Abstract
Intracranial xanthogranulomas (XGs) have been found at various sites, but xanthogranuloma of the sellar region is extremely rare. We report about a case of sellar XG in a 34-year-old female. Magnetic resonance imaging showed a solid-cystic mass located at the sella turcica. The cystic component was hyperintense on the T1-weighted image (WI) and T2WI. The solid component was hyperintense on T1WI and hypointense on T2WI. There was peripheral enhancement after gadolinium administration. The diagnosis of cystic macroadenoma was considered before surgery. Final diagnosis of XG was confirmed by histopathological examination after surgical resection. Gross total resection of the lesion was achieved using the microscope through endoscopic endonasal transsphenoidal approach. The patient had a good outcome and no symptom of diabetes insipidus, hormonal evaluation did not show any alterations compatible with hypopituitarism and prolactin levels were normal XG should receive diagnostic consideration for the sellar mass lesions with cystic components hyperintense on T1WI and T2WI, solid components hyperintense on T1WI and hypointense on T2WI, and CT without evidence of calcifications. It is important to consider the possibility of XG when pertinent, as it facilitates a proper surgical approach strategy.
Introduction
XG of the sellar region is exceedingly rare and has been reported only in a few case reports, with a prevalence of around 0.6% amongst pituitary tumors.1–3
The World Health Organization characterized XG of the sellar region as an independent entity in 2000. 4 Here, we report a case of sellar XG that simulated a macroadenoma. Histopathology showed the typical features of a sellar XG with cholesterol clefts, marked hemosiderin deposits, multinucleated foreign body giant cells around cholesterol clefts, and fibrosis. 5 XGs of the sellar region are rarely diagnosed and should be considered in the differential diagnosis of cases presenting with cholesterol tissue properties in the sellar region.
XG of the sellar region is a chronic inflammatory lesion resulting from secondary hemorrhage, inflammation, infarction, and necrosis of an existing Rathke’s cleft cyst, craniopharyngioma, or pituitary adenoma, and presents a diagnostic challenge due to its difficulty to differentiate from other more common sellar lesions . There are no specific radiological features for an XG of the sellar region. Thus, the final diagnosis is made histologically, showing a granulomatous reaction characterized by cholesterol clefts, hemosiderin deposits, accumulation of multinucleated foreign body giant cells and foamy macrophages, necrotic debris, and fibrous proliferation.6,7
The first-line treatment is surgical resection. The endoscopic endonasal approach is the preferred technique for surgery of the sellar region in most centers, and gross total resection typically provides good prognosis with low relapse rates. Endocrine deficiency is a common finding and often aggravated by surgery. For better postoperative outcomes and reduction of endocrinological morbidity, two surgical strategies are recommended: first, early surgery may improve pituitary function and avoid degenerative changes. Second, conservative microsurgical tumor resection may preserve normal pituitary tissue that is found on the tumor margins. Subtotal resection may also be considered, depending on tumor dimensions and adhesions to the normal gland. 7
The preoperative suspicion of XG might facilitate the surgical approach strategy. Although no radical changes to the approach strategy would come about, since the endoscopic endonasal approach with eventual variations would be the best suited for almost all sellar and suprasellar lesions, independent of the pathology. Nonetheless, when confronted to craniopharyngiomas, that would be the main differential diagnosis, XG are expected to be more circumscribed to the sellar region and less adherent to the hypothalamic structures, what might allow surgeons to program a less invasive endonasal approach, maybe sparing for example, at least initially, extended endonasal skull base bone removal and anti-CSF leak measures such as external lumbar drainage (and reserving these procedures to be performed only in case needed and not at the beginning of the procedure as frequently done for craniopharyngioma endoscopic endonasal resections).
Methods
This study was approved by the ethics review board of the Clinical Hospital of Campinas, University of Campinas. Informed written consent was obtained from the patient for publication of this case report.
Case report and results
We report a 34-year-old female who presented with amenorrhea and headache of 6 months duration. Neurological deficits were not detected. Pituitary hormone assessment was normal, with the exception of elevated prolactin levels (108.94 ng/mL; reference value: 3.3–26.7 ng/ml. Magnetic resonance imaging (MRI) showed a solid-cystic mass located at the sella turcica measuring 2.4 × 1.8 × 1.7 cm. The cystic component was hyperintense on the T1-weighted image (WI) and T2WI due to high protein level (Figure 1(a) and (b)). The solid component was hyperintense on T1WI and hypointense on T2WI (Figure 1(a) and (b)). There was peripheral enhancement after gadolinium administration (Figure 1(c) in the coronal plane, Figure 1(d) in the sagittal plane). The diagnosis of cystic macroadenoma was considered. The patient was submitted to endoscopic endonasal surgery and had the lesion completely removed. Intraoperatively, the cystic part of the lesion was a slightly xanthochromic fluid showing cholesterol crystals, very similar to the characteristics of cystic craniopharyngiomas, with the only difference of being clearer than the typical “motor oil” aspect of most craniopharyngiomas. The postoperative course was uneventful. Three months after surgery, hormonal evaluation did not show any alterations compatible with hypopituitarism and prolactin levels were normal, the patient was asymptomatic. One year after surgery, MRI showed no residual lesion. Histopathological examination showed cholesterol clefts, chronic inflammatory cells (Figure 2), and macrophages containing hemosiderin. The histopathological examination did not show any findings corresponding to a pituitary adenoma. Neither squamous epithelium nor ciliated cuboidal epithelium fragments were observed. These microscopic findings supported a diagnosis of sellar XG.
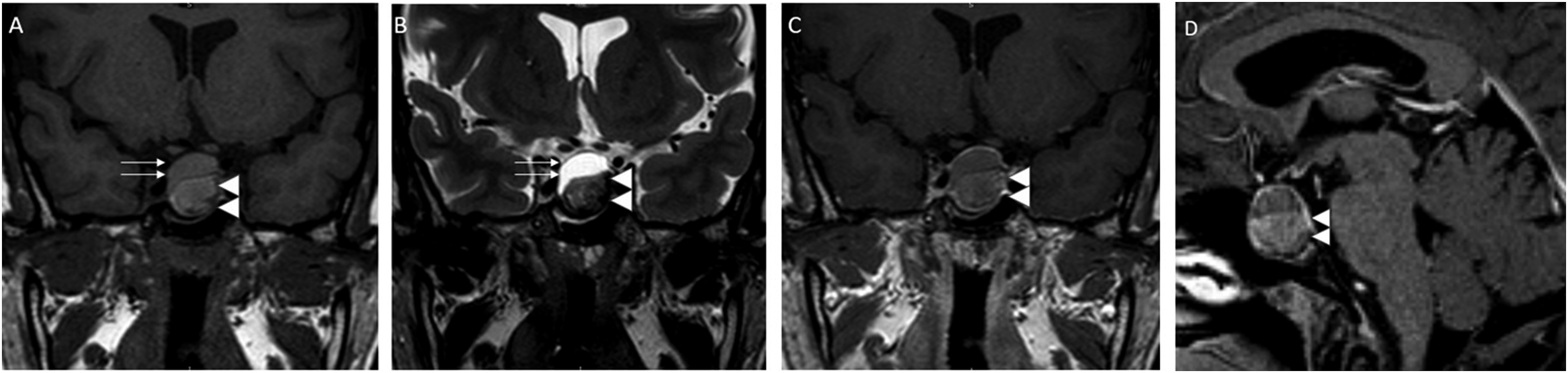
Pituitary MRI revealed a solid-cystic mass located at the sella turcica. The cystic (upper) component was hyperintense on T1WI (a; arrows) and T2WI (b; arrows) due to high protein level. The solid component was hyperintense on T1WI (a; double arrowhead) and hypointense on T2WI (b; double arrowhead). There was peripheral enhancement after gadolinium administration (c and d; double arrowhead). MRI = magnetic resonance imaging, T1WI = T1-weighted image, T2WI = T2-weighted image.
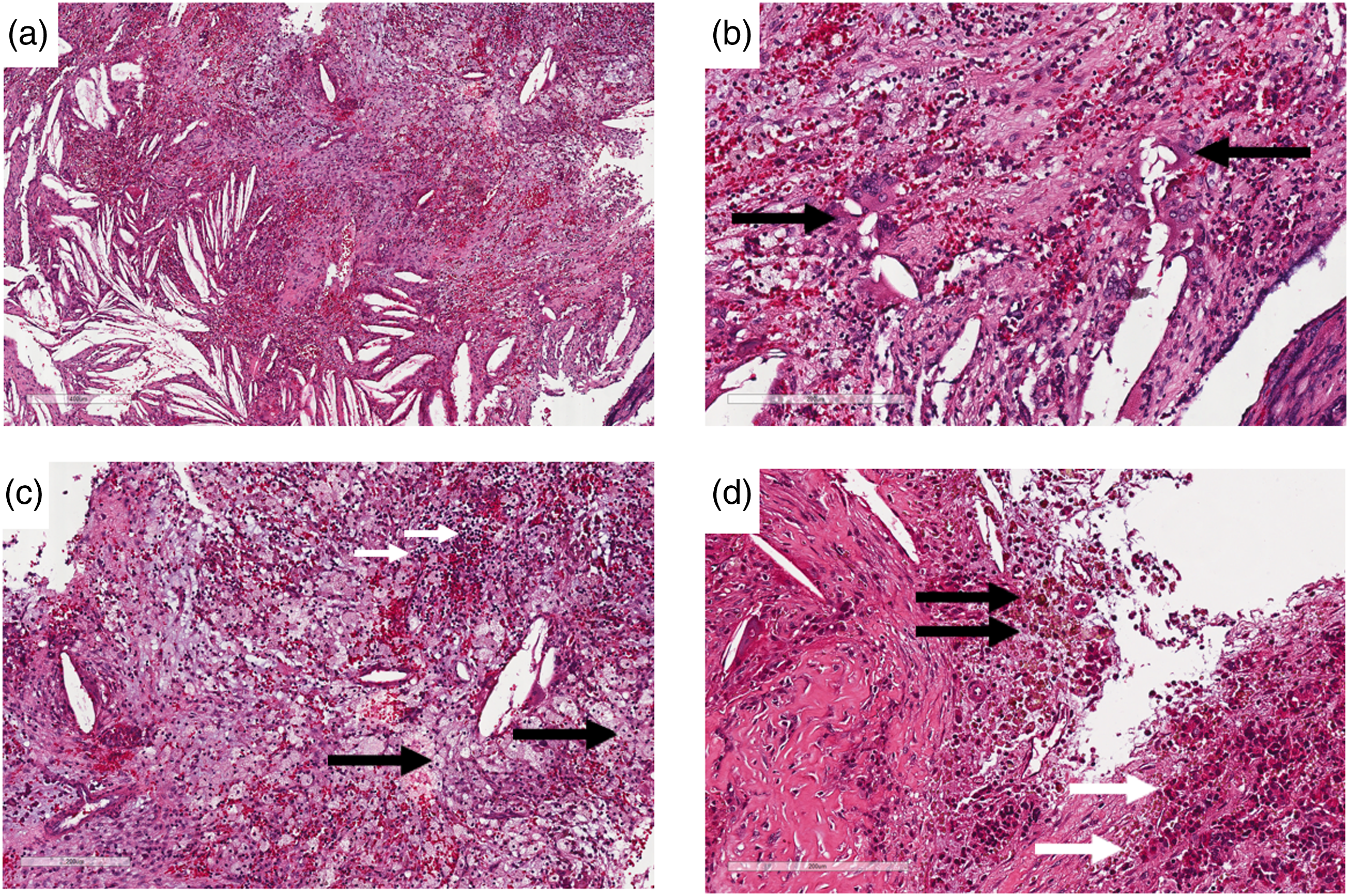
Histopathological evaluation of the xanthogranulomatous tissue shows features compatible with sellar XG. (a) Evidence of chronic inflammation with cholesterol clefts (mainly on the left). (b) Showing multinucleated giant cells with cholesterol clefts (black arrows). (c) Macrophage-rich region (cells indicated by black arrows) intermingled with lymphocytes (white arrows) and recent hemorrhage. (d) Hemosiderophages (hemosiderin-laden macrophages; black arrows) adjacent to a fibrotic region in which normal adenohypophysis cells (mainly eosinophils; white arrows) are observed. No histopathological findings were observed that would suggest distinct sellar region lesions (e.g., pituitary adenoma, craniopharyngioma, or Rathke cleft cyst). Scale bars: 400 μm (a) and 200 μm (b – d).
Discussion
There is no standard MRI finding for sellar XG. It has been shown that cholesterol clefts within these lesions show hyperintensity on T1WI and hypointensity on T2WI;1,8,9 fluid components within the cystic lesions can appear hyperintense on T2WI, and the development of severe fibrosis and hemorrhage can appear with hypointensity on both T1WI and T2WI.8,9 Such mixed findings may be due to severe granulomatous inflammation within XGs. In our case, the upper part of the lesion showed hyperintensity on T1WI and T2WI, which may be due to cholesterol clefts, and the lower part of the lesion showed hyperintensity on T1WI and hypointensity on T2WI, which may be due to hemorrhage and fibrosis. Céspedes et al. reviewed the MRI scans of 33 cases of sellar XG, 30 of which also performed post-contrast T1WI scans, and found that 50% of cases had no contrast enhancement, 30% showed peripheral rim enhancement (as did this case), and 20% appeared with heterogeneous enhancement. 10 Craniopharyngioma often includes calcification within the cystic lesion, and shows heterogeneous enhancement on the post-contrast T1WI, so susceptibility weighted imaging and computed tomography (CT) may demonstrate these calcifications. Considering the heterogeneity of these lesions, CT is very useful for distinguishing between craniopharyngioma and XG. Rathke cleft cysts typically show no contrast enhancement and are homogeneous on both T1WI and T2WI.
Cystic sellar lesions are a potential diagnostic challenge, and the correct diagnosis may be crucial for clinical and surgical management. Conventional MRI can be used for the diagnosis of the most common lesions (Rathke cleft cyst, cystic pituitary adenoma, and craniopharyngioma) using the following radiologic patterns: wall enhancement and the presence of intracystic nodules, fluid–fluid levels, and calcifications. 11 However, a diagnosis of XG may also be considered when the lesion has some of these features.
Conclusion
A diagnosis of XG mostly depends on histopathological examination. XGs should be kept in mind when considering the differential diagnosis of sellar mass lesions with cystic components hyperintense on T1WI and T2WI, solid components hyperintense on T1WI and hypointense on T2WI, and CT without evidence of calcifications. It is important to consider the possibility of XG when pertinent, as it facilitates a proper surgical approach strategy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
