Abstract
Spontaneous epidural (SEH) and subdural hematomas (SSH) of the spine are a rare cause of spinal injury and morbidity. They often present in the emergency setting, though magnetic resonance imaging is the gold-standard for diagnosis. Knowledge of anatomy, and in particular of the dural layers of the spine, is crucial to understand the location of SEH and SSH and their relationship with spinal structure. In this pictorial review, we aim to explain imaging features of the SEH and SSH, and to rule out their main differential diagnosis.
Keywords
Introduction
Spontaneous epidural (SEH) and subdural hematomas (SSH) of the spine may be a cause of acute or progressive paraparesis or tetraparesis with or without sphincter dysfunction in the emergency department, frequently requiring prompt diagnosis and surgical treatment to avoid permanent damage of the spinal cord. 1
SEH and SSH are rare, therefore their exact incidence and prevalence are not available. 2
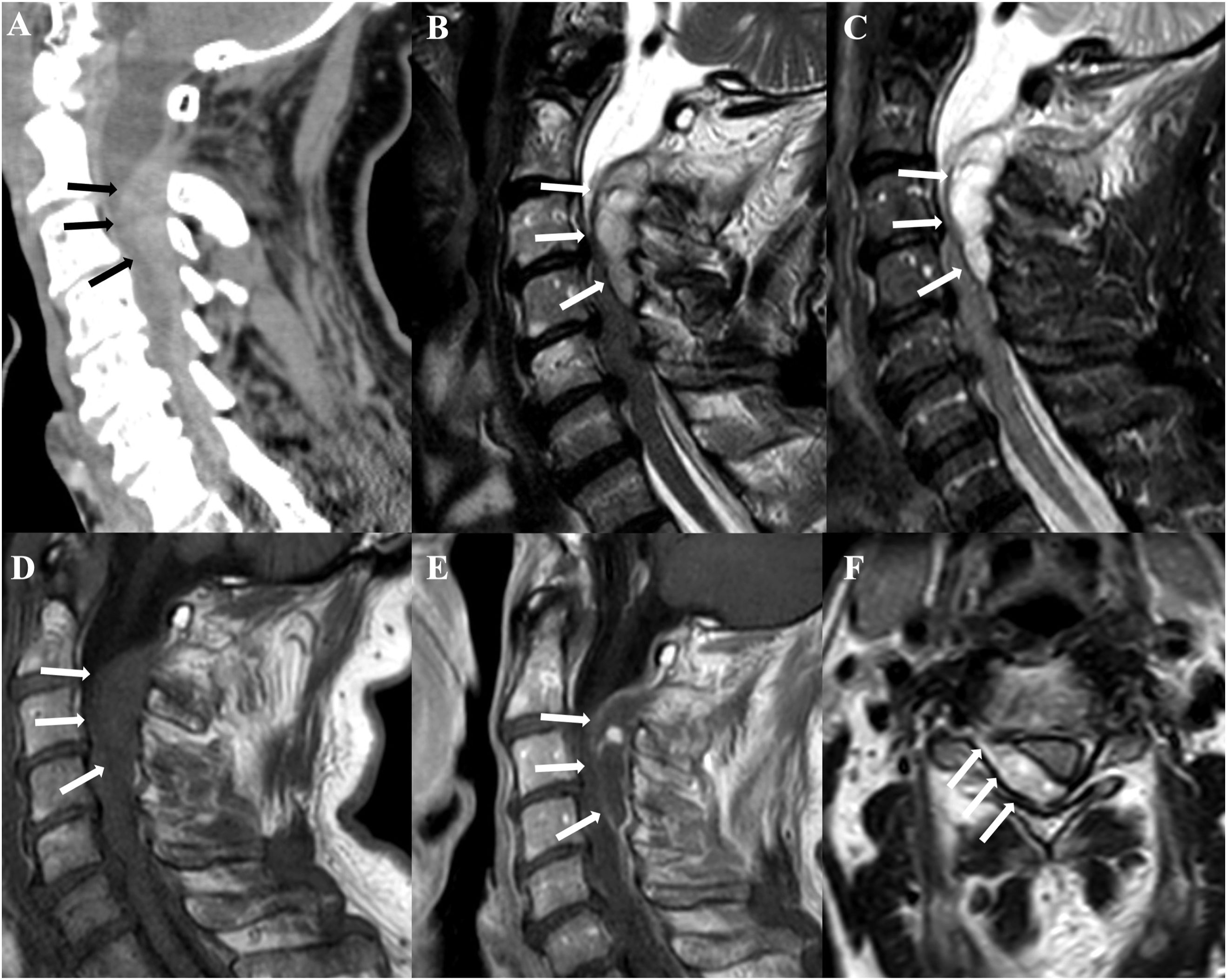
They are often idiopathic or can occur after trivial trauma; other possible causes are major trauma (Figure 1), spinal interventions (e.g. lumbar puncture or anesthetic procedures), and use of anticoagulant drugs or anticoagulant state.
3
Sagittal reformat of volumetric unenhanced CT of the spine (a), sagittal T2 (b), STIR (c) and unenhanced T1 (d), axial T2 FFE (e) and enhanced T1 (f) show a post-traumatic SEH (arrows).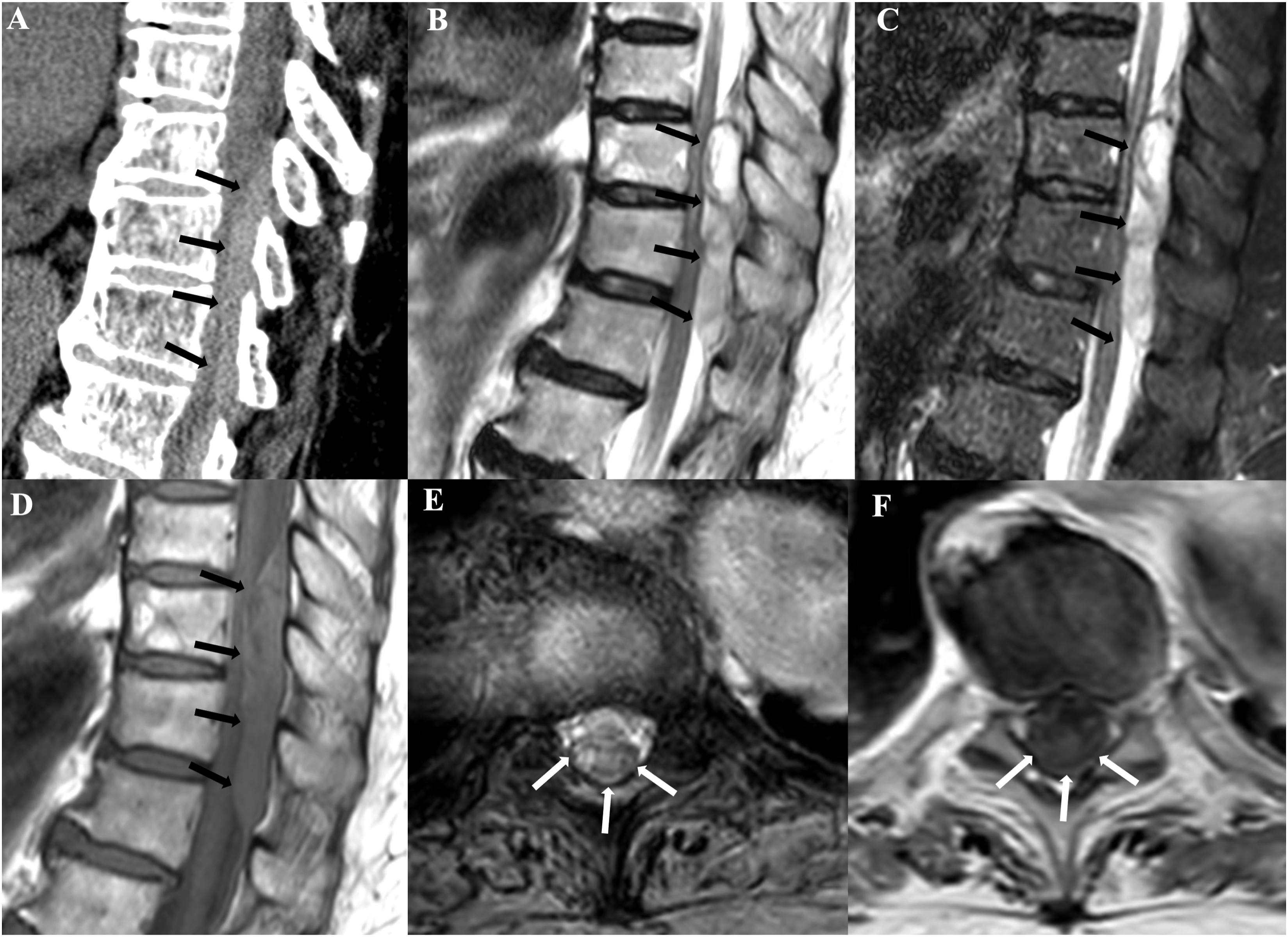
Clinical onset is frequently acute, with sudden onset of back pain, neurological deficits and paralysis, whose intensity and characteristics depend on the location of the hematoma. 4
Magnetic resonance imaging (MRI) of the spine is the imaging modality of choice as it enables correct anatomic localization of the hematoma and its extent. Furthermore, it enables depiction of cord compression and spinal cord edema. MRI is useful to follow the time frame of the bleeding as the signal of the hematoma changes over time, with some degree of variation in the population:
5
• In the hyperacute phase (less than 12 h after onset), blood appears similar to cerebrospinal fluid as it is isointense on T1 and hyperintense in T2-weighted sequences due to the presence of intracellular oxyhemoglobin, which has a diamagnetic magnetization state.
6
• In the acute phase (between 12 h and 3 days), intracellular paramagnetic deoxyhemoglobin begins to form. It is hypointense on T2 and hypo-/isointense on T1-weighted imaging. • In the early subacute phase (between 3 to 7 days), deoxyhemoglobin gradually becomes intracellular methemoglobin. It appears hyperintense on T1 and hypointense on T2-weighted sequences. • In the late subacute phase (between 7 to 30 days), blood cells begin to lyse, and methemoglobin goes outside the cell; this process results in hyperintensity on T2, whereas the signal on T1 does not change. • In the chronic stage (beyond 30 days), ferritin and hemosiderin are present. They are super-paramagnetic, and therefore they cause marked loss of signal on both T1 and T2 sequences.
Susceptibility-weighted sequences may be useful in the diagnosis and timing of SEH and SSH, as they are sensitive to paramagnetic substances. 7
Management of SEH and SSH depends on different factors, including medical history and comorbidities, time of onset, evolution of symptoms, extent of neurological deficits, position and size of hematoma. 8 Mild cases might be treated conservatively as spontaneous resolution is described in these cases. 9 Unfortunately, most of SEH and SSH tend to progress over time, therefore surgical removal of the hematoma is the treatment of choice. In particular, the outcome of surgery mainly depends on the neurological preoperative status and the operative time interval (the latter should be less than 12 h after onset). 10 Correct anatomic localization of blood, and in particular differential diagnosis between SEH and SSH is helpful for the surgeon, as it indicates whether the dura needs to be incised or not to evacuate blood. 1
Anatomy
Exact anatomic localization of spinal hematomas is extremely important for the management and treatment. In the adult age, the spinal cord ends at the level of T12-L1 with the conus medullaris and cauda equina. 11 There are three meningeal layers in the spine, which are the dura mater (the most external one), arachnoid mater and the pia mater (which is adjacent to the spinal cord) and cerebrospinal fluid is present between the second and the latter layers. 12
The epidural space (Figure 2) is located between the dura mater and ligamentous component of the spinal canal and it extends superiorly to the foramen magnum, inferiorly to the sacrococcygeal ligament and laterally into the neuroforamina.
13
It contains fat, connective tissue, internal vertebral venous plexus, small arteries, and spinal nerve roots.
12
It is divided into anterior, lateral and posterior compartments by meningovertebral ligaments, though material within the epidural space can freely move between these recesses.
14
Sagittal T2 depicts the epidural space in red and the subdural space in blue.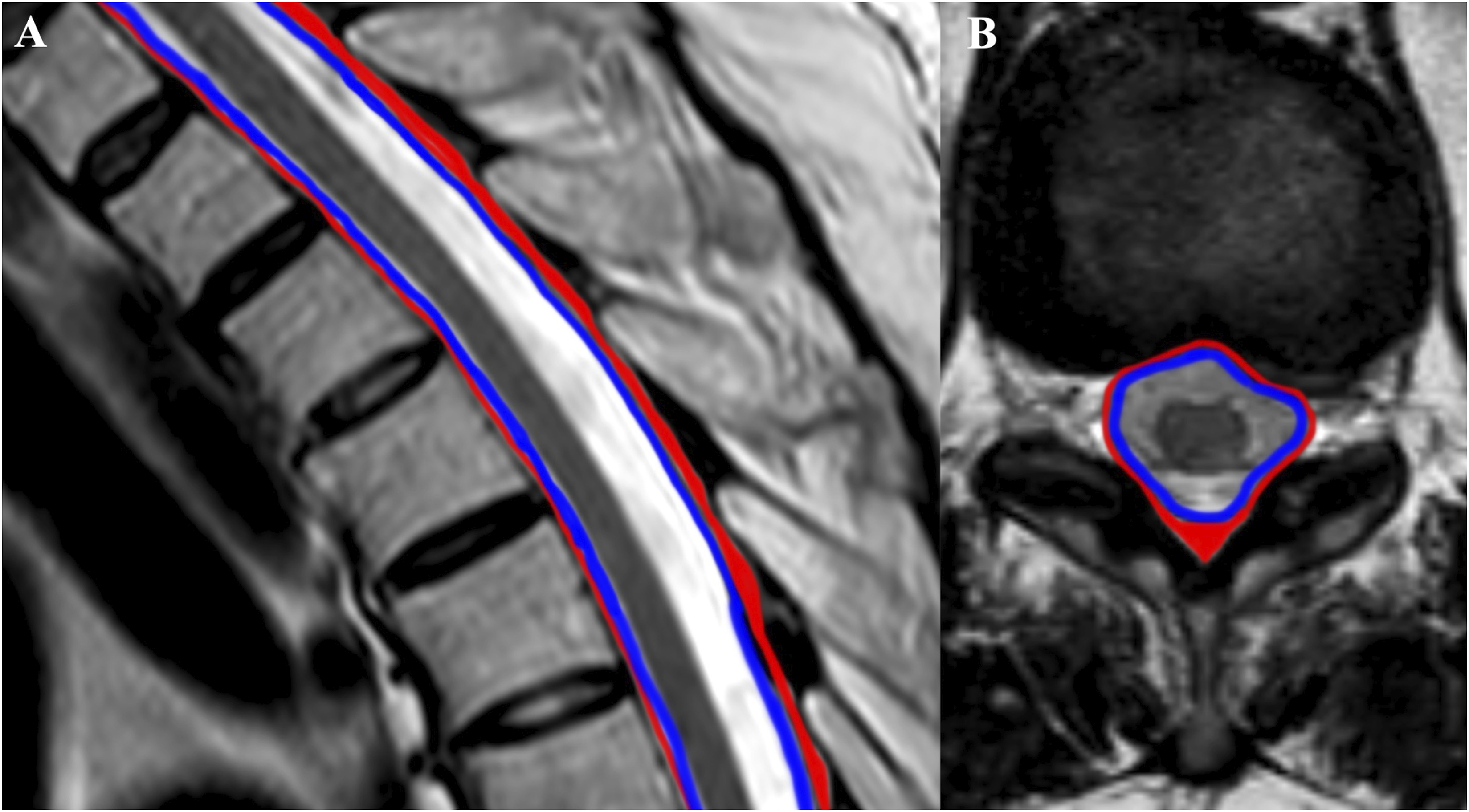
The subdural space (Figure 2) is located between the arachnoid and dura mater but its existence is controversial since there are multiple firm trabeculae between these two meningeal layers.
15
Thus the space between them might be potential in physiological conditions, and it is possible that the subdural space forms as consequence of trauma on the trabeculae.
4
In the subdural space, there are three fibrous elements (two lateral denticulate ligaments and the midline dorsal septum), which may limit the spread of blood, delineating the typical “inverted Mercedes-Benz sign” (Figure 3).
16
The schematic drawing shows the «inverted Mercedes-Benz sign», typical of subdural hematoma. It is determined by the presence of two lateral denticulate ligaments and the midline dorsal septum.
Finally, there is the subarachnoid space, which is immediately under the arachnoid mater, contains cerebrospinal fluid and it is continuous with intracranial subarachnoid space. 4
Spontaneous Epidural Hematoma
SEH is the most common type of spinal hematoma due the rich blood supply of this space, though it is overall a rare condition (incidence of 0.1 case per 100.000 per year).
17
In the majority of cases it is idiopathic or follows trivial trauma.
2
The most frequent locations are the cervicothoracic (Figure 4) and thoraco-lumbar regions.
2
Clinical manifestations depend on the spinal levels compressed and may include abrupt onset of back pain and rapidly progressive neurological deficits, with variable degrees of paralysis.
4
It is not always easy to suspect of SEH on the base of the clinical signs, as it is a very rare entity. Therefore, Computed Tomography (CT) may be performed in the acute setting and, even though it is not the modality of choice, hyperdense material may been in the spinal canal
4
(Figure 5). Sagittal TSE T2 (a), TSE T1 (b), STIR (c) and axial unenhanced TSE T1 show an acute SEH in the cervical region (white arrows). Note also the extension of the blood into the neuroforamina (black arrows). Sagittal reformat of 3D unenhanced CT (a), sagittal T2 (b), STIR (c), T1 (d), enhanced T1 (e) and axial T2 (f) show an acute SEH of the cervical spine (arrows), which causes marked compression of the spinal cord (f).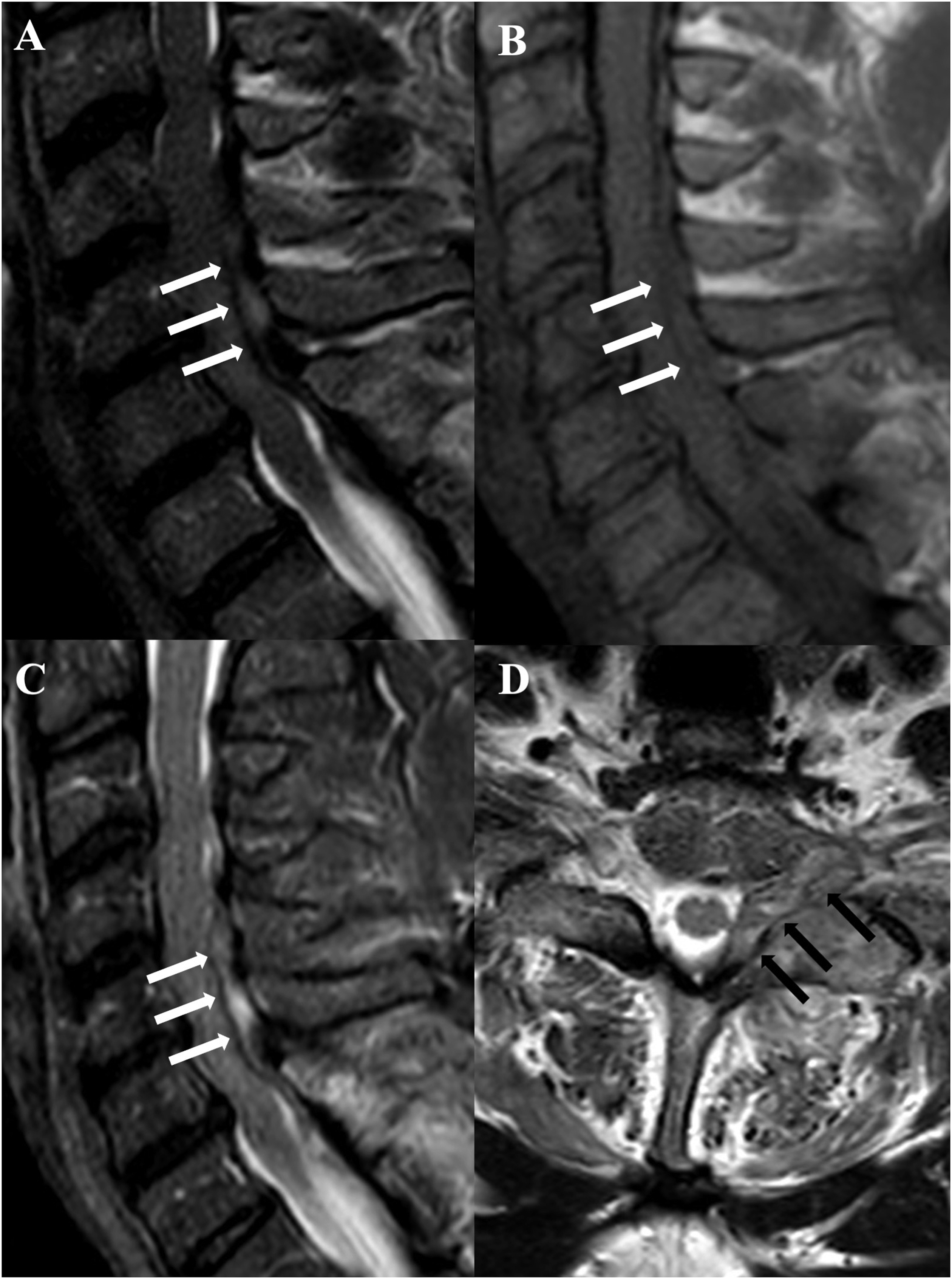
MRI of the spine in the acute setting is crucial to make the correct diagnosis, and to identify the levels of the spinal cord that are compressed. SEH is external to the dura mater; therefore, it causes compression of the thecal sac.
17
SEH may also extend into the neuroforamina (Figure 4).
18
In more than 75% of cases, it is located dorsally in the spinal canal due the more tight adhesion of the dura mater to the posterior longitudinal ligament when compared to that on the ligamenta flava (Figure 5). Nevertheless, may it also occur anteriorly in the spinal canal (Figure 6). The signal of hematoma depends on the timing of bleeding, as described in the introduction. After the injection of intravenous contrast, there is usually peripheral enhancement of the collection, whereas the collection itself does not enhance. Sagittal T2 (a), T1 (b), STIR (c) and axial T2 (d) show an acute SEH in the anterior epidural space (arrows), causing compression of the roots of the cauda equina (d).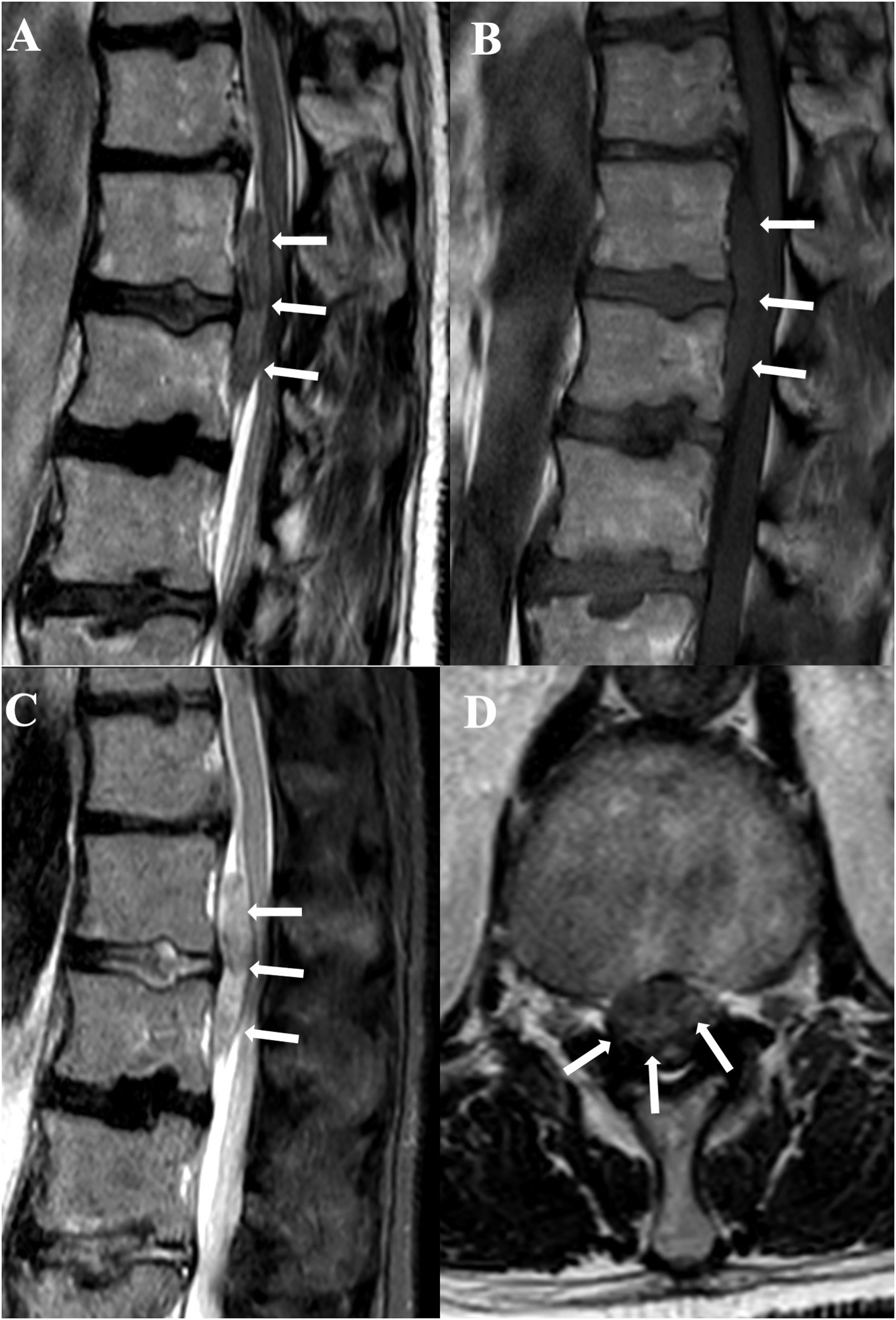
Spontaneous Subdural Hematoma
SSH are rarer than SEH and there are only few cases described in the scientific literature so far. 2 Risk factor are coagulopathy, use of anticoagulation drugs, and spinal percutaneous procedures. 19
Clinical manifestations are similar to those associated with SEH, and they are mainly characterized by acute onset of back pain and spinal cord and/or cauda equina compression. 19
MRI is the modality of choice for the diagnosis and it shows a collection of blood delimitated by the thecal sac, without displacement of the dura or compression of the epidural fat.
6
As described earlier, the presence of the denticulate ligament and the dorsal septum are responsible of the delimitation of the hematoma, resulting in the classic “inverted Mercedes-Benz sign.” The sign may also be incomplete in case of small collections (Figure 7).
4
Sagittal T2 (a), T1 (b), STIR (c), and axial T1 (d) show a subacute SSH (arrows), which determines an incomplete « inverted Mercedes-Benz sign» (arrows in D).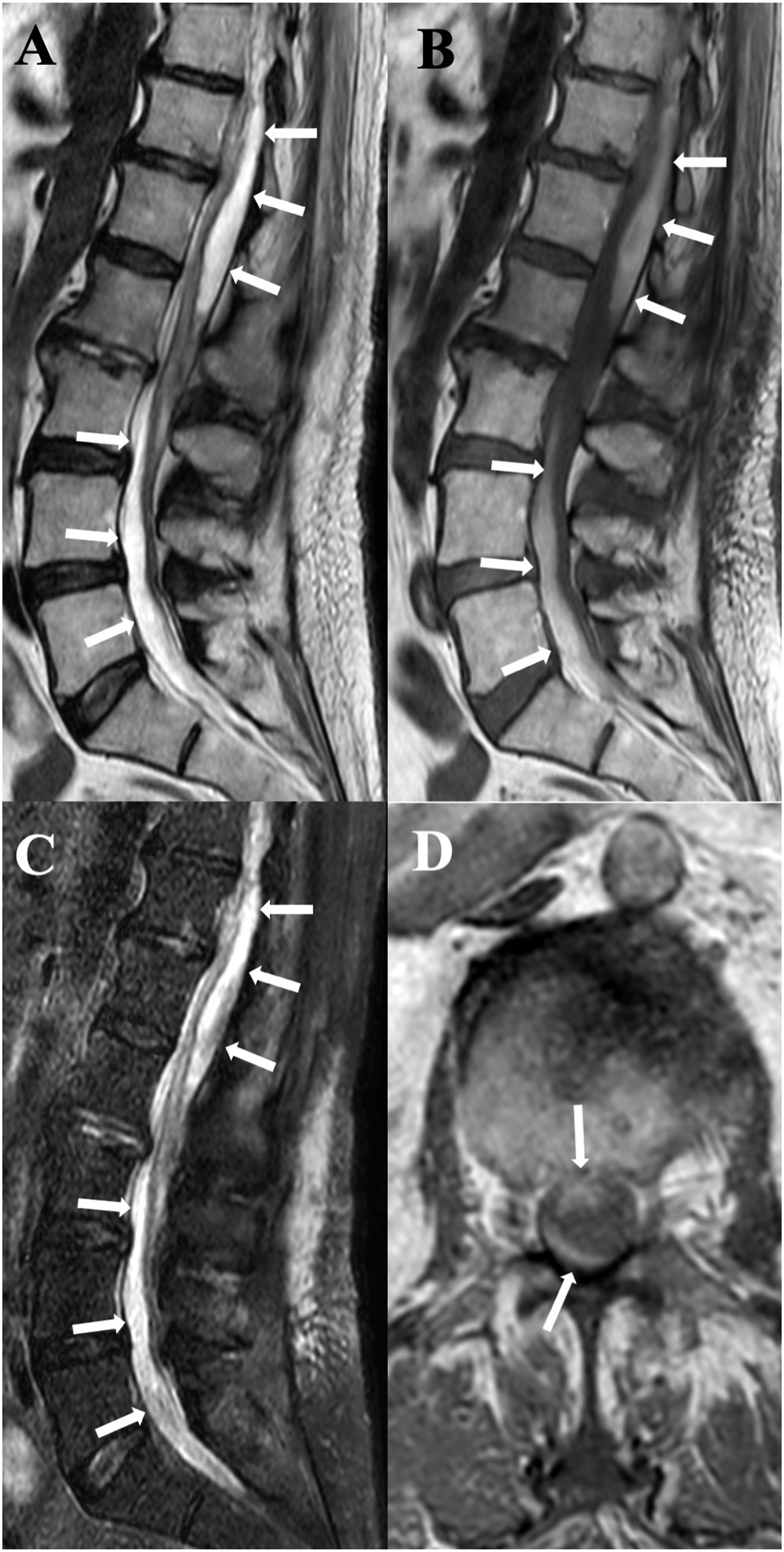
Differential diagnosis
In case of suspected SEH or SSH, there are some major differential diagnosis to rule out.
Metastasis may mimic SEH and SSH, in particular when hemorrhagic. Neoplastic tissue usually show more solid post-contrast enhancement and is accompanied by osseous involvement (Figure 8).
20
Similarly, meningiomas and nerve sheath tumors typically enhance avidly with contrast material, whereas hematoma does not.
4
Sagittal T2 (a), STIR (b), unenhanced T1 (c), enhanced T1 with fat suppression on the sagittal (d) and axial (e) planes show a metastasis from breast cancer, which has both a bony and epidural extension (arrows in a–e).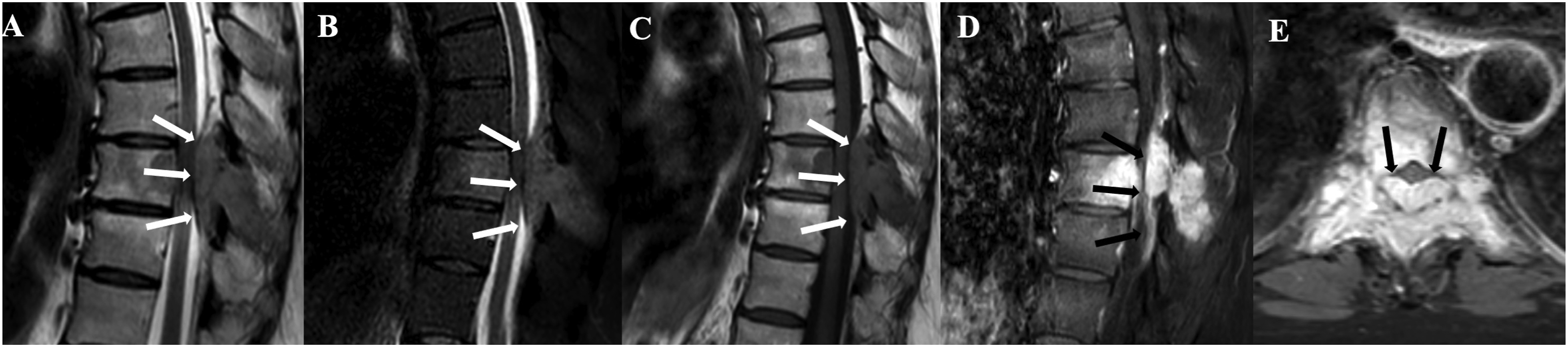
Spinal abscesses appear as fluid collections with heterogeneous signal intensity characteristics, usually hypointense on T1-weighted sequences and hyperintense on T2 images, and they can be epidural or subdural.
21
After contrast-injection, they show contrast enhancement of the solid components (Figure 9) whereas the fluid collection does not enhance.
21
They frequently show in the context of bacterial spondylodiscitis.
22
Subdural abscesses are an extremely rare entity, and they usually differ from SSH as they are hypointense on T1-images, unless they contain blood.
23
Sagittal T2 (a), STIR (b) and enhanced T1 (c) show a bacterial spondylodiscitis of the disco-somatic unit D5-D7. Note the epidural abscess (arrows in a–c), which show intense and homogeneous enhancement.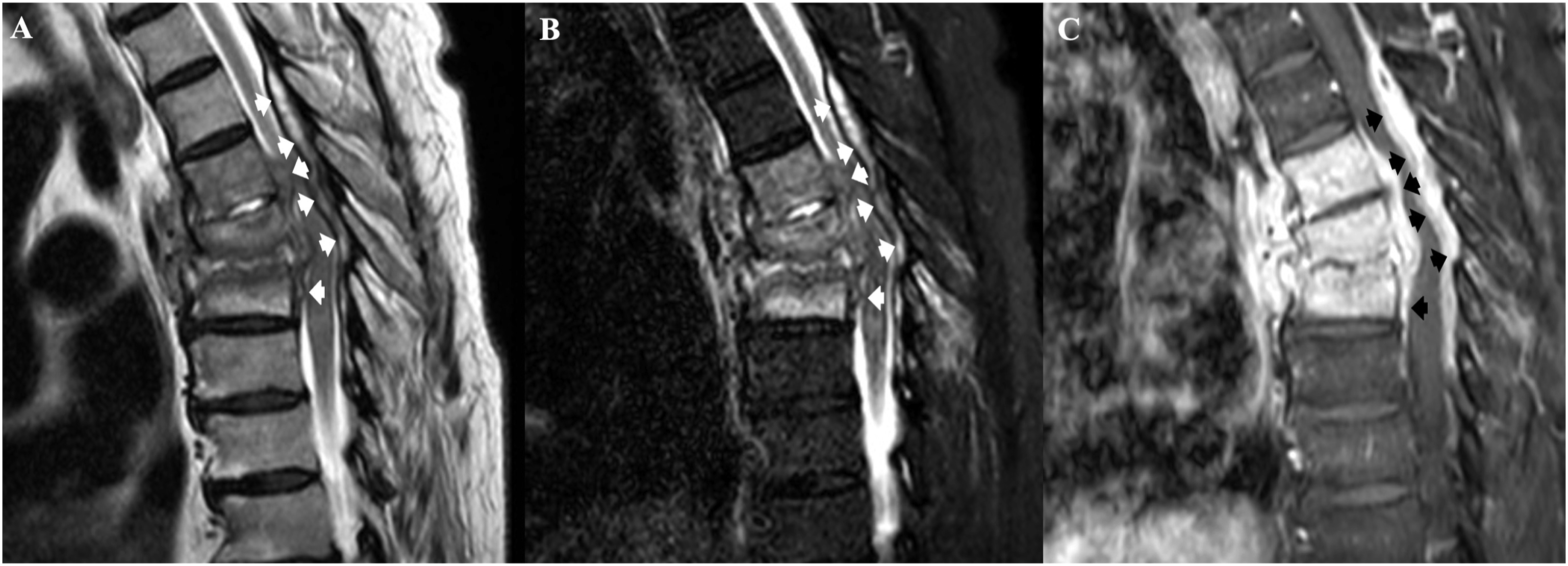
Epidural lipomatosis consists of increased fat content in the posterior epidural (Figure 10) or subdural space and therefore it can be confused with the T1 hyperintensity of SSH.
24
Fat-suppressed sequences on MRI can help differentiate this entity from blood, as the abnormal signal intensity from hemorrhage will not suppress.
4
Sagittal unenhanced T1 (a) and STIR (b) show epidural lipomatosis (arrows in a) with complete suppression of the signal of the fat in STIR (b).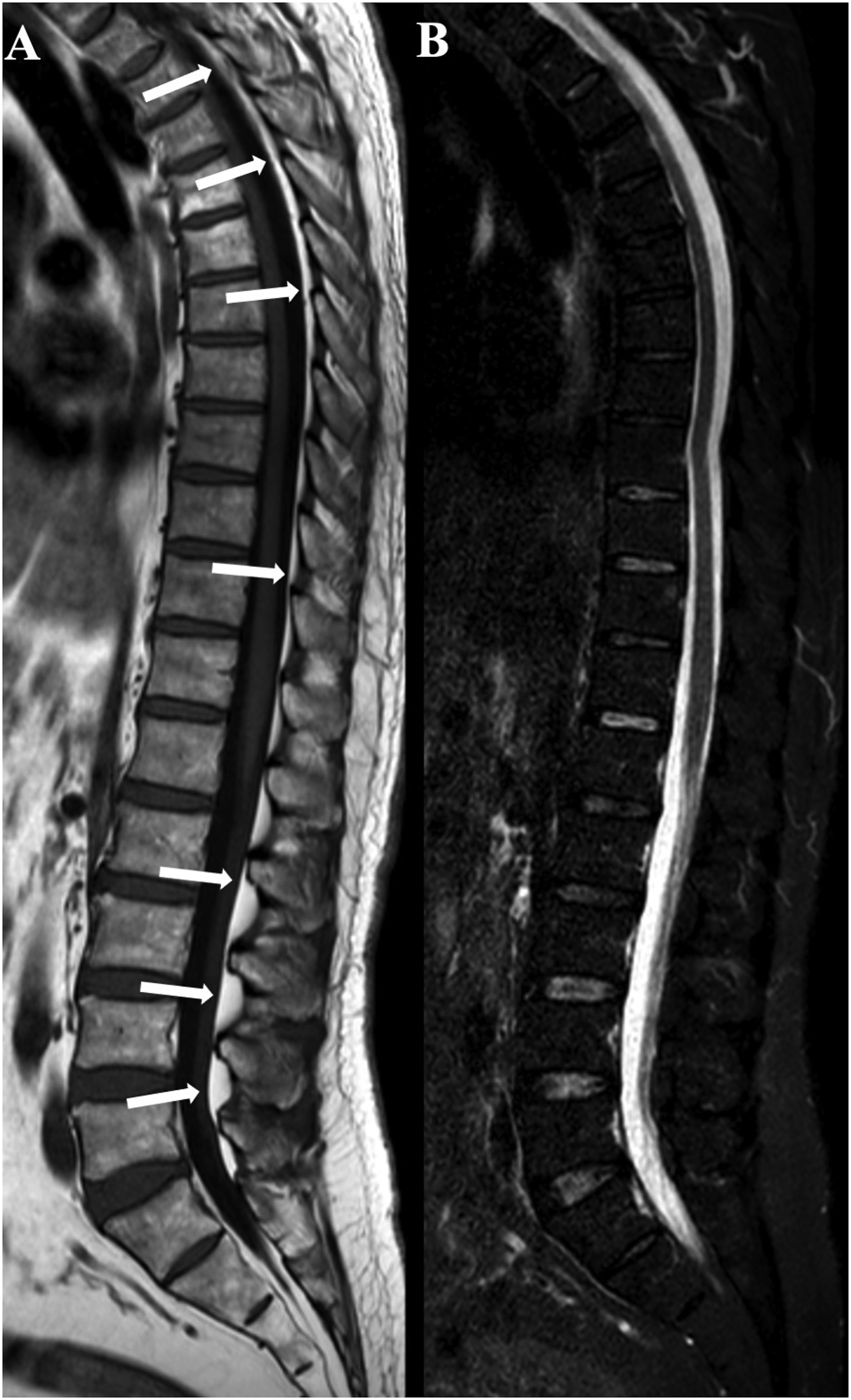
Disc herniations and protrusions usually follow the signal of the disk on MRI and there is usually a connection with the disk itself (Figure 11), except in case of sequestra.
16
Furthermore, there are also other concomitant degenerative alterations of the spine, such as bone hypertrophy or osteophytes, joint space narrowing, joint effusion, or subchondral cysts.
25
Sagittal (a) and axial (b) T2 show an intra-foraminal disc herniation at the L4–L5 level on the right, which extends superiorly (b).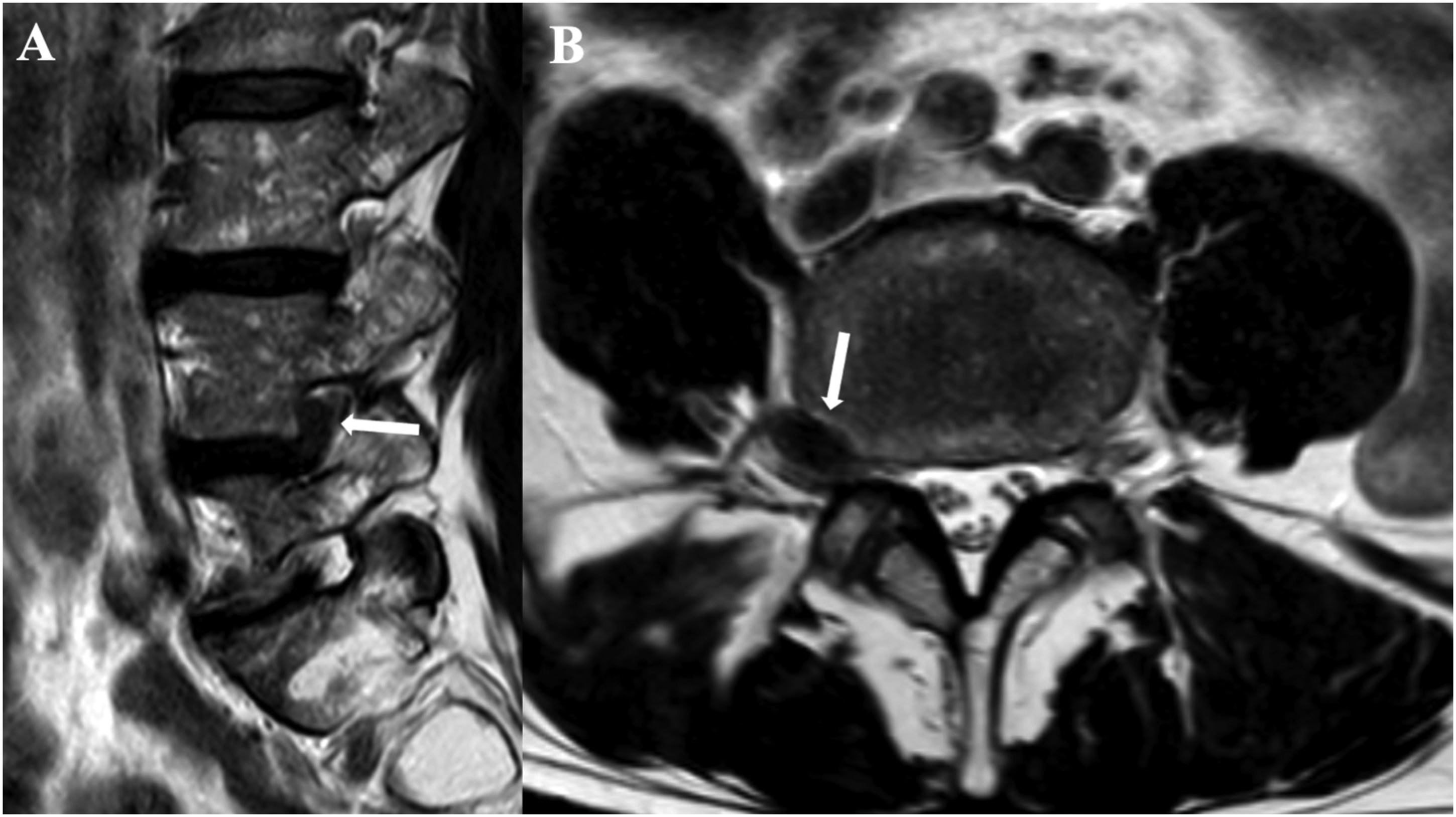
Subdural hygromas are collection of cerebrospinal fluid in the subdural space caused by dural injury, and thus they are usually seen in patients with history of recent trauma or intervention. 26 This collections of cerebrospinal fluid may also contain some blood; therefore, it is crucial to look for other signs of hypotension to make the correct diagnosis. 27
Arachnoid cysts (Figure 12) usually have a homogeneous fluid content which follows the signal of the cerebrospinal fluid and they appear unilocular.
28
Sagittal T2 (a), T1 (b), STIR (c), and axial T2 (d) show an epidural arachnoid cyst (arrows in a–d).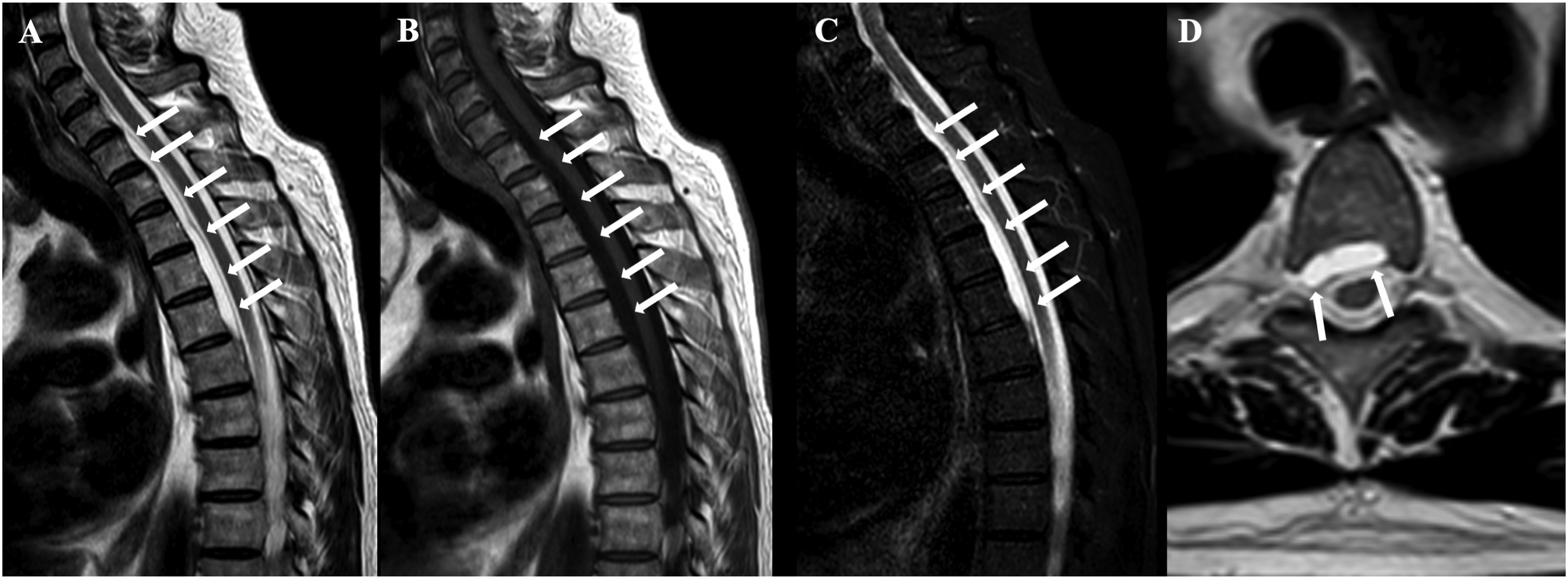
Arachnoiditis may determine central clumping of the nerve roots within the thecal sac, in a configuration that can sometimes mimic the displacement of nerve roots from a subdural hematoma. 29 In cases of arachnoiditis, the cerebrospinal fluid completely surrounds the clumped nerve roots, whereas displacement due to a subdural collection occurs with the inverted Mercedes-Benz sign. 4
Conclusion
SEH and SSH of the spine are a rare cause of acute neurological impairment, which requires rapid diagnosis and treatment especially in case of spinal cord compression.
MRI allows recognition of SEH and SSH, differentiation from other conditions that can cause this type of symptomatology and identification of spinal cord compression and/or edema.
Management depends on the etiology and the clinical manifestation, even though prompt surgical intervention is usually required.
Therefore, even though SEH and SSH are extremely rare conditions, it is important for the neuroradiologist to take them into account in case of acute paraparesis or tetraparesis, because exact diagnosis is crucial for the treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
