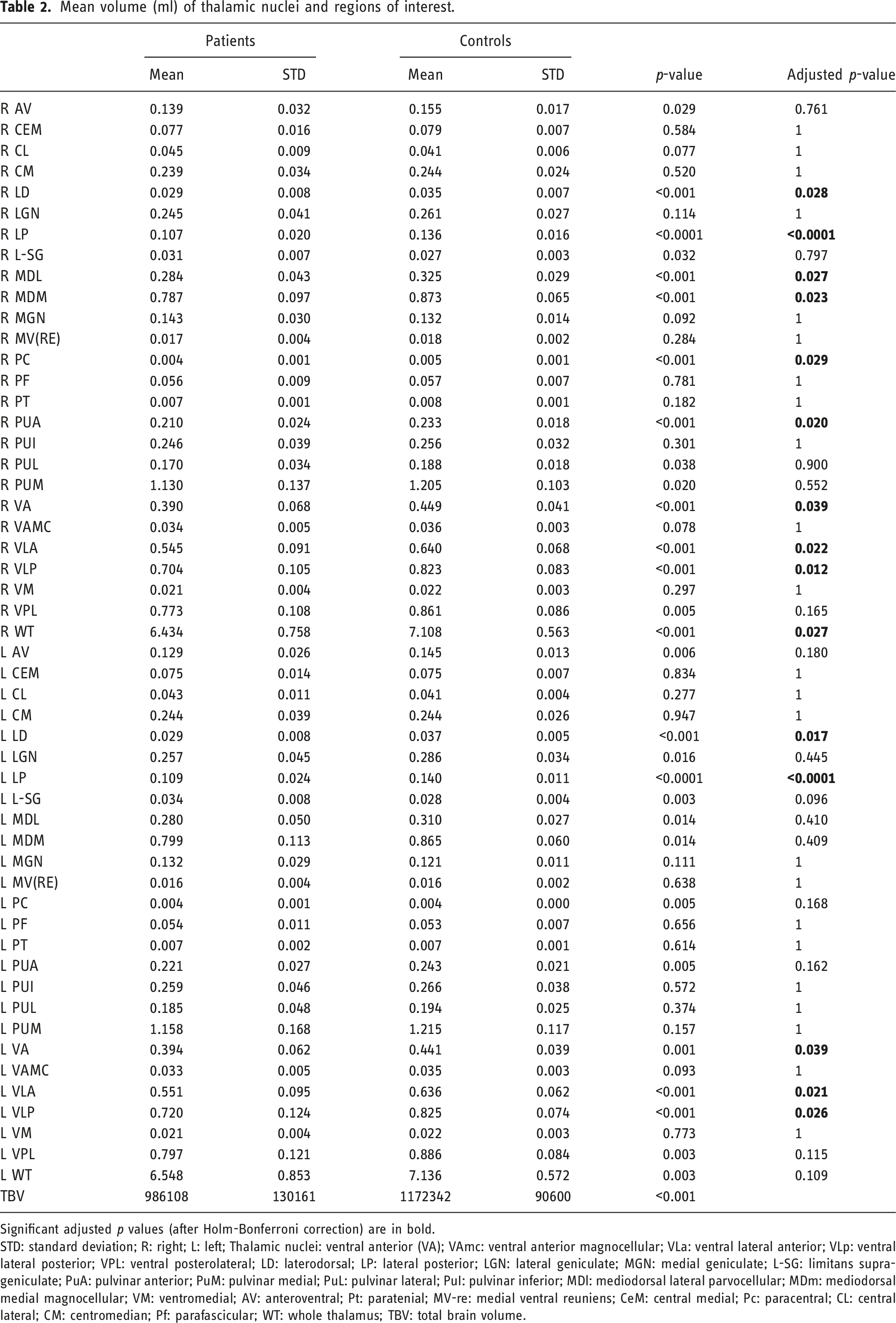
Significant adjusted p values (after Holm-Bonferroni correction) are in bold.
STD: standard deviation; R: right; L: left; Thalamic nuclei: ventral anterior (VA); VAmc: ventral anterior magnocellular; VLa: ventral lateral anterior; VLp: ventral lateral posterior; VPL: ventral posterolateral; LD: laterodorsal; LP: lateral posterior; LGN: lateral geniculate; MGN: medial geniculate; L-SG: limitans suprageniculate; PuA: pulvinar anterior; PuM: pulvinar medial; PuL: pulvinar lateral; PuI: pulvinar inferior; MDl: mediodorsal lateral parvocellular; MDm: mediodorsal medial magnocellular; VM: ventromedial; AV: anteroventral; Pt: paratenial; MV-re: medial ventral reuniens; CeM: central medial; Pc: paracentral; CL: central lateral; CM: centromedian; Pf: parafascicular; WT: whole thalamus; TBV: total brain volume.
RN-lateral group
The lateral group consists of the VA, VAmc, VLa, VLp, VPL, LD, LP, LGN, MGN, L-Sg, PuA, PuM, PuL, and PuI nuclei. Significant volumetric differences between patients and controls were found for the bilateral VA, VLa, VLp, LD, LP, and right PuA nuclei (Table 2). Mean right VA volume for patients was 0.39 ± 0.068 ml and for controls 0.449 ± 0.041 ml, adjusted p < 0.04. Mean left VA volume for patients was 0.394 ± 0.062 ml and for controls 0.441 ± 0.039 ml, adjusted p < 0.04. Mean right VLa volume for patients was 0.545 ± 0.091 ml and for controls 0.64 ± 0.068 ml, adjusted p = 0.02. Mean left VLa volume for patients was 0.55 ± 0.095 ml and for controls 0.636 ± 0.062 ml, adjusted p = 0.02. Mean right VLp volume for patients was 0.704 ± 0.105 ml and for controls 0.823 ± 0.083 ml, adjusted p = 0.01. Mean left VLp volume for patients was 0.72 ± 0.125 ml and for controls 0.825 ± 0.073 ml, adjusted p < 0.03. Mean right LD volume for patients was 0.029 ± 0.008 ml and for controls 0.035 ± 0.007 ml, adjusted p < 0.03. Mean left LD volume for patients was 0.029 ± 0.008 ml and for controls 0.037 ± 0.005 ml, adjusted p < 0.02. Mean right LP volume for patients was 0.107 ± 0.02 ml and for controls 0.136 ± 0.016 ml, adjusted p < 0.0001. Mean left LP volume for patients was 0.109 ± 0.024 ml and for controls 0.14 ± 0.011 ml, adjusted p < 0.0001. Mean right PuA volume for patients was 0.21 ± 0.024 ml and for controls 0.233 ± 0.018 ml, adjusted p = 0.02.
RN-medial group
The medial group consists of the MDl, MDm, and VM nuclei. Significant volumetric differences between patients and controls were found for the right MDl and MDm nuclei. Mean right MDl volume for patients was 0.284 ± 0.043 ml and for controls 0.325 ± 0.029 ml, adjusted p < 0.03. Mean right MDm volume for patients was 0.787 ± 0.097 ml and for controls 0.873 ± 0.065 ml, adjusted p = 0.02.
RN-anterior group
No significant differences were found for the AV nucleus.
RN-midline group
The midline group consists of the Pt and MV-re nuclei. No significant differences were found for these two nuclei.
IN-rostral group
The rostral group consists of the CeM, Pc, and CL nuclei. Significant volumetric differences between patients and controls were found for the right Pc nucleus. Mean right Pc volume for patients was 0.004 ± 0.001 ml and for controls 0.005 ± 0.001 ml, adjusted p < 0.03.
IN-caudal group
The caudal group consists of the CM and Pf nuclei. No significant differences were found for these two nuclei.
Discussion
In this retrospective case–control study, we observed significant volumetric differences of several thalamic nuclei between children with DS and age- and sex-matched controls. Our results confirm prior neuroimaging2,7 and histological13,14 studies, while extending the range of involved thalamic nuclei in pediatric DS to include the medial, intralaminar, and lateral group.
The thalamus is a key structure of the cortico-striato-thalamo-cortical circuits. 13 It also serves as the gateway of sensory input from the brainstem and sensory areas to the cortex and other subcortical regions 14 and plays a crucial role in attention and arousal. 13 Thalamic nuclei are divided into RN (lateral, medial, anterior, midline groups), IN (rostral, caudal nuclei), and the reticular nucleus. 12 They are fundamental for higher-order control processes including problem-solving, inhibition, and planning. 13 In the medial group, the MD nucleus is connected to the prefrontal cortex, amygdala, hippocampal region, and basal ganglia. 13 It has more neurons at birth than in the adult brain and connections are believed to be adjusted by retrograde degeneration to match the number of prefrontal cells.34,35 Stagni et al. and Karlsen et al. have shown that the right MD nucleus has a reduced cell density in fetuses with DS and a reduced number of neurons in adults with DS, respectively.13,14 Similar to their findings, we observed a significantly lower volume of the MDl and MDm nuclei on the right in our DS cohort. Additionally, Stagni et al. found a reduced cellularity of the right CM and Pf nuclei in the IN group. 13 The CM nucleus projects to the basal ganglia and sends diffuse excitatory projections to the cerebral cortex. 13 It plays a role in the processing of verbal information, and integrates activity conveying both interoceptive and exteroceptive information to cortical systems involved in reward and cognition.13,36 The Pf nucleus influences behavioral flexibility and is involved in object recognition tasks.37,38 Although both nuclei were smaller in DS in our study cohort, the differences did not yield statistical significance.
In the lateral group, we found significantly decreased volumes in patients VA, VLa, and VLp nuclei. These nuclei receive input from structures associated with motor functions. Projections arise from the deep cerebellar nuclei and basal ganglia including fibers from the dentate nucleus of the contralateral cerebellum.39,40 The VL and VA nuclei also receive inputs from the medial pallidal segment and substantia nigra and project to motor regions of the cortex including the primary motor and premotor cortices and the supplementary and premotor areas, respectively.40,41 Given the lower volume of the basal ganglia, 7 cerebellar gray, and white matter7,11,31,32,42,43 and frontal lobe structures2,29,31,44 in children with DS, it is conceivable that the decreased volume of the VA, VLa, and VLp nuclei is secondary to this.
The LP nucleus receives inputs mainly from the parietal lobe and projects into the superior parietal lobule. These regions are associated with complex sensorimotor integration helping to program the sequences of motor responses involved in complex motor processes. 40 The pulvinar is comprised of several subnuclei that interact with several different cortical and subcortical regions. The PuA (sub-)nucleus is a polysensory section, with connections to parietal and temporal cortices as well as to visual cortices. 45 The pulvinar and LP nuclei determine visual saliency and allocate attentional resources during visual processing by synchronizing the activity across visual cortices.46–48 We found a significantly lower volume of the LP and right PuA nuclei in children with DS. It is known that visual-motor integration is impaired to varying degrees in individuals with DS. 49
The LD nucleus receives visual and somatosensory inputs indirectly via the pretectal nuclei, the superior colliculus, or the ventral geniculate nucleus and projects to limbic cortical areas and to the posterior parietal cortex. 50 As per Bezdudnaya and Keller, the LD is a multisensory nucleus that integrates multimodal information. 50 It is often considered to be involved in spatial learning and memory through its interactions with the hippocampus. 50 The hippocampus is a deep gray matter region known to be critical for learning and memory and is known to be reduced in volume in children with DS.29,31,32 Thus, the significantly smaller LD volume noted in our study may be related to smaller hippocampal volumes. Clinically, it is know that almost all children with DS have cognitive deficits that affect their learning and memory. 1
Certain limitations need to be taken into account when interpreting the results of our study. First, our sample size was relatively limited and a relatively large number of MRI datasets needed to be excluded from analysis. This was related to the exclusion of patients where FreeSurfer failed to provide accurate parcellations or segmentations resulting in a lower-than-expected TBV and where MRI signal abnormalities secondary to hemorrhage and ischemia interfered with adequate FreeSurfer processing. Second, due to the retrospective nature of our study, a variety of MRI scanners and field strengths were used for data acquisition, which reflects clinical practice. Others have investigated the impact of field strength and imaging parameters on FreeSurfer. Heinen et al. 51 compared FreeSurfer volume estimates between 1.5T and 3T scanners and found robust volume estimates between field strength, with differences in total GM volume of only 1%. In addition, the percentage of patients and controls scanned on a 3 Tesla MRI scanner was similar (6 of 21 patients (29%) and 23 of 63 controls (37%)).
In conclusion, we found significant volumetric alterations of thalamic nuclei in pediatric patients with DS compared with age- and sex-matched controls. Our findings confirm prior neuroimaging and histological studies in fetal and adult populations and extend the range of involved thalamic nuclei in pediatric DS from the medial and intralaminar to the lateral group.
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Matthias W Wagner, Nirit Bernhard, Gopolang Mdebele, Logi Vidarsson, and Birgit B Ertl-Wagner. The first draft of the manuscript was written by Matthias W Wagner and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. This study was approved by the research ethics board of The Hospital for Sick Children, Toronto, Canada (REB# 1000078916).
Informed consent
Because of the retrospective nature of the study, informed consent was waived by the local research ethics board.
Data availability
The datasets generated during the current study are not publicly available but are available from the corresponding author on reasonable request.
