Abstract
Objective
Our review aims to summarize the current literature on skull base infections (SBIs) and retrospectively analyze any such cases encountered at our institution.
Design
A literature search was conducted using online databases PubMed, MEDLINE, and ResearchGate with the terms “skull base osteomyelitis,” “temporal bone osteomyelitis,” “skull base infections,” “necrotizing otitis media,” and “SBO”. References from the resulting manuscripts were reviewed for relevant articles. A search of our electronic health records using the same key terms was also performed to identify patients with a tissue biopsy-confirmed diagnosis of skull base infections. Patients with an indeterminate diagnosis or inaccessible/poor imaging were excluded.
Setting
A level one trauma and major tertiary academic medical center.
Participants
All patients treated at the University of California Davis Health System with a confirmed diagnosis of skull base infections from January 2005 to November 2020.
Main Outcome Measures
Imaging results, symptoms, treatment, morbidity, and mortality.
Results
Our literature search yielded 59 articles ranging from 1982 to 2021. A retrospective search of our electronic health records identified two cases of skull base infections.
Conclusion
Skull base infections have no pathognomonic findings. A multimodal approach with computed tomography (CT), magnetic resonance imaging (MRI), and nuclear medicine is necessary to characterize the disease process in addition to a biopsy for definitive diagnosis. Other diagnoses can mimic SBI on imaging, such as nasopharyngeal carcinoma and inflammatory pseudotumor. Culture-guided antimicrobial treatment and surgery are mainstay therapies. Other adjuvant strategies currently lack the robust evidence necessary to characterize their risks and benefits.
Introduction
Skull base infections (SBIs) are clinically significant diagnoses that require a high degree of clinical suspicion as death and neurologic dysfunction may occur. 1 However, diagnostic challenges often delay therapy due to SBI’s non-specific presentation on imaging and laboratory work-up.2–5 If left untreated, local extension of the infection into neighboring vascular and intracranial structures can lead to complications such as empyema, abscesses, cerebritis, and venous sinus thromboses, with neurologic dysfunction or disseminated infection being the ultimate causes of death.1,2,4,6–11 Therefore, efforts to improve morbidity and mortality are placed on acquiring a timely diagnosis before these complications arise.
Imaging studies such as computed tomography (CT), magnetic resonance imaging (MRI), and nuclear medicine are used to evaluate and monitor the location and extent of infection. Bony demineralization, erosions, and soft tissue swelling are often seen on CT. MRI will demonstrate osseous and soft tissue enhancement. Tracer uptake with scintigraphy using gallium-67 positively correlates with disease activity and is useful for monitoring and follow-up during treatment.3,7,10–17 Bone and soft tissue biopsy with immunohistochemistry also helps to differentiate between infection and other diagnostic mimics such as nasopharyngeal carcinoma, inflammatory pseudotumor, lymphoma, osteoradionecrosis, and granulomatous diseases.1,3,4,11,12,18–22
Laboratory findings are non-specific, with an elevated erythrocyte sedimentation rate and C-reactive protein typically being the only abnormalities.4,7,12,14,23,24 Cultures of the bone or nearby soft tissue may identify the causative organism and guide the choice of antimicrobial therapy. SBIs are most commonly bacterial in origin with Pseudomonas aeruginosa and Staphylococcus species being the likely causative organisms.3,7,18,24 Fungal infections are less common, but Aspergillus species are the most likely isolated pathogens in such cases.2,25–28
Diagnostic and treatment recommendations have yet to be rigorously validated due to the lack of randomized control trials and the limited number of cases overall. As such, there is no established consensus on the management of SBI. Studies have recommended different treatment durations ranging from six to twenty weeks of culture-guided antimicrobial therapy with the first six to eight being intravenously administered. 7 The role of surgical management remains controversial regarding improved patient outcomes but is typically utilized early and aggressively for debridement.
Given the difficulty of identifying SBIs in clinical practice and a lack of consensus in its management, the goal of our review is to highlight the constellation of clinical and imaging findings which should alert clinicians to the potential for SBI. A comparison of different imaging findings of SBIs and their mimics will also help to distinguish SBIs from other similar appearing pathologies. We also retrospectively review our experience with two cases of SBI, including initial presentation, clinical progression, imaging findings, and therapeutic management.
Method
An extensive literature review was conducted using online databases PubMed, MEDLINE, and ResearchGate using the terms “skull base osteomyelitis,” “temporal bone osteomyelitis,” “skull base infections,” “necrotizing otitis media,” and “SBO”. References from the resulting manuscripts were cross-checked and reviewed for relevant articles. No date or geographical restrictions were applied for our search. Determination of the level of relevancy of each article was discussed and agreed upon between the authors. Approval was obtained from our Institutional Review Board to conduct a search of our electronic health records. Using the same key terms, we identified any patients who were referred to the UC Davis Health System from January 2005 to November 2020 that had a tissue biopsy-confirmed diagnosis of skull base infections. Patients with an indeterminate diagnosis or inaccessible or poor imaging were excluded from our analysis. Patients who met our inclusion criteria were retrospectively reviewed for their clinical presentations, laboratory and pathologic findings, imaging findings, and therapeutic approaches.
Results
Our literature search yielded 59 relevant articles for review ranging from 1982 to 2021. A retrospective search of our electronic health record yielded two patient cases which met our inclusion and exclusion criteria for further analysis.
Case one
A 74-year-old man with uncontrolled insulin-dependent diabetes, chronic heart failure, and renal failure initially complained of left ear pain with drainage. Over several months, he also developed a left Bell’s palsy and lower cranial neuropathies with dysphagia, dysphonia, and aspiration. Culture of the drainage was performed, which revealed pan-sensitive Pseudomonas aeruginosa. The patient was then treated with one week of ciprofloxacin, two weeks of cefepime, and two weeks of posaconazole. However, these were unsuccessful, and the patient soon began complaining of systemic symptoms such as nausea, fatigue, and 20 kilograms of unintentional weight loss.
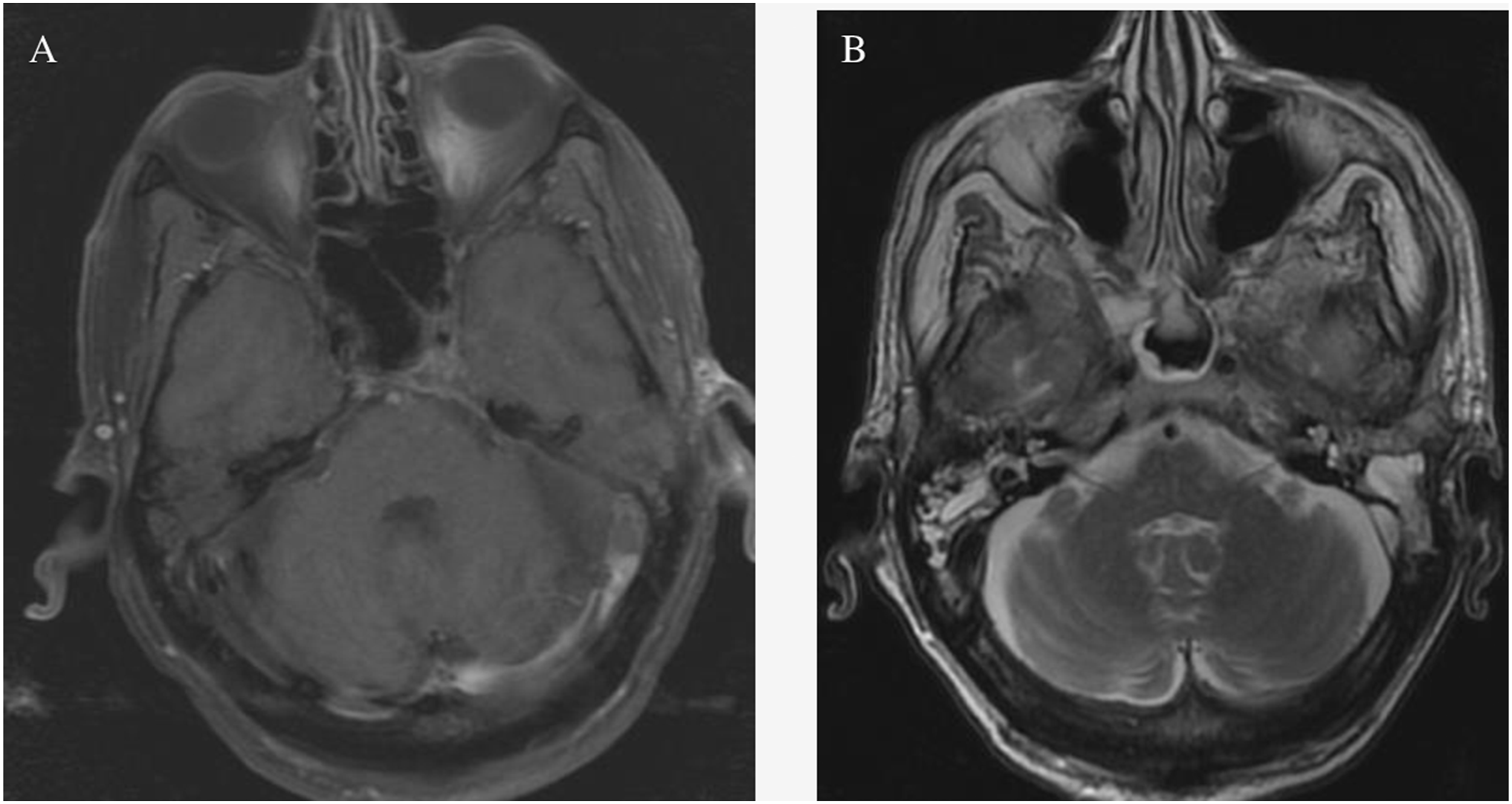
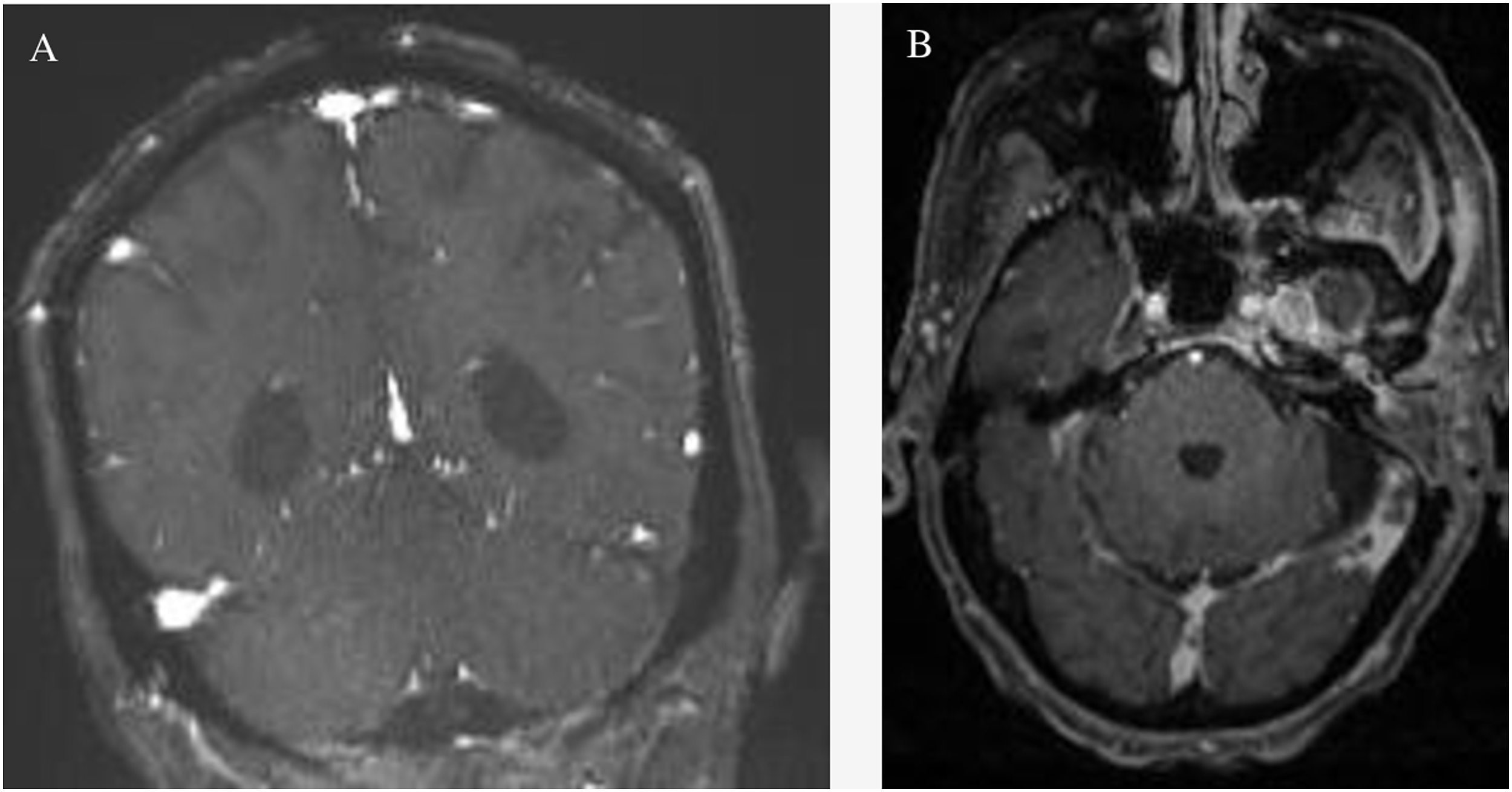
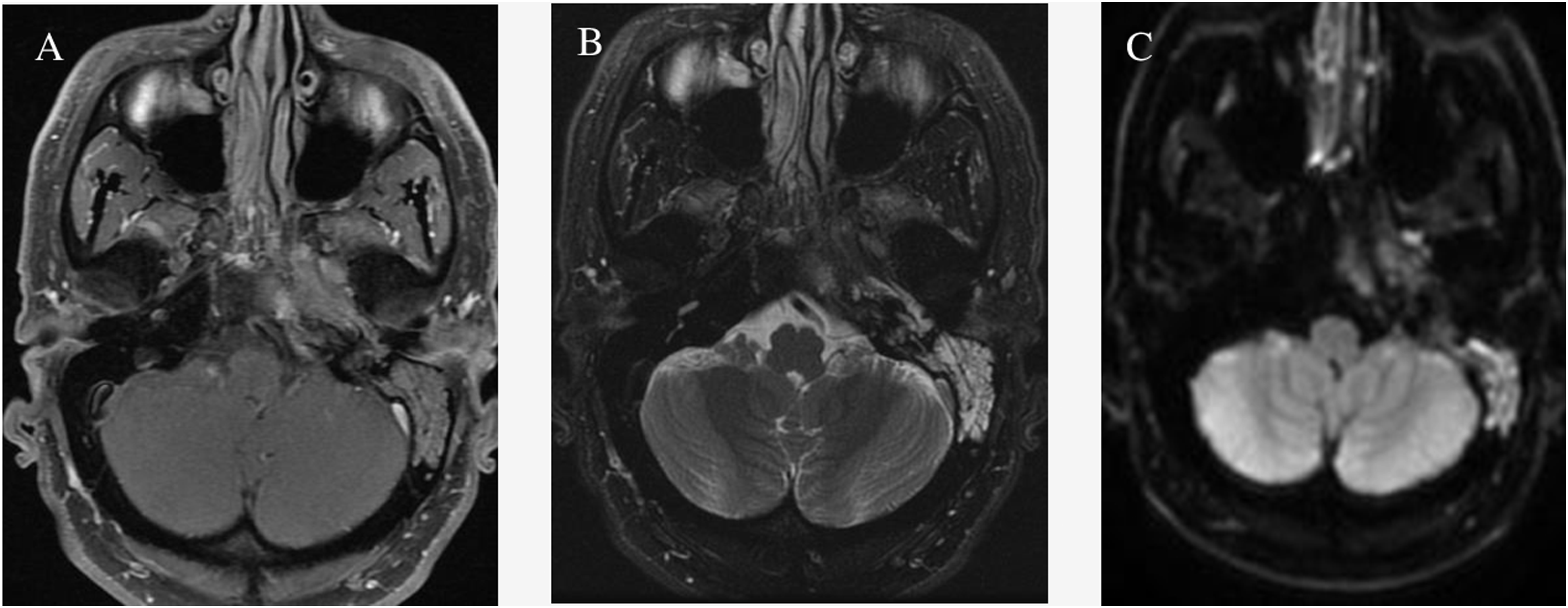
Imaging was ordered due to his worsening clinical status. Axial fluid attenuated inversion recovery (FLAIR) demonstrated subtle T2 hyperintensity in the left petrous apex extending into the nasopharynx. Axial T1 showed low signal and marrow replacement in the left petrous apex and clivus with blurring of the left superior parapharyngeal space. Diffusion-weighted image (DWI) at the same level demonstrated restricted diffusion in the left petrous apex (Figure 1). Additionally, gallium-avid foci were noted in the left skull base in the region of the left petrous apex and clivus extending to the nasopharynx (Figure 2). (a) Axial fluid attenuated inversion recovery demonstrates subtle T2 hyperintensity in the left petrous apex extending into the nasopharynx. (b) Axial T1 demonstrates low signal and marrow replacement in the left petrous apex and clivus with blurring of the left superior parapharyngeal space. (c) Diffusion weighted image at the same level demonstrating restricted diffusion in the left petrous apex. (a) Gallium-avid foci in the left skull base in the region of the left petrous apex and clivus extending to the nasopharynx.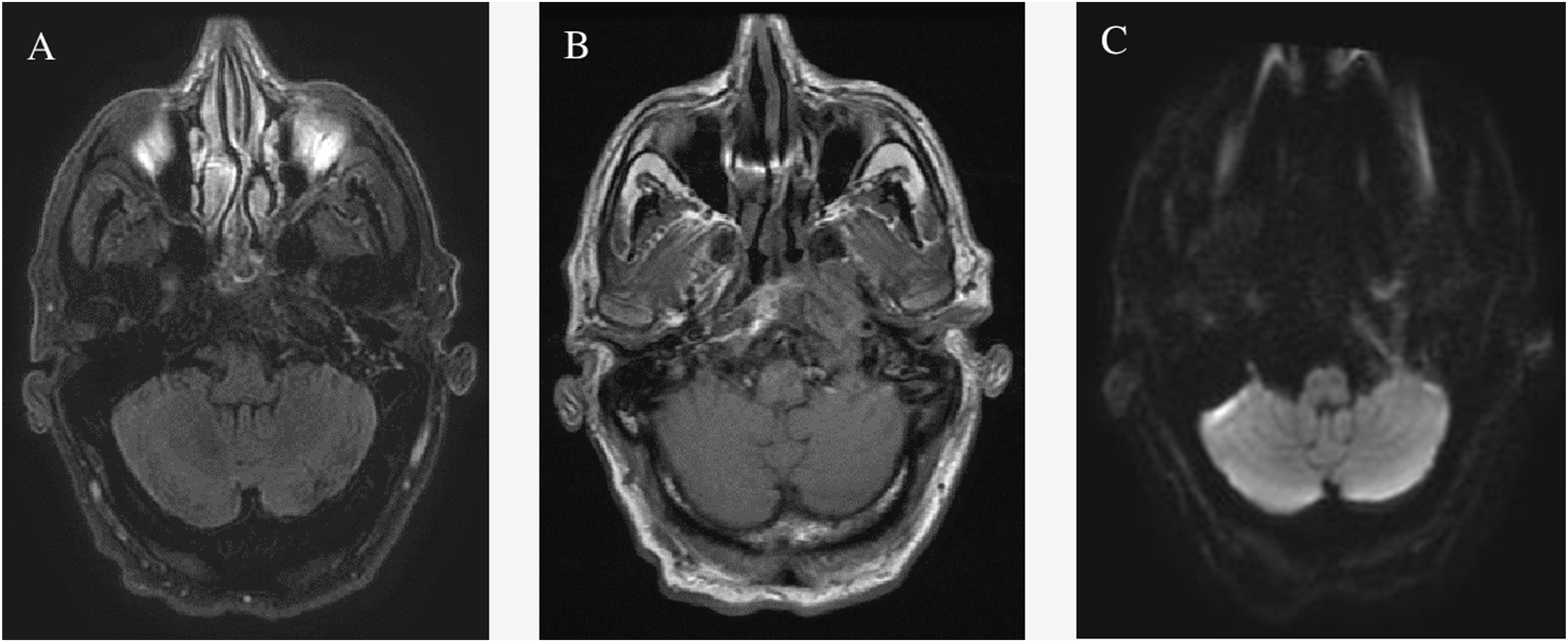
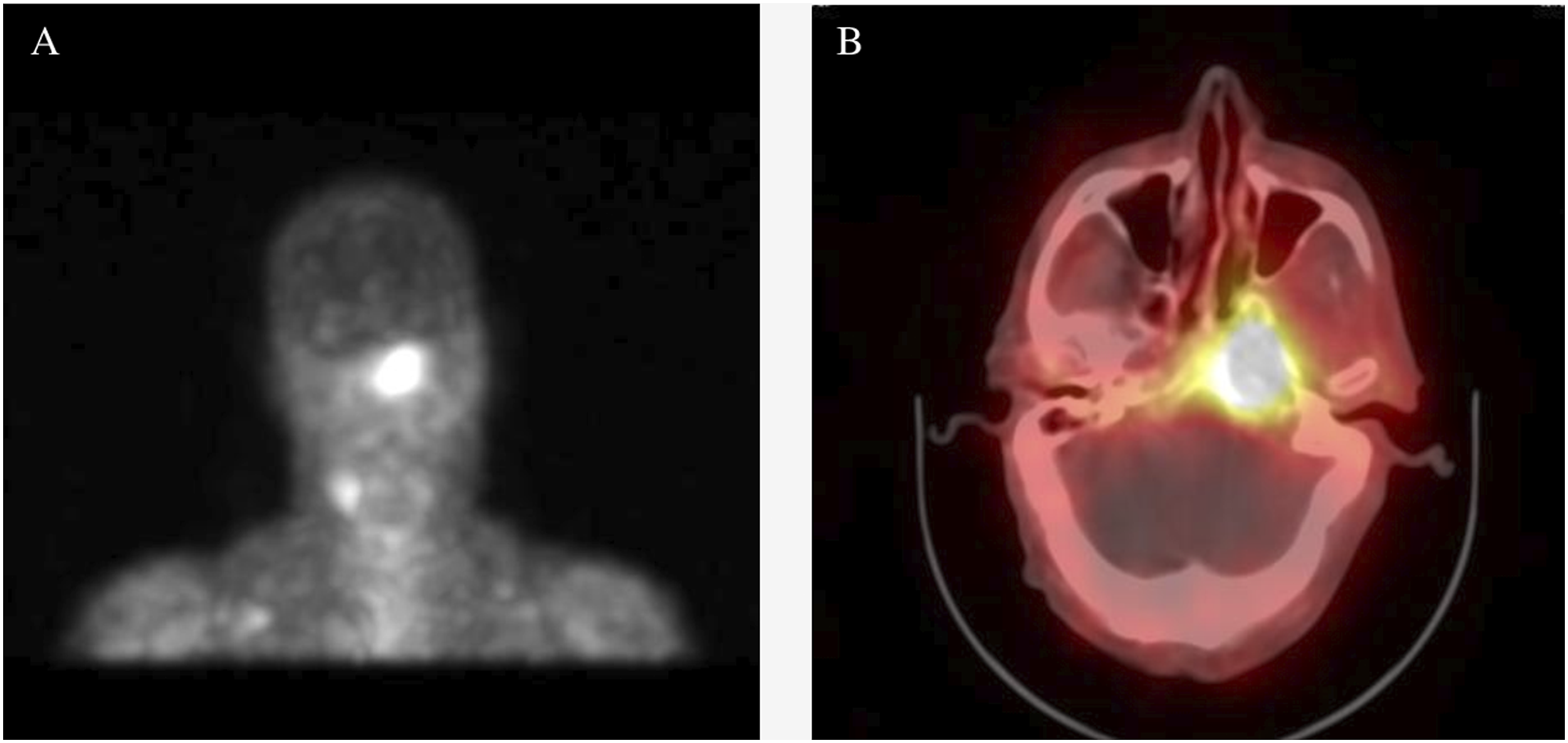
His diagnosis was determined to be chronic and relapsing skull base osteomyelitis based on clinical presentation, culture, and imaging findings. Surgical management was determined to be appropriate, and a left total ethmoidectomy, sphenoidotomy with removal of the sphenoid contents, complete mastoidectomy, and myringotomy with the insertion of a tympanostomy tube were performed in addition to a nasopharyngeal biopsy. The patient was then treated empirically based on common pathogens associated with SBIs—ceftazidime for Pseudomonas and posaconazole for Aspergillus—and he was discharged with 6 to 12 weeks of antibiotic therapy. Upon follow-up with outpatient otolaryngology and infectious disease clinics after treatment, the patient showed no further signs of recurrent osteomyelitis, and his most recent gallium scan revealed no significant soft tissue infection or osseous radiotracer activity.
Case two
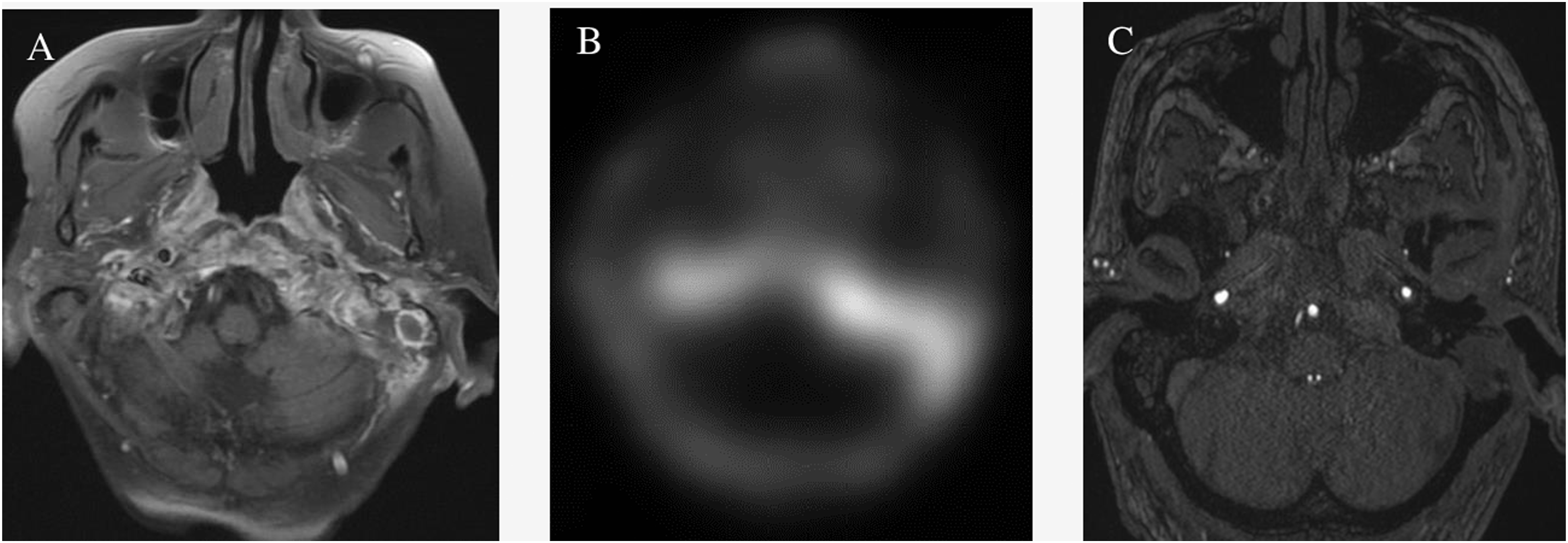
A 76-year-old man with type 2 diabetes mellitus and heart failure presents with left ear pain, purulent drainages, and post-auricular swelling. His otolaryngologist previously treated him with several courses of oral antibiotic and antibiotic ear drops. Still, his symptoms continue to return despite brief 1-to-2-month periods of improvement. His initial CT demonstrated erosive changes in the clivus and right occipital condyle. MRI showed mild osseous enhancement of basisphenoid and left greater than right enhancing inflammatory tissues in the infratemporal fossa (Figure 3). The patient agreed to an elective canaloplasty of the left ear and the resection of a biopsy-proven granulomatous mass within the left external auditory canal. He was discharged home after the surgery with improvement in his pain and swelling and no postoperative complications. (a,b) Computed tomography demonstrates erosive changes in the clivus and right occipital condyle. (c) Initial magnetic resonance imaging shows mild osseous enhancement of the basisphenoid and left greater than right enhancing inflammatory tissues in the infratemporal fossa.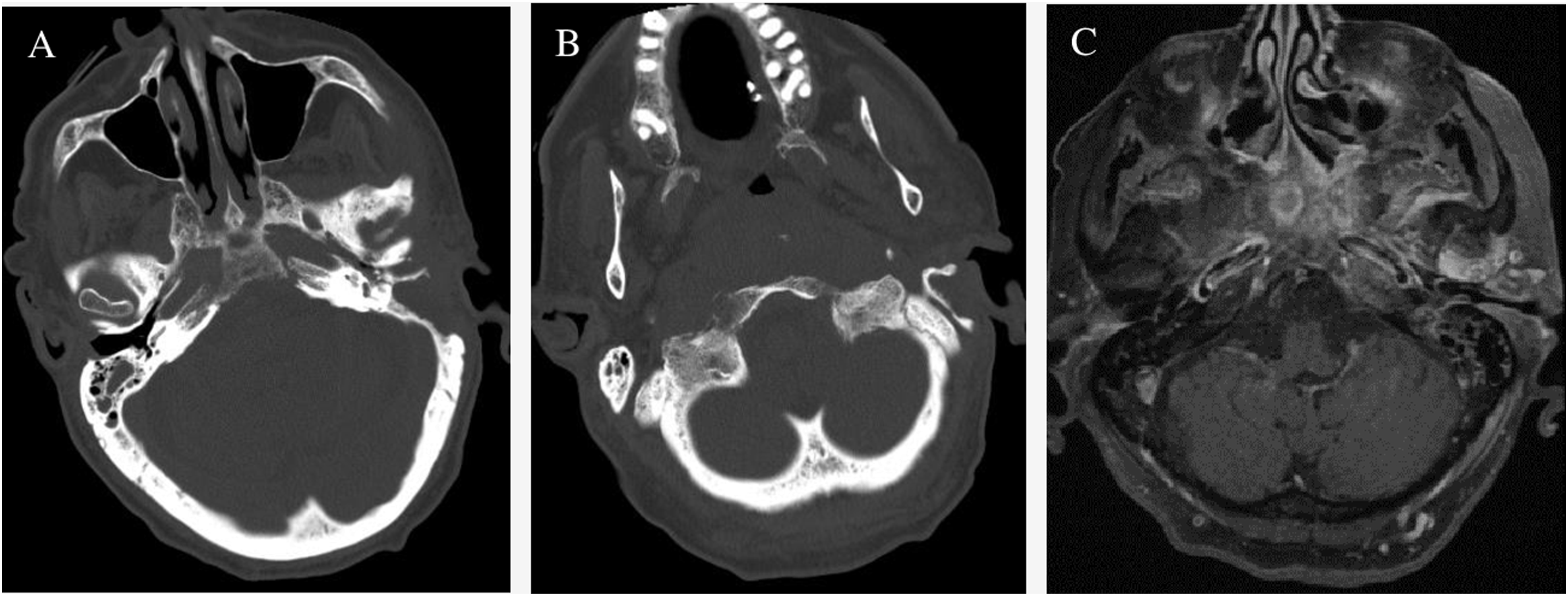
Two months later, the patient returned with increasing left ear pain and new onset of left hypoglossal paralysis. Due to cranial nerve involvement, a left mastoidectomy and myringotomy with tympanostomy tube were performed and intraoperative culture was taken which was positive for Staphylococcus epidermidis and Aspergillus niger. A swab was also positive for methicillin-resistant Staphylococcus aureus. After the surgery, the patient again had improvement in his symptoms and was discharged home with vancomycin, voriconazole, and tobramycin.
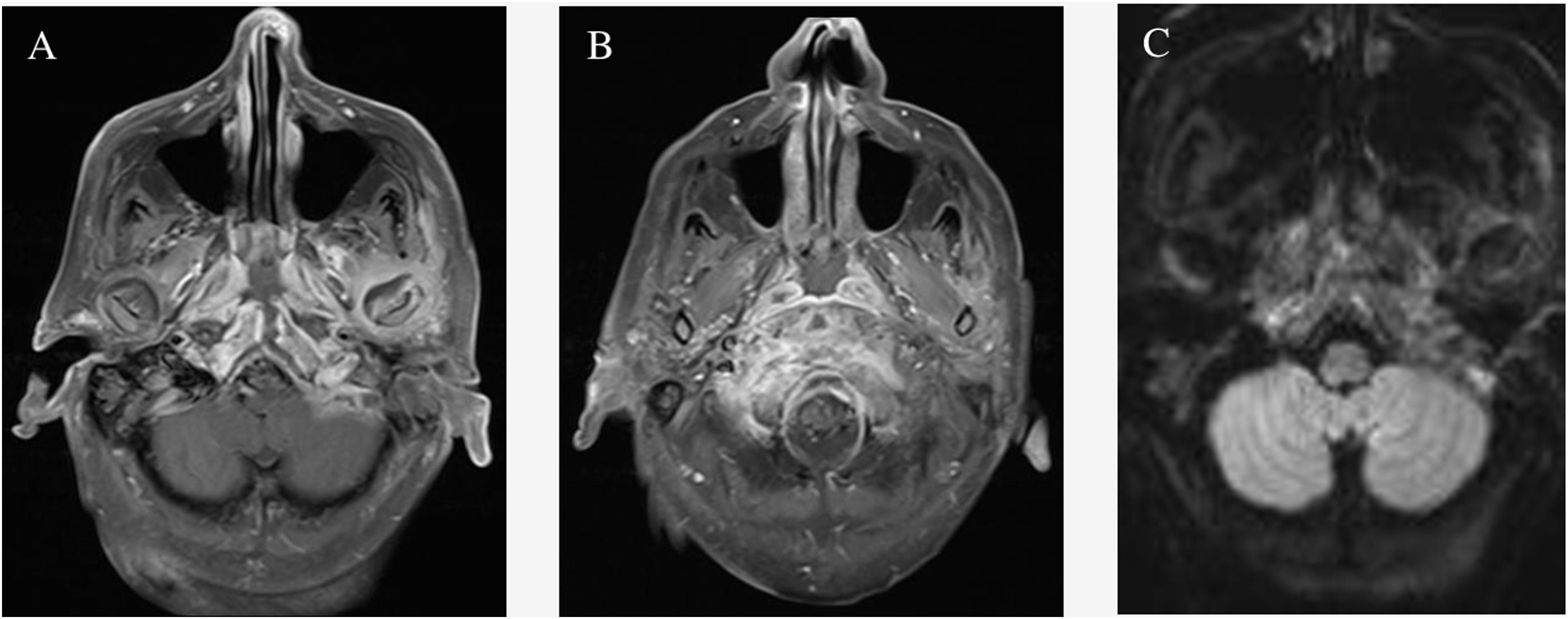
Another two months later, the patient re-presented with worsening ear pain, new-onset headache, and progressively poor appetite, dysphagia, slurring, and difficulty ambulating. His CT now demonstrated worsening erosions of the left petrous apex and central skull base osteolysis with left opacification of the mastoids consistent with clinical otomastoiditis and retroauricular soft tissue swelling and cellulitis (Figure 4). Post-contrast fast spin echo (FSE) T1 sequences demonstrated lack of opacification of the left distal transverse sinus with corresponding T2 weighted images demonstrating lack of the flow void in this same region (Figure 5). Magnetic resonance cerebral venography (MRV) demonstrates lack of flow-related enhancement in the left distal transverse sinus. Post-contrast fast spoiled gradient echo (FSPGR) sequences confirm the thrombosis of the left sigmoid sinus (Figure 6). Follow-up MRI showed increased enhancement of the skull base and surrounding tissue and wall thickening and enhancement of the traversing internal carotid arteries with left coalescent mastoiditis. Uptake on follow-up Gallium-67 single-photon emission computed tomography (SPECT) displayed an extensive and progressive infection. Magnetic resonance angiography (MRA) revealed subtle narrowing of the left petrous internal carotid artery secondary to soft tissue involvement from SBI (Figure 7). Post-contrast images of the nasopharynx and skull base demonstrated increased enhancement and worsening extension of SBI. Diffusion-weighted images showed restricted diffusion in this same region (Figure 8). Computed tomography demonstrates worsening erosions of the left petrous apex and central skull base osteolysis. Left opacification of the mastoids and retroauricular soft tissue swelling and cellulitis is consistent with clinical otomastoiditis. (a,b) Post-contrast fast spin echo T1 sequences demonstrate lack of opacification of the left distal transverse sinus with corresponding T2 weighted images demonstrating lack of the flow void in this same region. (a,b) Magnetic resonance venography demonstrates lack of flow-related enhancement in left distal transverse sinus. Post-contrast fast spoiled gradient echo sequences (right) demonstrate lack of opacification in the left sigmoid sinus. (a) Follow-up magnetic resonance imaging demonstrates increased enhancement of skull base and surrounding tissue, wall thickening and enhancement of the traversing internal carotid arteries, and left coalescent mastoiditis. (b) Uptake on follow-up Gallium-67 single photon emission computed tomography displays the extent and progression of infection. (c) Magnetic resonance angiography demonstrates subtle narrowing of the left petrous internal carotid artery secondary to soft tissue involvement from skull base infection. (a,b,c) Post-contrast images of the nasopharynx and skull base demonstrating increased enhancement and worsening extension of skull base infection. Diffusion weighted images show restricted diffusion in this same region.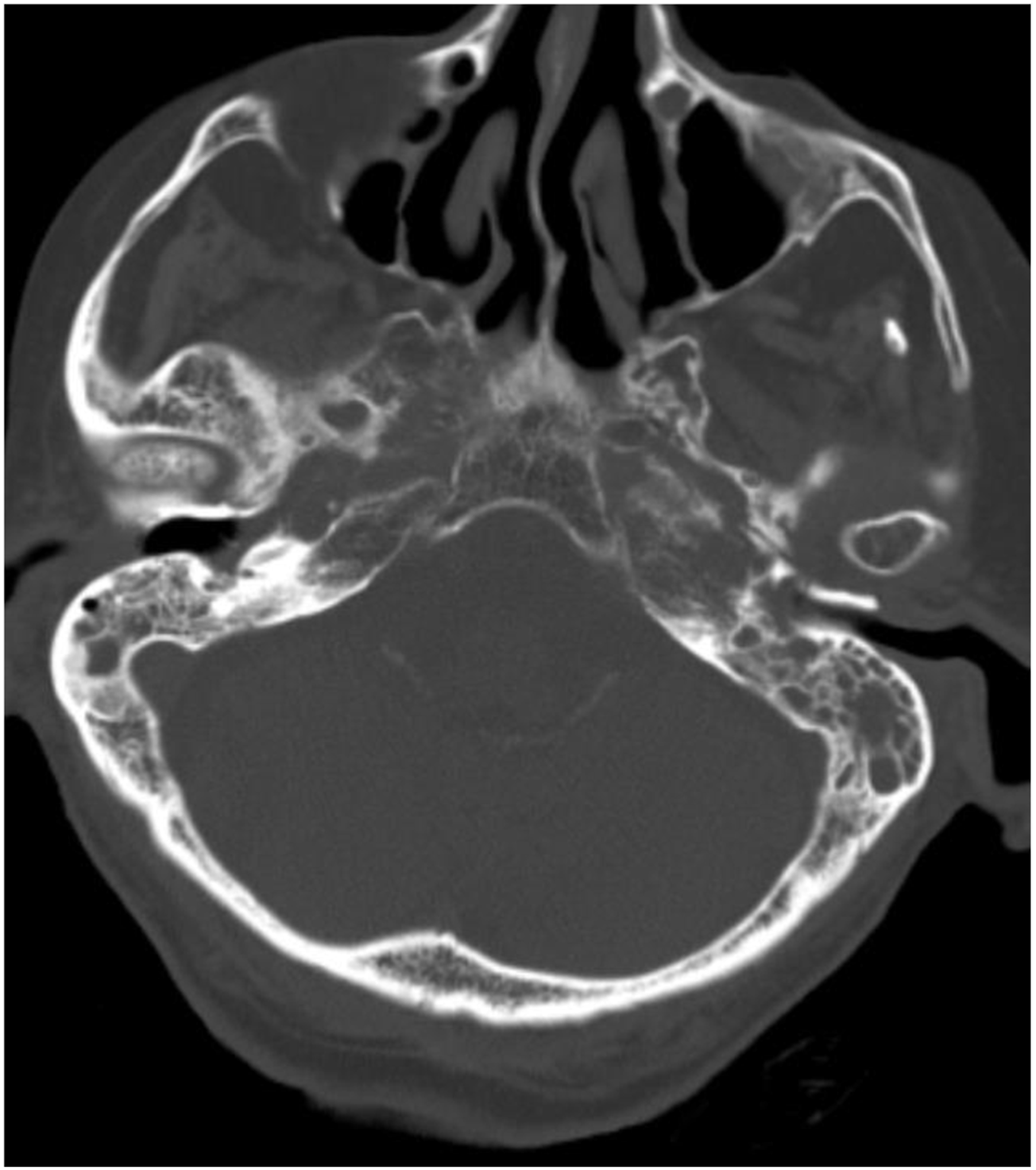
The patient’s diagnosis was determined to be chronic and progressive skull base osteomyelitis based on his persistent symptoms and corroborative imaging findings. Additionally, biopsies of the nasopharyngeal fascia were positive for MRSA, Pseudomonas aeruginosa, and Corynebacterium species. A peripheral catheter was inserted for further broad-spectrum antibiotics, with posaconazole, ciprofloxacin, piperacillin–tazobactam, and vancomycin. There was no further surgical management due to the patient’s poor prognosis in the setting of advanced infection. After a prolonged period of intensive level care, our patient was transitioned to comfort care and expired during hospitalization.
Pathophysiology
Certain etiologies have a strong association with their primary anatomical site of infection. Classifications are therefore an important discussion within the literature. Infections that start laterally in the temporal bone are coined “typical” skull base infections. These include conditions affecting the external auditory canal, mastoid, and middle ear cavity.2,29,30 Suppurative otitis media, malignant otitis externa, and acute mastoiditis are pathologies in this anatomical region that can progress to SBI and a diverse array of complications depending on the route of spread. Anterior extension from the external auditory canal through the fissures of Santorini can involve the parotid gland, temporomandibular joint, and stylomastoid foramen, leading to xerostomia, trismus, and ipsilateral facial nerve palsy, respectively. 2 If the infection progresses posteriorly, involvement of the jugular foramen and hypoglossal canal may lead to cranial nerves IX–XII neuropathy along with thrombosis of the jugular bulb and sigmoid venous sinus via the mastoid emissary veins.6,30 A more medial spread has the potential of involving the internal carotid artery, clivus, petrous apex with cranial nerves V and VI, retropharyngeal space, parapharyngeal space, and contralateral cervical spine.3,11,31,32 The most common organisms in these “typical” SBIs are Pseudomonas aeruginosa and Staphylococcus aureus. Other reported bacterial species include Klebsiella, Salmonella, Staphylococcus epidermidis, and Proteus mirabilis.2,33 While fungal infections are less common in lateral SBIs compared to medial SBIs, Aspergillus species are the most likely culprit in such rare instances.25,27,28,33
Primary sites of infection within the nasal cavity and paranasal sinuses that progress to medial skull base infections involving the sphenoid and occipital bones are considered “atypical.” The most common route of intracranial spread is through bony erosion across the involved paranasal sinuses and into the anterior or middle cranial fossa or through the adjacent venous channels. The frontal, ethmoid, and sphenoid sinuses are particularly vulnerable to these mechanisms owing to their intimate relationship with the cranial vault and cavernous sinuses.2,34–36 Like typical SBIs, infections originating from the sinuses and nasal cavity are predominantly bacterial and often due to Pseudomonas and Staphylococcus species.4,37–39 Other routes of intracranial spread to consider include the round and oval windows, endolymphatic sac, cochlear aqueduct, and hematogenous seeding and sutures. 9
Clinical presentation
Individuals who are elderly, diabetic, or immunocompromised are most commonly affected. Their susceptibility is likely secondary to compromised bone vascularity and a poor host immune defense.29,40 Owing to this mechanism, other known comorbid conditions associated with SBI also include renal failure, liver failure, and small vessel diseases. 33 Locoregional processes should also be considered as osteoradionecrosis, surgical site infections, foreign bodies, and tissue scarring have also been previously documented in cases of SBI. Postoperative craniotomy-related infections are the predominant cause of cranial osteomyelitis in developed countries, whereas paranasal sinusitis, scalp infections, and direct head injuries are the most common cause in developing nations.7,29,41
Clinical characteristics of SBI.

Imaging
Unenhanced CT is often the first-line modality for evaluation of a suspected SBI. As demonstrated by our two patient cases, CT may demonstrate demineralization and bony erosions. Opacification of the mastoid air cells and external auditory canal with new or increasing bony erosions in the walls of the surrounding temporal bone, petrous apex, clivus, jugular foramen, and skull base foramina should raise suspicion for SBI. Similarly, the walls of the paranasal sinuses should be meticulously examined for evidence of erosions in the presence of opacification, mucosal thickening, and air-fluid levels. Soft tissue swelling, infiltrations of fat planes, and enhancement are best shown on contrast-enhanced CT with soft tissue windows. Asymmetric soft tissue thickening of the nasopharynx may mimic an infiltrating neoplasm, such as nasopharyngeal carcinoma. The soft-tissue involvement may precede frank bony erosions and can occasionally evolve to abscess formation. Involvement of neurovascular structures can result in cavernous sinus thrombosis or stroke. 30 Vascular complications can be further assessed by CT angiography or CT venography.
MRI is superior to CT for depicting soft-tissue and marrow involvement in addition to intracranial complications. The use of pre-contrast spin-echo T-weighted imaging sequences is an excellent starting point for review. Replacement of high T1 signal of the marrow and fascial fat planes with intermediate to hypointense T1 signal should be considered suspicious for SBI. Detection of skull base foramina involvement is facilitated on pre-T1 images by noticing fat replacement surrounding these structures. T2 and short tau inversion recovery (STIR) can assist in defining the disease spread pattern with delineation of edema, inflammation, and phlegmon in the affected soft tissues and marrow adjacent to primary sources such as the ears and paranasal sinuses. DWI and apparent diffusion coefficient (ADC) maps are helpful tools for differentiating edema and phlegmon as well as identifying complications such as abscesses. After initiation of treatment, DWI is the most reliable MR sequence in distinguishing active infection and post-inflammatory fibrosis and granulation tissue, which can persist for more than a year after the initial insult. 46 Furthermore, DWI is demonstrated to be diagnostically helpful in differentiating between SBI versus lymphoma and nasopharyngeal carcinoma, with higher ADC values seen in the former. 21 T1-weighted fat-saturated contrast-enhanced images can delineate the extent of inflammatory tissue. It is worth noting that the skull base and surrounding tissues have a rich vascular network which should be differentiated from enhancing inflammatory tissues. Additionally, the lack of normal enhancement of these structures and bone marrow may reflect developing necrosis which can ultimately lead to abscess formation. MR angiography and MR venography are complementary methods to investigate vascular complications of SBI including occlusion, thrombosis, and mycotic pseudoaneurysm.
Imaging findings of skull base infections.

The 18F-FDG (fluorodeoxyglucose) is the main β-emitting tracer for infection assessment. FDG positron emission tomography (PET) can aid in delineating disease extent and evaluation of treatment response. However, FDG is not a specific tracer for infection and can show increased uptake in other conditions such as malignancy. Hybrid imaging techniques such as PET-CT or PET-MRI can improve spatial resolution and anatomic details. 47
Management
Complications of skull base infections.

A regimen covering both bacteria and fungus was initiated in our second case due to culture-positive MRSA, P. aeruginosa, and A. niger. Regarding the duration of therapy, there is no difference in clinical outcomes between ceasing antibiotics based on clinical, laboratory, or imaging evidence of disease resolution. However, there may not be enough cases in previous studies to detect such differences, and further research is needed to validate this non-difference. At a baseline, six to eight weeks of initial intravenous therapy is recommended with a switch to oral formulation after there is a stable clinical improvement.24,26–28,51,52 Finally, intensive level care may be necessary for those with a severe disease burden affecting their neurologic function or if there is significant end-organ damage.
Surgery is appropriate when there is a need for biopsy, culture, abscess drainage, or debridement of necrotic tissue, or in patients with progressive neurologic dysfunctions. Various skull base approaches can be utilized to achieve these goals, and neurological function can sometimes be protected or recovered in cases where decompression is needed. 43 It is important to note that aggressive removal of infected and necrotic soft tissue and skull base bone can result in destabilization, significant dead space, and/or cosmetic deformity, each of which may require additional surgical procedures. The goals of radical resection must be weighed against the potential consequences of aggressive soft tissue removal. For the patients in our series, surgery was performed to eliminate the nidus of infection, reduce inflammatory processes, and improve antimicrobial penetration to de-vascularized tissue. Intraoperative cultures were also performed to clarify antibiotic therapy and confirmed the presence of P. aeruginosa and MRSA.
Lastly, while no substantial evidence yet exists for hyperbaric oxygen therapy, and there is a risk for barotrauma, oxygen toxicity, and tympanic membrane perforation, its use as an adjunctive treatment is generally well tolerated and may be considered in severe or refractory cases.53,54
Overlap with common mimics
Commonly considered differentials in skull base infections.

(a) Post-contrast axial T1 with fat saturation shows abnormal enhancement of the left petrous apex with extension to the nasopharynx. (b) Axial T2 with fat saturation shows opacification of the left mastoid air cells and abnormal signal in the left petrous apex. (c) Diffusion weighted imaging shows abnormal restricted diffusion in the left petrous apex.
Discussion
Skull base infections are strongly associated with diabetics and immunocompromised patients—individuals susceptible to persistent infections that normally would have resolved after a course of antimicrobial therapy. Their susceptibility to osteomyelitis is theorized to be due to decreased bone vascularity and altered host immune defenses secondary to systemic disease.3,29,40,42,55 These cases may often present as mild local infections originating from the external auditory canal or paranasal sinuses. Their persistence, however, over several months or years eventually leads to involvement of the surrounding vascular and bony anatomy. Neurologic dysfunction is a concerning feature once there is extension into the brain parenchyma or cranial nerves traversing the skull base foramen.
As demonstrated by our patient cases, common imaging findings on CT may include soft tissue swelling, demineralization, and bony erosions. Such skull base erosive changes can be subtle, and attention should be given to the skull base foramina as their involvement is implicated in cranial nerve deficits. MRI will demonstrate marrow replacement with hypointense T1 signal and osseous and soft tissue enhancement. Vascular structures may also be involved, and flow voids should be carefully appreciated, and early venous sinus thrombosis, as seen in our patient, must be considered. If there is a high enough clinical suspicion, post-contrast FSPGR sequences can complement MRV in diagnosing dural venous sinus thrombosis. Gallium-67 scintigraphy will positively correlate with the progression of infection and can help monitor treatment response. Finally, DWI is diagnostically useful in differentiating skull base infections from lymphoma and malignancies, with a higher ADC associated with SBI.(11,16,19–21,34,56)
Culture-guided antimicrobial therapy ranging from six to twenty weeks is the mainstay treatment for SBI alongside early surgical debridement. Since the most common pathogens are Pseudomonas and Staphylococcus species, SBIs are typically treated intravenously for the first six to eight weeks with an aminoglycoside and β-lactamase, third-generation cephalosporin, or fluoroquinolone. While combination antibiotic therapy is generally not recommended for other kinds of P. aeruginosa infections such as pneumonia, dual coverage is appropriate in cases of P. aeruginosa-identified SBI.3,7,57 As seen in both of our cases, P. aeruginosa was treated with ciprofloxacin in combination with cefepime and piperacillin–tazobactam for cases one and two, respectively. If the patient is stable enough for outpatient therapy, then an oral formulation of ciprofloxacin may be used, though it must be initiated at a full dose for at least six to eight weeks. Follow-up for improvements in symptoms and inflammatory markers are essential in these instances to assess for treatment response as failure would require re-culturing or biopsy. 49 Generally, initial preservation of extra specimens for polymerase chain reaction (PCR) analysis should also be considered given the importance of an early and accurate diagnosis in SBI and to avoid delays in appropriately targeted therapy. This is especially prudent if cultures were collected in patients who have already received antimicrobial treatment which could erroneously influence results to be negative—especially in cases where purulent discharge may be lacking.
If the infection is of fungal origin, then Aspergillus species are the most likely pathogens identified. While intravenous voriconazole is recommended for patients with significant disease burden from Aspergillus species, oral formulations may be given for patients who are stable or who improved after intravenous treatment.25,58 For second-line therapy, a high-dose amphotericin B is the preferred agent. It has a more extensive side effect profile, including renal toxicity, hypokalemia, hypomagnesemia, anemia, and thrombocytopenia when compared to voriconazole’s mild effect on the liver. However, a new lipid formulation of amphotericin B has gained recent favor due to its comparable efficacy and lower adverse effect profile than high-dose amphotericin B. 7
Early and aggressive surgical management is recommended alongside systemic antimicrobial therapy. Although there has been an association found between surgery, extended hospital stays, and an increased likelihood for relapsing SBI, a cause-and-effect relationship is difficult to establish since patients with advanced skull base infections are more likely to have required surgery and a prolonged hospitalization in the first place regardless of the initial strategy.1,43 As a result of this bias, the evidence arguing against early and aggressive surgical management is not definitive and should not delay treatment.
SBIs are difficult to diagnose definitively due to their considerable overlap in presentation with other critical differentials, such as nasopharyngeal carcinoma and metastatic disease. A multimodal approach could facilitate the correct diagnosis, and clinicians must carefully consider the need for biopsy if appropriate.6,16,20,30,59 It is also critical to assess for dural or leptomeningeal involvement and any other associated intracranial complications such as cerebritis or empyema.
Finally, there is a lack of rigorously validated treatment recommendations which we believe may benefit from additional research. For example, no substantial evidence exists yet which supports the use of hyperbaric oxygen therapy. Additionally, debate persists regarding when it may be optimal to conclude antimicrobial therapy and whether that should be based on clinical, imaging, or laboratory evidence of disease resolution.
Conclusion
Skull base infections are life-threatening diseases requiring early interventions that are often delayed due to diagnostic difficulties on imaging. No pathognomonic imaging findings are associated. A multimodal approach with CT, MR, and nuclear medicine is necessary to characterize soft tissue and bony involvement, the extent of intracranial invasion, and therapeutic response. Careful consideration and work-up should be performed for other potential diagnoses such as nasopharyngeal carcinoma and inflammatory pseudotumor as they may mimic skull base infections on imaging.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
